Copyright: ©Author(s) 2026.
World J Exp Med. Mar 20, 2026; 16(1): 117125
Published online Mar 20, 2026. doi: 10.5493/wjem.v16.i1.117125
Published online Mar 20, 2026. doi: 10.5493/wjem.v16.i1.117125
Figure 1 Preparation of indocyanine green-doped chitosan nanoparticles.
A: Chitosan powder from shrimp shells 75% (deacetylated) (LOBA CHEMIE PVT. LTD.); B: Indocyanine Green Powder (Tokyo Chemical Industry Co., Ltd); C: Chitosan nanoparticles and indocyanine green dye is being mixed in magnetic stirrer; D: Digital pH meter showing pH of 4.8 of the mixed dye; E: Mixed photosensitizer is proceeded for lyophilization; F: 100 mg of indocyanine green-doped chitosan nanoparticles and 1000 mL of distilled water; G: Stirred with stirrer; H: Photosensitizer solution stored in amber coloured bottle to protect it from natural light; I: Photosensitizer solution loaded in 3 mL disposable syringe.
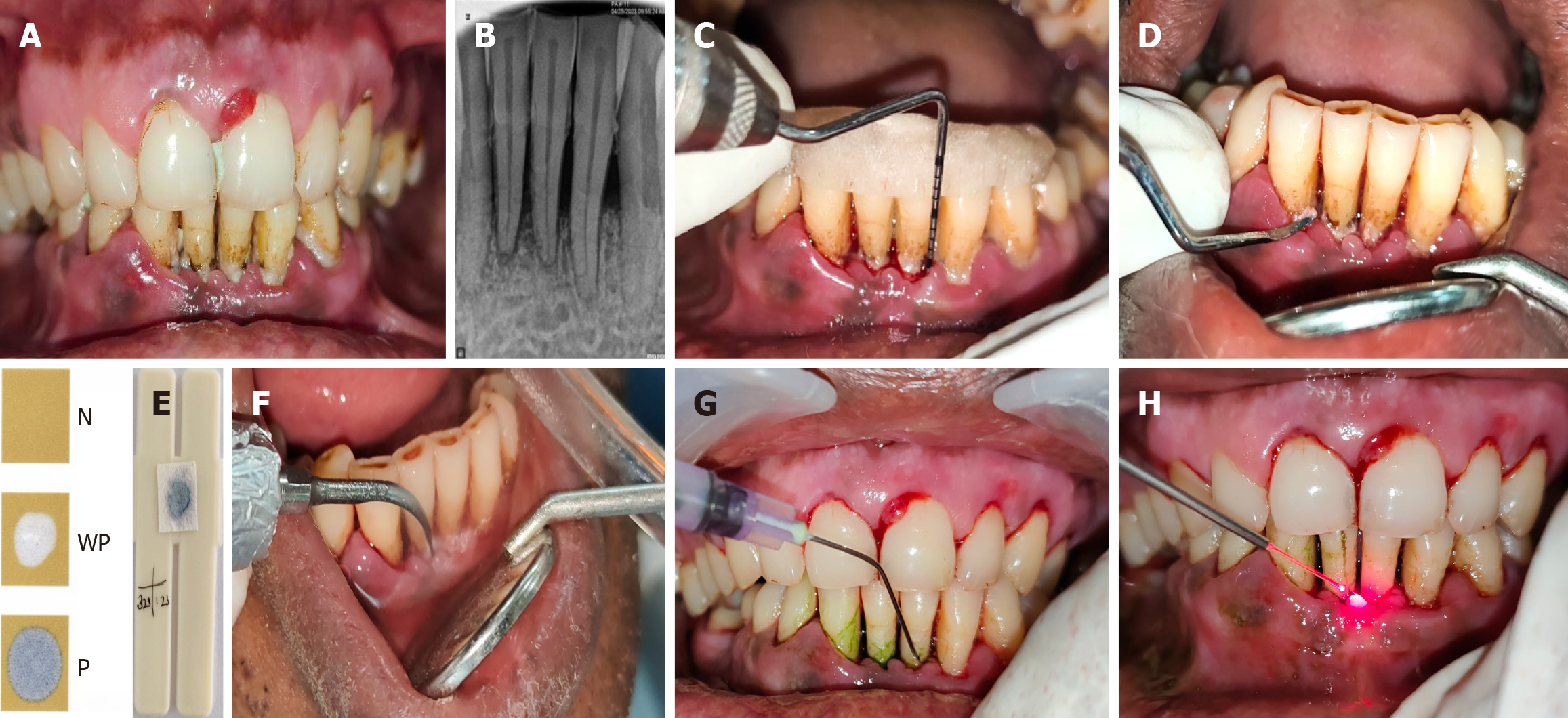
Figure 2 Test site.
A: Pre-operative clinical picture of test site; B: Pre-operative radiograph of test site; C: Customized acrylic stent in test site with baseline parameters recorded pocket probing depth - from gingival margin, relative clinical attachment level - from apical end of the groove made in stent; D: Collection of plaque sample from test site for microbiological evaluation; E: Test result being compared with interpretation chart (BANA-zyme test kit) showing blue colour- positive result[27]; F: Scaling and root planing being performed at the test site; G: Application of photosensitizer at test site; H: Application of laser irradiation at test site.
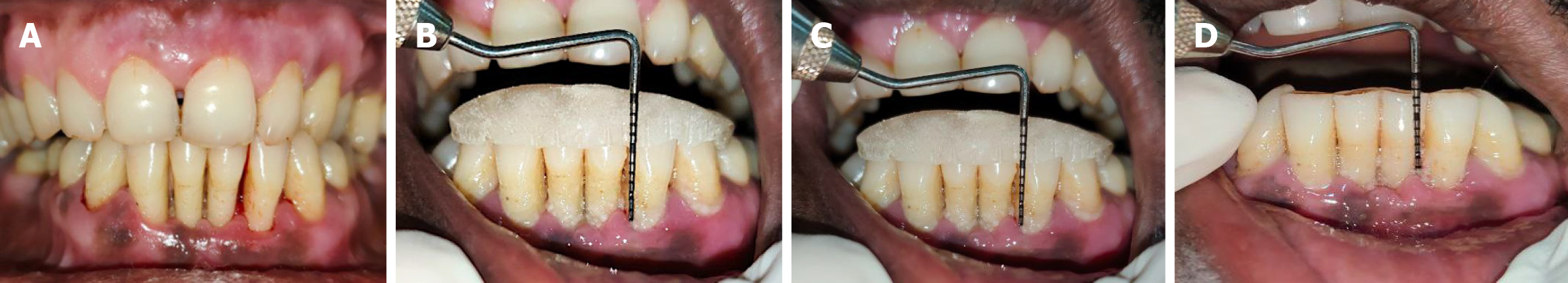
Figure 3 Test site follow-up.
A: Follow-up of test site (7 days post-operative); B: Follow-up of test site (1 month post-operative) with evaluation of clinical periodontal parameters; C: Follow-up of test site (3 months post-operative) with evaluation of relative clinical attachment level; D: Follow-up of test site (3 months post-operative) with evaluation of pocket probing depth.
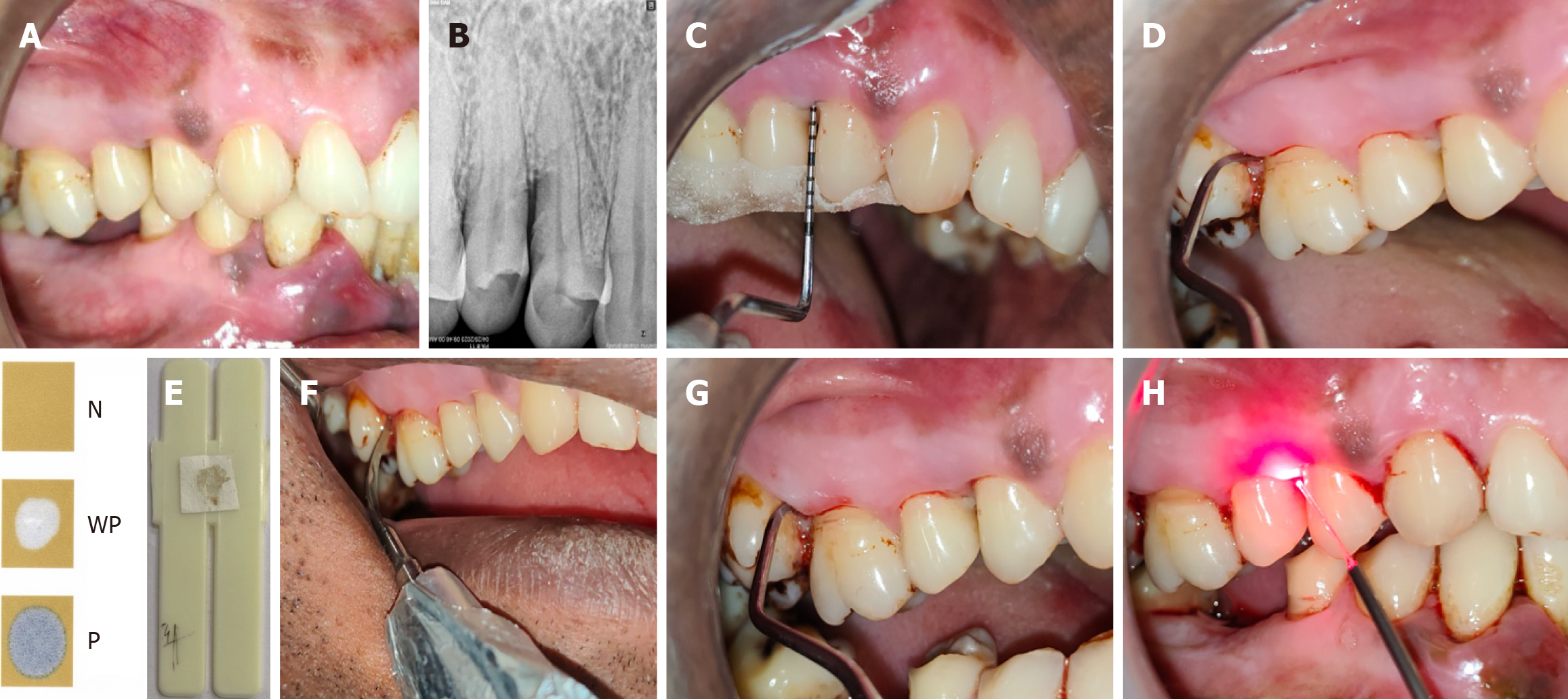
Figure 4 Control site.
A: Pre-operative clinical picture for control site; B: Pre-operative radiograph of control site; C: Customized acrylic stent in control site with baseline parameters recorded pocket probing depth - from gingival margin, relative clinical attachment level - from apical end of the groove made in stent; D: Collection of plaque sample from control site for microbiological evaluation; E: Test result being compared with interpretation chart (BANA-zyme test kit) showing faint blue colour- weakly positive result[27]; F: Ultrasonic scaling being performed at the control site; G: Root planing being performed at the control site; H: Application of laser irradiation at control site.
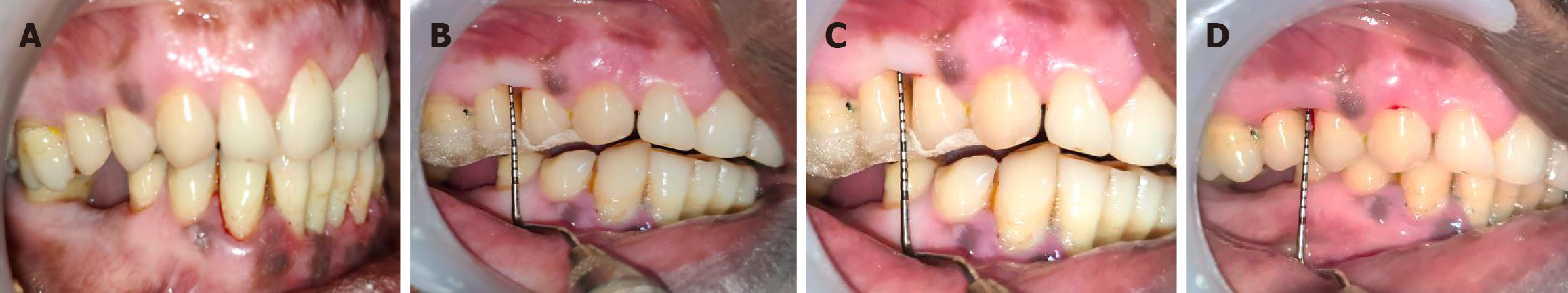
Figure 5 Control site follow-up.
A: Follow-up of control site (7 days post-operative); B: Follow-up of control site (1 month post-operative) with evaluation of clinical periodontal parameters; C: Follow-up of control site (3 months post-operative) with evaluation of relative clinical attachment level; D: Follow-up of control site (3 months post-operative) with evaluation of pocket probing depth.
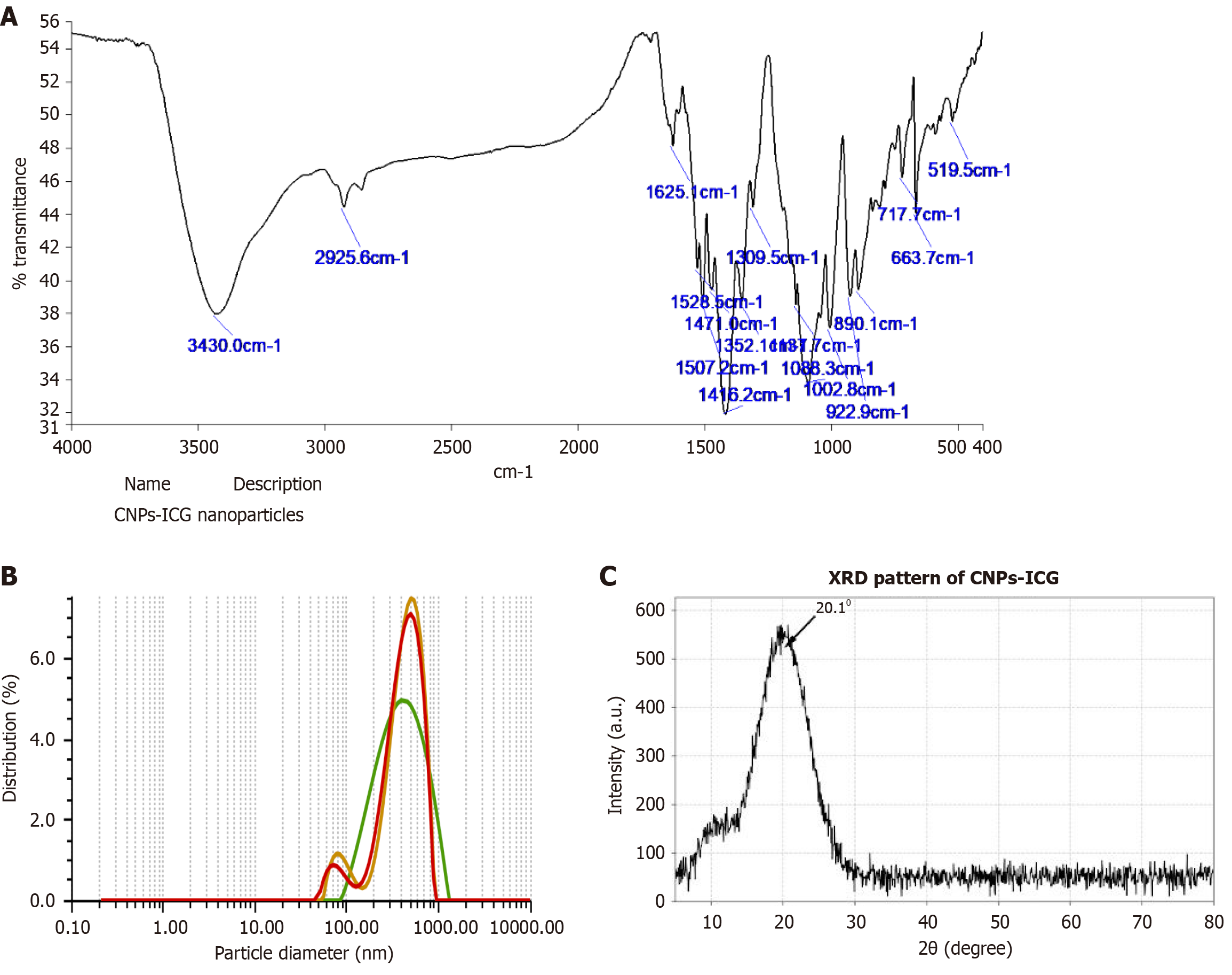
Figure 6 Confirmation of synthesized indocyanine green-doped chitosan nanoparticles.
A: Fourier transform infrared spectrum of synthesized indocyanine green-doped chitosan nanoparticles (CNPs-ICG); B: Dynamic light scattering of synthesized CNPs-ICG; C: X-ray diffraction pattern of CNPs-ICG. CNPs-ICG: Indocyanine green-doped chitosan nanoparticles.
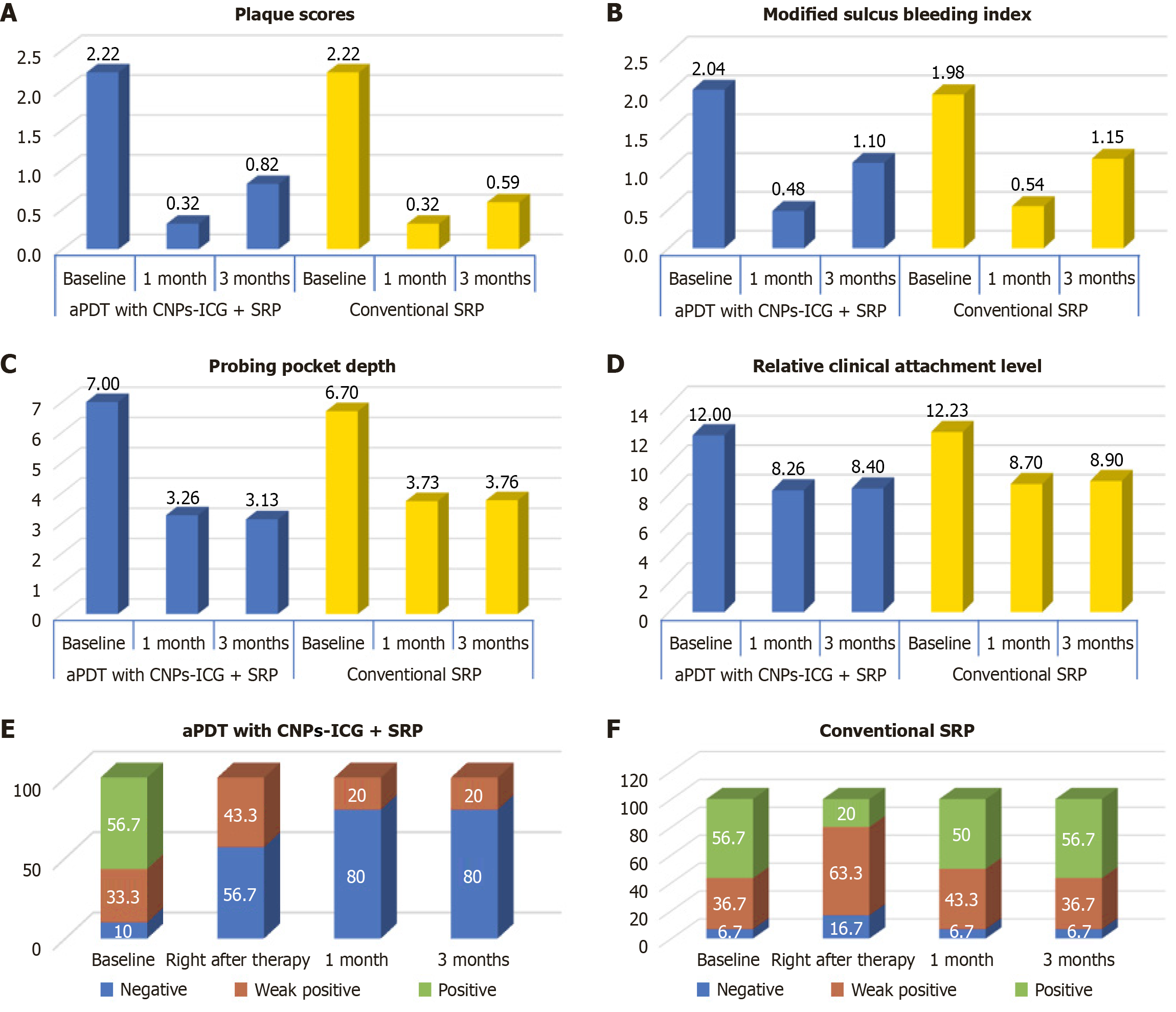
Figure 7 Comparison of variables within test and control sites (intra-group comparison).
A: Mean plaque scores at different time intervals in both the groups; B: Mean sulcular bleeding scores at different time intervals in both the groups; C: Mean probing pocket depth at different time intervals; D: Comparison of Mean relative clinical attachment level at different time intervals in both the groups; E: Distribution of BANA scores at different time intervals in antimicrobial photodynamic therapy with CNPs-ICG + scaling and root planing (SRP) group; F: Distribution of BANA scores at different time intervals in conventional SRP group. CNPs-ICG: Indocyanine green-doped chitosan nanoparticles; aPDT: Antimicrobial photodynamic therapy; SRP: Scaling and root planning.
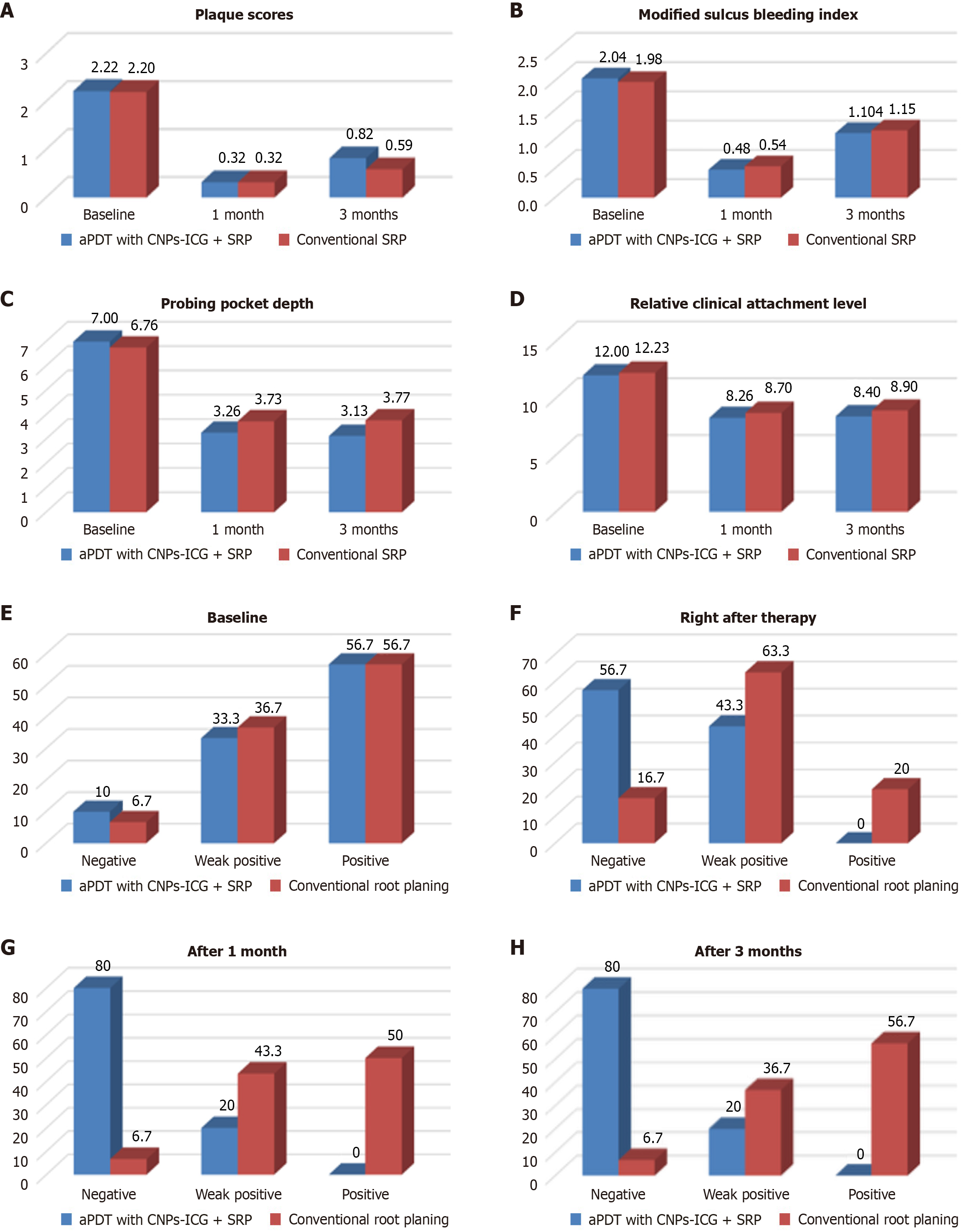
Figure 8 Comparison of variables between test and control sites (inter-group comparison).
A: Mean plaque scores between the groups at different time intervals; B: Mean modified sulcus bleeding scores between the groups at different time intervals; C: Mean probing pocket depth between the groups at different time intervals; D: Mean relative clinical attachment level between the groups at different time intervals; E: Comparison of BANA test scores at baseline between the groups; F: Comparison of BANA test scores at right after therapy between the groups; G: Comparison of BANA test scores after 1 month between the groups; H: Comparison of BANA test scores after 3 months between the groups. CNPs-ICG: Indocyanine green-doped chitosan nanoparticles; aPDT: Antimicrobial photodynamic therapy; SRP: Scaling and root planning.
- Citation: Raj SC, Sen S, Baral D, Mohanty D, Tabassum S, Sil S, Sahu M. Indocyanine green doped with chitosan nanoparticles based antimicrobial photodynamic therapy in periodontitis patients: A randomized clinical trial. World J Exp Med 2026; 16(1): 117125
- URL: https://www.wjgnet.com/2220-315x/full/v16/i1/117125.htm
- DOI: https://dx.doi.org/10.5493/wjem.v16.i1.117125