Published online Mar 9, 2026. doi: 10.5492/wjccm.v15.i1.110552
Revised: August 19, 2025
Accepted: November 14, 2025
Published online: March 9, 2026
Processing time: 264 Days and 18.4 Hours
Acute liver failure (ALF) is a devastating condition that primarily affects young adults. This often-lethal condition involves a rapid loss of hepatic function, that then leads to multiple organ failure. The accumulation of numerous toxins, espe
Core Tip: The early initiation of continuous renal replacement therapy in patients admitted to the intensive care unit with acute liver failure is safe and provides a range of benefits that are likely neuroprotective. These include control of hyperammonaemia, prevention of fever, control of acidaemia, control of fluid balance and control of electrolyte derangement. Continuous renal replacement therapy can be started in all acute liver failure patients intubated for hepatic encephalopathy and should not be delayed until evidence of renal failure becomes apparent.
- Citation: Fisher C, Warrillow S. Continuous renal replacement therapy in acute liver failure. World J Crit Care Med 2026; 15(1): 110552
- URL: https://www.wjgnet.com/2220-3141/full/v15/i1/110552.htm
- DOI: https://dx.doi.org/10.5492/wjccm.v15.i1.110552
Acute liver failure (ALF) is a rare condition with a high mortality and is characterized by rapid loss of liver function leading to multiple organ failure. Many definitions have been proposed over the years, but the original definition by Trey and Davidson[1] highlights the key elements of encephalopathy, coagulopathy (commonly assessed by prolongation of pro-thrombin time or the international normalized ration), and the absence of pre-existing liver disease. The absence of chronic liver disease differentiates of ALF from decompensated chronic liver disease, hepato-renal syndrome, and acute-on-chronic liver failure, all of which have different management priorities[2-4].
The causes of ALF are diverse (Table 1), with considerable regional differences in cause. In English speaking countries acetaminophen is the largest single cause while hepatotropic viruses dominate in Africa and Asia[4]. It is important that an extensive effort is made to establish the cause of ALF, as some have specific therapies (such as N-acetyl cysteine in paracetamol overdose; antiviral therapy for viral hepatitis; or silibinin for amatoxin ALF) and other potential etiologies may be contra-indications for liver transplantation (such as malignant infiltration, hemophagocytic lymphangitis, or ischemic hepatitis secondary to advanced cardiogenic shock).
| Aetiology | Examples |
| Metabolic | Wilsons disease, fatty liver disease of pregnancy |
| Viral | HAV, HBV, HDV, HEV, HSV, EBV, CMV |
| Drugs | Paracetamol, aspirin, niacin, MDMA |
| Toxins | Amanita mushroom, yellow phosphorous |
| Vascular | Budd-Chiari syndrome |
The length of time from the onset of jaundice to the development of hepatic encephalopathy is important. All classification systems are based upon dividing ALF patients into hyperacute ALF (< 7 days), acute ALF (7-21 days), and sub-acute ALF (> 2 days), albeit with subtle variation in terminology[5]. This approach assists in identifying the cause, the presenting illness’s characteristics and severity, development of extra-hepatic failures (hyperacute > acute > sub-acute), and mortality in the absence of liver transplantation (sub-acute > acute > hyperacute)[6].
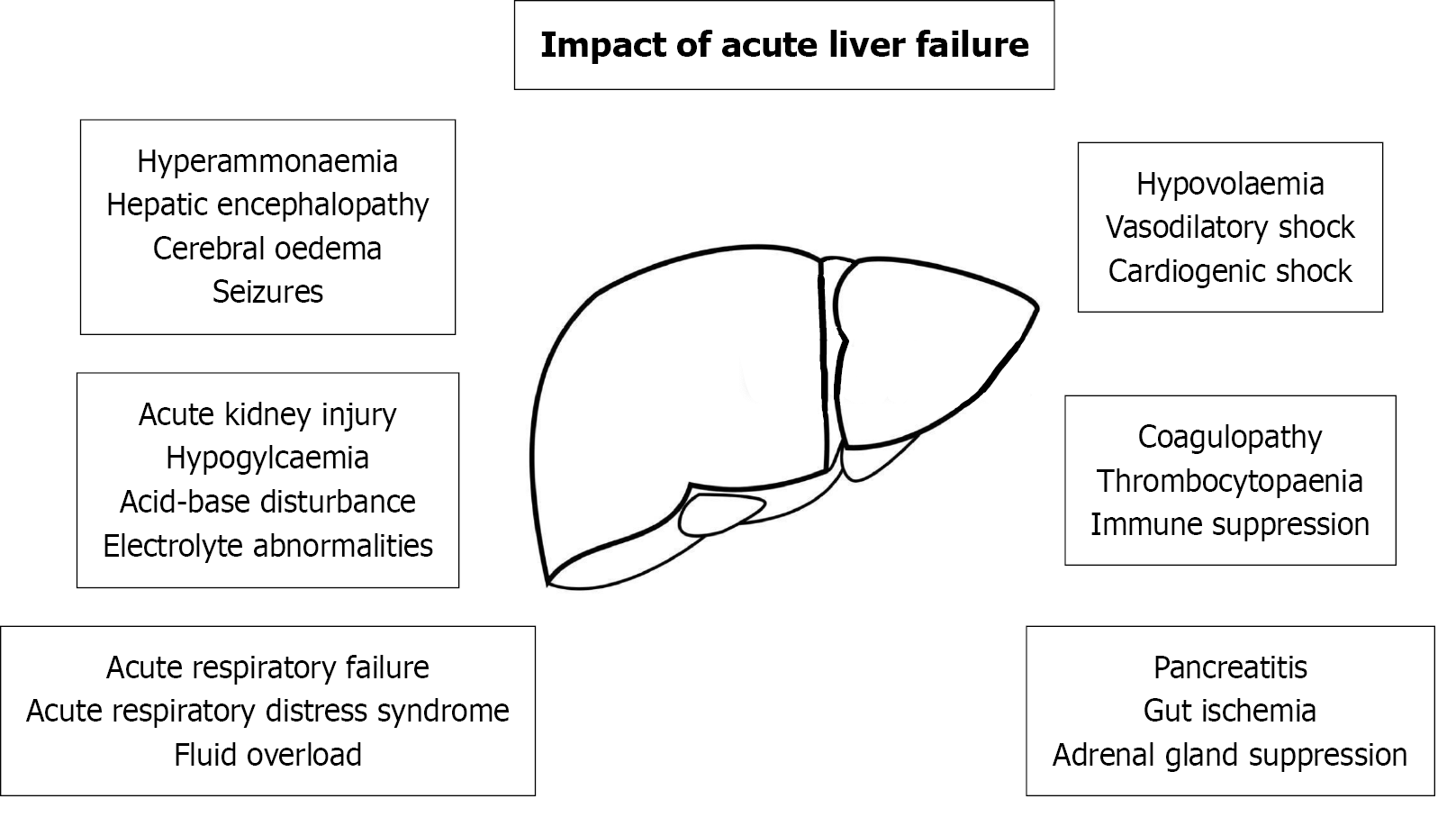
ALF frequently presents with multi-organ failure and management priorities largely centre on providing high quality supportive care (Figure 1). Many of the complications in ALF have either underlying causes or consequences, that can be addressed by the timely provision of continuous renal replacement therapy (CRRT) (Table 2). The presence and impact of these challenges has seen the conceptual evolution of CRRT from providing support based upon traditional renal-centric indications to a view of CRRT providing ALF specific “metabolic-toxin-fluid balance” management.
| Metabolic | Toxin | Fluid |
| Correction of electrolyte imbalance | Ammonia removal | Maintenance of euvolemia |
| Maintenance of normothermia | Potential removal of additional inflammatory mediators | - |
| Control of acid-base balance | - | - |
| Prevention of hyponatremia | - | - |
Acute kidney injury (AKI) is a frequent complication of ALF and an important indication for renal replacement therapy (RRT)[7,8]. Our understanding of the incidence and impact of AKI in ALF has matured in conjunction with the con
The pathophysiology of AKI in ALF is complex and often includes direct renal injury from the same insult that caused hepatic injury. Direct nephrotoxicity has been implicated in patients with paracetamol toxicity, via processes parallel to the hepatotoxic N-acetyl-p-benzoquinone imine pathway[11], and in amanita mushroom poisoning as a direct effect of amanita toxin[12]. The secondary effect of sustained multi-organ dysfunction also plays a significant role in the development of AKI in ALF. Patients with ALF manifest a shocked state that is similar in many ways to severe sepsis and is likely to share some of the same microcirculatory and macrocirculatory pathological processes. Early hepatocyte necrosis is associated with release of pathogen-associated molecular patterns and damage-associated molecular patterns that trigger an innate immune system response via recognition by toll-like receptors and a pro-inflammatory response ensues. This disordered inflammatory response result in renal microcirculation shunting as well as direct oxidative stress and mitochondrial dysfunction in renal tubular cells that is similar to sepsis related AKI[13,14].
Other possible contributing mechanisms for ALF associated AKI impact the macrocirculation. Systemic inflammation, vasodilation and hypovolemia from commonly associated gastrointestinal disturbances, coupled with variable cardiac depression related to ALF can lead to renal hypoperfusion and associated ischemic kidney injury[8]. Whilst the literature is sparse, it appears that age, requirement for vasopressor therapy, systemic inflammation, concurrent sepsis, and para
Severe cerebral oedema and associated raised intracranial hypertension are a feared and potentially fatal complication of ALF. The pathophysiology of cerebral oedema involves complex interactions between toxic metabolites, direct neu
Ammonia has several properties that promote its removal by CRRT. It is a small (17 kilodalton), water soluble, non-protein bound molecule; properties attractive for clearance by standard dialysers/membranes in both intermittent hemo
In the adult critically ill population, specifically ALF, there have been few studies looking at the clearance of ammonia. Despite the important differences between ALF and in-born errors of metabolism, the available evidence suggests a similar efficacy of ammonia clearance. Slack et al[24] looked at a mixed population of ALF, decompensated chronic liver disease and post-operative patients, who were hyperammonaemic, finding greater ammonia clearance with high intensity continuous veno-venous haemofiltration (CVVH) 90 mL/kg/hour compared with low intensity CVVH 35 mL/kg/hour. The authors concluded that ammonia clearance was related closely related to effluent flow rate, although the reported clearance rate was greater than the effluent rate, suggesting methodological challenges may have affected reported clearance values[24].
Other studies have looked at changes in ammonia levels in response to the CRRT. Warrillow et al[25] retrospectively reviewed 45 ALF patients with high grade encephalopathy and ammonia > 150 μmol/L. In this population, all patients received CRRT as either CVVHDF or CVVH, with a median effluent dose of 43 mL/kg/hour, and a median duration of CRRT of 75-hours. The authors found a significant reduction in ammonia levels over time, and that the reduction of ammonia levels was associated with the cumulative dose of CRRT rather than the intensity of therapy, (r = 0.299; P = 0.03)[25].
These findings have further supported by a large multi-center review from the America Liver Failure Study Group by Cardoso et al[26]. The authors retrospectively looked at 340 ALF patients who received RRT and had their ammonia status recorded. When they compared CRRT, intermittent dialysis, and no RRT; CRRT between the days 1-3 resulted in significant greater decrease in ammonia compared to no RRT (37.9% vs 18.6%; P < 0.007), but no significant difference was evident between intermittent and continuous modalities. However, this study is limited by the small number of CRRT patients (n = 56/340, 17%) and the absence of data regarding total duration of CRRT therapy, intensity of CRRT therapy, or modality of CRRT[26]. These papers suggest that the early aggressive initiation of CRRT is effective in facilitating ammonia clearance and may improve outcomes for patients with elevated ammonia levels and hepatic encephalopathy.
One challenge that is yet to be answered is the potential impact of the different CRRT modalities on hyperammonaemia. Whilst ammonia can be removed by all modalities it is unclear whether (CVVH - based on pressure gradient over the membrane), continuous veno-venous haemodiaylsis (CVVHD - based on concentration gradient across the membrane), or continuous veno-venous haemodiafiltration (CVVHDF - based on a combination of both modalities), offer any specific benefits. Based on ammonia’s chemical properties, it is possible that a purely diffusive technique, such as continuous veno-venous haemodiaylsis, may be more effective than other techniques using pre-dilution fluid, such as CVVH[27]. Alternatively, CVVHDF by offering combination of both may provide additional clearance of toxic small and middle-sized molecules, in addition to ammonia removal[28]. Evidence to guide decision making in ALF may be extrapolated from the studies above. Slack et al[24] used CVVH for all their patients, whereas in Warrillow et al[25] 42/54 (78%) used CVVHDF and 12/54 (22%) used CVVH, with no reported clearance difference between the modalities[24,25]. Looking outside the ALF population, Fisher et al[29] looked at differing CRRT modalities in a small population of mild hyperammonaemic patients with decompensated chronic liver disease, median ammonia 95 (interquartile range: 70-99) μmol/L. In this study, Fisher et al[29] found using a fixed 3000 mL/hour effluent rate, no significant difference in am
Another area of the uncertainty in the literature is the optimal intensity of CRRT for ammonia clearance. Slack et al[24] when comparing high volume exchange (90 mL/kg/hour) and low (35 mL/kg/hour) found a significantly higher clearance rate. Warrillow et al[25] reported that despite variation in effluent dose from 2000 mL/hour (24%), 3000 mL/hour (35%), 4000 mL/hour (33%), and 5000 mL/hour (9%) for a median dose 43 mL/kg/hour, there was a trend towards significance relationship between dose and decreasing ammonia levels (Spearman’s rank correlation coefficient rs = 0.249; P = 0.07), however this was significantly related to duration of therapy over days one and two (rs = 0.159; P = 0.26, rather than the intensity[24,25]. The current evidence would suggest that higher doses of CRRT therapy facilitate greater ammonia clearance, but this needs to be balanced by therapy duration. In their scoping review of ammonia clearance, Naorungroj et al[22], found that in the 13 studies (11 paediatric and 2 adult) ammonia clearance was weakly associated with effluent dose rs = 0.584; P < 0.001.
Given the importance of cerebral oedema and raised intracranial pressure in early ALF mortality, and the key role of ammonia in its pathogenesis, aggressive lowering of ammonia levels by CRRT is an attractive therapeutic option. How
The largest study to date attempting to answer this question has come from Cardosa and the America Liver Failure Study Group. Across 880 patients, when corrected for year of admission, age, aetiology, and disease severity, they found that CRRT was associated with 21-day transplant free survival, with an odds ratio of 0.47 (0.26-0.82) P = 0.008[26]. Although over this time there was a significance increase in use of other supporting therapies such as N-acetylcysteine, intravenous antibiotics and a preference for CRRT over intermittent hemodialysis. Of note is the finding that intermittent haemodialysis was associated with increased mortality, despite efficacy in ammonia clearance. One possible explanation is that the significant haemodynamic and fluid fluctuations that characterise intermittent techniques can potentially precipitate cerebral oedema and elevate intra-cranial pressure, directly leading to serious neurological complications[31].
ALF is also frequently accompanied by profound metabolic and electrolyte imbalance. Acidosis in ALF is usually multifactorial in nature related to systemic shock, impaired hepatic lactate clearance, and kidney dysfunction[32]. The importance of these factors is highlighted by their inclusion in the King’s College Listing for Liver Transplant Criteria in both paracetamol and non-paracetamol ALF. Hyponatraemia is known to be detrimental in the neurosurgical/traumatic brain injured population where it is associated with poor outcomes[33]; and should be carefully avoided in ALF patients. Further adding to the metabolic chaos characterizing ALF is the impact of temperature and positive fluid balance on cerebral oedema and raised intracranial pressure. Whilst a randomized multicenter trial looking at prophylactic hypothermia (33 degrees vs 36 degrees) failed to demonstrate a survival benefit, extrapolation from other neurological populations suggests that the avoidance of fever may be beneficial with patients at risk of cerebral oedema[34]. Addi
Over the last decade, the evidence supporting the early role of CRRT in ALF has evolved significantly. Cumulatively, this evidence suggests that CRRT should be viewed as a “metabolic-toxin-fluid” intervention, with early initiation aiming to minimize the impact of hepatic dysfunction by ammonia level control, acid-base and electrolyte abnormalities, maintenance of normothermia, and strict regulation of fluid balance. As such, CRRT has a unique and specific role in ALF that extends considerably beyond the traditional “renal” indications where the goals are fluid management and solute clearance. Further research is required to determine the optimal CRRT dose, duration and modality in its use in ALF.
We gratefully acknowledge the past support and inspiration of our great friend and mentor, Professor Rinaldo Bellomo.
| 1. | Trey C, Davidson CS. The management of fulminant hepatic failure. Prog Liver Dis. 1970;3:282-298. [PubMed] |
| 2. | Bernal W, Wendon J. Acute liver failure. N Engl J Med. 2013;369:2525-2534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1040] [Cited by in RCA: 885] [Article Influence: 68.1] [Reference Citation Analysis (0)] |
| 3. | Moreau R, Jalan R, Gines P, Pavesi M, Angeli P, Cordoba J, Durand F, Gustot T, Saliba F, Domenicali M, Gerbes A, Wendon J, Alessandria C, Laleman W, Zeuzem S, Trebicka J, Bernardi M, Arroyo V; CANONIC Study Investigators of the EASL–CLIF Consortium. Acute-on-chronic liver failure is a distinct syndrome that develops in patients with acute decompensation of cirrhosis. Gastroenterology. 2013;144:1426-1437, 1437.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2562] [Cited by in RCA: 2292] [Article Influence: 176.3] [Reference Citation Analysis (4)] |
| 4. | European Association for the Study of the Liver; Clinical practice guidelines panel, Wendon, J, Cordoba J, Dhawan A, Larsen FS, Manns M, Samuel D, Simpson KJ, Yaron I; EASL Governing Board representative, Bernardi M. EASL Clinical Practical Guidelines on the management of acute (fulminant) liver failure. J Hepatol. 2017;66:1047-1081. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 816] [Cited by in RCA: 695] [Article Influence: 77.2] [Reference Citation Analysis (4)] |
| 5. | O'Grady JG, Schalm SW, Williams R. Acute liver failure: redefining the syndromes. Lancet. 1993;342:273-275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 446] [Cited by in RCA: 414] [Article Influence: 12.5] [Reference Citation Analysis (3)] |
| 6. | Bernal W, Lee WM, Wendon J, Larsen FS, Williams R. Acute liver failure: A curable disease by 2024? J Hepatol. 2015;62:S112-S120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 219] [Cited by in RCA: 193] [Article Influence: 17.5] [Reference Citation Analysis (2)] |
| 7. | O'Riordan A, Brummell Z, Sizer E, Auzinger G, Heaton N, O'Grady JG, Bernal W, Hendry BM, Wendon JA. Acute kidney injury in patients admitted to a liver intensive therapy unit with paracetamol-induced hepatotoxicity. Nephrol Dial Transplant. 2011;26:3501-3508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 50] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 8. | Leithead JA, Ferguson JW, Bates CM, Davidson JS, Lee A, Bathgate AJ, Hayes PC, Simpson KJ. The systemic inflammatory response syndrome is predictive of renal dysfunction in patients with non-paracetamol-induced acute liver failure. Gut. 2009;58:443-449. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 89] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 9. | Tujios SR, Hynan LS, Vazquez MA, Larson AM, Seremba E, Sanders CM, Lee WM; Acute Liver Failure Study Group. Risk factors and outcomes of acute kidney injury in patients with acute liver failure. Clin Gastroenterol Hepatol. 2015;13:352-359. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 124] [Cited by in RCA: 102] [Article Influence: 9.3] [Reference Citation Analysis (1)] |
| 10. | Warrillow S, Tibballs H, Bailey M, McArthur C, Lawson-Smith P, Prasad B, Anstey M, Venkatesh B, Dashwood G, Walsham J, Holt A, Wiersema U, Gattas D, Zoeller M, Garcia Alvarez M, Bellomo R; Australasian Management of Acute Liver Failure Investigators (AMALFI). Characteristics, management and outcomes of patients with acute liver failure admitted to Australasian intensive care units. Crit Care Resusc. 2019;21:188-199. [PubMed] [DOI] [Full Text] |
| 11. | Moore JK, Love E, Craig DG, Hayes PC, Simpson KJ. Acute kidney injury in acute liver failure: a review. Expert Rev Gastroenterol Hepatol. 2013;7:701-712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 53] [Article Influence: 4.1] [Reference Citation Analysis (1)] |
| 12. | Kirchmair M, Carrilho P, Pfab R, Haberl B, Felgueiras J, Carvalho F, Cardoso J, Melo I, Vinhas J, Neuhauser S. Amanita poisonings resulting in acute, reversible renal failure: new cases, new toxic Amanita mushrooms. Nephrol Dial Transplant. 2012;27:1380-1386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 37] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 13. | Dong V, Nanchal R, Karvellas CJ. Pathophysiology of Acute Liver Failure. Nutr Clin Pract. 2020;35:24-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 114] [Article Influence: 19.0] [Reference Citation Analysis (1)] |
| 14. | Zarbock A, Gomez H, Kellum JA. Sepsis-induced acute kidney injury revisited: pathophysiology, prevention and future therapies. Curr Opin Crit Care. 2014;20:588-595. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 250] [Cited by in RCA: 270] [Article Influence: 22.5] [Reference Citation Analysis (1)] |
| 15. | Scott TR, Kronsten VT, Hughes RD, Shawcross DL. Pathophysiology of cerebral oedema in acute liver failure. World J Gastroenterol. 2013;19:9240-9255. [PubMed] [DOI] [Full Text] |
| 16. | Warrillow SJ, Bellomo R. Preventing cerebral oedema in acute liver failure: the case for quadruple-H therapy. Anaesth Intensive Care. 2014;42:78-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 28] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 17. | Bernal W, Hall C, Karvellas CJ, Auzinger G, Sizer E, Wendon J. Arterial ammonia and clinical risk factors for encephalopathy and intracranial hypertension in acute liver failure. Hepatology. 2007;46:1844-1852. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 405] [Cited by in RCA: 312] [Article Influence: 16.4] [Reference Citation Analysis (1)] |
| 18. | Clemmesen JO, Larsen FS, Kondrup J, Hansen BA, Ott P. Cerebral herniation in patients with acute liver failure is correlated with arterial ammonia concentration. Hepatology. 1999;29:648-653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 579] [Cited by in RCA: 449] [Article Influence: 16.6] [Reference Citation Analysis (1)] |
| 19. | Gupta S, Fenves AZ, Hootkins R. The Role of RRT in Hyperammonemic Patients. Clin J Am Soc Nephrol. 2016;11:1872-1878. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 71] [Article Influence: 7.1] [Reference Citation Analysis (1)] |
| 20. | Ronco C, Bellomo R, Ricci Z. Critical Care Nephrology. Amsterdam: Elsevier, 2019. |
| 21. | Raina R, Bedoyan JK, Lichter-Konecki U, Jouvet P, Picca S, Mew NA, Machado MC, Chakraborty R, Vemuganti M, Grewal MK, Bunchman T, Sethi SK, Krishnappa V, McCulloch M, Alhasan K, Bagga A, Basu RK, Schaefer F, Filler G, Warady BA. Consensus guidelines for management of hyperammonaemia in paediatric patients receiving continuous kidney replacement therapy. Nat Rev Nephrol. 2020;16:471-482. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 67] [Article Influence: 11.2] [Reference Citation Analysis (1)] |
| 22. | Naorungroj T, Yanase F, Eastwood GM, Baldwin I, Bellomo R. Extracorporeal Ammonia Clearance for Hyperammonemia in Critically Ill Patients: A Scoping Review. Blood Purif. 2021;50:453-461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 27] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 23. | Häberle J, Burlina A, Chakrapani A, Dixon M, Karall D, Lindner M, Mandel H, Martinelli D, Pintos-Morell G, Santer R, Skouma A, Servais A, Tal G, Rubio V, Huemer M, Dionisi-Vici C. Suggested guidelines for the diagnosis and management of urea cycle disorders: First revision. J Inherit Metab Dis. 2019;42:1192-1230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 380] [Cited by in RCA: 349] [Article Influence: 49.9] [Reference Citation Analysis (1)] |
| 24. | Slack AJ, Auzinger G, Willars C, Dew T, Musto R, Corsilli D, Sherwood R, Wendon JA, Bernal W. Ammonia clearance with haemofiltration in adults with liver disease. Liver Int. 2014;34:42-48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 117] [Article Influence: 9.8] [Reference Citation Analysis (5)] |
| 25. | Warrillow S, Fisher C, Bellomo R. Correction and Control of Hyperammonemia in Acute Liver Failure: The Impact of Continuous Renal Replacement Timing, Intensity, and Duration. Crit Care Med. 2020;48:218-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 53] [Article Influence: 8.8] [Reference Citation Analysis (2)] |
| 26. | Cardoso FS, Gottfried M, Tujios S, Olson JC, Karvellas CJ; US Acute Liver Failure Study Group. Continuous renal replacement therapy is associated with reduced serum ammonia levels and mortality in acute liver failure. Hepatology. 2018;67:711-720. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 192] [Cited by in RCA: 174] [Article Influence: 21.8] [Reference Citation Analysis (1)] |
| 27. | Cerdá J, Ronco C. Modalities of continuous renal replacement therapy: technical and clinical considerations. Semin Dial. 2009;22:114-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 41] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 28. | Ricci Z, Ronco C, Bachetoni A, D'amico G, Rossi S, Alessandri E, Rocco M, Pietropaoli P. Solute removal during continuous renal replacement therapy in critically ill patients: convection versus diffusion. Crit Care. 2006;10:R67. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 87] [Cited by in RCA: 83] [Article Influence: 4.2] [Reference Citation Analysis (1)] |
| 29. | Fisher C, Baldwin I, Fealy N, Naorungroj T, Bellomo R. Ammonia Clearance with Different Continuous Renal Replacement Therapy Techniques in Patients with Liver Failure. Blood Purif. 2022;51:840-846. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 20] [Reference Citation Analysis (0)] |
| 30. | Chaba A, Warrillow SJ, Fisher C, Spano S, Maeda A, Phongphithakchai A, Pattamin N, Hikasa Y, Kitisin N, Warming S, Michel C, Eastwood GM, Bellomo R. Severely Hyperammonemic Acute Liver Failure due to Paracetamol Overdose: The Impact of High-Intensity Continuous Renal Replacement Therapy. Blood Purif. 2025;54:111-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 31. | Davenport A. Renal replacement therapy in the patient with acute brain injury. Am J Kidney Dis. 2001;37:457-466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 56] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 32. | Gyorgypal A, Chaturvedi A, Chopda V, Zhang H, Chundawat SPS. Evaluating the impact of media and feed combinations on CHO cell culture performance and monoclonal antibody (trastuzumab) production. Cytotechnology. 2025;77:40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 33. | Cook AM, Morgan Jones G, Hawryluk GWJ, Mailloux P, McLaughlin D, Papangelou A, Samuel S, Tokumaru S, Venkatasubramanian C, Zacko C, Zimmermann LL, Hirsch K, Shutter L. Guidelines for the Acute Treatment of Cerebral Edema in Neurocritical Care Patients. Neurocrit Care. 2020;32:647-666. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 356] [Cited by in RCA: 258] [Article Influence: 43.0] [Reference Citation Analysis (1)] |
| 34. | Bernal W, Murphy N, Brown S, Whitehouse T, Bjerring PN, Hauerberg J, Frederiksen HJ, Auzinger G, Wendon J, Larsen FS. A multicentre randomized controlled trial of moderate hypothermia to prevent intracranial hypertension in acute liver failure. J Hepatol. 2016;65:273-279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 67] [Article Influence: 6.7] [Reference Citation Analysis (2)] |
| 35. | van der Jagt M. Fluid management of the neurological patient: a concise review. Crit Care. 2016;20:126. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 71] [Article Influence: 7.1] [Reference Citation Analysis (1)] |
| 36. | Vincent JL. Fluid management in the critically ill. Kidney Int. 2019;96:52-57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 82] [Article Influence: 11.7] [Reference Citation Analysis (1)] |
| 37. | Michel C, Warming S, Neto AS, Abeygunawardana V, Fisher C, Kishore K, Warrillow S, Bellomo R. Early and prolonged continuous hypertonic saline infusion in patients with acute liver failure. J Crit Care. 2023;76:154289. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/