Published online Mar 9, 2026. doi: 10.5492/wjccm.v15.i1.110763
Revised: August 18, 2025
Accepted: November 25, 2025
Published online: March 9, 2026
Processing time: 258 Days and 15.3 Hours
Tuberculous meningitis (TBM) is a severe extrapulmonary complication of tuber
To describe existing TBM diagnostic scores with the primary focus being on scores with external validation.
PubMed and EMBASE were searched from inception to 21 March 2024. Cross-sectional studies with numerical TBM scoring systems were included. Literature pertaining to non-human, non-live subjects, single diagnostic tests, papers with no gold standard diagnosis, and non-English papers were excluded.
We reviewed 21 unique scoring systems, of which 4 have been externally vali
We suggest Thwaites (2002) and Marais (2010) as first-line TBM screening tests due to high sensitivities and extensive external validation, however clinicians should consider scores specific to their patient population.
Core Tip: Tuberculous meningitis (TBM) is a severe complication of tuberculosis. Early diagnosis is crucial in improving patient outcomes, albeit difficult given the non-specific signs and symptoms of early TBM. Numerical diagnostic scoring systems may be a useful early diagnostic tool for clinicians, particularly in resource-constrained settings. Of all the scores currently published, we suggest using Thwaites (2002) and Marais (2010) as first line TBM screening tests due to their high sensitivities and extensive external validation. Nevertheless, clinicians are encouraged to select scores in accordance with the patient characteristics of the population of interest.
- Citation: Teo E, Kung K, Chen S, See KC. Clinical scoring systems for diagnosing tuberculous meningitis: A systematic review. World J Crit Care Med 2026; 15(1): 110763
- URL: https://www.wjgnet.com/2220-3141/full/v15/i1/110763.htm
- DOI: https://dx.doi.org/10.5492/wjccm.v15.i1.110763
Tuberculosis (TB) is the second leading cause of death from a single infectious agent, as reported in 2023[1]. Tuberculous meningitis (TBM) is the most severe form of central nervous system TB, with high morbidity and risk of neurodevelopmental sequelae. Even amongst patients receiving appropriate treatment, the mortality of the disease remains high. A 2021 modelling study investigating TBM globally found that TBM case fatality among patients undergoing treatment in 2019 was on average 27%[2]. An estimated 48% of incident TBM cases in 2019 resulted in death[2]. Timely diagnosis and treatment is key to improving patient outcomes[3]. Unfortunately, the diagnosis of TBM remains difficult. Early prodromal stage TBM presents with non-specific symptoms that frequently overlap with non-TBM (including bacterial, viral, fungal and aseptic causes)[4]. Furthermore, the time course of disease is variable, ranging from several days to six months[5].
World Health Organization-recommended gold standard diagnostic tests using cerebrospinal fluid (CSF) include: (1) Acid-fast bacilli (AFB) smear; (2) M.TB cultures; and (3) Nucleic acid amplification tests (NAATs), such as Xpert MTB/RIF Ultra. However, these tests can also contribute to diagnostic challenges. Sensitivities of AFB smears and cultures are suboptimal. Conventional cultures can take up to months in certain areas. NAATs, while more efficient, have higher processing costs and limited availability in resource-constrained areas[6].
Given these challenges, various numerical diagnostic scores have been produced with the aim of aiding diagnosis through a combination of clinical, laboratory, microbiological, histopathological and radiological criteria. Some notable scoring systems include that of the Lancet/Marais score (area under the receiver operating characteristic curve 0.90 to 0.979) and Thwaites (area under the receiver operating characteristic curve 0.666 to 0.99) In this study, we aim to describe key features of available TBM diagnostic scores, focusing on externally validated scores. Features described include accuracy (sensitivity, specificity), strengths and limitations.
This study seeks to serve as an overview of the scoring systems that are currently available for TBM diagnosis. In comparing the various systems, we seek to answer the following questions: (1) How do the scores vary in terms of sensitivity, specificity and applicability to various populations? (2) What are the strengths and limitations of existing scores affecting their utility in the real world? and (3) What is the role of diagnostic scoring systems in today’s setting and the future?
The literature search was conducted in accordance with the PRISMA guidelines. We searched PubMed and EMBASE from conception to 21 March 2024, using combinations of: “tuberculosis meningitis”, “central nervous system tu
Articles were screened independently by three researchers (Teo E, Kung K, and Chen S) first by title and abstract, then by full-text using eligibility criteria described below. The references of included papers were screened for relevant articles. Disagreement was resolved through discussion, reanalysis of data and consensus. A systematic review of literature was conducted for all existing cross sectional studies containing data on TBM scoring systems.
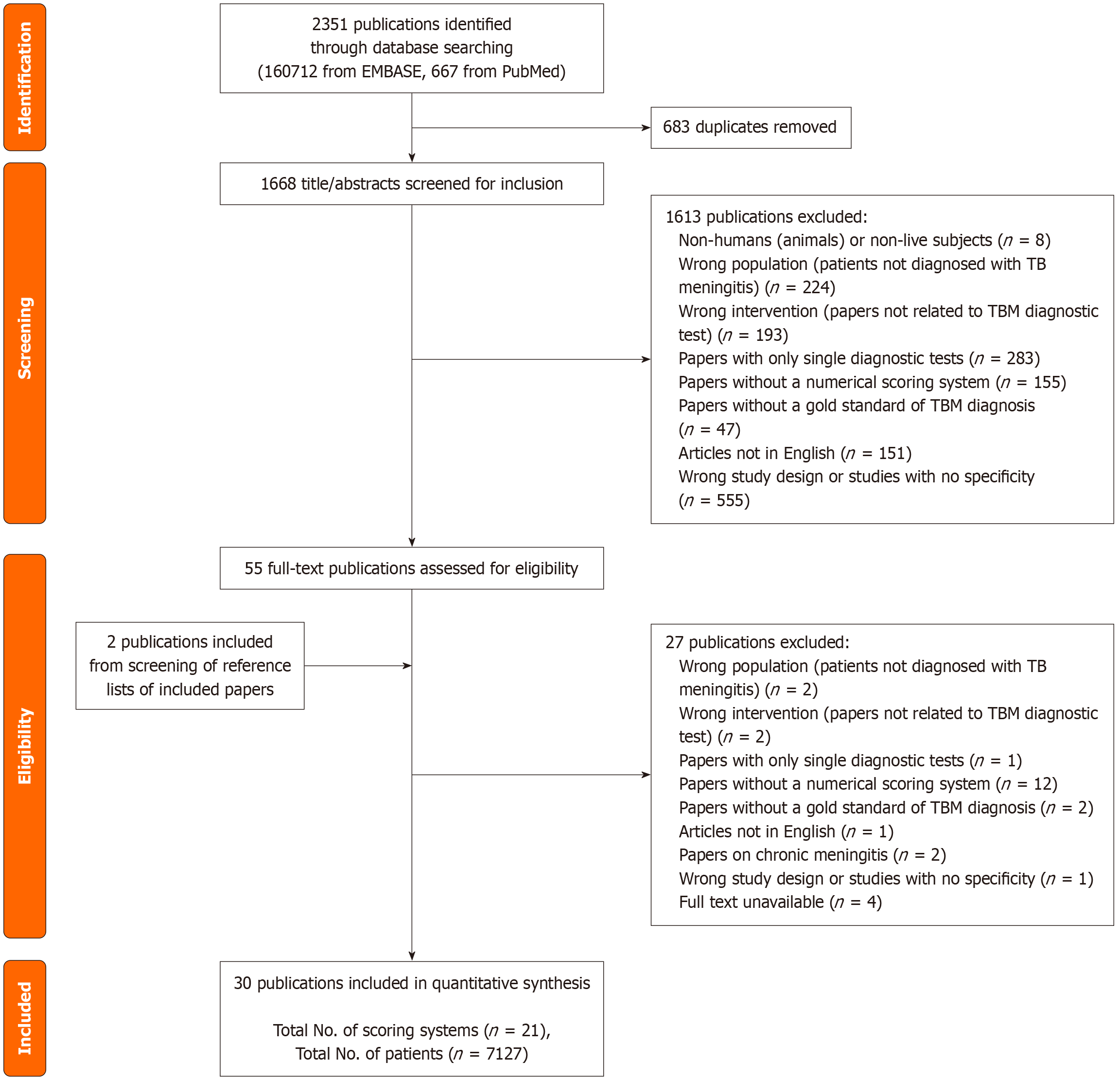
Exclusion criteria was: (1) Non-human or non-live subjects; (2) Patients not diagnosed with TBM; (3) Papers not related to TBM diagnostic tests; (4) Papers with only single diagnostic tests; (5) Papers without a numerical scoring system; (6) Papers without a gold standard of TBM diagnosis (positive CSF culture/polymerase chain reaction/NAATs); (7) Articles not in English; (8) Papers with chronic meningitis as the main patient population; (9) Systematic reviews, review articles, editorials, letters, single case studies, studies with no specificity; and (10) Papers where the full text was unavailable. 4 studies were excluded as the full text was unavailable. Figure 1 represents a summary of the study selection process.
The Joanna Briggs Institute Critical Appraisal tool (Supplementary Table 1) was used to assess studies’ methodological quality and determine the extent of possible bias in terms of design, conduct and analysis. The tools were applied by three researchers independently, with disagreements resolved through discussion and consensus.
Numerical scoring systems refer to a set of criteria with a numerical value per component totaling to a final score. Modifications to scoring systems include alterations to cut-off values of individual components which originally involve a range of values. Papers which added extra components with their own numerical values to existing scores were assigned their own score. External validation is defined as the process of validating results using a separate group of patients from a different state or country from the original derivation cohort.
30 publications describing 21 scoring systems and 7127 patients were included for final analysis.
We reviewed 4 externally validated scoring systems: Thwaites (2002), Hristea (2012), Marais (2010); and Kumar (1999), with the year of development in parentheses. Throughout literature, Thwaites (2002) is also referenced as the Vietnam rule and Marais (2010) as the Lancet scoring classification; the former names will be used in this review for consistency. Details of each score and their interpretation are in Table 1. Notably, only Marais (2010) included radiological and microbiological criteria. Other scores only required simple laboratory investigations (CSF and serum analysis). Cohort populations also varied in age - Kumar (1999) used a paediatric population, Hristea (2012) a mixed paediatric and adult population and Thwaites (2002) an adult population.
| Ref. | Country | Internal validation | Clinical criteria | Laboratory criteria | Microbiological/histopathological criteria | Radiological criteria | Interpretation |
| Thwaites et al[31], 2002 | Vietnam | AUC of ROC: 0.990 | Age: (1) ≥ 36 years: 2 points; and (2) < 36 years: 0 points | CSF analysis (103/mL) | None | None | TDI: DI (age) + DI (blood white-cell count) + DI (history of illness) + DI (cerebrospinal fluid white-cell count) + DI (cerebrospinal fluid% neutrophils) |
| Sensitivity 97.0%; specificity: 91.0% | CSF total white cell count | ||||||
| ≥ 900: 3 points; < 900: 0 points | TBM: TDI ≤ 4 | ||||||
| CSF% neutrophils: ≥ 75: 4 points; < 75: 0 points | |||||||
| After internal validation with test data population (n = 75) | History of illness: (1) ≥ 6 days: -5 points; and (2) < 6 days: 0 points | Blood white cell count (103/mL): (1) ≥ 15000: 4 points; and (2) < 15000: 0 points | BM: TDI > 4 | ||||
| Sensitivity: 86.0%; specificity: 79.0% | |||||||
| Marais et al[32], 2010 | NIL | NIL | Entry criteria: Symptoms and signs of meningitis including at least one: Headache, irritability, vomiting, fever, neck stiffness, convulsions, focal neurological deficits, altered consciousness, or lethargy | CSF analysis 1 point each, maximum score: 4 points | Extraneural TB: Maximum score: 4 points. Imaging suggestive of extraneural TB (see next column). AFB identified or M.TB cultured from another source (e.g., sputum, lymph node, gastric washing, urine, blood culture): 4 points. Positive commercial M.TB NAAT from extra-neural specimen: 4 points | Cerebral imaging: Maximum score: 6 points: (1) Hydrocephalus: 1 point; (2) Basal meningeal enhancement: 2 points; (3) Tuberculoma: 2 points; (4) Infarct: 1 point; (5) Pre-contrast basal hyperdensity: 2 points; and (6) Imaging suggestive of extraneural TB: Chest radiograph suggestive of active TB: Signs of TB: 2 points. Miliary TB: 4 points. CT/MRI/ultrasound evidence for TB outside CNS: 2 points | Definite TBM |
| Clinical entry criteria | |||||||
| At least 1: (1) AFB seen in CSF; (2) M.TB cultured from CSF; and (3) CSF positive commercial NAAT | |||||||
| Clinical criteria: Maximum score: 6 points | Clear appearance | “AND/OR” AFB seen in context of histological changes consistent with TB in the brain or spinal cord with suggestive symptoms or signs and CSF changes, or visible meningitis (on autopsy) | |||||
| Symptom duration > 5 days: 4 points | Cells: 10-500 per μL | Probable TBM | |||||
| Clinical entry criteria: (1) Total diagnostic score (with ≥ 2 point from laboratory or radiological criteria); (2) Score ≥ 10 (when cerebral imaging unavailable); and (3) Score ≥ 12 (when cerebral imaging available) | |||||||
| Systemic symptoms suggestive of TB (at least 1): 3 points: (1) Weight loss or poor weight gain in children; (2) Night sweats; and (3) Persistent cough > 2 weeks | Lymphocytic predominance (> 50%) | Exclusion of alternative diagnoses | |||||
| History of recent (within past year) close contact with an individual with pulmonary TB or positive TST or IGRA (only in children < 10 years old): 2 points | Protein concentration > 1 g/L | Probable TBM | |||||
| Clinical entry criteria: (1) Total diagnostic score (with ≥ 2 points from laboratory or radiological criteria); (2) Score ≥ 10 (when cerebral imaging unavailable); and (3) Score ≥ 12 (when cerebral imaging available) | |||||||
| Focal neurological deficit (excluding cranial nerve palsies): 1 point | CSF: Plasma glucose ratio < 50% or an absolute CSF glucose concentration < 2.2 mmol/L | Exclusion of alternative diagnoses | |||||
| Cranial nerve palsy: 1 point | Possible TBM: (1) Clinical entry criteria; (2) Total diagnostic score: Score: 6-9 (when cerebral imaging is not available); score: 6-11 (when cerebral imaging is available); and (3) Exclusion of alternative diagnoses. Possible TB cannot be diagnosed or excluded without doing a lumbar puncture or cerebral imaging | ||||||
| Altered consciousness: 1 point | Non-TBM: Alternative diagnosis established, without a definitive diagnosis of TBM or other convincing signs of dual disease | ||||||
| Hristea et al[33], 2012 | Romania | AUC of ROC: 0.977 | DSBA ≥ 5 days: Fever; headache; vomiting; neck stiffness | CSF analysis | None | None | For patients with clear CSF meningitis, use this equation to calculate the probability of TBM: Ln (ODDS) = 3.615 × DSBA + 2.680 × (neurological stage) + 3.414 × (glucose ratio) + 1.669 × (CSF protein) - 7.612 |
| Sensitivity: 92.0%; specificity: 94.0% | Neurological stage II and III (defined based on Medical Research Council Definitions) | CSF/blood glucose ratio < 0.5 | |||||
| Stage II: Lethargy, confusion, mild focal signs (cranial nerve palsy, hemiparesis) | CSF protein level > 100 mg/dL | Scoring system: (1) DBSA ≥ 5 days: 3 points; (2) Neurological stages II/III (instead of I): 2 points; (3) Glucose ratio < 0.5: 3 points; and (4) CSF protein > 100 mg/dL: 1 point | |||||
| PPV: TBM: 89.1%; VM: 95.6% | Stage III: Delirium, stupor, coma, seizures, multiple cranial nerve palsies, dense hemiplegia or paraparesis | ||||||
| Kumar et al[34], 19991 | India | One or more predictor variables present: Total n = 100; TBM n = 65; non-TBM n = 35; sensitivity: 98.4%; specificity: 43.5% | Prodromal stage ≥ 7 days | CSF analysis: CSF leukocytes < 50% polymorphs | None | None | TBM ruled in with high degree of certainty: ≥ 3 of 5 criteria present |
| Two or more predictor variables present: Total n = 59; TBM n = 51; non-TBM n = 8; sensitivity: 77.2%; specificity: 87.0% | Fundal optic atrophy | Highly unlikely TBM: None of the 5 criteria present | |||||
| Three or more predictor variables present: Total n = 37; TBM n = 36; non-TBM n = 1; sensitivity: 54.5%; specificity: 98.3% | Focal neurological deficits | Use scoring system and ROC plot if ≥ 2 out of 5 criteria present (best cut off: 34): (1) Prodromal stage ≥ 7 days: 23; (2) Fundal optic atrophy: 2; (3) Focal deficits: 18; (4) EP movements: 29; and (5) CSF leucocytes < 50% polymorphs: 34 | |||||
| Extrapyramidal movements |
Thwaites (2002), Hristea (2012), and Kumar (1999) were internally validated with their respective test data populations. Internal validation refers to using patients from the developmental population to investigate the reproducibility of the score[7]. Of the 3 internally validated scoring systems, Kumar (1999) had the highest sensitivity of 98.4%.
An overview of the external validations of the 4 scores were detailed in Table 2. External validation looks into the generalisability of scores to various populations[7]. Each score provided a different interpretation of final numerical values. For instance, Marais distinguished between definite, probable and possible TBM. Sensitivity and specificity in Table 2 were calculated based on the score that reflected the lowest threshold for treatment, i.e., the score at which clinicians would be likely to initiate anti-TBM treatment.
| Ref. | Sensitivity (%) | Specificity (%) | Country of external validation population | Clinical criteria | Laboratory criteria | Microbiological/histopathological criteria | Radiological criteria |
| Thwaites et al[31], 2002 | Cut off of ≤ 3.5: TBM vs non-TBM: 78.0 | Cut off of ≤ 3.5: TBM vs non-TBM: 63.0 | Malawi | √ | √ | X | X |
| TBM vs PM: 95.0 | TBM vs PM: 23.5 | Pakistan | |||||
| Cut off of ≤ 4: TBM vs non-TBM: 78.0-100 | Cut off of ≤ 4: TBM vs non-TBM: 40.8-43.0 | Malawi, Turkey, Qatar | |||||
| TBM vs BME: 95.8 | TBM vs BME: 0.680 | Turkey | |||||
| TBM vs PM: 96.0 | - | India | |||||
| TBM vs BM: 95.8-98.2 | TBM vs BM: 43.6-87.9 | China, Turkey, India | |||||
| Marais et al[32], 2010 | Cut off of ≥ 6: TBM vs non-TBM: 66.7-100 | Cut off of ≥ 6: TBM vs non-TBM: 48.7-85.5 | Brazil, Turkey, China, Qatar | √ | √ | √ | √ |
| TBM vs BM: 93.0-97.0 | TBM vs BM: 56.0-68.0 | China | |||||
| TBM vs BME: 96.8 | TBM vs BME: 3.06 | Turkey | |||||
| Cut off of ≥ 8: TBM vs non-TBM: 84.9 | Cut off of ≥ 8: TBM vs non-TBM: 77.2 | Brazil | |||||
| Cut off of ≥ 10: TBM vs non-TBM: 100 | Cut off of ≥ 10: TBM vs non-TBM: 40.8 | Brazil | |||||
| Hristea et al[33], 20121 | Cut off of ≥ 6: TBM vs non-TBM: 100 | Cut off of ≥ 6 | Turkey | √ | √ | X | X |
| TBM vs VM: 96.7 | TBM vs VM: 81.1 | Romania | |||||
| TBM vs CM: 86.6 | TBM vs CM: 27.7 | Romania | |||||
| Cut off of ≥ 9: TBM vs VM: 51.6 | Cut off of ≥ 9: TBM vs VM: 100 | Romania | |||||
| Kumar et al[34], 19991 | Cut off of ≥ 3: TBM vs non-TBM: 37.0 | Cut off of ≥ 3: TBM vs non-TBM: 58.1 | India | √ | √ | X | X |
Generally, Thwaites (2002) was most sensitive (78.0%-100.0%), while Kumar (1999) was the least (37.0%). Some studies modified the existing score to better suit local populations. Thwaites (2002) was frequently modified in externally validated cohorts. An adjustment in cut-off from ≤ 4 to ≤ 3.5, where patients who scored ≤ 3.5 points were more likely to have TBM, was noted in 2 papers with patients from Malawi and Pakistan[8,9]. The cut-off of each perimeter of Thwaites (2002) was also changed in an Indian population in a study by Vibha et al[10], seen in Supplementary Table 2.
The remaining 17 scores were not externally validated as of time of writing. Table 3 provides an overview of the scores from highest to lowest sensitivity. Of the 17, Yang (2019) had the highest sensitivity (97.0%-98.0%). Further details of each score are in Supplementary Tables 3-24. Of note, Solomons created 2 different scoring systems, Solomons A (2016) and Solomons B (2016), the latter being a simplified 3-variable score. Sensitivity and specificity in Table 3. Lu (2021), Luo (2021) and Yang (2020) had additional patient cohorts internally validating their scores. These additional cohorts comprised patients in the same province as the original cohort. The accuracy of these additional cohorts are provided in Supplementary Tables 13, 14, and 23.
| Ref. | Sensitivity | Specificity | Clinical criteria | Laboratory criteria | Microbiological/histopathological criteria | Radiological criteria | Country |
| Yang et al[35], 2019 | 97.0-98.0 | 81.0-82.0 | √ | √ | X | X | China |
| Goenka et al[36], 2018 | 93.0-100 | 89.0-93.0 | √ | √ | √ | X | South Africa |
| Wen et al[37], 2022 | 96.0 | 86.0 | √ | √ | X | X | China |
| Vibha et al[10], 2012 | 95.7 | 97.6 | √ | √ | X | X | India |
| Ersoy et al[38], 2012 | 95.5 | 100 | √ | √ | X | X | Turkey |
| Solari et al[39], 2018 | 94.9 | 52.6-94.7 | √ | √ | X | X | Peru |
| Chusri et al[40], 2018 | 92.0 | 89.0 | √ | √ | X | √ | Thailand |
| Lee et al[41], 2018 | 89.4 | 80.4 | √ | √ | X | X | South Korea |
| Solomons et al[42] 20161 | 97.0 | 48.0-78.0 | √ | √ | √ | √ | South Africa |
| 79.0 | 48.0-78.0 | √ | √ | √ | √ | ||
| He et al[43], 2021 | 87.0 | 94.0 | √ | √ | X | X | China |
| Dendane et al[44], 2013 | 86.0-88.0 | 93.0-95.0 | √ | √ | √ | √ | Morocco |
| Lu et al[45], 2021 | 81.0-85.8 | 86.3-87.7 | √ | √ | X | √ | China |
| Luo et al[46], 2021 | 81.6 | 91.8 | X | √ | X | X | China |
| Fong et al[24], 2023 | 81.5 | 88.1 | √ | √ | X | √ | China |
| Handryastuti et al[47], 20231 | 47.1 | 95.1 | √ | X | X | √ | Indonesia |
| Patel et al[48], 2010 | 38.0 | 93.0 | √ | √ | X | X | South Africa |
We reviewed 21 existing numerical TBM diagnostic scoring systems. Encouragingly, the externally validated scores display high sensitivity. Across the board, Thwaites (2002) was the most sensitive, achieving 86.0%-97.0% sensitivity with internal validation and 78.0%-100% sensitivity with external validation in 9 different study settings in 6 countries. Sensitivity of Marais (2010) was also relatively high, ranging from 66.7%-100% in 6 study settings in 4 countries. These high sensitivities render them suitable screening tests that can potentially rule out TBM when used in conjunction with clinical acumen, which is particularly important in a high mortality and morbidity disease[11].
TB incidence rises as gross domestic product per capita falls and rates of poverty increase[12]. In such settings, scores are especially useful in establishing a quick diagnosis for early initiation of empirical treatment - the most significant factor in reducing mortality[13]. A significant advantage of 3 of 4 externally validated scores -Thwaites (2002), Hristea (2012) and Kumar (1999) is that they use a combination of clinical criteria and readily-available biochemical tests. Marais (2010) accounts for its use of less available microbiological and radiological criteria by adjusting score cut-off values for when cerebral imaging is unavailable, therefore keeping it accessible for both resource-constrained and non-resource-constrained settings. Microbiological findings are also not essential to diagnose possible or probable TBM. Even in the case where such investigations are unavailable, a likely TBM diagnosis can still be made, and empirical treatment can be started. Furthermore, the scores do not require complex digital programmes for application or interpretation. They can thus be integrated into clinical practice seamlessly without skill training or technology upgrades.
Firstly, scoring systems cannot replace definitive tests, and should act as a guiding tool instead. Some patients will inevitably be classified as having intermediate risk of TBM or have an indeterminate score[6]. In such cases, diagnostic scores may ultimately be of little utility as further evaluation and/or testing by clinicians will still be necessary. Additionally, score models only take into consideration the sign or symptom at the time of presentation. Most of the scores do not include a follow-up component, although clinical, laboratory, microbiological and radiological findings may vary over the disease course and alter scores, particularly in ambiguous presentation[3]. Thus, in ambiguous presentations, we propose clinicians re-calculate patients’ numerical scores throughout admission for disease progression or atypical findings pointing to other diagnoses.
Next, not all scores accounted for significant TBM determinants in formulation and validation. Known risk factors associated with TB include undernourishment, human immunodeficiency virus (HIV) co-infection, alcohol use, smoking and diabetes[1,12]. Of these, HIV co-infection is the most significant risk factor - HIV-positive patients are 15 times to 21 times more likely to develop TB disease[14]. However, only Goenka (2018), which has yet to be externally validated, included HIV co-infection as a criterion. Several studies listed HIV co-infection as an exclusion criteria, limiting their utility in HIV-positive patients. One study in Malawi investigating Thwaites concluded that the score was not clinically appropriate in settings with high HIV-prevalence[8]. The score performed sub-optimally (78% sensitive, 43% positive) in their cohort, with all 12 patients with cryptococcal meningitis giving false positives, a finding attributed to the high prevalence of HIV and HIV-associated cryptococcal meningitis in Malawi. Age is another significant factor in TBM. Young children infected with TB are at high risk of disseminated TB and TBM incidence peaks in children between the ages of 2 years and 4 years[15]. Scores formulated with adult populations may perform differently and have yet to be validated with paediatric populations and vice versa. Of the externally validated scores, only Hristea (2012) was formulated with a population of both adults and children. Therefore, further research on the use of scores in these vulnerable populations should be done. Clinicians operating in settings with high HIV prevalence or treating paediatric patients may consider scores that have been validated in similar populations.
Additionally, scores may be limited by the heterogeneity of the papers and their respective study locations and populations. TB prevalence and strains vary greatly from country to country (and within countries, from region to region). Certain strains of M.TB have been reported to be more likely than others to progress to TBM[16]. Similarly, the prevalence of non-TBM aetiologies varies in different regions. Cryptococcus, which causes cryptococcal meningitis is endemic in tropical, subtropical and temperate climatic regions[17]. Meanwhile, streptococcus pneumoniae is the most common bacterial cause of meningitis in the African and East Mediterranean region as compared to Neisseria menin
Finally, 18 of 30 included studies were carried out retrospectively using medical records. Data collection was not stan
Further validation with different study populations should be done to improve credibility. Existing scoring systems may be mostly applicable to the paper’s study populations with specific demographics and not generalisable to other populations. This is particularly relevant in the case of the 17 scores lacking external validation reviewed in this study. As aforementioned, an immunocompromised state alters the differential diagnoses in a patient with meningitis, which is not accounted for in some of the existing scoring systems[19]. Validation of scores with HIV-positive patients and other immunocompromised cohorts, such as organ transplant patients, is another possibility.
Incorporating CSF and serum biomarkers in scoring systems is an avenue for further research[20]. For example, adenosine deaminase (ADA) was found to be elevated in the CSF of both adult and paediatric TBM patients[21]. 2 scores without external validation, Chusri (2018) and Solari (2018), use CSF ADA as a criterion. A study by Bhatnagar et al[22] also reported that including positive CSF ADA improved the sensitivity and specificity of Kumar (1999) for all TBM stages. Another promising example is tuberculostearic acid, which can promptly detect the presence of M.TB with high sensitivity and specificity in one to two days[23]. When incorporated into a diagnostic model, diagnostic accuracy was found to be comparable to Marais (2010)[24]. As of the time of writing, tuberculostearic acid is only incorporated in Fong (2023). Serum tests can also support TBM diagnosis. One 2019 study found that the combination of 3 serum biomarkers (adipsin, Aβ42, and interleukin-10) was 82.6% sensitive in diagnosing TBM in children[25]. A similar 2016 study found that a 7-marker host serum protein biosignature (C-reactive protein, transthyretin, interferon-γ, complement factor H, apolipoprotein-A1, inducible protein 10 and serum amyloid A) was 93.8% sensitive in diagnosing TB in adults[26].
The role of other supporting tests can also be explored further. Tests such as tuberculin skin test and interferon-gamma release assays support TB diagnosis, although their role in definitive diagnosis is controversial[27-29]. Due to less cross-reactivity with prior bacillus Calmette-Guérin vaccination, interferon-gamma release assays are more specific than tuberculin skin test and can thus serve as a possible criterion in diagnostic scores[30].
Numerical diagnostic scoring systems are easily accessible and useful adjuncts to definitive TBM tests. We suggest using Thwaites (2002) and Marais (2010) as screening tests, as they have been externally validated in multiple settings and show the highest sensitivities. Nevertheless, there is much room for improvement in terms of additional external validation in other specific patient cohorts, especially HIV-positive and paediatric populations. Clinicians should exercise their best judgement in selecting an appropriate scoring system based on the characteristics of the population of interest.
| 1. | World Health Organization. Global tuberculosis report 2023. [cited 14 June 2025]. Available from: https://www.who.int/publications/i/item/9789240083851. |
| 2. | Dodd PJ, Osman M, Cresswell FV, Stadelman AM, Lan NH, Thuong NTT, Muzyamba M, Glaser L, Dlamini SS, Seddon JA. The global burden of tuberculous meningitis in adults: A modelling study. PLOS Glob Public Health. 2021;1:e0000069. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 99] [Cited by in RCA: 72] [Article Influence: 14.4] [Reference Citation Analysis (1)] |
| 3. | Thwaites GE, Tran TH. Tuberculous meningitis: many questions, too few answers. Lancet Neurol. 2005;4:160-170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 102] [Article Influence: 4.9] [Reference Citation Analysis (1)] |
| 4. | Basu Roy R, Thee S, Blázquez-Gamero D, Falcón-Neyra L, Neth O, Noguera-Julian A, Lillo C, Galli L, Venturini E, Buonsenso D, Götzinger F, Martinez-Alier N, Velizarova S, Brinkmann F, Welch SB, Tsolia M, Santiago-Garcia B, Krüger R, Tebruegge M; ptbnet TB Meningitis Study Group. Performance of immune-based and microbiological tests in children with tuberculosis meningitis in Europe: a multicentre Paediatric Tuberculosis Network European Trials Group (ptbnet) study. Eur Respir J. 2020;56:1902004. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 27] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 5. | Schurz H, Glanzmann B, Bowker N, van Toorn R, Solomons R, Schoeman J, van Helden PD, Kinnear CJ, Hoal EG, Möller M. Deciphering Genetic Susceptibility to Tuberculous Meningitis. Front Neurol. 2022;13:820168. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 6. | Thwaites GE, van Toorn R, Schoeman J. Tuberculous meningitis: more questions, still too few answers. Lancet Neurol. 2013;12:999-1010. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 379] [Cited by in RCA: 302] [Article Influence: 23.2] [Reference Citation Analysis (1)] |
| 7. | Ramspek CL, Jager KJ, Dekker FW, Zoccali C, van Diepen M. External validation of prognostic models: what, why, how, when and where? Clin Kidney J. 2021;14:49-58. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 734] [Cited by in RCA: 624] [Article Influence: 124.8] [Reference Citation Analysis (5)] |
| 8. | Checkley AM, Njalale Y, Scarborough M, Zjilstra EE. Sensitivity and specificity of an index for the diagnosis of TB meningitis in patients in an urban teaching hospital in Malawi. Trop Med Int Health. 2008;13:1042-1046. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 9. | Khan SS, Ali Z. Tuberculosis versus pyogenic meningitis in a Pakistani population. Indian J Tuberc. 2017;64:276-280. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 10. | Vibha D, Bhatia R, Prasad K, Srivastava MV, Tripathi M, Kumar G, Singh MB. Validation of diagnostic algorithm to differentiate between tuberculous meningitis and acute bacterial meningitis. Clin Neurol Neurosurg. 2012;114:639-644. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 21] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 11. | Lee HG, William T, Menon J, Ralph AP, Ooi EE, Hou Y, Sessions O, Yeo TW. Tuberculous meningitis is a major cause of mortality and morbidity in adults with central nervous system infections in Kota Kinabalu, Sabah, Malaysia: an observational study. BMC Infect Dis. 2016;16:296. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 12. | Slane VH, Unakal CG. Tuberculous Meningitis. 2024 Sep 2. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025. [PubMed] |
| 13. | Marx GE, Chan ED. Tuberculous meningitis: diagnosis and treatment overview. Tuberc Res Treat. 2011;2011:798764. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 83] [Cited by in RCA: 93] [Article Influence: 6.2] [Reference Citation Analysis (1)] |
| 14. | Qi CC, Xu LR, Zhao CJ, Zhang HY, Li QY, Liu MJ, Zhang YX, Tang Z, Ma XX. Prevalence and risk factors of tuberculosis among people living with HIV/AIDS in China: a systematic review and meta-analysis. BMC Infect Dis. 2023;23:584. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 19] [Article Influence: 6.3] [Reference Citation Analysis (1)] |
| 15. | van Well GT, Paes BF, Terwee CB, Springer P, Roord JJ, Donald PR, van Furth AM, Schoeman JF. Twenty years of pediatric tuberculous meningitis: a retrospective cohort study in the western cape of South Africa. Pediatrics. 2009;123:e1-e8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 229] [Cited by in RCA: 194] [Article Influence: 11.4] [Reference Citation Analysis (1)] |
| 16. | Caws M, Thwaites G, Dunstan S, Hawn TR, Lan NT, Thuong NT, Stepniewska K, Huyen MN, Bang ND, Loc TH, Gagneux S, van Soolingen D, Kremer K, van der Sande M, Small P, Anh PT, Chinh NT, Quy HT, Duyen NT, Tho DQ, Hieu NT, Torok E, Hien TT, Dung NH, Nhu NT, Duy PM, van Vinh Chau N, Farrar J. The influence of host and bacterial genotype on the development of disseminated disease with Mycobacterium tuberculosis. PLoS Pathog. 2008;4:e1000034. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 385] [Cited by in RCA: 362] [Article Influence: 20.1] [Reference Citation Analysis (1)] |
| 17. | do Carmo FN, de Camargo Fenley J, Garcia MT, Rossoni RD, Junqueira JC, de Barros PP, Scorzoni L. Cryptococcus spp. and Cryptococcosis: focusing on the infection in Brazil. Braz J Microbiol. 2022;53:1321-1337. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 18. | Oordt-Speets AM, Bolijn R, van Hoorn RC, Bhavsar A, Kyaw MH. Global etiology of bacterial meningitis: A systematic review and meta-analysis. PLoS One. 2018;13:e0198772. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 210] [Cited by in RCA: 171] [Article Influence: 21.4] [Reference Citation Analysis (1)] |
| 19. | Pruitt AA. Central Nervous System Infections in Immunocompromised Patients. Curr Neurol Neurosci Rep. 2021;21:37. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (1)] |
| 20. | Cao D, Wang T, Wang Y, Han J. Analysis of Cases with Cerebrospinal Fluid Characteristics Similar to Tuberculous Meningitis. Biomed Res Int. 2022;2022:9692804. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 21. | Chander A, Shrestha CD. Cerebrospinal fluid adenosine deaminase levels as a diagnostic marker in tuberculous meningitis in adult Nepalese patients. Asian Pac J Trop Dis. 2013;3:16-19. [RCA] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 9] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 22. | Bhatnagar S, Beig FK, Malik A. Adenosine deaminase and C-reactive protein in cerebrospinal fluid for differential diagnosis of tubercular meningitis in children. Indian J Clin Biochem. 2008;23:299-301. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (1)] |
| 23. | Mårdh PA, Larsson L, Høiby N, Engbaek HC, Odham G. Tuberculostearic acid as a diagnostic marker in tuberculous meningitis. Lancet. 1983;1:367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 34] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 24. | Fong TH, Shi W, Ruan G, Li S, Liu G, Yang L, Wu K, Fan J, Ng CL, Hu Y, Jiang H. Tuberculostearic acid incorporated predictive model contributes to the clinical diagnosis of tuberculous meningitis. iScience. 2023;26:107858. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 25. | Manyelo CM, Solomons RS, Snyders CI, Mutavhatsindi H, Manngo PM, Stanley K, Walzl G, Chegou NN. Potential of Host Serum Protein Biomarkers in the Diagnosis of Tuberculous Meningitis in Children. Front Pediatr. 2019;7:376. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 29] [Article Influence: 4.1] [Reference Citation Analysis (1)] |
| 26. | Chegou NN, Sutherland JS, Malherbe S, Crampin AC, Corstjens PL, Geluk A, Mayanja-Kizza H, Loxton AG, van der Spuy G, Stanley K, Kotzé LA, van der Vyver M, Rosenkrands I, Kidd M, van Helden PD, Dockrell HM, Ottenhoff TH, Kaufmann SH, Walzl G; AE-TBC consortium. Diagnostic performance of a seven-marker serum protein biosignature for the diagnosis of active TB disease in African primary healthcare clinic attendees with signs and symptoms suggestive of TB. Thorax. 2016;71:785-794. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 126] [Article Influence: 12.6] [Reference Citation Analysis (1)] |
| 27. | Zubair M, Sergent SR. Interferon Test. 2025 Aug 2. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025. [PubMed] |
| 28. | Hirabayashi R, Nakayama H, Yahaba M, Yamanashi H, Kawasaki T; Japanese TB Clinical Practice Guideline Systematic Review Task Force. Utility of interferon-gamma releasing assay for the diagnosis of active tuberculosis in children: A systematic review and meta-analysis. J Infect Chemother. 2024;30:516-525. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 29. | Campbell JI, Tabatneck M, Sun M, He W, Musinguzi N, Hedt-Gauthier B, Lamb GS, Goldmann D, Sabharwal V, Sandora TJ, Haberer JE. Increasing Use Of Interferon Gamma Release Assays Among Children ≥2 Years of Age in a Setting With Low Tuberculosis Prevalence. Pediatr Infect Dis J. 2022;41:e534-e537. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 30. | Farhat M, Greenaway C, Pai M, Menzies D. False-positive tuberculin skin tests: what is the absolute effect of BCG and non-tuberculous mycobacteria? Int J Tuberc Lung Dis. 2006;10:1192-1204. [PubMed] |
| 31. | Thwaites GE, Chau TT, Stepniewska K, Phu NH, Chuong LV, Sinh DX, White NJ, Parry CM, Farrar JJ. Diagnosis of adult tuberculous meningitis by use of clinical and laboratory features. Lancet. 2002;360:1287-1292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 275] [Cited by in RCA: 240] [Article Influence: 10.0] [Reference Citation Analysis (1)] |
| 32. | Marais S, Thwaites G, Schoeman JF, Török ME, Misra UK, Prasad K, Donald PR, Wilkinson RJ, Marais BJ. Tuberculous meningitis: a uniform case definition for use in clinical research. Lancet Infect Dis. 2010;10:803-812. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 755] [Cited by in RCA: 679] [Article Influence: 42.4] [Reference Citation Analysis (1)] |
| 33. | Hristea A, Olaru ID, Baicus C, Moroti R, Arama V, Ion M. Clinical prediction rule for differentiating tuberculous from viral meningitis. Int J Tuberc Lung Dis. 2012;16:793-798. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 30] [Article Influence: 2.1] [Reference Citation Analysis (1)] |
| 34. | Kumar R, Singh SN, Kohli N. A diagnostic rule for tuberculous meningitis. Arch Dis Child. 1999;81:221-224. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 80] [Cited by in RCA: 67] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 35. | Yang Y, Qu XH, Zhang KN, Wu XM, Wang XR, Wen A, Li LJ. A Diagnostic Formula for Discrimination of Tuberculous and Bacterial Meningitis Using Clinical and Laboratory Features. Front Cell Infect Microbiol. 2019;9:448. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 36. | Goenka A, Jeena PM, Mlisana K, Solomon T, Spicer K, Stephenson R, Verma A, Dhada B, Griffiths MJ. Rapid Accurate Identification of Tuberculous Meningitis Among South African Children Using a Novel Clinical Decision Tool. Pediatr Infect Dis J. 2018;37:229-234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 37. | Wen A, Liu SM, Cao WF, Zhou YL, Luo CQ, Xiang ZB, Hu F, Zhang P, Leng EL. A New Scoring System to Differentially Diagnose and Distinguish Tuberculous Meningitis and Bacterial Meningitis in South China. Front Neurol. 2022;13:830969. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 38. | Ersoy Y, Yetkin F, Bayraktar MR, Ersoy Y, Yologlu S. A new diagnostic scoring for discrimination of tuberculous and bacterial meningitis on the basis of clinical and laboratory findings. Med Princ Pract. 2012;21:259-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 39. | Solari L, Soto A, Van der Stuyft P. Development of a clinical prediction rule for tuberculous meningitis in adults in Lima, Peru. Trop Med Int Health. 2018;23:367-374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 40. | Chusri S, Hortiwakul T, Sathaporn N, Charernmak B, Phengmak M, Jitpiboon W, Geater AF. Diagnostic scoring system for tuberculous meningitis among adult patients with non-suppurative and non-bacterial meningitis. J Infect Chemother. 2018;24:648-653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 41. | Lee SA, Kim SW, Chang HH, Jung H, Kim Y, Hwang S, Kim S, Park HK, Lee JM. A New Scoring System for the Differential Diagnosis between Tuberculous Meningitis and Viral Meningitis. J Korean Med Sci. 2018;33:e201. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 28] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 42. | Solomons RS, Visser DH, Marais BJ, Schoeman JF, van Furth AM. Diagnostic accuracy of a uniform research case definition for TBM in children: a prospective study. Int J Tuberc Lung Dis. 2016;20:903-908. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 18] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 43. | He H, Zou Y, He J, Bu H, Liu Y. A Diagnostic Scoring System for Distinguishing between Tuberculous and Bacterial Meningitis Based on Clinical and Laboratory Findings. Biomed Res Int. 2021;2021:1220650. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 44. | Dendane T, Madani N, Zekraoui A, Belayachi J, Abidi K, Zeggwagh AA, Abouqal R. A simple diagnostic aid for tuberculous meningitis in adults in Morocco by use of clinical and laboratory features. Int J Infect Dis. 2013;17:e461-e465. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 45. | Lu Y, Ma C, Chen R, Hu Z, Yao H, Zhang Q, Zhu H, Wang Z, Song Z, Zhang C, Liu D. Development and validation of a new scoring system for the early diagnosis of tuberculous meningitis in adults. Diagn Microbiol Infect Dis. 2021;101:115393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 46. | Luo Y, Xue Y, Lin Q, Mao L, Tang G, Song H, Liu W, Wu S, Liu W, Zhou Y, Xu L, Xiong Z, Wang T, Yuan X, Gan Y, Sun Z, Wang F. Diagnostic Model for Discrimination Between Tuberculous Meningitis and Bacterial Meningitis. Front Immunol. 2021;12:731876. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (1)] |
| 47. | Handryastuti S, Latifah D, Bermanshah EK, Gunardi H, Kadim M, Iskandar RATP. Development of clinical-based scoring system to diagnose tuberculous meningitis in children. Arch Dis Child. 2023;108:884-888. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 48. | Patel VB, Singh R, Connolly C, Kasprowicz V, Zumla A, Ndungu T, Dheda K. Comparison of a clinical prediction rule and a LAM antigen-detection assay for the rapid diagnosis of TBM in a high HIV prevalence setting. PLoS One. 2010;5:e15664. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 44] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/