Published online Mar 9, 2026. doi: 10.5409/wjcp.v15.i1.113478
Revised: September 28, 2025
Accepted: December 15, 2025
Published online: March 9, 2026
Processing time: 192 Days and 0.6 Hours
Evidence supports that biliary atresia (BA) in Egyptian infants is an aflatoxin-induced cholangiopathy acquired perinatally in infants with glutathione S-transferase M1 deficiency: The Kotb disease BA variant. This inability to detoxify aflatoxins leads to progressive inflammatory adhesions and obliterative cholangiopathy early in life.
To investigate the cytochrome P450 1A2 (CYP1A2) detoxification enzyme levels in neonates with confirmed BA.
This prospective descriptive analytical study estimated CYP1A2 detoxification enzyme levels using enzyme-linked immunosorbent assay in 20 neonates with confirmed BA. The level CYP1A2 was also estimated in a control group of 20 infants with neonatal hepatitis (NH) and 20 age- and sex-matched healthy infants.
The mean age at enrollment in the study was 2.16 ± 0.58 months, 2.51 ± 1.05 months and 1.9 ± 0.81 months for the BA, NH and control group (P = 0.42). The mean ± SD of CYP1A2 among BA, NH and control group was 13.58 ± 9.73, 9.59 ± 9.95 and 31.68 ± 12.33 (P = 0.001). Within 3 months of follow up, of those with BA who underwent portoenterostomy 12 (60%) had successful outcome and cleared the jaundice, 5 (25%) had a stationary course and did not clear the jaundice while 3 (15%) died. The cholestasis resolved in 5 (25%) of the NH group, and improved in 15 (75%). CYP1A2 level did not correlate with age (r = 0.1559, P = 0.336), or total bilirubin level (r = 0.156, P = 0.336), alanine aminotransferase (r = -0.166, P = 0.305), aspartate aminotransferase (r = 0.091, P = 0.576), gamma glutamyl transferase (r = -0.0027, P = 0.99), alkaline phosphatase (r = 0.1059, P = 0.515), or outcome (r = 1.565, P = 0.23). A low CYP1A2 predicted a susceptibility to cholestasis by logistic regression (P = 0.000, 95% confidence interval: 0.8224-0.9327).
Significantly low serum CYP1A2 was frequent among infants with BA and NH compared to the control group. The low serum CYP1A2 compromises the detoxification ability of neonates with cholestasis and may alter the pharmacokinetics of commonly used drugs. Careful consideration to drug dosing and toxicity risk should be given to those neonates. Further research on drug pharmacokinetics and detoxification in neonates with cholestasis is recommended.
Core Tip: This study investigated cytochrome P450 1A2 (CYP1A2) detoxification enzyme levels in neonates with confirmed biliary atresia (BA), neonatal hepatitis and age and sex matched healthy infants. Significantly low serum CYP1A2 was frequent among BA and neonatal hepatitis patients compared to the control group. The synergism of null glutathione S-transferase and low serum CYP1A2 in BA; the Kotb disease maybe an additional factor that compromises the aflatoxins detoxification with subsequent acceleration of the progressive inflammatory adhesions and obliterative cholangiopathy early in life. Reduced CYP1A2 seems to be an inherent susceptibility to hepatitis in the neonatal period and may alter drugs’ pharmacokinetics. Careful consideration to drug dosing and toxicity risk should be given.
- Citation: Kotb MA, Wilson MM, Shafik AN, Ahmed NA, Fahim VF, Lotfi NA, Eissawy YM. Neonatal cholestasis is associated with cytochrome P450 1A2 detoxification defect: A single center pilot study. World J Clin Pediatr 2026; 15(1): 113478
- URL: https://www.wjgnet.com/2219-2808/full/v15/i1/113478.htm
- DOI: https://dx.doi.org/10.5409/wjcp.v15.i1.113478
Biliary atresia (BA) among Egyptian neonates proved to be aflatoxin induced cholangiopathy among neonates with glutathione S-transferase (GST) Mu 1 (M1) deficiency. This BA variant is named Kotb disease BA variant[1]. GST is a super family responsible for second tier detoxification of a group of chemicals that affect cellular replication and DNA fidelity. Cytosolic GSTM1 is a member of the GST super family[2,3]. Neonates with BA have typical homozygous null GSTM1, and all their mothers have heterozygous GSTM1 mutation[4]. The neonates with BA are exposed to the aflatoxin B1 and to a less extent B2 perinatally[5]. All through the pregnancy and prior to delivery, the placenta detoxifies the aflatoxins, but during labor the placenta stops this detoxification process and the aflatoxins reach the porta-hepatis of the newborn through the umbilical cord. When the newborn is deficient in GSTM1, they are unable to detoxify the accumulating aflatoxins. Aflatoxins cause massive hepatocellular and cholangiopathic inflammatory response, with neutrophil-elastase induced tissue damage, that ends in accelerated cirrhosis within 3 months of life, edema, inflammation, adhesions and fibrotic obstruction of major extrahepatic bile ducts and small caliber intrahepatic bile ducts[6].
Yet, GSTM1 is not the only enzyme responsible for detoxification of aflatoxins. Cytochrome P450 super family of enzymes - cytochrome P450 1A2 (CYP1A2) in particular - is the responsible first-tier detoxification enzyme of aflatoxin[7]. CYP1A2 is ontogenically programmed to function after the initial 8-12 weeks of life. Hence, GSTM1-deficient neonates are susceptible to toxic effect of aflatoxins during the early 2-3 months of life. Among those with BA and detoxification defects, cirrhosis is accelerated and is established by the earliest 3 months of life. Even after successful Kasasi portoenterostomy, children with BA fall prey to repeated attacks of cholangitis, which is related to more exposure to aflatoxin M1[5] and others drugs as ursodeoxycholic acid[8,9]. By the age of 2 years, almost 50% of children with BA will need liver transplantation[10]. We aimed to investigate the CYP1A2 detoxification enzyme levels in neonates with confirmed BA.
This prospective analytical descriptive study included 60 neonates and infants with cholestasis who were enrolled at Pediatric Hepatology and Pediatric Surgery Departments, Children Cairo University Hospitals. The study included 3 groups: (1) Group 1 included neonates and infants with confirmed BA (n = 20); (2) Group 2 included neonates and infants with cholestasis due to neonatal hepatitis (NH) (n = 20); and (3) Group 3 included healthy, sex - and age-matched control group (n = 20).
Inclusion criteria included infants of both sexes up to 3 months of age. Infants with other surgical causes of neonatal cholestasis were excluded. An informed consent was obtained from parents or relatives of all patients. The study complied with Helsinki Declaration of study that includes human subjects. The study was approved by Committee for Research and Higher Education of Faculty of Medicine, Cairo University, and Institution Ethical Committee of Faculty of Medicine, Cairo University, approval No. MS-487-2021.
History, clinical examination and confirmatory diagnostic tests of all patients were tabulated. Serum CYP1A2 level was assessed by enzyme-linked immunosorbent assay using human CYP1A2 enzyme-linked immunosorbent assay reagent kit (Catalogue No. 201 12-2282), SunRed, Shanghai, China following manufacturer instructions. Blood samples were allowed to clot for 10-20 minutes at room temperature and centrifuged at 2000-3000 rpm for 20 minutes and the supernatant was collected without sediment.
Data was fed into the computer and analyzed using IBM SPSS for Windows, Version 22.0. Armonk, NY, United States. Qualitative data was described using number and percent. Quantitative data was described using median and inter quartile range for nonparametric data, and mean, standard deviation for parametric data after testing normality using Kolmogrov-Smirnov test. Significance of the obtained results was judged at the (0.05) level. For qualitative data, we employed χ2 test for comparison of 2 or more groups. For quantitative data Student, t-test. Analysis of variance was employed to compare the means of the 3 groups according to age. Receiver operating characteristic curve analysis was employed to study diagnostic accuracy. Sensitivity and specificity were detected from the curve while positive predictive value, negative predictive value and accuracy were calculated through cross tabulation. Binary stepwise logistic regression analysis was employed to predict independent variables of binary outcome. Significant predictors in the Univariate analysis were entered into the regression model using forward Wald method/Enter. Adjusted odds ratios and their 95% confidence interval (CI) were calculated.
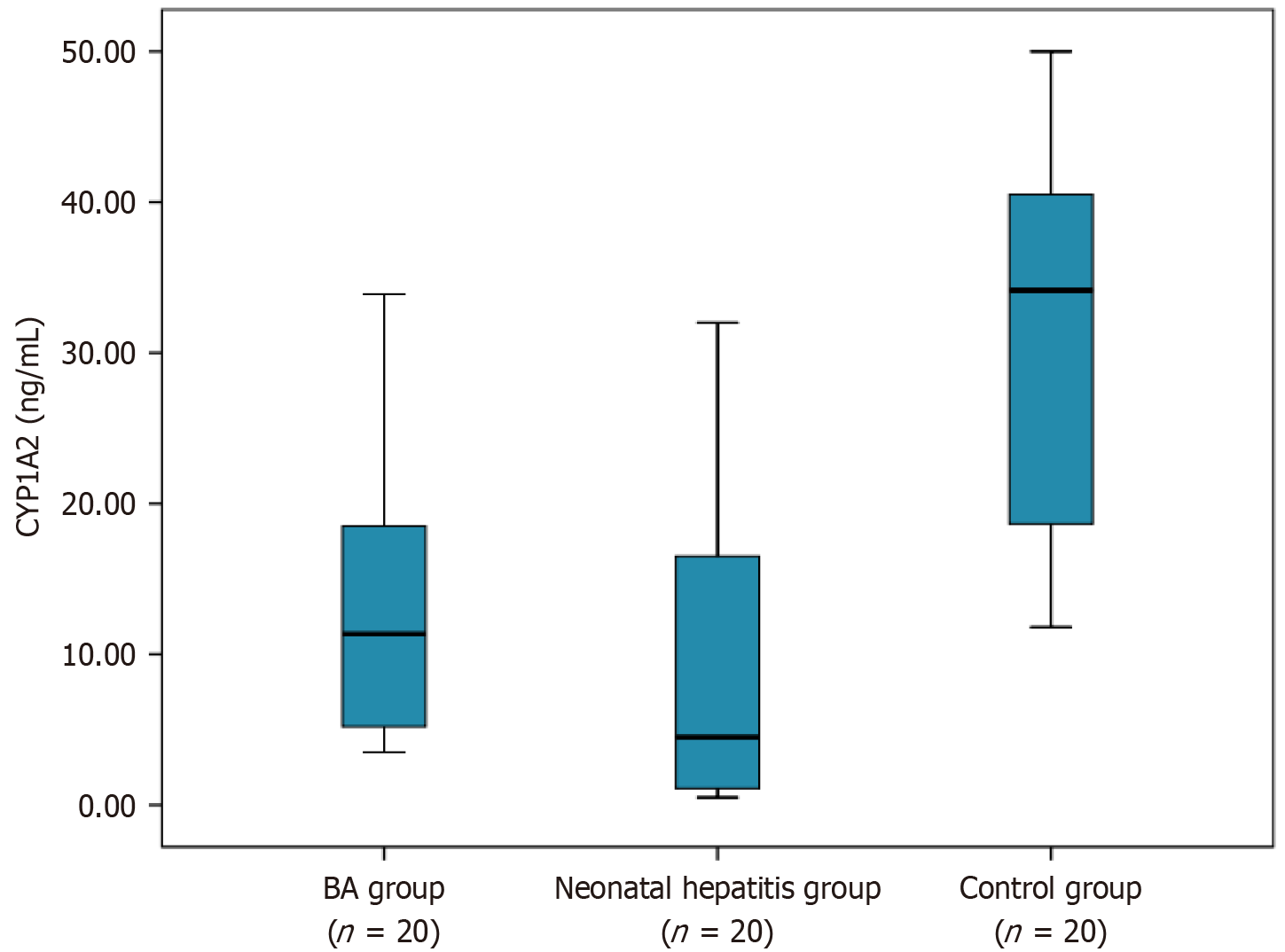
The mean age at enrollment in the study was 2.16 ± 0.58 months, 2.51 ± 1.05 months and 1.9 ± 0.81 months for the BA, NH and control group (P = 0.42). The BA group underwent timely portoenterostomy at a mean ± SD was 84 ± 11 days. 12 (60%) of this group had successful outcome with clearing of jaundice, 5 (25%) had a stationary course and did not clear the jaundice while 3 (15%) died within 18 months of the portoenterostomy. The NH group comprised 16 (80%) with idiopathic NH, 2 (10%) with galactosemia and 2 (10%) with tyrosinemia. They all cleared the jaundice. The mean serum ± SD of CYP1A2 among BA, NH and control group was 13.58 ± 9.73, 9.59 ± 9.95 and 31.68 ± 12.33 (P = 0.001). The demographic and clinical characteristics of the studied groups are shown in Table 1. The characteristics of both the BA and NH groups are illustrated in Table 2 and Figure 1. The mean ± SD serum CYP1A2 was significantly lower among BA and NH patients compared to the control group: 11.35 ± 9.72 (range = 5.2-19.25) ng/mL, 4.5 ± 9.956 (1-16.75) ng/mL, 34.15 ± 12.02 (18.48-40.9) ng/mL, respectively, (P = 0.001). The total number of those with normal level of CYP1A2 among those with BA and NH were 3 and 1 respectively (P = 0.001). Only 2 of those with normal CYP1A2 among those with BA resolved the cholestasis and the third died. CYP1A2 was not a predictor of outcome in the BA group according to the logistic regression analysis (P = 0.2828, 95%CI: 0.9505-1.1728), and was not a predictor of outcome in the NH group according to the logistic regression analysis (P = 0.2828, 95%CI: 0.3341-6.3919). CYP1A2 levels were lower among those with BA and NH during the first month of life, the second month of life and the third month (P = 0.006, P = 0.007 and P = 0.01 respectively) as shown in Table 3. None of the studied parameters correlated with the CYP1A2 level. CYP1A2 level did not correlate with age (r = 0.1559, P = 0.336), or total bilirubin level (r = 0.156, P = 0.336), alanine aminotransferase (r = -0.166, P = 0.305), aspartate aminotransferase (r = 0.091, P = 0.576), gamma glutamyl transferase (r = -0.0027, P = 0.99), alkaline phosphatase (r = 0.1059, P = 0.515), or outcome (r = 1.565, P = 0.23).
| Laboratory investigations | BA group (n = 20) | NH group (n = 20) | P value | |
| Age (months) | mean ± SD | 2.16 ± 0.58 | 2.51 ± 1.05 | 0.42 |
| Range | 1.2-3 | 0.83-6 | ||
| Sex | Male | 16 | 11 | 0.09 |
| Female | 4 | 9 | ||
| Age onset of disease (days) | Median (IQR) | 3 (1.25-3) | 3 (2.25-10) | 0.21 |
| Range | 1-60 | 1-165 | ||
| Age at diagnosis (months) | mean ± SD | 2 (1.5-2.5) | 2 (1.13) | 0.78 |
| Range | 1-3 | 0.75-6 | ||
| Weight percentile | Median (IQR) | 25 (10-25) | 10 (10-25) | 0.48 |
| Min-Max | 3-75 | 3-50 | ||
| Height percentile | Median (IQR) | 10 (3-25) | 25 (5-50) | 0.24 |
| Min-Max | 3-50 | 3-75 | ||
| TB (mg/dL) | mean ± SD | 10.96 ± 3.46 | 9.49 ± 5.86 | 0.34 |
| Range | 5.2-17.9 | 7-21.7 | ||
| DB (mg/dL) | mean ± SD | 7.69 ± 2.85 | 4.68 ± 3.42 | 0.004a |
| Range | 2.5-12.5 | 3-11.7 | ||
| ALT (IU/L) | mean ± SD | 213.9 ±133.5 | 225.6 ± 193.7 | 0.82 |
| Range | 9-563 | 29-790 | ||
| AST (IU/L) | mean ± SD | 238.66 ± 118.2 | 228.65 ± 263.8 | 0.88 |
| Range | 41-452 | 64-1320 | ||
| GGT (IU/L) | mean ± SD | 880.5 ± 863.7 | 225.7 ± 164 | 0.006a |
| Range | 35-1022 | 36-710 | ||
| ALP (IU/L) | mean ± SD | 478.2 ± 403 | 234.2 ± 215.3 | 0.025a |
| Range | 104-1980 | 18-967 | ||
| CYP1A2 (ng/mL) | mean ± SD | 13.58 ± 9.73 | 9.59 ± 9.95 | 0.1 |
| Range | 4.3-33.9 | 1.3-19 | ||
| Age groups | CYP1A2 (ng/mL) | BA group (n = 20) | NH group (n = 20) | Control group (n = 20) | P value |
| 1 month subgroup | n | n = 7 | n = 2 | n = 6 | 0.006a |
| mean ± SD | 13.58 ± 9.73 | 9.59 ± 9.95 | 31.68 ± 12.33 | ||
| Range | 4.3-33.9 | 1.3-19 | 18.3-50 | ||
| 2 months subgroup | n | n = 8 | n = 4 | n = 8 | 0.007a |
| mean ± SD | 12.84 ± 11.91 | 6.72 ± 6.4 | 34.29 ± 13.63 | ||
| Range | 3.6-33.5 | 0.5-15.7 | 11.8-47.7 | ||
| 3 months subgroup | n | n = 5 | n = 14 | n = 6 | 0.01 |
| mean ± SD | 12.84 ± 8.73 | 10.8 ± 11 | 27.1 ± 11.49 | ||
| Range | 3.5-24 | 0.5-26 | 12-39.7 |
The current study provides evidence that BA and NH are associated with low serum CYP1A2 levels. CYP1A2 is a member of the cytochrome p450 superfamily responsible for first tier detoxification. CYP1A2 becomes active after birth, and this induction of activity gradually increases over the first 8 months of life[11]. The CYP1A2 reduction was found to be lower than expected for age among those with BA and NH. The cause of lack of induction of CYP1A2 among those with BA and NH was not within the scope of this study. The lack of induction among those with NH seems to be more pronounced than among those with BA, but did not reach statistical significance (P = 0.1).
We did not study the gene coding for CYP1A2, hence we do not know if this lack of CYP1A2 induction is genetic or acquired. Yet, it is known that caffeine, aflatoxins and other chemicals induce CYP1A2. It is not clear why the aflatoxins that induce cholangiopathy in Kotb disease BA variant did not induce CYP1A2. It is not clear if NH is induced by toxins in those with CYP1A2 detoxification defect, yet evidence supports that BA is an aflatoxin-induced cholangiopathy in those with GSTM1 null deficient Egyptian neonates[12]. It seems that the detoxification genomics in BA and NH is not a mono-enzyme defect, but rather a pleuri-enzyme defect. This assumption is supported by our current work, and the evidence that those with BA have GSTM1, and GST Pi deficiencies as well[1,4].
The lack of significant correlation of levels of CYP1A2 and outcome among those with BA and NH suggests that the decreased CYP1A2 is not the only factor that controls the outcome in BA and NH. It suggests that the amount of toxin, the efficiency of the other detoxification enzymes and immune response all interact and contribute to defining the outcome.
The low CYP1A2 was predictive of cholestasis among our studied group (P = 0.0001), irrespective of the outcome, indicated that the low CYP1A2 is a susceptibility factor for the development of the cholestasis. We did not study any drug levels in our studied cohort, but this susceptibility seems to be a constant feature among neonates with cholestasis. This detoxification defect might hinder detoxification of natural components of metabolomics in the early life, and does not necessarily mean that these neonates are exposed to exogenous chemicals that are of maternal or otherwise exposure. Mice with low maternal CYP1A2 are known to have compromised learning[13], despite the lack of toxin exposure. Hence, this reduction of CYP1A2 below the already low threshold known for this early period of life challenges the mistakenly accepted myth that drug trials in adults maybe a justification for off-label use in pediatric age groups. Our work defies this concept and alerts to the importance of conducting pharmacokinetic drug trials in pediatric age group. We are not sure if this detoxification incapacitation contributes to the detrimental effect of ursodeoxycholic acid in neonatal cholestasis[9].
The poor induction of CYP1A2 renders neonates susceptible to Escherichia coli (E. coli) toxins, which may be a susceptibility factor to cholangitis known to complicate BA post-portoentersotomy. Yet, non-toxigenic E. coli is known to express human CYP1A2 that has a potential to be harnessed as a probiotic in future research[14].
The poor induction of CYP1A2 among those with BA and NH should alert the treating physicians that the pharmacokinetics of medications in BA and NH are different, and drug toxicities should be sought. Hence, the detoxification of clozapine, lidocaine, mirtazapine, naproxen, propranolol, triamterene, theophylline and others will be compromised among those with BA and NH[15].
The diminished CYP1A2 may reflect secondary dysfunction resulting from the liver injury among our studied cohorts with NH and BA. Yet, the levels of CYP1A2 were lower earlier in their lives, when the hepatocyte injury was less severe. Hence, the diminished CYP1A2 may be a cause and an effect of the hepatocyte injury.
It is interesting however, that higher CYP1A2 expression is associated with better prognosis and survival among patients with biliary tract cancer[16]. Accumulated nitrosamines and other chemicals are known to cause cholangiocarcinoma[17]. Hence, it seems that the efficient CYP1A2 detoxification is essential for detoxification, recovery and survival.
Limitations of the study include the small sample size of the studied cohorts, and the longitudinal study of CYP1A2 beyond the third month of life among our studied cohort. It is not clear if they have a delay of maturation of the CYP1A2, or the reduced levels of CYP1A2 are permanent among those with NH and BA. It is not clear if those with poorer outcomes had more sustained CYP1A2 reductions. Longer-duration studies are needed to address this gap of knowledge. Another limitation was the lack of assessment of aflatoxin half-life among those with BA to study the impact of reduced on CYP1A2 on the aflatoxin detoxification. The third limitation is the lack of CYP1A2 genotyping. It would differentiate between congenital dysregulation and secondary suppression due to hepatic pathology.
Significantly low serum CYP1A2 was frequent among patients with BA and NH compared to the control group. The low serum CYP1A2 compromises the detoxification ability of neonates with cholestasis. The synergism of null GST and low serum CYP1A2 in BA variant; the Kotb disease maybe an additional factor that compromises the aflatoxins detoxification, with subsequent acceleration of the progressive inflammatory adhesions and obliterative cholangiopathy early in life. Reduced CYP1A2 below the known accepted threshold during this neonatal period seems to be an inherent susceptibility to hepatitis in the neonatal period. Additionally, it may alter the pharmacokinetics of commonly used drugs. Careful consideration to drug dosing and toxicity risk should be given to neonates with cholestasis. Future research incorporating aflatoxin biomarkers assessment, CYP1A2 functional assays and genetic sequencing of the CYP1A2 coding region is recommended to elucidate the underlying mechanisms and differentiate between congenital dysregulation and se
| 1. | Kotb MA, Kotb A, Talaat S, Shehata SM, El Dessouki N, ElHaddad AA, El Tagy G, Esmat H, Shehata S, Hashim M, Kotb HA, Zekry H, Abd Elkader HM, Kaddah S, Abd El Baky HE, Lotfi N. Congenital aflatoxicosis, mal-detoxification genomics & ontogeny trigger immune-mediated Kotb disease biliary atresia variant: SANRA compliant review. Medicine (Baltimore). 2022;101:e30368. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 2. | Kotb M, Hamdy M, Eid K, Aziz M, Abd El Salam M, Abd El Baky H, Lotfi N, Salama N. Detoxification Genomics in Children with β-Thalassemia Major: Pilot Study of Glutathione S Transferase M1, Pi & Methyltetrahydrofolate Reductase Gene Polymorphisms Combinations in β- Thalassemia Major. Pediatr Sci J. 2021;1:98-106. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 3. | Vaish S, Gupta D, Mehrotra R, Mehrotra S, Basantani MK. Glutathione S-transferase: a versatile protein family. 3 Biotech. 2020;10:321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 119] [Article Influence: 19.8] [Reference Citation Analysis (0)] |
| 4. | Kotb MA. Glutathione S Transferase M1 Polymorphism in Extrahepatic Biliary Atresia. Med J Cairo Univ. 2015;83:109-112. |
| 5. | Kotb MA. Aflatoxins in Infants with Extrahepatic Biliary Atresia. Med J Cairo Univ. 2015;83:207-210. |
| 6. | Kotb MA. Neutrophil elastase mediated damage in infants with extrahepatic biliary atresia: A prospective cohort study. Med J Cairo Univ. 2014;82:233-237. |
| 7. | Bbosa GS, Kitya D, Lubega A, Ogwal-Okeng J, Anokbonggo WW, Kyegombe DB. Review of the Biological and Health Effects of Aflatoxins on Body Organs and Body Systems. In: Razzaghi-Abyaneh M, editor. Aflatoxins - Recent Advances and Future Prospects. Florida: InTech, 2013: 239-265. |
| 8. | Kotb MA. Review of historical cohort: ursodeoxycholic acid in extrahepatic biliary atresia. J Pediatr Surg. 2008;43:1321-1327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 25] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 9. | Kotb MA, Mosallam D, Basanti CWS, El Sorogy STM, Badr AM, Abd El Baky HEH, Draz IH. Ursodeoxycholic acid use is associated with significant risk of morbidity and mortality in infants with cholestasis: A strobe compliant study. Medicine (Baltimore). 2020;99:e18730. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 10. | Antala S, Taylor SA. Biliary Atresia in Children: Update on Disease Mechanism, Therapies, and Patient Outcomes. Clin Liver Dis. 2022;26:341-354. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 48] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 11. | Aldridge A, Aranda JV, Neims AH. Caffeine metabolism in the newborn. Clin Pharmacol Ther. 1979;25:447-453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 202] [Cited by in RCA: 179] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 12. | Li XC. Glutathione and Glutathione‐S‐Transferase in Detoxification Mechanisms. Gen Appl Syst Toxicol. 2011. [DOI] [Full Text] |
| 13. | Curran CP, Vorhees CV, Williams MT, Genter MB, Miller ML, Nebert DW. In utero and lactational exposure to a complex mixture of polychlorinated biphenyls: toxicity in pups dependent on the Cyp1a2 and Ahr genotypes. Toxicol Sci. 2011;119:189-208. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 17] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 14. | Kranendonk M, Mesquita P, Laires A, Vermeulen NP, Rueff J. Expression of human cytochrome P450 1A2 in Escherichia coli: a system for biotransformation and genotoxicity studies of chemical carcinogens. Mutagenesis. 1998;13:263-269. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 41] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 15. | Guo J, Zhu X, Badawy S, Ihsan A, Liu Z, Xie C, Wang X. Metabolism and Mechanism of Human Cytochrome P450 Enzyme 1A2. Curr Drug Metab. 2021;22:40-49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 36] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 16. | Yu J, Xia X, Dong Y, Gong Z, Li G, Chen GG, Lai PBS. CYP1A2 suppresses hepatocellular carcinoma through antagonizing HGF/MET signaling. Theranostics. 2021;11:2123-2136. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 17. | She T, Shah N, Jacob B, Starkman N, Lieman J, Kaur A, Shah N, Wilkenfeld M. Occupational and Environmental Cholangiocarcinoma-Related Toxic Exposures. GE Port J Gastroenterol. 2025;32:151-160. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (4)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/