Published online Mar 9, 2026. doi: 10.5409/wjcp.v15.i1.113430
Revised: November 7, 2025
Accepted: November 27, 2025
Published online: March 9, 2026
Processing time: 193 Days and 4.8 Hours
Recurrent wheezing following acute lower respiratory tract infection (ALRTI) in early childhood is a common clinical problem and may be an early indicator of chronic respiratory diseases. Early identification of associated risk factors is essential for early intervention and prevention.
To investigate the risk factors and timing of recurrent wheezing episodes following ALRTI in children.
A retrospective cohort study was conducted among pediatric patients ≤ 5 years old admitted with ALRTI and wheezing at Naresuan University Hospital between July 1, 2020 and June 30, 2023. Participants were followed for 12 months. Data from electronic and paper records were analyzed using STATA 18.0. Multi
Significant predictors of recurrent wheezing included age 12-24 months [odds ratio (OR): 2.38; 95%CI: 1.50-3.78, P < 0.001], prematurity (OR: 1.66; 95%CI: 1.07-2.58, P = 0.024), allergic rhinitis (OR: 1.50; 95%CI: 1.04-2.17, P = 0.031), urban residency (OR: 1.68; 95%CI: 1.19-2.38, P = 0.003), eosinophilia (absolute eosinophil count > 500 cells/μL) (OR: 3.29; 95%CI: 1.57-6.91, P = 0.002), and prior lower respiratory tract infection (OR: 1.82; 95%CI: 1.20-2.76, P = 0.005). The median time to recurrence was 100 days (interquartile range: 43-125), varying across clinical and environmental subgroups.
Children with certain demographic and clinical features have higher recurrent wheezing risk after ALRTI, highlighting the need for closer monitoring and early preventive care.
Core Tip: This study examined children hospitalized with acute lower respiratory tract infections and wheezing, identifying several independent risk factors for recurrent wheezing, including age 12-24 months, prematurity, allergic rhinitis, urban residence, eosinophilia, and prior lower respiratory tract infection. Notably, the median time to recurrence was 100 days, providing practical insight for optimizing follow-up timing and early intervention strategies.
- Citation: Kiatvitchukul T, Dokkham P, Jeephet K, Srisingh K. Understanding recurrent wheezing: A parent’s guide. World J Clin Pediatr 2026; 15(1): 113430
- URL: https://www.wjgnet.com/2219-2808/full/v15/i1/113430.htm
- DOI: https://dx.doi.org/10.5409/wjcp.v15.i1.113430
Acute lower respiratory tract infections (ALRTIs) are among the most common causes of hospitalization in young children and remain a major contributor to pediatric morbidity and mortality worldwide. While most children recover without long-term complications, a substantial proportion experience recurrent wheezing after an initial episode of ALRTI, a condition with important clinical and public health implications.
Recurrent wheezing is more than a transient respiratory symptom; it is often considered an early clinical manifestation of chronic respiratory disorders, particularly asthma[1]. Repeated episodes of wheezing can negatively impact a child's quality of life by limiting physical activity, increasing school absenteeism, and requiring frequent medication use and healthcare visits. Moreover, recurrent wheezing imposes a substantial burden on families and healthcare systems due to repeated consultations, hospital readmissions, and associated medical costs.
Identifying children at risk of developing recurrent wheezing following ALRTI is crucial for early intervention and long-term disease prevention. Previous studies have reported a variety of potential risk factors, including young age at first infection, specific viral pathogens [e.g., respiratory syncytial virus (RSV), rhinovirus], family history of atopy or asthma, environmental exposures (e.g., tobacco smoke, air pollution), and socioeconomic status[2-9]. However, current evidence on the precise timing between an ALRTI with wheezing and the onset of recurrent wheezing remains limited and inconsistent. This knowledge gap underscores the need for further investigation across diverse populations and settings.
This study aims to evaluate the clinical, demographic, and environmental risk factors associated with recurrent wheezing episodes, as well as the timing of these recurrences, in children hospitalized with ALRTI. A better un
This retrospective cohort study was conducted at Naresuan University Hospital, a 484-bed tertiary-care teaching hospital in the lower northern region of Thailand, between July 1, 2020 and June 31, 2023. The pediatric ward has 85 beds and 8 single rooms for patient isolation. All admissions are documented in the hospital’s database, with clinical diagnoses coded according to the International Classification of Diseases, 10th Revision.
Children aged ≤ 5 years who were admitted to the pediatric ward with a primary diagnosis of ALRTI with wheezing were eligible for inclusion. ALRTI was defined by clinical presentation, including cough, difficulty breathing, and auscultatory wheezing, consistent with pneumonia, bronchiolitis, or bronchitis, with or without radiologic confirmation.
Patients were excluded from this study if they had underlying asthma or pre-existing chronic lung diseases (e.g., cystic fibrosis, bronchopulmonary dysplasia), congenital airway or lung abnormalities, known congenital heart disease or congestive heart failure, a history of foreign body aspiration or respiratory system surgery, or were lost to follow-up within 12 months after hospital discharge.
The primary exposure variables included demographic factors (age and sex) and potential risk factors. Perinatal factors included prematurity, route of delivery, and birth weight. Environmental factors included residential location (urban/rural), exposure to smoking during pregnancy or postnatally, presence of pets, and daycare attendance. Personal and family history included underlying diseases (e.g., allergic rhinitis, atopic dermatitis, hematologic disorders), history of food or drug allergies, and parental asthma. Prior infections assessed included RSV, upper respiratory tract infection, and lower respiratory tract infection (LRTI). Laboratory parameters included complete blood count (CBC) indices.
The primary outcome was the occurrence of recurrent wheezing episodes, defined as ≥ 2 physician-diagnosed wheezing episodes within 12 months following hospital discharge, as documented in follow-up records.
Data were extracted from electronic inpatient and outpatient medical records using a structured data abstraction form. Collected variables collected included demographic characteristics, perinatal factors, environmental exposures, personal and family medical history, prior infections, laboratory parameters from CBC, and follow-up information such as outpatient visits, readmissions, and physician-documented wheezing episodes within 12 months.
All data were anonymized prior to analysis. To ensure accuracy, 10% of charts were randomly reviewed by a second investigator. For cases with incomplete records, additional information was obtained through follow-up phone interviews with parents or guardians.
The sample size was calculated using pilot data from 20 pediatric patients with a history of ALRTI at the Department of Pediatrics, Naresuan University Hospital. Of these, 10 patients experienced recurrent wheezing and 10 did not. Among the variables analyzed, residential location was identified as the most influential factor for determining the largest sample size and was therefore selected for estimation.
Based on the pilot data (30% vs 10% recurrent wheezing among those with and without the risk factor), a two-sample comparison of proportions with a type I error (α) of 0.05, a statistical power of 80%, and a group ratio of 1:3 (P1 = 0.30, P2 = 0.10) yielded a required sample size of 108 patients (27 with recurrent wheezing and 81 without).
Categorical data were summarized as frequencies and percentages, and analyzed using the exact probability test. Normally distributed continuous data were described with the mean and standard deviation, and compared using the independent t-test. Non-normally distributed continuous data were presented as the median and interquartile range (IQR).
Statistical uncertainty was expressed as two-sided 95% confidence intervals (95%CIs). A P value < 0.05 was considered statistically significant. Multivariable binary logistic regression was used to identify independent predictors of recurrent wheezing, including potential risk factors identified in the literature (e.g., prematurity, family history of atopic disease, prior respiratory illness, exposure to individuals with respiratory tract infections, and underlying disease). Kaplan-Meier survival analysis was conducted to compare the time to recurrence between the recurrence and non-recurrence groups, and survival curves were generated accordingly. The log-rank test was used to assess the statistical differences between the curves.
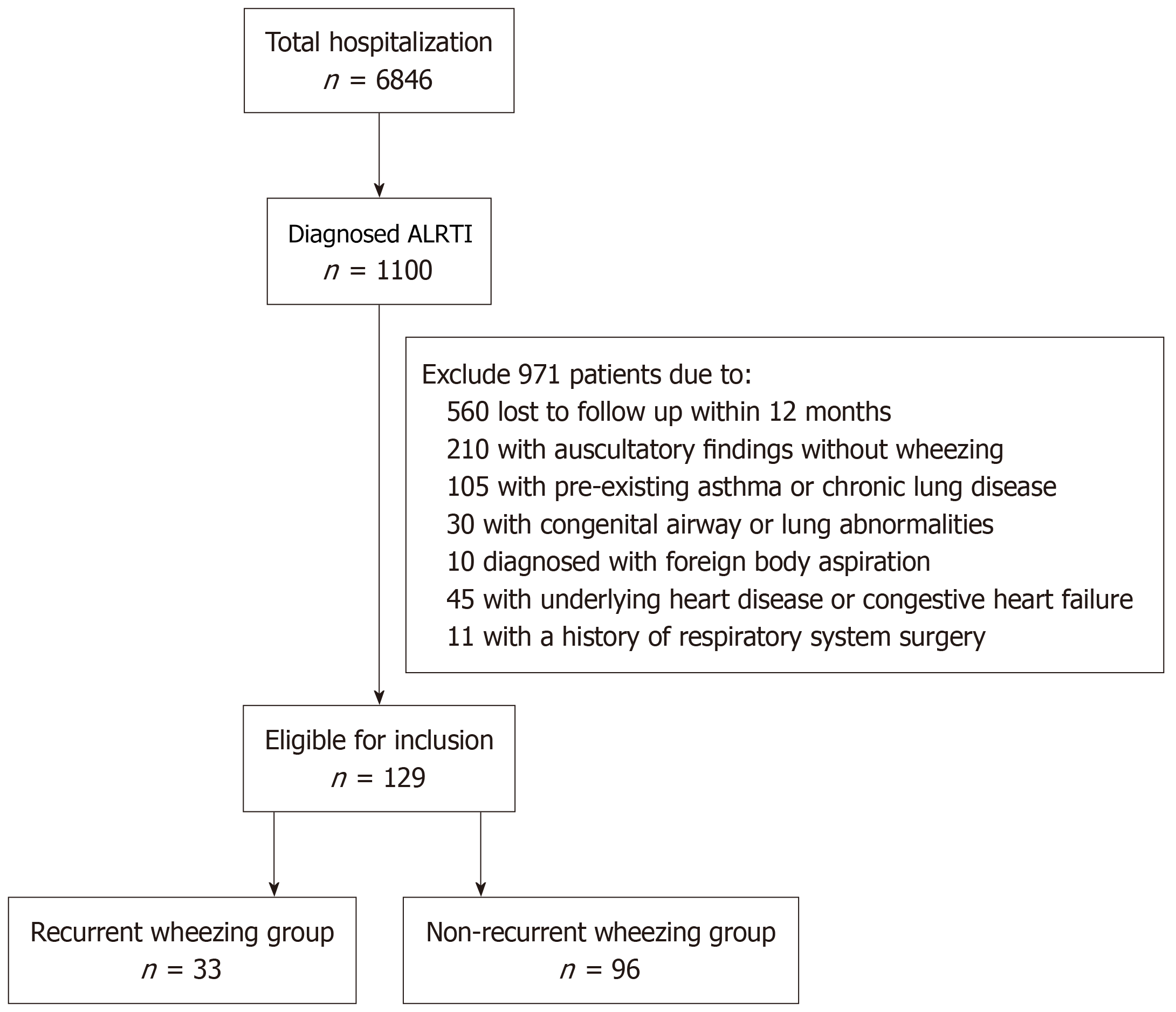
During the study period, a total of 6846 pediatric patients were hospitalized, of whom 1100 were diagnosed with ALRTI. Following a review, 971 patients were excluded from the study for the following reasons: 560 were lost to follow-up within 12 months after hospital discharge, 210 had medical records showing auscultatory findings without wheezing, 105 had a pre-existing diagnosis of asthma or chronic lung disease, 30 had congenital airway or lung abnormalities, 45 had congenital heart disease or congestive heart failure, 10 were diagnosed with foreign body aspiration, and 11 had a history of respiratory surgery. The final study cohort comprised 129 eligible patients, who were categorized into two groups: 33 patients who experienced recurrent wheezing episodes within 12 months (recurrent group) and 96 who did not (non-recurrent group) (Figure 1). The groups were comparable in terms of gender, with 13 males (39.39%) in the recurrent group and 46 (47.92%) in the non-recurrent group (P = 0.425). The mean age was also comparable between the recurrent and non-recurrent groups (27.68 ± 14.95 months vs 30.61 ± 14.52 months; P = 0.330).
Comparison of clinical characteristics revealed several statistically significant differences between the groups (Table 1). The recurrent group exhibited a higher prevalence of allergic rhinitis [7 (21.21%) vs 4 (4.17%), P = 0.006], parental asthma [4 (16.00%) vs 0 (0.00%), P = 0.002], underlying disease [14 (42.42%) vs 23 (23.96%), P = 0.048], eosinophilia [absolute eosinophil count (AEC) > 500 cells/μL] [9 (27.27%) vs 3 (3.12%), P < 0.001] and post-lower respiratory tract infection complications [21 (63.64%) vs 20 (20.83%), P < 0.001]. This group also had a higher mean white blood cell count (14420 ± 5269 vs 11058 ± 4466 cells/μL; P < 0.001). Conversely, post-RSV infection was less frequent in the recurrent group [12 (38.71%) vs 58 (65.91%), P = 0.011].
| Characteristics | Recurrent wheezing (n = 33) | Non-recurrent wheezing (n = 96) | P value |
| Male | 13 (39.39) | 46 (47.92) | 0.425 |
| Age (months) | 27.68 ± 14.95 | 30.61 ± 14.52 | 0.330 |
| Age category (months) | |||
| > 36-60 | 10 (30.30) | 26 (27.08) | 0.477 |
| > 24-36 | 13 (39.39) | 30 (31.25) | |
| > 12-24 | 8 (24.24) | 24 (25.00) | |
| ≤ 12 | 2 (6.06) | 16 (16.67) | |
| Preterm birth | 2 (7.69) | 8 (9.20) | 0.585 |
| Low birth weight | 2 (7.69) | 9 (10.47) | 0.425 |
| Cesarean birth | 15 (60.00) | 50 (60.98) | 0.555 |
| Allergic rhinitis | 7 (21.21) | 4 (4.17) | 0.006 |
| Atopic dermatitis | 4 (12.12) | 12 (12.50) | 0.612 |
| History of food/drug allergy | 2 (6.06) | 3 (3.12) | 0.602 |
| Urban residency | 23 (69.70) | 73 (76.04) | 0.493 |
| Underlying disease | 14 (42.42) | 23 (23.96) | 0.048 |
| Hematologic disease | 4 (12.12) | 4 (4.17) | 0.202 |
| Maternal smoking during pregnancy | 2 (9.09) | 11 (11.58) | 0.542 |
| Secondhand smoke | 52 (22.73) | 23 (4.21) | 0.560 |
| Parental asthma | 4 (16.00) | 0 (0.00) | 0.002 |
| Daycare attendance | 17 (70.83) | 53 (55.79) | 0.247 |
| Hct | 33 ± 35.55 | 96 ± 36.43 | 0.129 |
| WBC | 14420.30 ± 5268.52 | 11057.81 ± 4466.33 | < 0.001 |
| Neutrophil (%) | 50.37 ± 20.89 | 50.37 ± 18.86 | 0.112 |
| ANC | 8965.52 ± 5118.23 | 5843.84 ± 3702.22 | |
| Lymphocyte (%) | 32.72 ± 18.43 | 39.76 ± 18.42 | 0.061 |
| ALC | 4240.18 | 4535.91 | 0.660 |
| Eosinophil (%) | 2.20 ± 1.83 | 1.03 ± 1.34 | < 0.001 |
| AEC | 322.85 ± 318.11 | 113.80 ± 151.50 | < 0.001 |
| Eosinophilia (AEC > 500) | 9 (27.27) | 3 (3.12) | < 0.001 |
| Platelet count | 361393.90 ± 106626.40 | 339863.20 ± 99551.72 | 0.295 |
| Anemia | 7 (21.21) | 14 (14.58) | 0.416 |
| Pet ownership | 0.250 | ||
| None | 17 (80.95) | 83 (89.25) | |
| Dog | 1 (4.76) | 6 (6.45) | |
| Cat | 3 (14.29) | 3 (3.23) | |
| Both dog and cat | 0 (0.00) | 1 (1.08) | |
| Post-RSV infection | 12 (38.71) | 58 (65.91) | 0.011 |
| Influenza A | 0 (0.00) | 2 (2.33) | 0.539 |
| Post-LRTI | 21 (63.64) | 20 (20.83) | < 0.001 |
| Post-URTI | 19 (57.58) | 47 (48.96) | 0.425 |
No statistically significant differences were observed between the groups for other baseline characteristics and clinical features, including preterm birth [2 (7.69%) vs 8 (9.20%), P = 0.585], low birth weight [2 (7.69%) vs 9 (10.47%), P = 0.425], cesarean delivery [15 (60.00%) vs 50 (60.98%), P = 0.555], atopic dermatitis [4 (12.12%) vs 12 (12.50%), P = 0.612], urban residency [23 (69.70%) vs 73 (76.04%), P = 0.493], maternal smoking during pregnancy [2 (9.09%) vs 11 (11.58%), P = 0.542], history of food or drug allergy [2 (6.06%) vs 3 (3.12%), P = 0.602], exposure to secondhand smoke [52 (22.73%) vs 23 (4.21%), P = 0.560], daycare attendance [17(70.83%) vs 53 (55.79%), P = 0.247], anemia [7 (21.21%) vs 14 (14.58%), P = 0.416], and post-upper respiratory tract infection [19 (57.58%) vs 47 (48.96%), P = 0.425].
Univariable analysis identified allergic rhinitis [odds ratio (OR): 6.19; 95%CI: 1.68-22.80; P = 0.006], eosinophilia (AEC > 500 cells/μL) (OR: 1.00; 95%CI: 1.00-1.01, P < 0.001), post-RSV infection (OR: 0.33; 95%CI: 0.14-0.76; P = 0.010) and post-LRTI (OR: 6.65; 95%CI: 2.80-15.77.06; P < 0.001) as significant risk factors for recurrent wheezing (Table 2).
| Risk factors | OR | 95%CI | P value |
| Male | 1.42 | 0.63-3.17 | 0.398 |
| Age (months) | |||
| > 36-60 | Reference | Reference | |
| > 24-36 | 1.23 | 0.43-2.99 | 0.811 |
| > 12-24 | 0.87 | 0.29-2.56 | 0.796 |
| ≤ 12 | 0.33 | 0.06-1.78 | 0.179 |
| Preterm birth | 1.22 | 0.24-6.11 | 0.813 |
| Low birth weight | 1.40 | 0.28-6.94 | 0.678 |
| Cesarean birth | 0.96 | 0.38-2.40 | 0.930 |
| Allergic rhinitis | 6.19 | 1.68-22.80 | 0.006 |
| Atopic dermatitis | 0.97 | 0.29-3.23 | 0.955 |
| History of food/drug allergy | 2.00 | 0.32-12.52 | 0.459 |
| Urban residency | 1.38 | 0.57-3.32 | 0.472 |
| Underlying disease | 2.34 | 1.02-5.39 | 0.046 |
| Hematologic disease | 3.17 | 0.75-13.48 | 0.118 |
| Maternal smoking during pregnancy | 0.76 | 0.16-3.72 | 0.739 |
| Secondhand smoke | 0.92 | 0.31-2.77 | 0.883 |
| Daycare attendance | 1.92 | 0.73-5.07 | 0.185 |
| Anemia | 1.58 | 0.58-4.32 | 0.376 |
| Eosinophilia (AEC > 500 cells/μL) | 1.00 | 1.00-1.01 | < 0.001 |
| Pet | 1.57 | 0.77-3.20 | 0.217 |
| Post-RSV infection | 0.33 | 0.14-0.76 | 0.010 |
| Post-LRTI | 6.65 | 2.80-15.77 | < 0.001 |
| Post-URTI | 1.41 | 0.64-3.14 | 0.394 |
Subsequent multivariable logistic regression analysis confirmed several potential risk factors for recurrent wheezing, including age between 12 months to 24 months (OR: 2.38; 95%CI: 1.50-3.78; P < 0.001), preterm birth (OR: 1.66; 95%CI: 1.07-2.58; P = 0.024), allergic rhinitis (OR: 1.50; 95%CI: 1.04-2.17; P = 0.031), urban residency (OR: 1.68; 95%CI: 1.19-2.38; P = 0.003), eosinophilia (AEC > 500 cells/μL) (OR: 3.29; 95%CI: 1.57-6.91; P = 0.002) and post-LRTI (OR: 1.82; 95%CI: 1.20-2.76; P = 0.005). Conversely, this analysis also identified atopic dermatitis (OR: 0.51; 95%CI: 0.26-0.98; P = 0.044) and secondhand smoke (OR: 0.50; 95%CI: 0.26-0.98; P = 0.045) as protective factors against recurrent wheezing (Table 3).
| Potential risk factors | Adjust odds ratio | 95%CI | P value |
| Male | 1.13 | 0.80-1.60 | 0.482 |
| Age (months) | |||
| > 36-60 | Reference | Reference | |
| > 24-36 | 1.12 | 0.71-1.75 | 0.625 |
| > 12-24 | 2.38 | 1.50-3.78 | < 0.001 |
| ≤ 12 | NA | NA | NA |
| Preterm birth | 1.66 | 1.07-2.58 | 0.024 |
| Allergic rhinitis | 1.50 | 1.04-2.17 | 0.031 |
| Atopic dermatitis | 0.51 | 0.26-0.98 | 0.044 |
| Urban residency | 1.68 | 1.19-2.38 | 0.003 |
| Hematologic disease | 2.43 | 0.78-7.57 | 0.126 |
| Maternal smoking during pregnancy | 1.60 | 0.71-3.60 | 0.254 |
| Secondhand smoke | 0.50 | 0.26-0.98 | 0.045 |
| Daycare attendance | 1.19 | 0.85-1.66 | 0.315 |
| Eosinophilia (AEC > 500 cells/μL) | 3.29 | 1.57-6.91 | 0.002 |
| Pet | 1.04 | 0.57-1.89 | 0.907 |
| Post-RSV infection | 1.22 | 0.83-1.81 | 0.312 |
| Post-LRTI | 1.82 | 1.20-2.76 | 0.005 |
| Post-URTI | 0.81 | 0.57-1.14 | 0.232 |
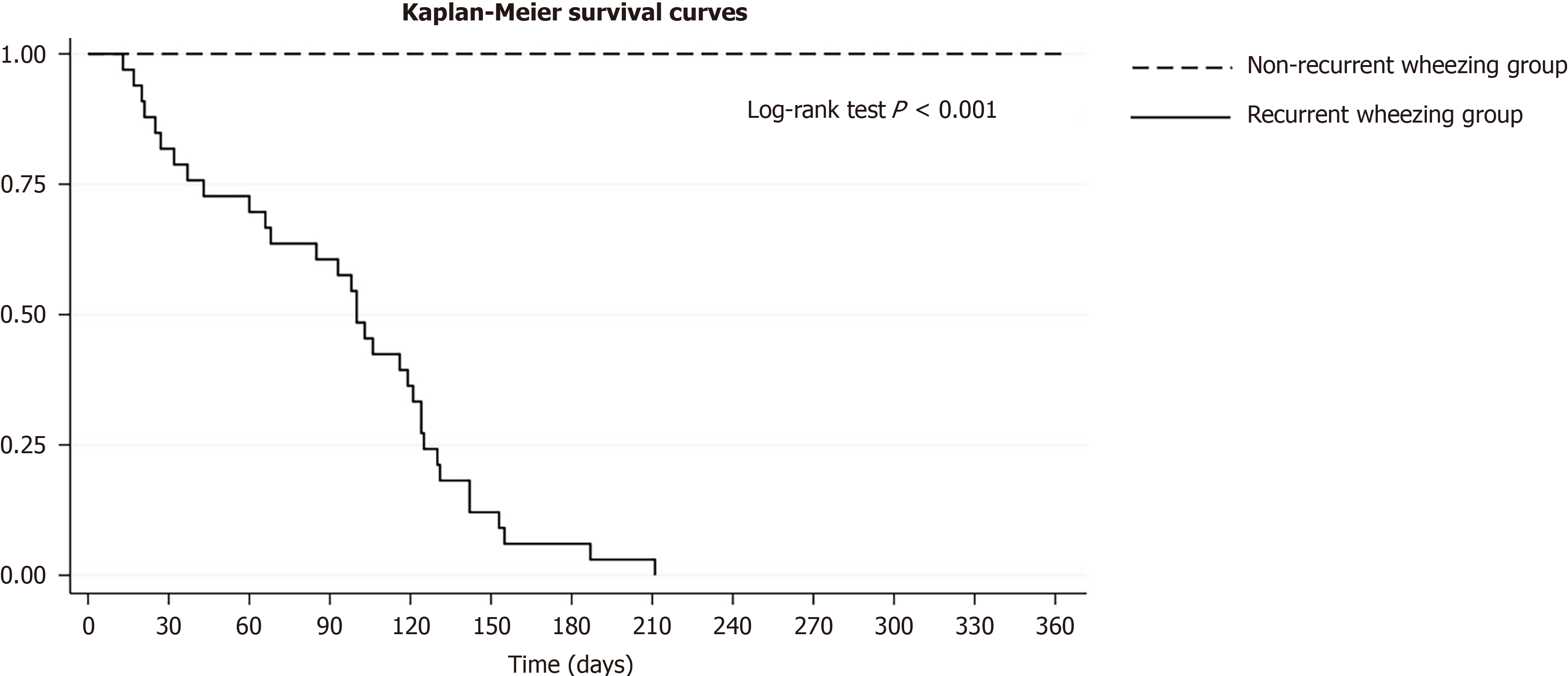
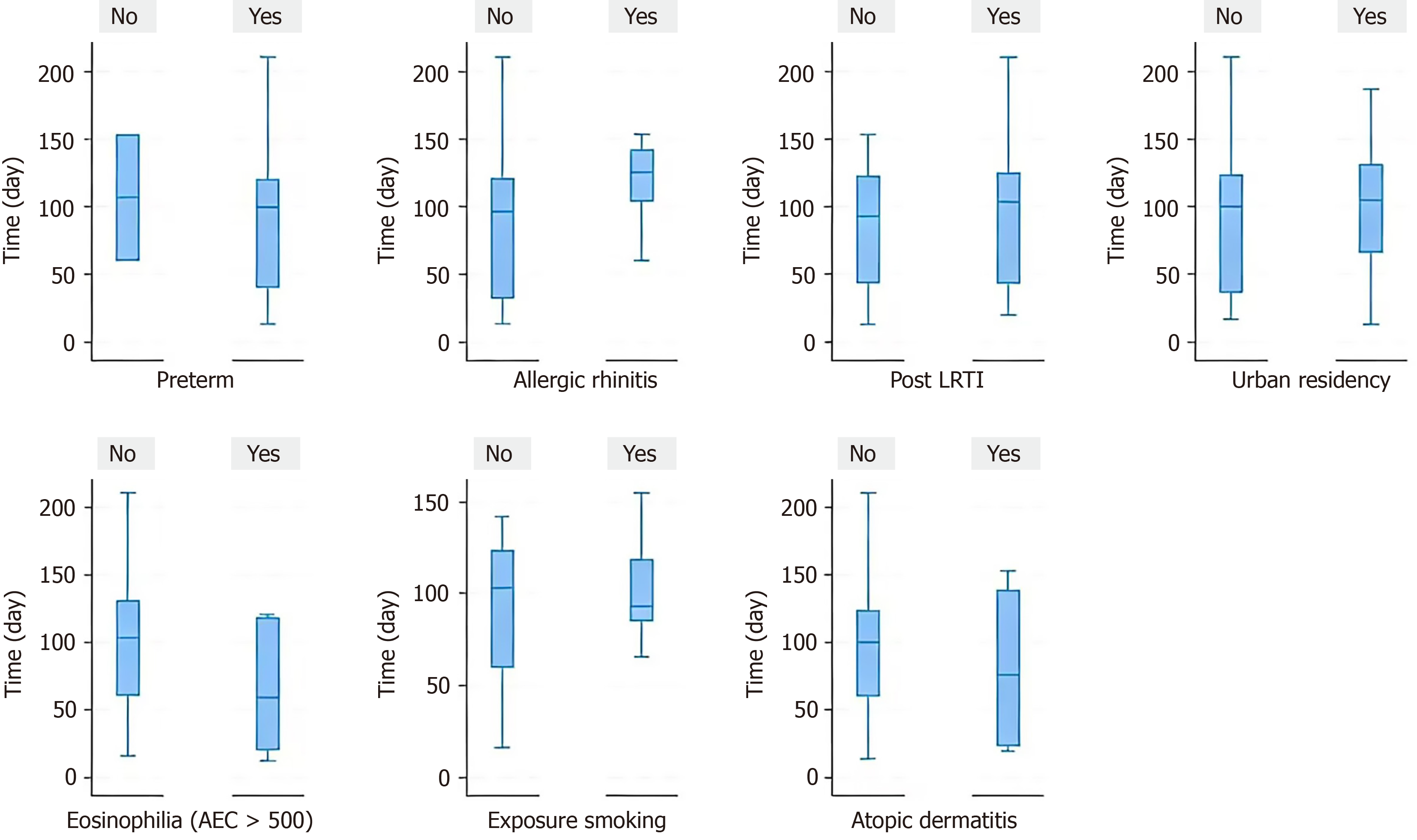
Kaplan-Meier analysis showed that the median recurrence time from the initial ALRTI hospitalization to the first episode of recurrent wheezing was 100 days (IQR: 43-125; P < 0.001) (Figure 2). Analysis by risk factors showed median recurrent times as follows: Age 12-24 months (100 days; IQR: 66-106), preterm birth (99 days; IQR: 40-120), allergic rhinitis (125 days; IQR: 103-142), atopic dermatitis (27 days; IQR: 20-125), urban residency (85 days; IQR: 66-131), maternal smoking during pregnancy (93 days; IQR: 85-119), eosinophilia (AEC > 500 cells/μL) (59 days; IQR: 21-119), and post-ALRTI complications (103 days; IQR: 43-125) (Figure 3).
Recurrent wheezing following an episode of ALRTI in early childhood represents a clinically relevant outcome, given its potential role as an early indicator of persistent airway disease, including asthma[10-12]. Beyond its long-term respiratory implications, recurrent wheezing places a substantial burden on healthcare systems and adversely affects the quality of life of affected children and their families. In this study, we investigated clinical and demographic determinants of recurrent wheezing after ALRTI and identified several significant risk factors, including age 12-24 months, prematurity, allergic rhinitis, urban place of residence, eosinophilia (AEC > 500 cells/μL) and post lower respiratory tract infection.
Age between 12 months and 24 months emerged as a potential significant risk factor for recurrent wheezing following ALRTI, consistent with earlier studies[2,13]. Children in this age group are anatomically and physiologically predisposed to wheezing: Their bronchi are narrower, airway smooth muscle remains immature, and airway reactivity is heightened compared to older children[14-16]. While children under 12 months were also included in our cohort, the subgroup was too small for meaningful statistical analysis, limiting conclusions regarding this younger population.
Prematurity similarly predisposed children to recurrent wheezing, in line with previous research[3,4,13,17,18]. Immaturity of lung structure and function in preterm infants—characterized by underdeveloped alveolar architecture, smaller airway caliber, and fragile connective tissue—renders them more vulnerable to obstruction when exposed to infections or environmental triggers. Additionally, their immunological immaturity increases susceptibility to respiratory infections, while bronchial hyperresponsiveness is more prevalent, all of which contribute to recurrent wheezing[19].
Allergic rhinitis also emerged as a contributing factor to recurrent wheezing, consistent with previous studies[5,20]. This finding is biologically plausible, given the shared inflammatory mechanisms between allergic rhinitis and wheezing, often conceptualized under the 'united airway disease’ spectrum. Eosinophils, mast cells, and cytokines active in allergic rhinitis contribute to lower airway inflammation and hyperresponsiveness[21]. Nasal obstruction further promotes mouth breathing, impairing air filtration of inhaled particles and increasing allergen and irritant exposure to the lower airways.
In addition, eosinophilia was strongly associated with recurrent wheezing, corroborating previous reports[6,20,22]. Eosinophils play a central role in allergic airway inflammation and the pathophysiology of wheezing and asthma. When activated, eosinophils release cytotoxic proteins—including major basic protein and eosinophil cationic protein that directly damage the airway epithelium, disrupt the airway barrier, and initiate an inflammatory response[23-25]. These mediators cause mucosal edema, increase airway permeability, and enhance bronchial hyperresponsiveness, increasing the likelihood of bronchospasm.
Urban residency was also found to be a significant risk factor for recurrent wheezing in this study, consistent with prior epidemiological findings[7,26]. Children in urban environments are exposed to higher levels of air pollutants [e.g., particulate matter (PM2.5), nitrogen dioxide (NO2), vehicle exhaust], which are known to induce airway inflammation and bronchial hyperresponsiveness[26,27]. Higher population density also facilitates the spread of respiratory infections, a common trigger for wheezing. Additionally, urban lifestyles may lead to more time spent indoors, where allergens and pollutants, such as dust mites, pet dander, and mold spores, can accumulate. According to the hygiene hypothesis, reduced exposure to environmental microbes, in sanitized urban settings may lead to an immune imbalance, increasing the risk of allergic sensitization and recurrent wheezing[28,29].
A history of previous LRTI was also identified as a significant risk factor for recurrent wheezing, consistent with prior research[8,9,20]. Viral infections, particularly from RSV, rhinovirus, and human metapneumovirus, can induce inflammation of the bronchial epithelium, leading to mucosal edema and increased airway hyperresponsiveness. Certain viruses, such as RSV and rhinovirus, are known to stimulate a Th2-skewed immune response, which promotes asthma-like airway inflammation and may predispose children to recurrent wheezing[30-33]. Severe or recurrent LRTIs can also cause structural changes in the airways, such as epithelial disruption, subepithelial fibrosis, and airway remodeling, which can narrow the airway lumen and contribute to long-term respiratory problems.
Unexpectedly, our study observed a negative association between both atopic dermatitis and secondhand smoke exposure with recurrent wheezing. These findings are inconsistent with previous studies[5,13,17,20,34,35], which generally report positive relationships between atopic conditions, environmental tobacco smoke exposure, and early childhood wheezing or asthma[5,20]. Several factors may account for these discrepancies. First, the characteristics of our study population and setting may differ from those in previous research. Conducted in a tertiary care hospital, our cohort comprised children admitted for ALRTI, who may have presented with more complex or acute conditions unrelated to atopic predisposition, such as RSV bronchiolitis, which may be less strongly linked to long-term atopic wheezing pathways. Second, the relatively short 12-month follow-up may have inadvertently selected for families with higher health-seeking behaviors and better healthcare access, who might have adopted protective measures (e.g., smoking cessation) after hospitalization. This could have led to underrepresentation of children with persistent environmental tobacco smoke exposure. Third, measurement limitations should be considered. Information on secondhand smoke exposure and atopic dermatitis relied on parental recall and medical records, which may have introduced potential misclassification, particularly of mild or intermittent cases. Although random verification of 10% of medical records was implemented, residual misclassification cannot be excluded. Fourth, further analysis of our dataset indicated that atopic dermatitis severity was generally mild across both recurrence and non-recurrence groups, and detailed information regarding the dose or duration of secondhand smoke exposure were not systematically collected. These factors suggest potential residual confounding or statistical chance (e.g., type II error) influencing the observed associations. Finally, children under 5 years of age, wheezing is often episodic and primarily virus-induced rather than driven by chronic atopic inflammation; hence, the atopy-related asthma phenotype may not yet be fully established. Therefore, these unexpected protective associations should be interpreted with caution and considered hypothesis-generating. Prospective studies with larger sample sizes and more detailed exposure assessments are warranted to confirm or refute these findings.
Our study also contributes to the understanding of the temporal pattern of recurrent wheezing following ALRTI. The median interval between the initial ALRTI hospitalization and the first recurrence was 100 days (IQR: 43-125 days), with variability across subgroups: 100 days in children aged 12-24 months, 99 days in preterm infants, 125 days in those with allergic rhinitis, 27 days in children with atopic dermatitis, 85 days in urban residents, 93 days in those with prenatal tobacco smoke exposure, 59 days in those with eosinophilia, and 103 days in children who developed postALRTI complications.
Compared with previous research—such as a Thai study reporting a mean interval of 4.7 ± 3.7 months (approximately 141 days) among hospitalized children aged 6 months to 5 years in Bangkok[36], and others documenting most recurrences within the first year postinfection[22,37]—our findings provide a more granular temporal framework by subgroup. Clinically, these data underscore the importance of intensive monitoring and early followup, particularly within the first 3 months to 4 months after ALRTI, among children with identified risk factors. For example, the notably short median interval (27 days) in children with atopic dermatitis suggests an accelerated risk trajectory. Tailored followup protocols—such as pediatric reassessment or targeted respiratory evaluation approximately 30 days postdischarge for atrisk children—may therefore be warranted.
However, these findings must be interpreted cautiously. Although our cohort size and followup duration were sufficient to identify median recurrence intervals, residual confounding and heterogeneity among wheezing phenotypes (e.g., viraltriggered vs. atopymediated) cannot be ruled out. Larger prospective studies with standardized definitions of wheezing onset and recurrence are needed to validate and refine these timelines.
Despite our efforts to minimize missing data, several limitations should be acknowledged. The retrospective design inherently restricts causal inference, and unmeasured confounders—such as environmental allergens, genetic predisposition, or socioeconomic factors—may have influenced the results. Moreover, the relatively small number of recurrence events (n = 33), particularly within certain subgroups (e.g., children under 12 months, those exposed to secondhand smoke, or those with atopic dermatitis), may have reduced statistical power, compromised the stability of multivariate analyses, and constrained generalizability of our findings.
Furthermore, the exclusion of up to 560 patients due to loss to follow-up may have introduced selection bias, particularly if these patients differed systematically from those included. Information on secondhand smoke exposure and history of allergic diseases also relied on parental recall and medical records, raising the possibility of measurement bias. Although random verification of 10% of medical records was conducted to mitigate the issue, residual bias cannot be completely excluded. Taken together, these factors underscore the need for cautious interpretation of our results and highlight the importance of future large-scale prospective studies to validate and expand upon our findings.
The implementation of findings from “Understanding recurrent wheezing: A parent’s guide” underscores the importance of translating clinical evidence into practical, evidence-based strategies that enable healthcare providers and parents to identify and manage risk factors at an early stage. The study highlights that children aged 12-24 months, those born preterm, and those with allergic rhinitis, eosinophilia, urban residence, or a prior history of lower respiratory infections are at a substantially increased risk of developing recurrent wheezing following ALRTI.
To operationalize these insights, pediatric care protocols should incorporate structured post-infection surveillance systems that include scheduled follow-up visits within the first 100 days—identified as the median recurrence interval—and intensified monitoring for children with allergic dermatitis, who may experience recurrence as early as 27 days post-infection.
For parents, early recognition of clinical warning signs plays a pivotal role. Educational interventions should equip caregivers with the ability to identify symptoms such as persistent coughing, noisy breathing, and manifestations of allergic conditions, while reinforcing the importance of regular pediatric assessments.
By fostering a collaborative parent-clinician partnership, these implementation strategies not only facilitate timely medical intervention but also promote long-term respiratory health. Ultimately, translating these findings into accessible, family-centered care frameworks may help prevent the progression of recurrent wheezing to chronic respiratory disorders such as asthma, thereby aligning scientific insight with practical, preventive healthcare delivery.
This study identifies key risk factors for recurrent wheezing following ALRTI in early childhood. Children aged 12-24 months, those born preterm, and those with allergic rhinitis, eosinophilia, urban residency, or a history of prior lower respiratory infections were at increased risk. The median time to recurrent wheezing was 100 days, with considerable variability across subgroups—ranging from as early as 27 days in children with allergic dermatitis to 125 days in those with allergic rhinitis. These findings underscore the importance of early identification and monitoring of high-risk children and support the development of targeted preventive and therapeutic strategies aimed at mitigating recurrent wheezing and its progression to chronic respiratory diseases such as asthma.
The authors would like to express their gratitude to Miss Daisy Gonzales of the International Relations Section, Faculty of Medicine, Naresuan University, for her valuable assistance in editing the manuscript.
| 1. | Ng MC, How CH. Recurrent wheeze and cough in young children: is it asthma? Singapore Med J. 2014;55:236-241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 12] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 2. | Teijeiro A, Badellino H, Raiden MG, Cuello MN, Kevorkof G, Gatti C, Croce VH, Solé D. Risk factors for recurrent wheezing in the first year of life in the city of Córdoba, Argentina. Allergol Immunopathol (Madr). 2017;45:234-239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 3. | Gustafson B, Britt RD Jr, Eisner M, Narayanan D, Grayson MH. Predictors of recurrent wheezing in late preterm infants. Pediatr Pulmonol. 2024;59:181-188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 4. | Chen X, Yang M, Xie J, Huang S, Qin X, Pan Z, Zhu M, Zeng D, Huang Y, Liang T, Liang C, Nong G. Risk factors for early wheezing in preterm infants: a retrospective cohort study. Front Pediatr. 2025;13:1555834. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 5. | Geng L, Tang X, Hua L, Liu H, Ren S, Zhong W, Zhang J. The analysis of risk factors for recurrent wheezing in infants and clinical intervention. Transl Pediatr. 2023;12:1810-1822. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 6. | Fitzpatrick AM, Grunwell JR, Cottrill KA, Mutic AD, Mauger DT. Blood Eosinophils for Prediction of Exacerbation in Preschool Children With Recurrent Wheezing. J Allergy Clin Immunol Pract. 2023;11:1485-1493.e8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 25] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 7. | Grant TL, Wood RA. The influence of urban exposures and residence on childhood asthma. Pediatr Allergy Immunol. 2022;33:e13784. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 55] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 8. | Zhu Y, Chen L, Miao Y, Chen J, Bai M, Gao H, Zhu Z, Zhang Y, Zhang J, Raza HK, Liu G. An analysis of risk factors associated with recurrent wheezing in the pediatric population. Ital J Pediatr. 2023;49:31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 9. | Srisingh K, Weerakul J, Srijuntongsiri S, Ngoenmak T. Risk Factors of Recurrent Wheezing in Children Under 5 Years of Age. J Med Assoc Thai. 2017;100:183-189. [PubMed] |
| 10. | Toivonen L, Forsström V, Waris M, Peltola V. Acute respiratory infections in early childhood and risk of asthma at age 7 years. J Allergy Clin Immunol. 2019;143:407-410.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 18] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 11. | Kenmoe S, Atenguena Okobalemba E, Takuissu GR, Ebogo-Belobo JT, Oyono MG, Magoudjou-Pekam JN, Kame-Ngasse GI, Taya-Fokou JB, Mbongue Mikangue CA, Kenfack-Momo R, Mbaga DS, Bowo-Ngandji A, Kengne-Ndé C, Esemu SN, Njouom R, Ndip L. Association between early viral lower respiratory tract infections and subsequent asthma development. World J Crit Care Med. 2022;11:298-310. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 6] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 12. | Zaitsu M, Morita S. Infants Hospitalized with Lower Respiratory Tract Infections Were More Likely to Develop Asthma. Adv Respir Med. 2022;90:246-253. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 13. | Bozaykut A, Paketci A, Sezer RG, Paketci C. Evaluation of risk factors for recurrent wheezing episodes. J Clin Med Res. 2013;5:395-400. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 10] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 14. | Trachsel D, Erb TO, Hammer J, von Ungern-Sternberg BS. Developmental respiratory physiology. Paediatr Anaesth. 2022;32:108-117. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 77] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 15. | Tepper RS, Stevens J, Eigen H. Heightened airway responsiveness in normal female children compared with adults. Am J Respir Crit Care Med. 1994;149:678-681. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 16. | Weist A, Williams T, Kisling J, Clem C, Tepper RS. Volume history and effect on airway reactivity in infants and adults. J Appl Physiol (1985). 2002;93:1069-1074. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 17. | Simões MCRDS, Inoue Y, Matsunaga NY, Carvalho MRV, Ribeiro GLT, Morais EO, Ribeiro MAGO, Morcillo AM, Ribeiro JD, Toro AADC. Recurrent wheezing in preterm infants: Prevalence and risk factors. J Pediatr (Rio J). 2019;95:720-727. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 13] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 18. | Hsu YL, Huang HC, Su TY, Chen IL. Early Life Factors Associated with Preschool Wheezing in Preterm Infants. Children (Basel). 2021;8:732. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 19. | Melville JM, Moss TJ. The immune consequences of preterm birth. Front Neurosci. 2013;7:79. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 222] [Cited by in RCA: 276] [Article Influence: 21.2] [Reference Citation Analysis (0)] |
| 20. | Kim JH, Choi JY, Kim NY, Kim JW, Baek JH, Baek HS, Yoon JW, Jee HM, Choi SH, Kim HY, Kim KE, Shin YH, Han MY. Clinical risk factors associated with the development of wheezing in children less than 2 years of age who required hospitalization for viral lower respiratory tract infections. Korean J Pediatr. 2015;58:245-250. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 21. | Pawankar R, Mori S, Ozu C, Kimura S. Overview on the pathomechanisms of allergic rhinitis. Asia Pac Allergy. 2011;1:157-167. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 138] [Cited by in RCA: 192] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 22. | Midulla F, Nicolai A, Ferrara M, Gentile F, Pierangeli A, Bonci E, Scagnolari C, Moretti C, Antonelli G, Papoff P. Recurrent wheezing 36 months after bronchiolitis is associated with rhinovirus infections and blood eosinophilia. Acta Paediatr. 2014;103:1094-1099. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 52] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 23. | Acharya KR, Ackerman SJ. Eosinophil granule proteins: form and function. J Biol Chem. 2014;289:17406-17415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 288] [Cited by in RCA: 400] [Article Influence: 33.3] [Reference Citation Analysis (0)] |
| 24. | Adamko D. Chapter 8 - Eosinophil Secretory Functions. In: Lee JJ, Rosenberg HF, editors. Eosinophils in Health and Disease. Academic Press, 2013: 229-275. [DOI] [Full Text] |
| 25. | Gigon L, Fettrelet T, Yousefi S, Simon D, Simon HU. Eosinophils from A to Z. Allergy. 2023;78:1810-1846. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 98] [Reference Citation Analysis (0)] |
| 26. | Son JY, Kim H, Bell ML. Does urban land-use increase risk of asthma symptoms? Environ Res. 2015;142:309-318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 27. | Vieira SE, Stein RT, Ferraro AA, Pastro LD, Pedro SS, Lemos M, da Silva ER, Sly PD, Saldiva PH. Urban air pollutants are significant risk factors for asthma and pneumonia in children: the influence of location on the measurement of pollutants. Arch Bronconeumol. 2012;48:389-395. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 18] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 28. | Liu AH. Revisiting the hygiene hypothesis for allergy and asthma. J Allergy Clin Immunol. 2015;136:860-865. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 127] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 29. | Le Cann P, Bonvallot N, Glorennec P, Deguen S, Goeury C, Le Bot B. Indoor environment and children's health: recent developments in chemical, biological, physical and social aspects. Int J Hyg Environ Health. 2011;215:1-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 40] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 30. | Bacharier LB. The recurrently wheezing preschool child-benign or asthma in the making? Ann Allergy Asthma Immunol. 2015;115:463-470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 31. | Malinczak CA, Lukacs NW, Fonseca W. Early-Life Respiratory Syncytial Virus Infection, Trained Immunity and Subsequent Pulmonary Diseases. Viruses. 2020;12:505. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 38] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 32. | Gern JE, Busse WW. Relationship of viral infections to wheezing illnesses and asthma. Nat Rev Immunol. 2002;2:132-138. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 87] [Cited by in RCA: 92] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 33. | Han M, Rajput C, Hershenson MB. Rhinovirus Attributes that Contribute to Asthma Development. Immunol Allergy Clin North Am. 2019;39:345-359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 34. | Kanchongkittiphon W, Gaffin JM, Phipatanakul W. The indoor environment and inner-city childhood asthma. Asian Pac J Allergy Immunol. 2014;32:103-110. [PubMed] |
| 35. | Magnus MC, Håberg SE, Karlstad Ø, Nafstad P, London SJ, Nystad W. Grandmother's smoking when pregnant with the mother and asthma in the grandchild: the Norwegian Mother and Child Cohort Study. Thorax. 2015;70:237-243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 88] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 36. | Chantawarangul K, Deerojanawong J, Sritippayawan S. First episode of preschool wheeze requiring hospitalization:a prospective study on the chance of recurrence and associated factors. Asian Pac J Allergy Immunol. 2018;36:232-237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 37. | Escobar GJ, Ragins A, Li SX, Prager L, Masaquel AS, Kipnis P. Recurrent wheezing in the third year of life among children born at 32 weeks' gestation or later: relationship to laboratory-confirmed, medically attended infection with respiratory syncytial virus during the first year of life. Arch Pediatr Adolesc Med. 2010;164:915-922. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 66] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/