Published online Mar 9, 2026. doi: 10.5492/wjccm.v15.i1.111114
Revised: August 14, 2025
Accepted: November 14, 2025
Published online: March 9, 2026
Processing time: 249 Days and 23.5 Hours
Acute kidney injury (AKI) affects 40%–80% of patients with acute liver failure (ALF) and arises from complex mechanisms, including hemodynamic instability, drug toxicity, and direct renal insults. Unlike chronic liver disease, AKI in ALF is poorly studied.
To consolidate current evidence and guide future research and interventions to improve outcomes in ALF patients with AKI.
This systematic review followed PRISMA guidelines, searching databases from 2000 to 2024 for studies on AKI in ALF. We included studies focused on adult, English-language, full-length epidemiological studies. Data were extracted on study design, patient demographics, risk factors, and outcomes. Methodological quality was assessed using a tailored 5-point tool. Data were summarized using descriptive statistics and analyzed with GraphPad Prism and MedCalc software.
This systematic review analyzed nine studies published between 2009 and 2024, encompassing 6031 patients with ALF, of whom 62.9% developed AKI. Most studies were retrospective and varied in methodology, setting, and population. Only three studies reported in
AKI is common in ALF, increasing morbidity and mortality; standardised diagnosis and targeted management strategies are urgently needed.
Core Tip: This systematic review of nine studies (6031 patients) found that acute kidney injury (AKI) occurs in 62.9% of acute liver failure (ALF) cases, with incidence varying from 37.9% to 79.1%. Drug-induced ALF, especially paracetamol-related, was the predominant etiology in most cohorts, while viral hepatitis was more common in Asia. Few studies iden
- Citation: Ahmed A, Gautam M, Azim A, Chakravarti B, Tiwari M. Burden and impact of acute kidney injury in acute liver failure: A systematic review. World J Crit Care Med 2026; 15(1): 111114
- URL: https://www.wjgnet.com/2220-3141/full/v15/i1/111114.htm
- DOI: https://dx.doi.org/10.5492/wjccm.v15.i1.111114
Acute kidney injury (AKI) is characterized by a sudden and often reversible decline in renal function. It is typically classified into prerenal, intrarenal, and postrenal categories based on the underlying pathophysiological mechanisms. The prevalence of AKI varies significantly, ranging from less than 1% to 66% across different studies, contingent upon the patient cohort, geographical location of the study, and the diagnostic criteria employed to define AKI[1].
AKI affects 40% to 80% of acute liver failure (ALF) patients[2]. Among cirrhosis patients, its prevalence can vary from 20% to 50%[3,4]. Irrespective of the cause, the onset of renal injury in patients with liver disease heralds increased mor
Multiple factors predispose liver failure patients to renal injury. The association between advanced cirrhosis and hepatorenal syndrome (HRS-1 or HRS-AKI) underscores a distinct form of AKI predominantly driven by systemic and renal hemodynamic disturbances[4]. In contrast to chronic liver failure, the development of AKI in patients with ALF is far more complex, multifaceted, and distinct in mechanisms[5]. The key mechanisms leading to AKI in ALF include pre-renal insults, such as poor oral intake, use of laxatives, and aggressive decongestant therapy for raised intracranial pressure; ischemic acute tubular injury resulting from hemorrhagic or septic shock, congestive nephropathy, raised intra-abdominal pressure, or prolonged pre-renal insult; toxic acute tubular injury due to bile cast nephropathy or nephrotoxic drugs; and direct renal injury from the same insult that caused liver failure, for example, paracetamol poisoning.
Renal injury in patients with liver disease has been well-documented in the medical literature for the past seven to eight decades. Still, the primary focus of these studies has typically been AKI in patients with chronic liver disease, with very few studies evaluating its pathogenesis and treatment strategies in ALF[6,7]. Without studies assessing the contributions of different factors to renal injury in this patient population, implementing effective prophylactic measures and treatment strategies is challenging.
This systematic review aims to comprehensively assess the prevalence of AKI in patients with ALF, identify associated risk factors, evaluate its impact on clinical outcomes, and highlight key gaps in the existing research. Systematic reviews are a valuable tool to synthesize and summarize available data in a given field, providing a foundation for evidence-based practice and guiding future research[8].
The primary objective of this systematic review was to consolidate the epidemiological evidence on AKI in the context of ALF, with a focus on estimating its prevalence, identifying the risk factors contributing to its development, the associated clinical characteristics and outcomes, and highlighting gaps in the existing literature and reporting practices.
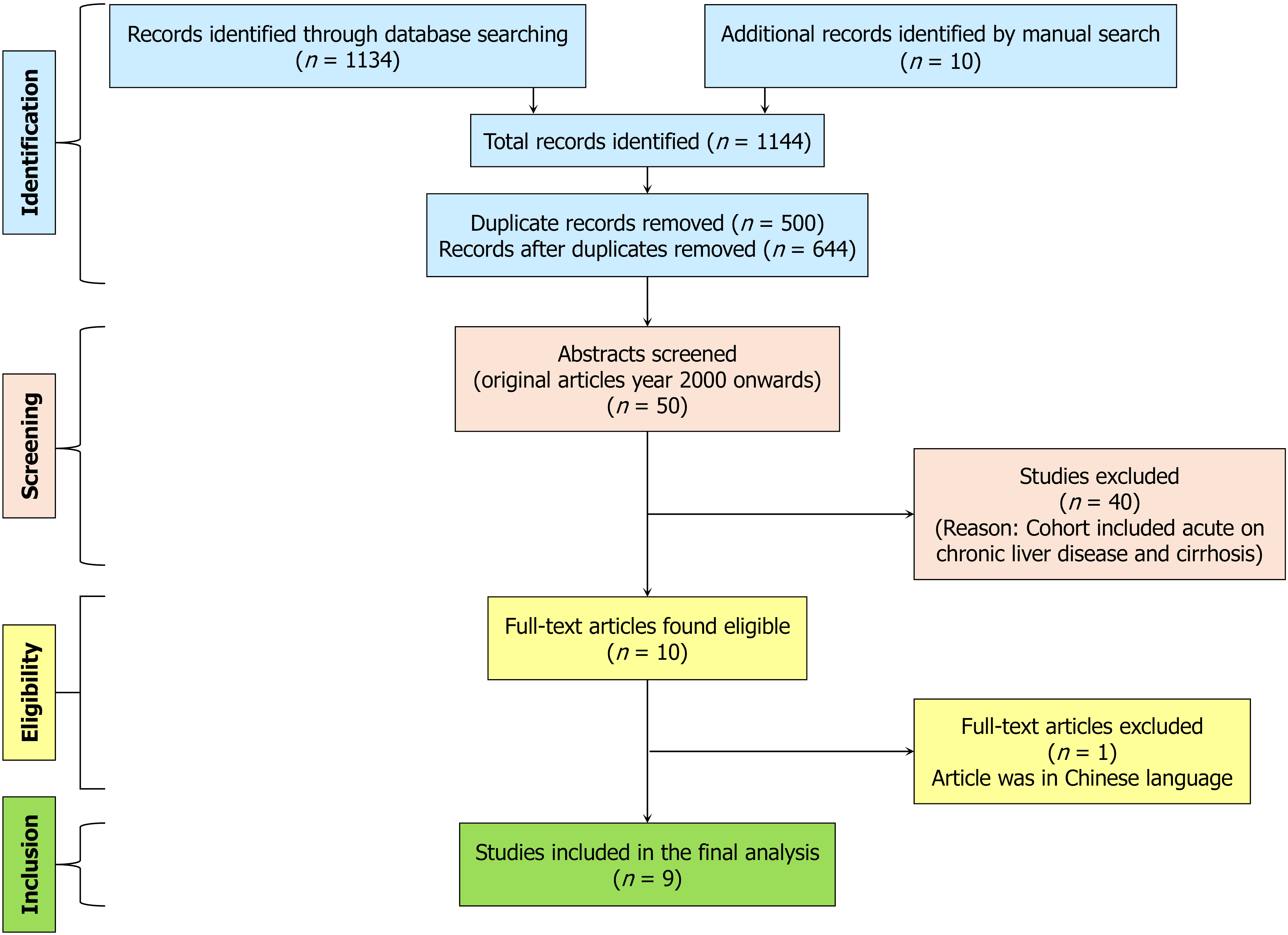
Two investigators (Ahmed A and Gautam M) independently conducted a comprehensive literature search from January 2000 to December 2024 using multiple databases, including PubMed, MEDLINE, EMBASE, and Google Scholar, to identify relevant studies on AKI in ALF patients. We performed search using specific keywords phrase “acute kidney injury in acute liver failure”, “acute kidney failure in acute liver failure”, “acute renal insufficiency in acute liver failure”, “acute kidney injury in acute hepatic failure”, “acute kidney failure in acute hepatic failure”, “acute renal insufficiency in acute hepatic failure”. Moreover, we manually searched the references cited in the relevant articles. This approach aimed to capture the breadth of literature addressing the interplay between AKI and ALF, including studies with varying methodologies, geographical contexts, and clinical insights. The titles and abstracts of all documents retrieved through the literature search were thoroughly screened to assess their relevance. Studies found irrelevant to the research objectives and duplicate records were excluded from further analysis to ensure the final selection comprised unique and pertinent manuscripts. This multi-step approach was designed to maximize the comprehensiveness and rigor of the systematic review process. The literature search results are outlined in the PRISMA flow chart, as shown in Figure 1.
We established explicit inclusion and exclusion criteria for selecting studies. The literature was screened for eligibility based on these predefined criteria.
The inclusion criteria: (1) Original and full-length articles that reported epidemiological data on AKI in patients with ALF in the adult population; and (2) Articles published in the English language to ensure accurate interpretation and consistency during the review process.
The exclusion criteria: (1) Review articles; (2) Studies on AKI in acute-on-chronic liver failure; (3) Studies on AKI in chronic liver failure or cirrhosis; (4) Studies on chronic kidney disease in liver failure; (5) Paediatric studies; (6) Case reports; (7) Non-epidemiological investigations; and (8) Studies that failed to provide the required information. Any discordance in the selection of studies was solved using consensus, predefined inclusion, and exclusion criteria.
Two investigators (Ahmed A and Gautam M) independently screened the literature, resolving disagreements through discussion with other authors. Data was numerically extracted from each article that qualified the inclusion criteria and then presented in a Microsoft Excel spreadsheet (Windows 10 edition; Microsoft Corporation, Lisbon, Portugal). The extracted information was divided into different categories. The baseline information included the study title, study objective, design, setting, duration, and data regarding the inclusion and exclusion criteria, length of data collection, year of publication, and department in which the study was conducted. The demographic description of study participants was noted along with etiologies of ALF and AKI, stage of AKI, timeframe of AKI assessment, and risk factors of AKI in univariate and multivariate analysis. Outcomes were documented, focusing on the need for renal replacement therapy, mortality rates, liver transplantation, and transplant-free survival.
AKI was defined as a sudden decline in kidney function occurring within hours to days, typically within 48 hours. It is diagnosed using Risk, Injury, Failure, Loss of kidney function, and End-stage kidney disease (RIFLE) and Acute Kidney Injury Network (AKIN) criteria, but most recently, the Kidney Disease Improving Global Outcomes (KDIGO) guidelines are used as a standardized definition and staging system (Stages 1–3)[9]. Persistent AKI was defined as AKI that persisted for more than 48 hours based on the KDIGO criteria, as outlined in the consensus report of the Acute Disease Quality Initiative (ADQI) 16 Workgroup[10]. Transient AKI was defined as AKI lasting less than 48 hours[10].
ALF was defined as a rapid loss of liver function characterized by severe hepatocellular injury, leading to coagulopathy (international normalized ratio ≥ 1.5) and hepatic encephalopathy in patients without preexisting liver disease. ALF is categorized into hyperacute (< 7 days), acute (8 to 28 days), and subacute (29 days to 3 months) forms based on the time from jaundice to hepatic encephalopathy interval[11].
The methodological quality of eligible studies was assessed to evaluate the potential risk of bias in the retrieved infor
Categorical data were expressed as counts and percentages. Information on the etiologies of AKI in ALF and hepatic and renal outcomes was extracted from individual studies and summarized using measures such as the mean, median, and range. Statistical analyses were conducted using GraphPad Prism version 5.0 (Carlsbad, CA, United States) and MedCalc software.
Our search identified a total of 1134 records. After screening and excluding articles that did not meet the study inclusion criteria, the full texts of 10 articles were assessed for eligibility. One study was excluded due to a language barrier, as shown in Figure 1. Ultimately, nine studies were included in the final analysis.
The methodological quality assessment revealed deficiencies in several aspects described in Table 1[12-20]. Four studies were rated as moderate quality, each scoring 3, while five studies were classified as high quality, with scores of 4 (2 studies) or 5 (3 studies). The systematic review included research articles published between January 2009 and December 2024. Studies were predominantly conducted by research groups in the United States and the United Kingdom[13-15,16,18]. Notably, research in this field has shown significant growth in recent years, with three articles published between 2022 and 2024 by research groups from various countries[18-20]. A total of nine studies were published over the last 15 years.
| Ref. | Study design and setting are clearly defined | Eligibility and recruitment criteria of patients are clearly described | Operational definitions of ALF and AKI as per standard guidelines | Outcome variables described | Limitations of the study are clearly described | Score |
| Leithead et al[12] | Yes | No | Yes | No | Yes | 3 |
| O’Riordan et al[13] | Yes | Yes | No | Yes | Yes | 4 |
| Tujios et al[14] | Yes | Yes | Yes | Yes | Yes | 4 |
| Urrunaga et al[15] | Yes | No | No | Yes | Yes | 3 |
| Coelho et al[16] | Yes | Yes | Yes | Yes | Yes | 5 |
| Hadem et al[17] | No | Yes | No | Yes | Yes | 3 |
| Cardoso et al[18] | Yes | Yes | Yes | Yes | Yes | 5 |
| Imoto et al[19] | Yes | No | Yes | Yes | No | 3 |
| Fidalgo et al[20] | Yes | Yes | Yes | Yes | Yes | 5 |
Out of the nine studies, eight were retrospective and one was a prospective study, with a total of 6031 participants. These studies comprised six single-center studies, two registry-based studies, and one multicenter study, as given in Table 2. Eight studies investigated both acetaminophen-related and non-acetaminophen-related ALF, while one study focused exclusively on acetaminophen-related ALF.
| Ref. | Home country | Study design | Length of data collection (years) | Number of centers | AKI criteria | Time frame of AKI assessment | Inclusion criteria | Exclusion criteria | Study location |
| Leithead et al[12] | United Kingdom | R | 14 | 1 | RIFLE | Hospital stay | ALF | MV, already on RRT | LTU |
| O’Riordan et al[13] | United Kingdom | R | 8 | 1 | AKIN | Hospital stay | Patients admitted to LITU with paracetamol poisoning | Liver transplant patients | LTU |
| Tujios et al[14] | United States | R | 12 | 23 | AKIN | Hospital stay | INR > 1.5, HE < 26 weeks of illness without liver disease | NA | NA |
| Urrunaga et al[15] | United States | R | 10 | Registry | Others | During the waitlisting period (median duration 3 days) | > 18 years ALF with status 1 or 1A for ALF transplant | NA | NA |
| Coelho et al[16] | Portugal | R | 20 | 1 | KDIGO | NA | 18 years with ALF | CLD, CKD on RRT, RRT need before ICU | Gastroenterology, liver ICU |
| Hadem et al[17] | Germany | R | 18 | 1 | AKIN | Hospital stay | Hepatic encephalopathy with available renal function data | Overt sepsis | ICU |
| Cardoso et al[18] | United States | R | 18 | Registry | KDIGO | On day 1 of enrolment | > 18 years and ALF | < 3 days, follow up without renal function test on day 3, cirrhosis | NA |
| Imoto et al[19] | Japan | R | 12 | 1 | KDIGO | NA | ALF admitted to hospital | Malignancy, HIV | NA |
| Fidalgo et al[20] | Portugal | P | 7 | 1 | KDIGO | 7 days of ICU stay | ALF/ALI > 16 years | NA | ICU |
The sample size in these studies varied significantly, ranging from 51 to 2280 participants, as shown in Table 3. The mean age of the study populations was 41.86 ± 4.3 years with 67% female participants (range: 45.4% to 76.1%).
| Ref. | Total number of patients | Female gender | Age in years (mean) | Co-morbidities | Severity score | Number of patients developing AKI | AKI with ALF patients requiring RRT | Mortality (%) |
| Leithead et al[12] | 308 | 175 (56.8) | 39.7 | NA | NA | 208 (67.5) | 145 (70) | 112 (26.9) |
| O’Riordan et al[13] | 302 | 178 (59) | 37 | NA | SOFA, MELD, APACHE II, KCH | 239 (79.1) | 182 (76) | 74 (25) |
| Tujios et al[14] | 1604 | 1106 (68.9) | NA | DM (n = 84; 5.2%), HTN (n = 93; 5.7%) | NA | 1122 (69.9) | 491 (43.7) | 478 (29.8) |
| Urrunaga et al[15] | 2280 | 1596 (70) | NA | NA | MELD | 1271 (55.7) | NA | NA |
| Coelho et al[16] | 51 | 28 (54.9) | NA | DM (n = 1; 5.9%), HTN (n = 8; 15.6%), CAD (n = 6; 9.8%), others (n = 5; 9.8%) | SOFA | 34 (66.7) | 5 (14.7) | 22 (43.1) |
| Hadem et al[17] | 134 | 102 (76.1) | 42.5 | NA | SOFA, SAPS III, KCH, BiLE | 54 (40.3) | 52 (96.2) | 45 (33.6) |
| Cardoso et al[18] | 1071 | 731 (68.3) | 39 | NA | MELD | 744 (69.4) | 221 (29.7) | 324 (33.5) |
| Imoto et al[19] | 174 | 79 (45.4) | 49 | NA | MELD | 66 (37.9) | NA | 20 (11.5) |
| Fidalgo et al[20] | 104 | 44 (57.7) | 44 | NA | SOFA | 57 (54.80) | 32 (56.1) | 32 (30.8) |
Data on co-morbidities were inconsistently reported across the studies. Diabetes mellitus was noted in up to 5.2% of patients in larger cohorts, while hypertension reached a prevalence of 15.6%. Coronary artery disease was less frequently observed. Notably, some studies lacked detailed information on co-morbid conditions.
The studies employed a variety of scoring systems to assess illness severity. The Sequential Organ Failure Assessment score was the most frequently used score in multiple studies. Other commonly applied tools included the Model for End-Stage Liver Disease score and the Acute Physiology and Chronic Health Evaluation score. Additionally, specific criteria like the King’s College Hospital criteria, Simplified Acute Physiology Score, and the Bilirubin Lactate and Etiology score were used in selected studies.
The studies had substantial heterogeneity in participant characteristics, co-morbidities, and AKI incidence, as shown in Table 3. The variability in retrieved data highlights the diverse methodologies employed in ALF-related AKI research, underscoring the need for standardized criteria to enhance comparability and facilitate clinical interpretation.
Table 4 summarizes the etiologies of ALF reported in different studies on AKI in ALF. Except for the Japanese cohort (Imoto et al[19]), drug-induced ALF was the predominant cause in most series, accounting for approximately one-quarter to all cases. Among drug-related etiologies, paracetamol was the most frequent offending agent, responsible for 100% of cases in O’Riordan et al[13] and Tujios et al[14], and for more than 80% of cases in Leithead et al[12] and Urrunaga et al[15] (86.7%).
| Ref. | Drugs | Viral hepatitis | AIH | Budd-Chiari syndrome | Pregnancy | Wilson’s disease | Shock/ischemia | Other/undetermined |
| Leithead et al[12] | 242 (78.5). PCM (89.6), non PCM (10.3) | 46 (14.9) | 5 (1.6) | 5 (1.6) | 3 (0.9) | 2 (0.6) | 0 (0) | 5 (1.6) |
| O’Riordan et al[13] | 302 (100). PCM (100) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Tujios et al[14] | 738 (46). PCM (100) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 84 (5.2) | 783 (48.8) |
| Urrunaga et al[15] | 777 (34). PCM (86.7), non PCM (13.2) | 216 (9.4) | 118 (5.1) | 3 (0.1) | 19 (0.8) | 13 (0.6) | 0 (0) | 1134 (49.7) |
| Coelho et al[16] | 12 (23.5) | 15 (29.4) | 3 (5.8) | 0 (0) | 2 (3.9) | 0 (0) | 8 (15.6) | 11 (21.6) |
| Hadem et al[17] | 45 (33.8). PCM (28.9), non PCM (71.1) | 31 (23.3) | 2 (1.5) | 9 (6.7) | 0 (0) | 3 (2.2) | 1 (0.7) | 43 (32.3) |
| Cardoso et al[18] | 497 (46.4) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 573 (53.5) |
| Imoto et al[19] | 5 (2.8). PCM (0), non PCM (100) | 71 (40.8) | 17 (9.8) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 81 (46.5) |
| Fidalgo et al[20] | 36 (34.6). PCM (50), non PCM (50) | 11 (10.5) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 8 (7.6) | 49 (47.1) |
Viral hepatitis was the second most common cause overall, accounting for 40.8% of cases in the Japanese study done by Imoto et al[19], 29.4% in Coelho et al[16], and 9.4% in Urrunaga et al[15]. Autoimmune hepatitis was reported in ≤ 9.8% of cases, Budd–Chiari syndrome in ≤ 6.7%, and pregnancy-related ALF in ≤ 3.9%. Wilson’s disease was rare (≤ 2.2%). “Other” or undetermined causes accounted for a substantial proportion of cases in several studies, including 48.8% in Tujios et al[14], 53.5% in Cardoso et al[18], and 47.1% in Fidalgo et al[20].
A total of 6031 ALF patients were studied in nine studies, out of which 3795 (62.9%) patients developed AKI. The percentage of patients developing AKI varied widely, from 37.9% Imoto et al[19], to 79.1% O’Riordan et al[13]. Among the seven studies explicitly reporting AKI staging, the need for renal replacement therapy ranged from 14.7% to 96.2%. None of the studies described the cause or mechanism of AKI in these patients.
Most studies did not differentiate between transient and persistent AKI. However, two studies, Coelho et al[16], and Cardoso et al[18], reported this distinction. In the study conducted by Coelho et al[16], 29.4% patients had transient AKI while 70.5% had persistent AKI. In the study done by Cardoso et al[18], 9.6% had transient AKI, 80.7% had persistent AKI, and 9.5% experienced late-onset AKI (AKI after one day of admission).
The study done by Imoto et al[19], has used only coagulopathy in patients with deranged liver function as the diagnostic criteria of ALF, and they have further divided the patients into ALF with hepatic coma and ALF without hepatic coma. Development of AKI was more common in patients with hepatic coma as compared to patients without hepatic coma, 66.0% vs 38.5% respectively.
Only three studies have described the risk factors for developing AKI among ALF patients. Additionally, there is significant variability in the risk factors evaluated by these studies.
The study done by Leithead et al[12], in United Kingdom found systemic inflammatory response syndrome [odds ratio (OR) = 2.42 (95%CI: 1.17-5.00)], paracetamol poisoning [OR = 10.72 (95%CI: 4.24-27.09)], shock [OR = 7.01 (95%CI: 3.06-16.07)], King's College Hospital criteria [OR = 6.33 (95%CI: 2.65-15.13)] and infection [OR = 1.93 (95%CI: 0.93-4.02)] as independent risk factors for AKI in multivariate analysis.
Another study from United Kingdom done on paracetamol poisoning patients reported need for mechanical ventilation [OR = 4.78 (95%CI: 4.19-5.45)], vasopressor support [OR = 4.95 (95%CI: 1.02-24.81)], high admission phosphate [OR = 15.18 (95%CI: 3.46-66.02)], admission sodium [OR = 0.86 (95%CI: 0.77-0.96)], day-3 haematocrit [OR = 0.86 (95%CI: 0.77-0.96), and high day-3 Lactate [OR = 3.74 (95%CI: 1.77-7.92)] as independent risk factors for development of AKI in patients with paracetamol induced ALF[13].
Additionally, a study from Japan including 174 ALF patients reported TNF-α [OR = 1.647 (95%CI: 1.012–2.682)] as an independent risk factor linked with AKI (P = 0.0103)[19].
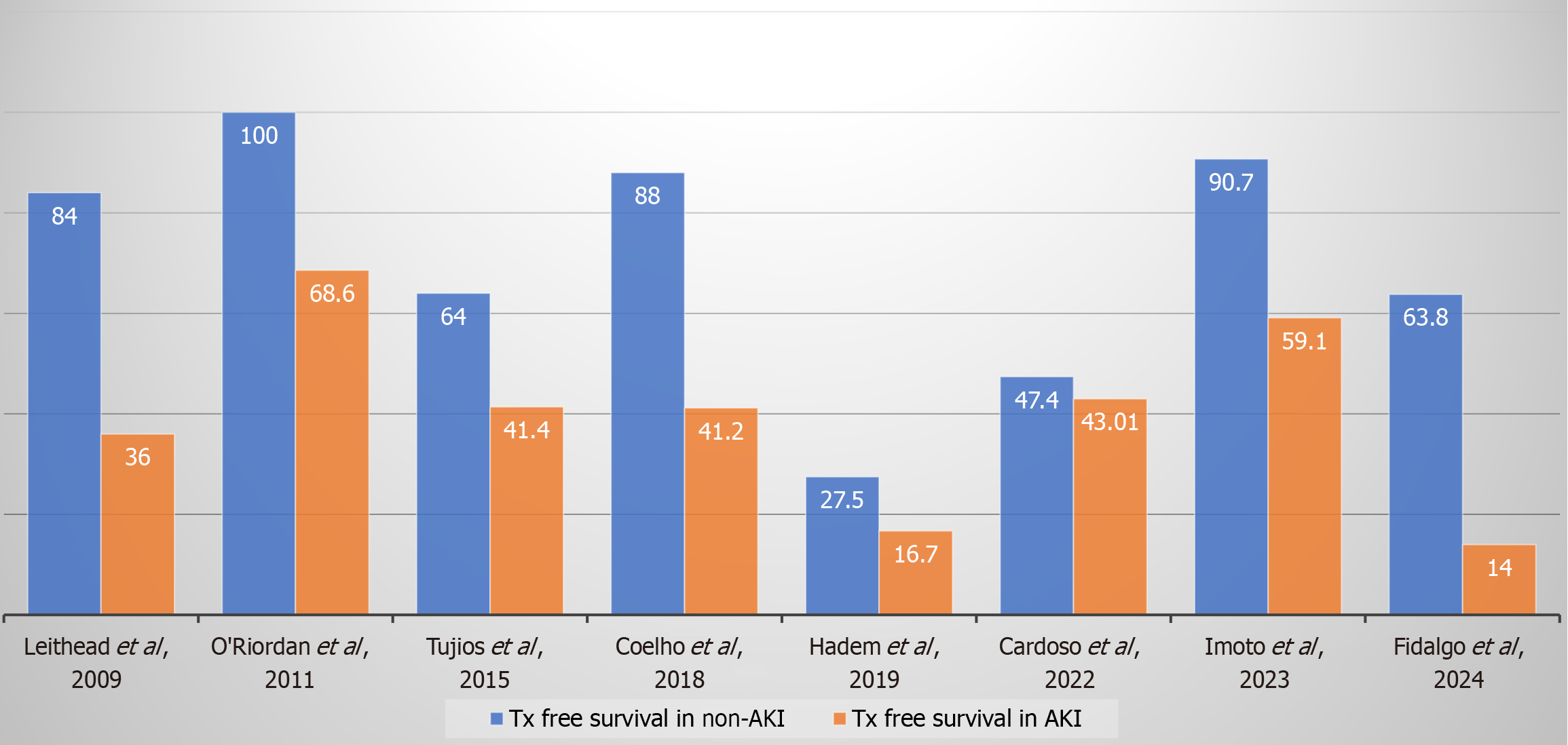
Across the studies included, the average mortality rate was 29.3%, ranging from 11.5% to 43.1%. Notably, transplant-free survival was consistently higher among patients without AKI compared to those with AKI, as shown in Figure 2.
Among the various risk factors independently associated with mortality in ALF patients, kidney injury emerged as a significant predictor in multiple studies. Leithead et al[12], reported that AKI was associated with increased mortality at hospital discharge, with a hazard ratio (HR) of 5.48 (95%CI: 2.0–13.64) in multivariate analysis. Similarly, Fidalgo et al[20], found AKI to be an independent risk factor for all-cause mortality within the first seven days of ICU stay, with an HR of 11.61 (95%CI: 1.49–90.34). Urrunaga et al[15], identified stage III AKI as a predictor of mortality within the first six weeks in patients waitlisted for liver transplantation, with an HR of 2.77 (95%CI: 2.071–3.705). Hadem et al[17], also reported stage III AKI to be significantly associated with 28-day mortality, with an HR of 3.48 (95%CI: 1.75–6.93).
Our study highlights a high rate of AKI (62.9%) among patients with ALF, a complication that often precedes a more prolonged and complex hospital course. This is the first systematic review to synthesise the available literature on AKI in ALF. The findings should be interpreted in light of substantial heterogeneity across studies, including differences in study design (retrospective vs prospective), patient populations (acetaminophen vs non-acetaminophen ALF), diagnostic criteria for AKI (RIFLE, AKIN, KDIGO), and ALF (O’Grady vs Japanese classification). Furthermore, the length of the data collection ranges from 7 to 20 years, with at least three studies having a data collection period of more than 15 years. During this duration, changes in risk factors, diagnostic criteria, supportive care, and transplant practices potentially limit direct comparability.
A unified framework for identifying risk factors is also lacking, representing a key research gap. Several studies had missing or incomplete critical data, and none reported the underlying etiology of AKI (e.g., hypoperfusion, nephrotoxicity). Moreover, the pathophysiological mechanisms were not explored in any study, underscoring the need for future research in this area. Knowing the mechanism is important to guide the preventive measures, therapeutic interventions, and improve clinical outcomes, for example, ischemic AKI has been shown to have a better prognosis than septic AKI[21].
In contrast to ALF, cirrhosis, and chronic liver disease-related AKI are well studied in the literature. Lekakis et al[22] conducted a meta-analysis of 5202232 cirrhotic patients in 73 studies. The authors reported that AKI was common in hospitalized cirrhotic patients (29%) as well as stable outpatients (28%)[22]. On admission, AKI was most frequent in patients with infection or sepsis/septic shock (47%), followed by those with hepatic encephalopathy (41%). Disease severity significantly influenced AKI risk, with ICU patients experiencing the highest incidence (61%). In 2023, the International Club of Ascites and the ADQI convened to refine diagnostic criteria, outline graded recommendations for evaluation and management, and define research priorities in cirrhosis-related AKI[23]. In contrast, there remains a notable lack of studies and clinical guidelines addressing AKI in the context of ALF within the medical literature.
Our study also underscores the critical lack of data regarding AKI in ALF from low- and middle-income countries (LMICs). The impact of this bias on pooled estimates could not be quantified due to the lack of comparable LMIC data; however, it is plausible that variations in disease etiology, healthcare resources, and management practices in LMICs may influence AKI incidence and outcomes. Future multicenter studies from diverse geographic and economic settings are warranted to address this gap.
Recognizing geographical variations in the etiology of ALF is essential, as these differences influence the underlying pathophysiology and the approach to AKI management[24,25]. In the Asia-Pacific region, viral hepatitis remains the most common cause of ALF, whereas paracetamol (acetaminophen) overdose is the leading cause in Europe and North Ame
The frequency and nature of AKI in ALF patients may vary depending on the underlying cause of liver failure and remain a subject insufficiently explored in the literature[28].
A notable example is obstetric ALF, a distinct and understudied clinical entity. In a study by Casey et al[29] conducted in the United States, the clinical features and outcomes of seventy pregnant patients who developed ALF were analyzed. The etiologies were categorized into pregnancy-associated acute liver disease (PAALD), acetaminophen poisoning (APAP), and other causes. Notably, admission serum creatinine levels were highest in the PAALD group (median 2.4 mg/dL, range 0.4–5.8 mg/dL), followed by the APAP group (median 1.4 mg/dL, range 0.3–5.4 mg/dL), and the 'other' group (median 0.7 mg/dL, range 0.3–2.7 mg/dL).
These findings suggest that PAALD-related ALF may be more frequently associated with kidney injury than other etiologies of ALF. One of the reasons for this could be a shared pathogenic pathway involving endothelial dysfunction and microcirculatory disturbances caused by elevated soluble fms-like tyrosine kinase-1 and soluble endoglin levels[30]. Recognizing this link is crucial for guiding timely pregnancy termination and initiating early, aggressive renal monitoring and nephroprotective strategies in pregnant patients with hepatic dysfunction.
As with many diseases, early identification of the pathological process enables timely intervention and improves outcomes. Biomarkers play a crucial role in early disease identification in its subclinical stage. In this regard, a note
The lack of data on specific causes and risk factors for AKI in this population constrains our findings. High heterogeneity, limited data on etiology-specific AKI, and the retrospective design of most studies were the key limitations of our study. The heterogeneity in reporting methods across various studies posed challenges for direct comparisons. Most available data came from Europe and the USA, leaving other geographical regions underrepresented. Given potential regional differences in the causes and risk factors for AKI in ALF, further research is needed to address these gaps.
In light of the high incidence and substantial clinical impact of AKI in ALF, there is an urgent need for standardized diagnostic criteria, uniform reporting practices, and targeted interventional studies to guide clinical decision-making.
AKI is a common and serious complication in patients with ALF, with a prevalence ranging from 25% to 70% across studies. The development of AKI in this population is associated with increased morbidity and mortality. Variability in definitions, diagnostic criteria, and reporting practices poses challenges in accurately assessing incidence and outcomes. This review also reveals a marked lack of data from LMICs, where differences in etiology, healthcare resources, and management practices may significantly influence AKI patterns. These gaps underscore the need for standardized AKI definitions in ALF, early recognition, and targeted management strategies. Future multicentre prospective studies should aim to define risk factors better, refine diagnostic tools, and develop effective preventive and therapeutic interventions.
| 1. | Hoste EAJ, Kellum JA, Selby NM, Zarbock A, Palevsky PM, Bagshaw SM, Goldstein SL, Cerdá J, Chawla LS. Global epidemiology and outcomes of acute kidney injury. Nat Rev Nephrol. 2018;14:607-625. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1144] [Cited by in RCA: 1002] [Article Influence: 125.3] [Reference Citation Analysis (1)] |
| 2. | Sharma B, Bhateja A, Sharma R, Chauhan A, Bodh V. Acute kidney injury in acute liver failure: A narrative review. Indian J Gastroenterol. 2024;43:377-386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (3)] |
| 3. | Nadim MK, Garcia-Tsao G. Acute Kidney Injury in Patients with Cirrhosis. N Engl J Med. 2023;388:733-745. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 150] [Cited by in RCA: 127] [Article Influence: 42.3] [Reference Citation Analysis (1)] |
| 4. | Attieh RM, Wadei HM. Acute Kidney Injury in Liver Cirrhosis. Diagnostics (Basel). 2023;13:2361. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 5. | Kanduri SR, Velez JCQ. Kidney Dysfunction in the Setting of Liver Failure: Core Curriculum 2024. Am J Kidney Dis. 2024;83:386-401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 17] [Article Influence: 8.5] [Reference Citation Analysis (1)] |
| 6. | Pose E, Piano S, Juanola A, Ginès P. Hepatorenal Syndrome in Cirrhosis. Gastroenterology. 2024;166:588-604.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 34] [Reference Citation Analysis (1)] |
| 7. | Cullaro G, Kanduri SR, Velez JCQ. Acute Kidney Injury in Patients with Liver Disease. Clin J Am Soc Nephrol. 2022;17:1674-1684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 47] [Article Influence: 11.8] [Reference Citation Analysis (4)] |
| 8. | White A, Schmidt K. Systematic literature reviews. Complement Ther Med. 2005;13:54-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 29] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 9. | Makris K, Spanou L. Acute Kidney Injury: Definition, Pathophysiology and Clinical Phenotypes. Clin Biochem Rev. 2016;37:85-98. [PubMed] |
| 10. | Chawla LS, Bellomo R, Bihorac A, Goldstein SL, Siew ED, Bagshaw SM, Bittleman D, Cruz D, Endre Z, Fitzgerald RL, Forni L, Kane-Gill SL, Hoste E, Koyner J, Liu KD, Macedo E, Mehta R, Murray P, Nadim M, Ostermann M, Palevsky PM, Pannu N, Rosner M, Wald R, Zarbock A, Ronco C, Kellum JA; Acute Disease Quality Initiative Workgroup 16. Acute kidney disease and renal recovery: consensus report of the Acute Disease Quality Initiative (ADQI) 16 Workgroup. Nat Rev Nephrol. 2017;13:241-257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1285] [Cited by in RCA: 1150] [Article Influence: 127.8] [Reference Citation Analysis (1)] |
| 11. | Maiwall R, Kulkarni AV, Arab JP, Piano S. Acute liver failure. Lancet. 2024;404:789-802. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 100] [Article Influence: 50.0] [Reference Citation Analysis (1)] |
| 12. | Leithead JA, Ferguson JW, Bates CM, Davidson JS, Lee A, Bathgate AJ, Hayes PC, Simpson KJ. The systemic inflammatory response syndrome is predictive of renal dysfunction in patients with non-paracetamol-induced acute liver failure. Gut. 2009;58:443-449. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 89] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 13. | O'Riordan A, Brummell Z, Sizer E, Auzinger G, Heaton N, O'Grady JG, Bernal W, Hendry BM, Wendon JA. Acute kidney injury in patients admitted to a liver intensive therapy unit with paracetamol-induced hepatotoxicity. Nephrol Dial Transplant. 2011;26:3501-3508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 50] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 14. | Tujios SR, Hynan LS, Vazquez MA, Larson AM, Seremba E, Sanders CM, Lee WM; Acute Liver Failure Study Group. Risk factors and outcomes of acute kidney injury in patients with acute liver failure. Clin Gastroenterol Hepatol. 2015;13:352-359. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 124] [Cited by in RCA: 102] [Article Influence: 9.3] [Reference Citation Analysis (1)] |
| 15. | Urrunaga NH, Magder LS, Weir MR, Rockey DC, Mindikoglu AL. Prevalence, Severity, and Impact of Renal Dysfunction in Acute Liver Failure on the US Liver Transplant Waiting List. Dig Dis Sci. 2016;61:309-316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 17] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 16. | Coelho S, Fonseca JN, Gameiro J, Jorge S, Velosa J, Lopes JA. Transient and persistent acute kidney injury in acute liver failure. J Nephrol. 2019;32:289-296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 15] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 17. | Hadem J, Kielstein JT, Manns MP, Kümpers P, Lukasz A. Outcomes of renal dysfunction in patients with acute liver failure. United European Gastroenterol J. 2019;7:388-396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 24] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 18. | Cardoso FS, Fidalgo P, Bagshaw SM, Gottfried M, Tujios S, Olson JC, Lee WM, Karvellas CJ; U. S. Acute Liver Failure Study Group. Persistent But Not Transient Acute Kidney Injury Was Associated With Lower Transplant-Free Survival in Patients With Acute Liver Failure: A Multicenter Cohort Study. Crit Care Med. 2022;50:1329-1338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (2)] |
| 19. | Imoto K, Tanaka M, Goya T, Azuma Y, Hioki T, Aoyagi T, Takahashi M, Kurokawa M, Kato M, Kohjima M, Ogawa Y. Acute kidney injury is an unfavorable prognostic factor in acute liver failure and is associated with tumor necrosis factor-alpha. Medicine (Baltimore). 2023;102:e35931. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 20. | Fidalgo P, Póvoa P, Germano N, Karvellas CJ, Cardoso FS. Long-Term Follow-Up of Kidney Function after Acute Liver Failure or Acute Liver Injury: A Cohort Study. GE Port J Gastroenterol. 2024;31:351-359. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 21. | Cruz MG, Dantas JG, Levi TM, Rocha Mde S, de Souza SP, Boa-Sorte N, de Moura CG, Cruz CM. Septic versus non-septic acute kidney injury in critically ill patients: characteristics and clinical outcomes. Rev Bras Ter Intensiva. 2014;26:384-391. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 29] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 22. | Lekakis V, Gkoufa A, Vlachogiannakos J, Papatheodoridis GV, Cholongitas E. Incidence and risk factors of acute kidney injury in cirrhosis: a systematic review and meta-analysis of 5,202,232 outpatients, inpatients, and ICU-admitted patients. Expert Rev Gastroenterol Hepatol. 2024;18:377-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (1)] |
| 23. | Nadim MK, Kellum JA, Forni L, Francoz C, Asrani SK, Ostermann M, Allegretti AS, Neyra JA, Olson JC, Piano S, VanWagner LB, Verna EC, Akcan-Arikan A, Angeli P, Belcher JM, Biggins SW, Deep A, Garcia-Tsao G, Genyk YS, Gines P, Kamath PS, Kane-Gill SL, Kaushik M, Lumlertgul N, Macedo E, Maiwall R, Marciano S, Pichler RH, Ronco C, Tandon P, Velez JQ, Mehta RL, Durand F. Acute kidney injury in patients with cirrhosis: Acute Disease Quality Initiative (ADQI) and International Club of Ascites (ICA) joint multidisciplinary consensus meeting. J Hepatol. 2024;81:163-183. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 93] [Cited by in RCA: 138] [Article Influence: 69.0] [Reference Citation Analysis (1)] |
| 24. | Biswas S, Shalimar. Definitions, etiopathogenesis and epidemiology of ALF. Best Pract Res Clin Gastroenterol. 2024;73:101959. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (2)] |
| 25. | Acharya SK. Acute Liver Failure: Indian Perspective. Clin Liver Dis (Hoboken). 2021;18:143-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 26. | Jindal A, Sarin SK. Epidemiology of liver failure in Asia-Pacific region. Liver Int. 2022;42:2093-2109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 27] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 27. | Devarbhavi H, Asrani SK, Arab JP, Nartey YA, Pose E, Kamath PS. Global burden of liver disease: 2023 update. J Hepatol. 2023;79:516-537. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1738] [Cited by in RCA: 1383] [Article Influence: 461.0] [Reference Citation Analysis (3)] |
| 28. | Moore JK, Love E, Craig DG, Hayes PC, Simpson KJ. Acute kidney injury in acute liver failure: a review. Expert Rev Gastroenterol Hepatol. 2013;7:701-712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 53] [Article Influence: 4.1] [Reference Citation Analysis (1)] |
| 29. | Casey LC, Fontana RJ, Aday A, Nelson DB, Rule JA, Gottfried M, Tran M, Lee WM; Acute Liver Failure Study Group. Acute Liver Failure (ALF) in Pregnancy: How Much Is Pregnancy Related? Hepatology. 2020;72:1366-1377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 44] [Article Influence: 7.3] [Reference Citation Analysis (4)] |
| 30. | Ives CW, Sinkey R, Rajapreyar I, Tita ATN, Oparil S. Preeclampsia-Pathophysiology and Clinical Presentations: JACC State-of-the-Art Review. J Am Coll Cardiol. 2020;76:1690-1702. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 553] [Article Influence: 110.6] [Reference Citation Analysis (0)] |
| 31. | Panackel C, Raja K, Fawas M, Jacob M. Prognostic models in acute liver failure-historic evolution and newer updates "prognostic models in acute liver failure". Best Pract Res Clin Gastroenterol. 2024;73:101957. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/