Published online Mar 20, 2026. doi: 10.5493/wjem.v16.i1.111034
Revised: August 14, 2025
Accepted: January 6, 2026
Published online: March 20, 2026
Processing time: 266 Days and 19.9 Hours
The ocular manifestations in acute leukemia can occur due to primary leukemic infiltration or secondary to the disease and chemotherapy complications. As the life expectancy has increased in leukemia cases due to the advent of modern chemotherapy, the incidence of ocular features is more evident.
To describe the clinical pattern of ocular manifestations at the time of diagnosis of acute leukemia in children.
This was a hospital-based, cross-sectional study conducted at a tertiary care academic Institute in eastern India between July 2016 to December 2019. All children below 15 years diagnosed with acute leukemia underwent a comprehensive eye checkup before initiation of chemotherapy. The demographic details, type of leukemia, hematological findings, and chemotherapy regimen were documented.
Among the 47 children diagnosed with acute leukemia only 19 cases (40.4%) had ocular involvement. There were 33 boys (70.2%) and 14 (29.8%) girls. Ocular involvement was more common in lymphoblastic leukemia in our study in 13 cases [39.4%; 11 B-acute lymphoblastic leukemia (B-ALL), 2 T-ALL] than in mye
Ophthalmological manifestations are more common in ALL patients (39.4%). Periodic ophthalmic evaluations of all patients must be made to detect ocular findings in asymptomatic cases. Early detection, risk stratification, and regular follow-up are also needed.
Core Tip: This study was undertaken to analyze the ocular features of cases of acute leukemia in children. Most of the cases are asymptomatic at presentation and can manifest with vision-threatening features in the form of vitreous hemorrhage and subhyaloid hemorrhage in the posterior pole, which needs immediate treatment. Hence, periodic ocular examinations are needed.
- Citation: Parija S, Palanisamy S. Acute childhood leukemia and ophthalmic manifestations: An observational study from Eastern India. World J Exp Med 2026; 16(1): 111034
- URL: https://www.wjgnet.com/2220-315x/full/v16/i1/111034.htm
- DOI: https://dx.doi.org/10.5493/wjem.v16.i1.111034
Leukemia is a malignancy of the hematopoietic stem cells affecting children as well as adults. It can have an acute or insidious onset, presenting as acute or chronic leukemia. Acute leukemia is a result of the clonal proliferation of blast cells, which are immature white blood cells (WBCs), whereas chronic leukemia is caused by abnormal proliferation of mature and maturing WBCs[1]. Based on the progenitor cell immunophenotype, they can be classified into two subtypes: Lymphoid and myeloid. Leukemia in children is predominantly acute. Acute lymphoblastic leukemia (ALL) is more common in children[2,3]. The leukemias are primarily present as high leucocyte count with a preponderance of blast in the bone marrow, peripheral blood, and lymphoid tissue, which are the major sites of involvement in leukemia. Visceral involvement is not uncommon in leukemias, and ocular involvement can range from 9% to 90% in all leukemias[4]. In the 1860s, the retina was detected to be the most commonly affected tissue, and the retinal findings were first described by Liebreich as leukemic retinopathy[3,5]. Later, the involvement of other structures of the eye was also reported. Ophthalmic involvement can be direct or indirect. Also, infiltration of leukemic blasts directly into the optic nerve and orbit results in several neurological and orbital findings[6]. Leukemia-associated changes, such as hemorrhages and coagulopathy can indirectly affect the retina.
With the advancement of therapeutics, there has been an increase in the mean life expectancy of children with acute leukemia. Thus, the incidence of ocular findings is more evident. The manifestations can be observed at the time of presentation, during treatment, or on follow-up. This present study aimed to describe the clinical pattern of ocular manifestations at the time of diagnosis of acute leukemia in children.
This is a descriptive cross-sectional hospital-based study. All the children below 15 years diagnosed with acute leukemia and admitted for chemotherapy to a tertiary hospital in Eastern India between July 2016 and December 2019 were included in the study. Institutional Ethical Committee approval was sought, and the study was conducted as per ethical standards outlined in the 1964 version of the Declaration of Helsinki. Written consent from the guardian or parents was obtained at the time of the ophthalmic examination of the child.
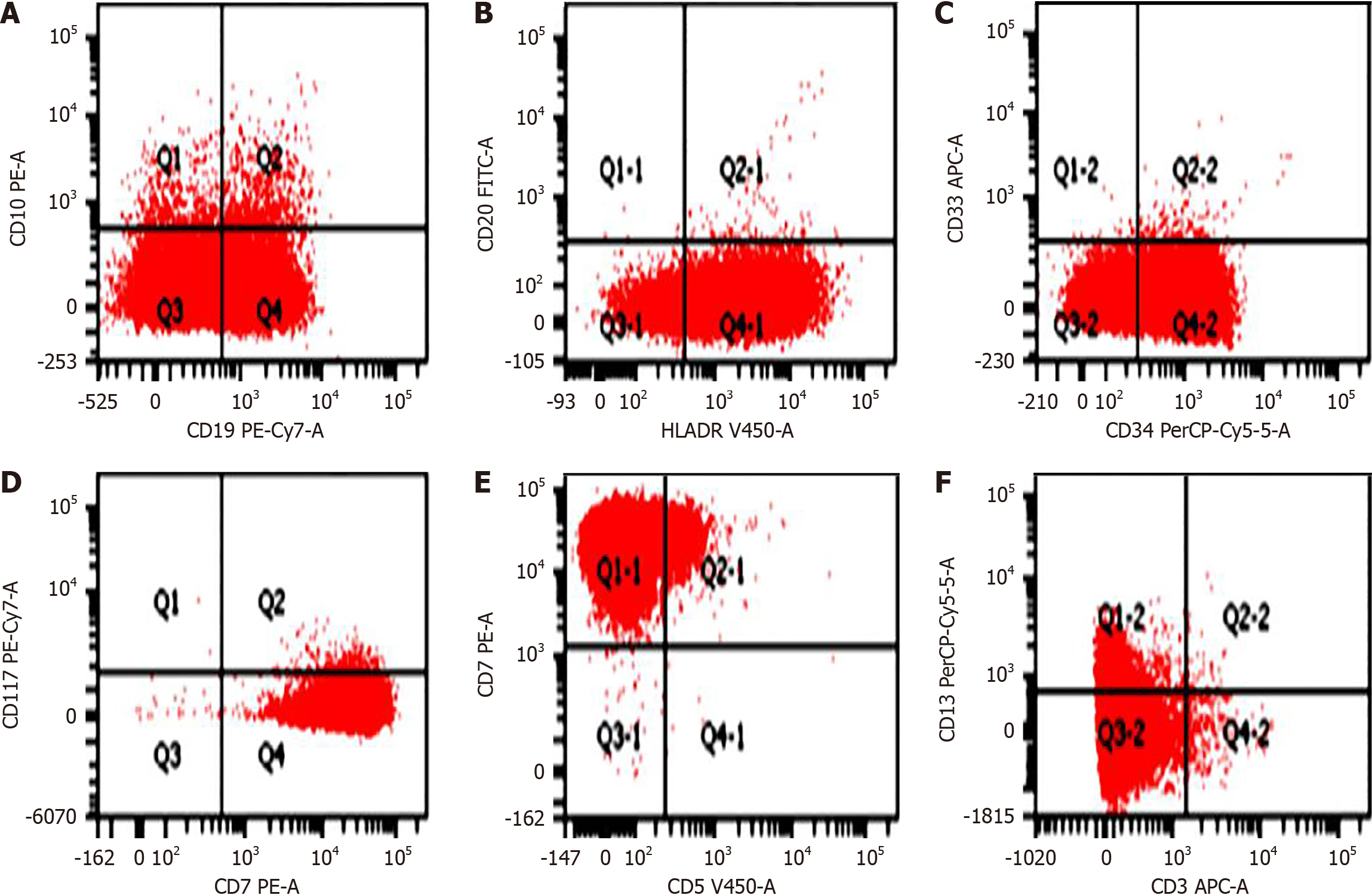
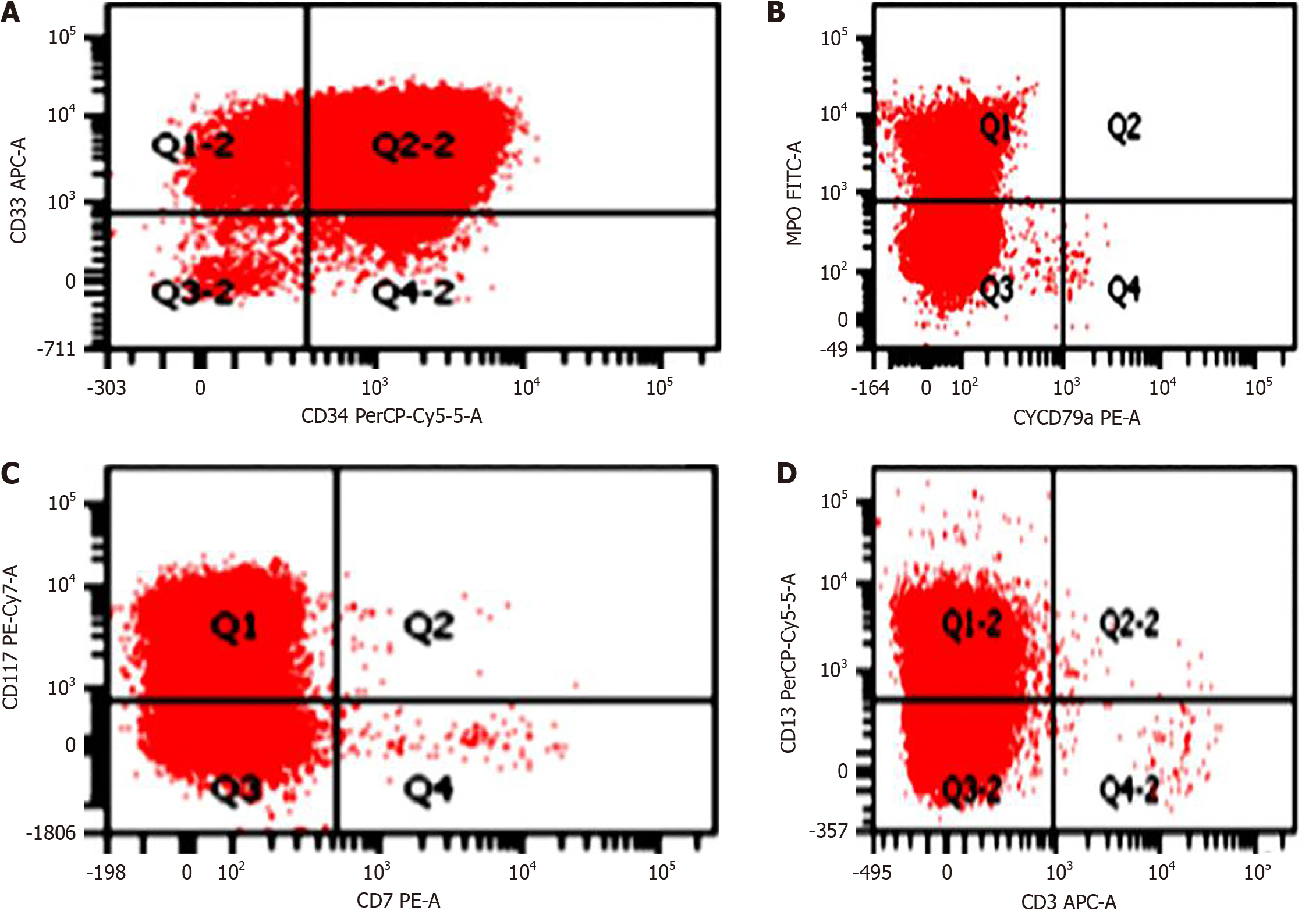
The diagnosis of leukemia was based on the history, clinical profile, complete hemogram, and peripheral blood smear morphology. This was further confirmed and classified by bone marrow aspiration to ascertain the blast morphology and enumeration (percentage of blasts). Flow cytometric immunophenotyping was performed for the diagnosis and subtyping of leukemia into acute myelogenous/myeloblastic/myeloid (AML), B-ALL, T-ALL, and mixed phenotype/Lineage acute leukemia (MPAL). Multicolor flow cytometric immunophenotyping of the bone marrow aspirate sample was performed in each case, using CD45 vs side scatter gating in the BD FACS Canto II/BD FACS DIVA (Version 8.0.2) instrument and software. The stain-lyse-wash method was used for the preparation of the sample. The patients with sickle cell anemia, diseases with ocular manifestations mimicking leukemia, any other associated comorbidities such as hypertension and diabetes, and very sick leukemic patients were excluded from the study. All the leukemic cases, regardless of their ocular complaints and before initiation of chemotherapy, were examined by the ophthalmologist. A protocol was maintained to record the demographic data, type of leukemia, duration of illness, ocular lesions, hematological parameters, radiological findings, and the chemotherapy regimen. A detailed ophthalmological examination included visual acuity testing where possible, ocular motility testing, anterior segment details by the slit lamp biomicroscopy, and fundus by indirect ophthalmoscopy. Special tests like Hertel exophthalmometry, intraocular pressure recording, and ultrasonography of the eye were performed as and where required. Fundus photographs were taken for documentation, where the child was cooperative. Primary tumor infiltration was defined if there was a cellular mass formation in the ocular tissues due to direct infiltration of leukemic cells, while secondary changes were attributed to predominantly hemorrhagic manifestations due to hematological changes. Systemic evaluation, hematological status, chemotherapy regimen, and radiation therapy were monitored by a pediatric oncologist and radiologist, respectively.
The data was entered in an Excel spreadsheet and underwent statistical analysis using Statistical Package for Social Sciences for Windows software, version 16 (SPSS Inc., Chicago, IL, United States). Quantitative data was measured using arithmetic mean and median values, whereas percentages and frequencies were used for qualitative data. The level of statistical significance was set at P < 0.05.
Among the 47 children with a diagnosis of acute leukemia, only 19 cases (40.4%) had ocular involvement. There were 33 boys (70.2%) and 14 (29.8%) girls in the study. The age ranged from 2 years 6 months to 15 years, with a peak between 5-10 years (44.7%); the mean age was 5.7 years. The demographic data of all patients suffering from acute leukemia is illustrated in Table 1. There were 36 newly diagnosed cases and 11 Leukemic children on follow-up. In total, 33 cases (70.2%) were suffering from ALL, and 14 cases (29.8%) were from AML. There was no case of MPAL. Among the ALL category, 28 were B-ALL, and 5 were T-ALL. The immunophenotyping details were described in Table 2.
| Sl No. | Age in years | Sex | Type of leukemia | Systemic features | Ocular lesions | Laterality of eye | Treatment |
| 1 | 12 | F | ALL | Present | SH + IRH | Both | Chemotherapy |
| 2 | 7 | M | ALL | Nil | 6th CN palsy | Left | Chemotherapy |
| 3 | 5 | M | T-ALL | Nil | Proptosis | Right | Chemotherapy |
| 4 | 6 | F | ALL | Present | VH + IRH | Both | Chemotherapy |
| 5 | 2.5 | M | ALL | Present | IRH | Both | Chemotherapy |
| 6 | 2.5 | M | ALL | Present | IRH | Right | Chemotherapy |
| 7 | 5 | M | ALL | Present | IRH | Both | Chemotherapy |
| 8 | 4 | F | T-ALL | Nil | Proptosis, disc edema | Left | Chemotherapy+ irradiation |
| 9 | 6 | M | AML | Present | VH | Right | Chemotherapy |
| 10 | 13 | F | AML | Nil | Chloroma, 6th CN Palsy, Proptosis, IRH | Left | Chemotherapy+ irradiation |
| 11 | 3 | F | AML | Present | IRH | Left | Chemotherapy |
| 12 | 6 | M | B-ALL | Nil | Anterior uveitis | Right | Chemotherapy |
| 13 | 4 | F | B-ALL | Present | IRH | Right | Chemotherapy |
| 14 | 7 | M | AML | Nil | Proptosis | Left | Chemotherapy |
| 15 | 3 | F | ALL | Present | IRH | Right | Chemotherapy |
| 16 | 6 | M | AML | Nil | Chloroma, IRH | Both | Chemotherapy |
| 17 | 7 | M | ALL | Nil | IRH | Right | Chemotherapy |
| 18 | 5 | F | AML | Nil | Proptosis, papilloedma | Right | Chemotherapy+ irradiation |
| 19 | 4 | M | ALL | Nil | IRH | Left | Chemotherapy |
| Patient | Age (years) | Gender | Initial presentation | Hemoglobin (g/dL) | Total platelet count (× 103/μL) | WBC count (× 103/μL) | Flow cytometric immunophenotyping of the blasts | Type of leukemia | Systemic features | Ocular lesions | Laterality of eye | Treatment |
| 1 | 12 | F | Fever | 3 | 20 | 120 | CD19+/CD10+/dim CD34+/HLA-DR/CD79a+/Tdt+/CD20-/aberrant CD33+/other markers negative | B-ALL | Present | SH and IRH | BE | Chemotherapy |
| 2 | 7 | M | Diplopia | 9.8 | 65 | 77 | CD10+/CD19+/CD20 dim+/CD34 dim+/HLA-DR+/CD79a+/Tdt+/other markers negative | B-ALL | Nil | 6th Cranial Nerve Palsy | LE | Chemotherapy |
| 3 | 5 | M | Proptosis | 5.5 | 45 | 54 | CD7+/CD34+/ HLA-DR+/ surface CD3-/surface CD5-/surface CD10-/cytoplasmic CD3+/cytoplasmic Tdt+/other markers negative | T-ALL | Nil | Proptosis | RE | Chemotherapy |
| 4 | 6 | F | Fever | 7.4 | 18 | 90 | CD10+/CD19+/CD20 dim+/CD34+/HLA-DR+/CD79a+/Tdt+/other markers negative | B-ALL | Present | VH and IRH | BE | Chemotherapy |
| 5 | 2.5 | M | Fever | 10.1 | 21 | 107 | CD10+/CD19+/CD34+/HLA-DR+/CD79a+/Tdt+/ CD20-/other markers negative | B-ALL | Present | IRH | BE | Chemotherapy |
| 6 | 2.5 | M | Fever | 6.5 | 34 | 95 | CD10+/CD19+/CD20 dim+/CD34 dim+/HLA-DR+/CD79a+/Tdt+/other markers negative | B-ALL | Present | IRH | RE | Chemotherapy |
| 7 | 5 | M | Fever | 4 | 23 | 83 | CD10+/CD19+/CD20 moderate+/CD34+/HLA-DR+/CD79a+/Tdt+/other markers negative | B-ALL | Present | IRH | BE | Chemotherapy |
| 8 | 4 | F | Fever | 9.4 | 29 | CD7+/CD5+/dim CD4/surface CD3+/ CD38+/cytoplasmic CD3+/Tdt+/dim CD34+/CD8-/HLA-DR-/other markers negative | T-ALL | Nil | Proptosis and Disc Edema | LE | Chemotherapy and irradiation | |
| 9 | 6 | M | Fever | 4.5 | 27 | 112 | CD34+/CD117+/CD33+/CD13+/HLA-DR-/cytoplasmic MPO+/other markers negative | AML | Present | VH | RE | Chemotherapy |
| 10 | 13 | F | Fever | 7.2 | 29 | 97 | CD38+/CD117+/CD33+/CD13+/ HLA-DR+/dim CD56/dim CD36/cytoplasmic MPO-/other markers negative | AML | Nil | Chloroma, 6th Cranial Nerve Palsy, Proptosis, and IRH | LE | Chemotherapy and Irradiation |
| 11 | 3 | F | Fever | 5.5 | 35 | 96 | CD34+/CD117+/HLA-DR+/CD33+/dim CD13/cytoplasmic MPO/other markers negative | AML | Present | IRH | LE | Chemotherapy |
| 12 | 6 | M | Fever | 3.8 | 32 | 105 | CD19+/CD10+/CD34+/ CD38+/HLA-DR+/CD79a+/CD20-/other markers negative | B-ALL | Nil | Anterior uveitis | RE | Chemotherapy |
| 13 | 4 | F | Fever | 7.5 | 29 | 98 | CD10+/CD19+/CD20 dim+/CD34 dim+/HLA-DR+/CD79a+/Tdt+/other markers negative | B-ALL | Present | IRH | RE | Chemotherapy |
| 14 | 7 | M | Proptosis | 5 | 78 | 75 | CD38+/CD33+/CD117+/dim CD4/dim CD64/dim CD15/ CD34+/HLA-DR-/CD14-/cytoplasmic MPO-/other markers negative | AML | Nil | Proptosis | LE | Chemotherapy |
| 15 | 3 | F | Fever and vomiting | 10.5 | 64 | 59 | CD7+/CD10+/dim CD5/surface CD3-/cytoplasmic CD3+/Tdt+/other markers negative | T-ALL | Present | IRH | RE | Chemotherapy |
| 16 | 6 | M | Fever | 7.8 | 31 | 101 | CD34+/CD117+/CD33+/CD13+/HLA-DR-/cytoplasmic MPO+/others markers negative | AML | Nil | Chloroma and IRH | BE | Chemotherapy |
| 17 | 7 | M | Fever | 4 | 35 | 94 | CD10+/CD19+/CD34+/HLA-DR+/CD79a+/Tdt+/variable CD20/other markers negative | B-ALL | Nil | IRH | RE | Chemotherapy |
| 18 | 5 | F | Proptosis | 3.7 | 54 | 123 | CD13+/CD33+/CD117+/CD38+/CD64+/HLA-DR+/cytoplasmic MPO+/other markers negative | AML | Nil | Proptosis and papilledma | RE | Chemotherapy and irradiation |
| 19 | 4 | M | Blurring of vision | 9.5 | 24 | 39 | CD19+/CD10+/HLA-DR+/dim CD34/CD79a+/Tdt+/CD20-/other markers negative | B-ALL | Nil | IRH | LE | Chemotherapy |
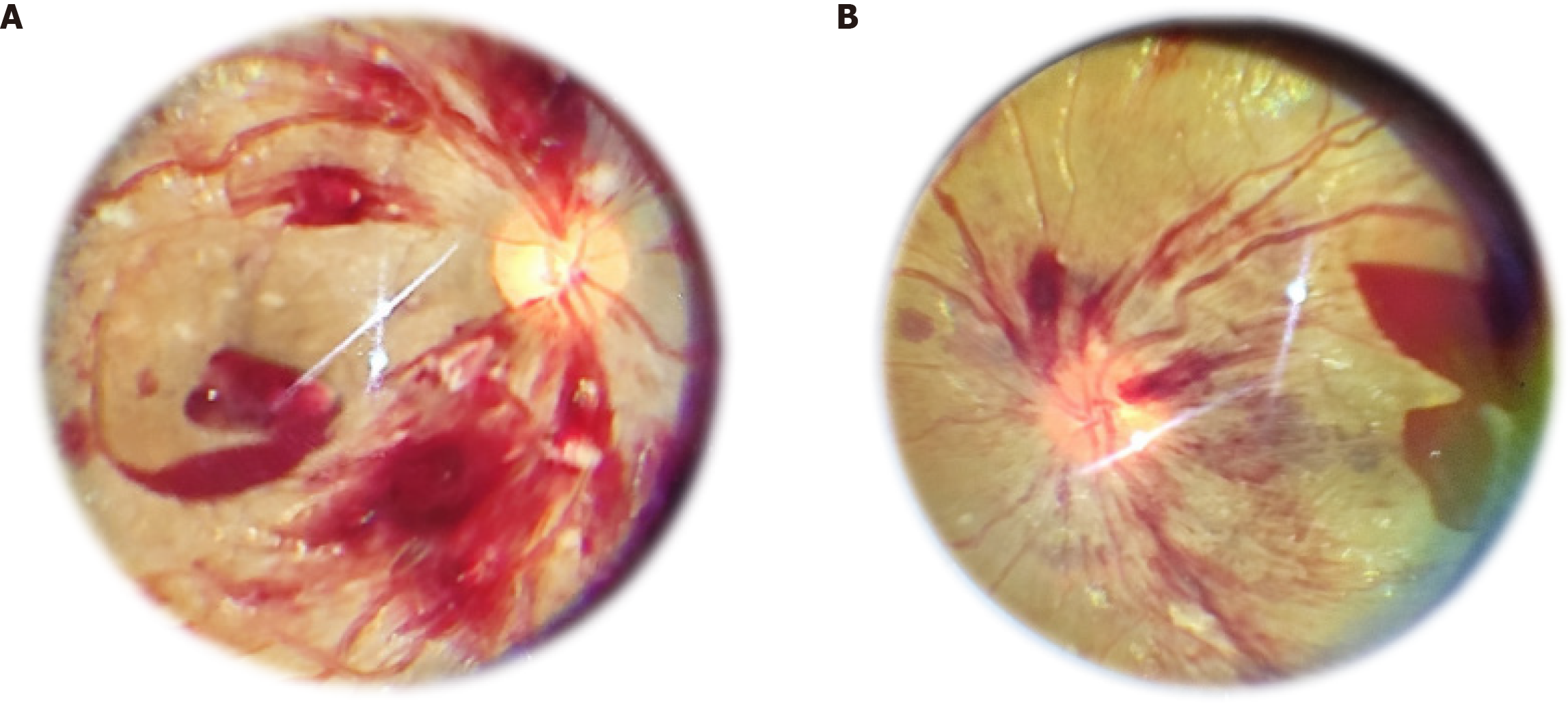
Ocular involvement was more common in lymphoblastic leukemia in our study in 13 cases (39.4%; 11 B-ALL, 2 T-ALL) than in myeloid leukemia in 6 cases (42.9%). There were 5 cases (10.6%) with ocular complaints and visual disturbances only in 2 cases (4.2%) as the initial manifestation in acute leukemia while 22 cases (46.8%) presented with systemic manifestations like fever and lymphadenopathy among all diagnosed leukemia patients. Four cases presented with proptosis out of which one case had bilateral orbital granulocytic sarcoma. The most common manifestation was leukemic retinopathy mainly intraretinal hemorrhages in 12 cases (25.5%; B-ALL-10, and T-ALL-2). There was a 6 year-old-male child with B-ALL, who had features of acute anterior uveitis in the right eye non-responsive to topical steroid therapy. Acute onset sixth cranial nerve palsy was observed in two patients (BLL-1, T ALL-1). The clinical and hematological profiles of patients with a spectrum of ocular lesions are shown in Figures 1, 2, 3, 4 and Table 1. All patients with ocular features received chemotherapy and three patients further received additional radiation therapy due to optic disc edema. Nine patients (19.1%) had ocular improvement six months post-chemotherapy. The hematological profile reported a low platelet count among ALL patients in comparison with AML cases (P = 0.006).
Leukemias are the malignant neoplasms of abnormal hematopoietic stem cells, the proliferation of which results in disruption of normal marrow function, thus causing marrow failure[7]. Acute leukemia is the most common presentation of childhood leukemia, with ALL accounting for 70%, as noted from various studies[8-10]. The ocular involvement was higher in the AML cases compared to ALL (42.9% vs 39.4%), respectively, in our study. This is comparable to the findings reported by Bitirgen et al[10] (AML: 36.4% vs ALL: 20.4%). The age of the patients ranged from 2.5 to 15 years in our study, and the male-to-female (M:F) ratio was 2.5:1, which is comparable to the study by Khadka et al[11], who reported a mean age of 8.5 ± 4.0 years and a M:F ratio of 2.3:1. Further studies by Bitirgen et al[10] (60%), Koshy et al[9] (63.5%) and Dhasmana et al[5] (64.7%) also reported a male majority.
The prevalence of ophthalmic findings in acute leukemia in childhood is variable. Ridgway et al[8] in an early study had reported ocular involvement in only 9% of children. Guyer et al[3], who studied only retinal changes at diagnosis, reported findings in 15.9% of children. Later, Ohkoshi and Tsiaras[2] reported ocular findings in 21.37% of children in their study group. Asian studies by Reddy et al[6], Koshy et al[9], and Bitirgen et al[10] have reported a prevalence of 16.5%, 29.4%, and 24.3%, respectively. In our study, the ocular involvement at the time of diagnosis was 40.4%. This variation among different studies may be due to the diversities in study population, age and duration of presentation.
The majority of the cases are asymptomatic at presentation[9,11]. Ocular findings in acute leukemia are presented as symptomatic in only 3%-15% of cases[5,6,8,9]. In our study, around 59.6% of the cases were asymptomatic, which was higher than Reddy et al[6] 28.0%, but lower than Bitirgen et al[10] 62.2%. Most retinal hemorrhages were asymptomatic except when they involved the macula, as observed in our study.
Clinical manifestations in childhood acute leukemia have often been assigned three groups: (1) Involvement of the orbit and ocular structures by direct leukemic infiltration; (2) Retinal involvement secondary to vascular abnormalities; and (3) Central nervous system (CNS) spread causing neuro-ophthalmic manifestations like papilledema or isolated cranial nerve palsies[12]. The most common tissue affected in both lymphoblastic and myeloid leukemia is the retina[5,6]. Fundus can present with intra-retinal hemorrhage, subhyaloid hemorrhage, retinal vascular changes, white-centered hemorrhage, and vitreous hemorrhage, with IRH being the most common of all as reported by several studies[5,6,7,13]. Subhyaloid hemorrhage at the posterior pole and vitreous hemorrhage are the most vision-threatening complications in patients with acute anemia[1]. Retinal manifestations in leukemia have been reported in ≤ 69% of all patients at some point in the course of the disease and are often attributed to the rheological changes caused by changing hematologic parameters[8]. Guyer et al[3] reported that thrombocytopenia leads to IRH formation and hematocrit changes, resulted in white-centered hemorrhage. Abu el-Asrar et al[14] also reported that in ALL cases, the retinal hemorrhages and cotton-wool spots were attributed to low hemoglobin (Hb) levels, while white-centered hemorrhages noted in AML patients were related to high WBC count. Further, Soman et al[1] established that the probability of developing sub-hyaloid hemorrhage was reduced by > 50% with an increase in Hb from 5 g/dL to 7 g/dL and platelet count to more than 50000 cells/mm3. Similarly, the incidence of leukemic infiltration of the retina increased with raised WBC count of > 100000 cells/mm3 and with a higher percentage of blast cells[10]. However, the studies of Orhan et al[4] reported no correlation between retinal manifestations and hematological parameters. In our study, low Hb and platelet counts resulted in retinal hemorrhages requiring frequent transfusion therapy, mostly observed in ALL cases.
Extramedullary orbital presentation is due to granulocytic sarcoma, which is a rare form of AML. The cells originate in the bone marrow and spread via Haversian canals to form soft deposits in the orbit[12]. Onset is variable and orbit can be involved before features of malignancy are seen in bone marrow or peripheral blood smear[12]. Orbital cellulitis as a secondary ophthalmic alteration has been observed in 2% of cases in a cohort study involving ALL patients[15]. In our study, we had one case who presented with bilateral proptosis with fever and was histologically proven by tissue biopsy to be suffering from AML.
Neuro-ophthalmic manifestations can be a result of direct infiltration of the optic nerve or due to CNS leukemia leading to papilledema and cranial nerve palsies. They are more commonly reported in ALL[2,11,16]. Khadka et al[11] reported optic nerve infiltration in 7%, cranial nerve palsies in 5.6%, and papilledema in 9.9%. The presence of blast cells in CSF was positive in the majority of cases[16]. In our study, all the cases presenting with headaches had papilledema on fundus examination, but none of them had CNS leukemia. This was comparable to the study by Karesh et al[17], who further stated that the relation of optic disc edema to CNS leukemia should only be established on histopathological confirmation.
Ocular manifestations carry a poor prognosis in the survival analysis of acute leukemia as reported in various studies[18-24]. Russo et al[18] established a significant difference in the frequency of relapses in specific or non-specific orbital or ocular manifestations and those without any ocular manifestations[18-24]. Ohkoshi and Tsiaras[2] reported a poor 5-year survival rate in patients with ocular manifestations. Curto et al[25] also reported that the presence of ocular features carries a poor prognostic sign for survival. However, Bitirgen et al[10] could not establish statistical significance. This difference may be due to different study designs and also the time of observation of ocular manifestations in the course of the disease, rather than at the time of diagnosis. Retinal hemorrhages due to hematological disorder are seen early in the disease, while disc edema due to leukemic infiltration is seen in later stages and hence carries a poor prognosis. Age is a major prognostic factor in ALL; most ophthalmic manifestations are seen in older ALL patients[17,18,26,27]. High WBC count (> 50000/mm3 in ALL and > 100000/ mm3 in AML) is also considered to be a poor prognostic factor[20-23,26].
The limitations of the study were a retrospective design, a small sample size, and short-term follow-up. Secondly, there was a lack of correlation with bone marrow aspirates for minimal residual disease, hematological parameters, and ocular features, which can be a future study to look more deeply into the molecular features in the disease severity and among subtypes of leukemia. Thirdly, very sick patients with leukemia were excluded from the study, and hence, there may be under-reporting in our study. Our findings call for a prospective randomized study on ocular manifestations of acute leukemia with long-term follow-up for prognostic information in the future.
There is a wide spectrum of ocular lesions presenting in a child suffering from leukemia at initial diagnosis, and often, most of these cases are asymptomatic. There is a need for routine ocular examination at the time of diagnosis of leukemia and also in the course of therapy, as the need arises, to help in preventing any vision-threatening complications and aid in risk stratification. Thus, having at least one examination at diagnosis and during relapse is essential. Multicentric and large-scale studies are necessary to find the association between the type of leukemia, hematological profile, survival factor, type of ocular features, and final visual outcome.
Special appreciation to Late Dr. Sambit Kumar Mohanty, MD, Pathology, for his help and contribution to the clinic-pathological work and in statistical work. We also thank Dr. Prabodh Kumar Das, HOD, Medical Hematology and Oncology, for his cooperation in data collection.
| 1. | Soman S, Kasturi N, Srinivasan R, Vinod KV. Ocular Manifestations in Leukemias and Their Correlation with Hematologic Parameters at a Tertiary Care Setting in South India. Ophthalmol Retina. 2018;2:17-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 24] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 2. | Ohkoshi K, Tsiaras WG. Prognostic importance of ophthalmic manifestations in childhood leukaemia. Br J Ophthalmol. 1992;76:651-655. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 78] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 3. | Guyer DR, Schachat AP, Vitale S, Markowitz JA, Braine H, Burke PJ, Karp JE, Graham M. Leukemic retinopathy. Relationship between fundus lesions and hematologic parameters at diagnosis. Ophthalmology. 1989;96:860-864. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 60] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 4. | Orhan B, Malbora B, Akça Bayar S, Avcı Z, Alioğlu B, Özbek N. Ophthalmologic Findings in Children with Leukemia: A Single-Center Study. Turk J Ophthalmol. 2016;46:62-67. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 19] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 5. | Dhasmana R, Prakash A, Gupta N, Verma SK. Ocular manifestations in leukemia and myeloproliferative disorders and their association with hematological parameters. Ann Afr Med. 2016;15:97-103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 26] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 6. | Reddy SC, Menon BS. A prospective study of ocular manifestations in childhood acute leukaemia. Acta Ophthalmol Scand. 1998;76:700-703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 39] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 7. | Paul VK, Bagga A. Ghai Essential Pediatrics. 8th ed. Delhi: CBS Publishers, 2014. |
| 8. | Ridgway EW, Jaffe N, Walton DS. Leukemic ophthalmopathy in children. Cancer. 1976;38:1744-1749. [PubMed] [DOI] [Full Text] |
| 9. | Koshy J, John MJ, Thomas S, Kaur G, Batra N, Xavier WJ. Ophthalmic manifestations of acute and chronic leukemias presenting to a tertiary care center in India. Indian J Ophthalmol. 2015;63:659-664. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 42] [Article Influence: 4.2] [Reference Citation Analysis (1)] |
| 10. | Bitirgen G, Belviranli S, Caliskan U, Tokgoz H, Ozkagnici A, Zengin N. Ophthalmic manifestations in recently diagnosed childhood leukemia. Eur J Ophthalmol. 2016;26:88-91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 23] [Article Influence: 2.1] [Reference Citation Analysis (3)] |
| 11. | Khadka D, Sharma AK, Shrestha JK, Shrestha GS, Shrestha PN, Pant SR, Pant BP. Ocular manifestations of childhood acute leukemia in a tertiiary level eye centre of Kathmandu, Nepal. Nepal J Ophthalmol. 2014;6:197-204. [PubMed] [DOI] [Full Text] |
| 12. | Sharma T, Grewal J, Gupta S, Murray PI. Ophthalmic manifestations of acute leukaemias: the ophthalmologist's role. Eye (Lond). 2004;18:663-672. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 148] [Article Influence: 6.7] [Reference Citation Analysis (1)] |
| 13. | Robb RM, Ervin LD, Sallan SE. A pathological study of eye involvement in acute leukemia of childhood. Trans Am Ophthalmol Soc. 1978;76:90-101. [PubMed] |
| 14. | abu el-Asrar AM, al-Momen AK, Kangave D, Harakati MS, Ajarim DS. Correlation of fundus lesions and hematologic findings in leukemic retinopathy. Eur J Ophthalmol. 1996;6:167-172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 20] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 15. | Murthy R, Vemuganti GK, Honavar SG, Naik M, Reddy V. Extramedullary leukemia in children presenting with proptosis. J Hematol Oncol. 2009;2:4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 28] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 16. | de Queiroz Mendonca C, Freire MV, Viana SS, Silva Tavares MKG, Almeida Silva WM, Cipolotti R. Ocular manifestations in acute lymphoblastic leukemia: A five-year cohort study of pediatric patients. Leuk Res. 2019;76:24-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 30] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 17. | Karesh JW, Goldman EJ, Reck K, Kelman SE, Lee EJ, Schiffer CA. A prospective ophthalmic evaluation of patients with acute myeloid leukemia: correlation of ocular and hematologic findings. J Clin Oncol. 1989;7:1528-1532. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 65] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 18. | Russo V, Scott IU, Querques G, Stella A, Barone A, Delle Noci N. Orbital and ocular manifestations of acute childhood leukemia: clinical and statistical analysis of 180 patients. Eur J Ophthalmol. 2008;18:619-623. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 66] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 19. | Lee JW, Cho B. Prognostic factors and treatment of pediatric acute lymphoblastic leukemia. Korean J Pediatr. 2017;60:129-137. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 42] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 20. | Meshinchi S, Arceci RJ. Prognostic factors and risk-based therapy in pediatric acute myeloid leukemia. Oncologist. 2007;12:341-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 62] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 21. | LaRiviere MJ, Avery RA, Dolan JG, Adamson PC, Zarnow DM, Xie Y, Avery SM, Kurtz GA, Hill-Kayser CE, Lustig RA, Lukens JN. Emergent Radiation for Leukemic Optic Nerve Infiltration in a Child Receiving Intrathecal Methotrexate. Pract Radiat Oncol. 2019;9:226-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (1)] |
| 22. | Ravisankar S, Levy Y, Shah M. Myeloid Sarcoma of Orbits: Effectiveness of a Low-Dose Radiation Regimen. Case Rep Hematol. 2018;2018:9071693. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 23. | Benvenuto F, Sgroi M, Guillen SS, Ancona D, Fandiño A. Ocular findings in children with acute leukemia at a tertiary care center in South America. Oman J Ophthalmol. 2022;15:159-162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 24. | Reinhardt D, Pekrun A, Lakomek M, Zimmermann M, Ritter J, Creutzig U. Primary myelosarcomas are associated with a high rate of relapse: report on 34 children from the acute myeloid leukaemia-Berlin-Frankfurt-Münster studies. Br J Haematol. 2000;110:863-866. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 25. | Curto ML, Zingone A, Acquaviva A, Bagnulo S, Calculli L, Cristiani L, Dini G, Di Tullio MT, Guazzelli C, Jancovic M. Leukemic infiltration of the eye: results of therapy in a retrospective multicentric study. Med Pediatr Oncol. 1989;17:134-139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 32] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 26. | Mirshahi R, Ghassemi F, Koochakzadeh L, Faranoush M, Ghomi Z, Mehrvar A, Mousavi SA, Safaee Nodehi SR. Ocular Manifestations of Newly Diagnosed Acute Leukemia Patients. J Curr Ophthalmol. 2022;34:100-105. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 27. | Hafeez MU, Ali MH, Najib N, Ayub MH, Shafi K, Munir M, Butt NH. Ophthalmic Manifestations of Acute Leukemia. Cureus. 2019;11:e3837. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (1)] |