Published online Mar 9, 2026. doi: 10.5409/wjcp.v15.i1.111304
Revised: July 24, 2025
Accepted: October 29, 2025
Published online: March 9, 2026
Processing time: 252 Days and 14 Hours
Fluid therapy is a cornerstone in the management of diabetic ketoacidosis (DKA). Normal saline (NS, 0.9%) has traditionally been the fluid of choice; however, con
To compare BES with NS in the initial resuscitation of children with DKA.
This double-blind, parallel, randomized controlled trial (RCT) was conducted in the Pediatrics department of a tertiary care teaching hospital over two-year pe
Sixty patients were enrolled. The mean (SD) age of the patients was 10.7 (1.42) years, and majority were female (73.3%). The time (hour) taken for resolution of acidosis was not significantly different (P = 0.16). The NS group had a higher incidence of hyperchloremia (P = 0.09), AKI (P = 0.07), and prolonged hospitalization (P = 0.23), but all these were not statistically significant. Other outcomes were comparable in both the groups.
This trial did not find any significant difference between BES and NS (0.9%) in the initial resuscitation of children with DKA.
Core Tip: The management of childhood diabetic keto-acidosis (DKA) is fluid resuscitation in addition to insulin infusion. There is a controversy regarding the ideal fluid for the management of DKA. This trial compared balanced electrolyte solution (BES) with normal saline (NS), and found no difference between the two. However, a trend towards lower complication rate was noted with BES. BES might be preferred over NS for initial management of children with DKA where resource constraint is not an issue.
- Citation: Sweety S, Panda S, Das RR. Balanced electrolyte solution vs isotonic saline in the resuscitation of children with diabetic ketoacidosis: A randomized controlled trial. World J Clin Pediatr 2026; 15(1): 111304
- URL: https://www.wjgnet.com/2219-2808/full/v15/i1/111304.htm
- DOI: https://dx.doi.org/10.5409/wjcp.v15.i1.111304
Diabetic ketoacidosis (DKA) is a serious and potentially life threatening complication of diabetes mellitus (DM), particularly type 1 DM (T1DM). It develops when the body starts breaking down fats at an excessive rate[1]. The liver processes the fat into ketone bodies, which causes the blood to become acidic. In children, DKA often is the initial presentation that unravels the underlying diagnosis of T1DM. In stressful situations, counter-regulatory hormones cause glycogenolysis, decreased glucose utilization, and increased lipolysis. This in turn aggravates hyperglycemia causing diuresis and dehydration, ketosis and increase lactate causing metabolic acidosis[2]. The treatment involves insulin infusion, and intravenous fluids to correct underlying dehydration[2].
Fluid replacement and maintenance remains the main study of therapy in addition to the insulin infusion. Fluid therapy restores circulatory volume, improves renal function, and facilitates the elimination of ketones and glucose from the body. The International Society for Pediatric and Adolescent Diabetes (ISPAD) in its latest guideline recommends use of crystalloids like normal saline (NS) or other balanced salt (electrolyte) solutions [Balanced salt solutions (BSS)/balanced electrolyte solution (BES)][2]. Of the types of NS, either 0.45% or 0.9% can be used in DKA. The later was based on the large multi-center Pediatric Emergency Care Applied Research Network DKA trial[3]. Of the two crystalloids, NS has long been debated to be an unbalanced fluid based on its higher chloride concentration than plasma (40% more), acidic pH, and a strong ion difference of “zero”[4]. NS in many studies has been shown to affect the microcirculation, immune complication causing hyperchloremic metabolic acidosis, acute kidney injury (AKI), and worse clinical outcomes[4-6].
The choice of fluid, mainly between NS and BES/BSS, has been a subject of ongoing debate among clinicians for fluid resuscitation in children with DKA. Published trials and systematic review/meta-analysis have found variable results with some favouring BES/BSS over NS, and others finding no difference between the two[7-11]. The aim of the present study was to compare the efficacy and safety of BES with that of NS in the initial resuscitation of children with DKA. The primary objective was the time taken to resolution of acidosis. Secondary objectives were-effect on electrolyte imbalances, incidence of cerebral edema, development of AKI, and other clinical outcomes.
This double-blind, parallel, randomized controlled trial (RCT) was conducted in the Pediatrics department (a 200 bedded hospital dedicated for children) of a tertiary care teaching institute in Eastern India. The study was conducted over two-year period (from July 2022 to June 2024). Eligible participants were randomly assigned to either the NS group or the BES group. For block randomization, an independent statistician prepared variable blocks with an allocation ratio of 1:1 (for the 2 fluid groups, NS and BES) using a web-based random table generator. Allocation of participants to the two groups was carried out by serially numbered opaque sealed envelopes. To maintain blinding, the individual fluid bag (500 mL of NS or BES) was packed in sealed opaque covers. The BES used was Ringer's Lactate (RL). Each of the covered fluid pack was then kept inside the variable randomization blocks prepared, which was again covered. These coverings were prepared by another person not involved in the study. After a patient was included in the study, the study personnel picked the fluid bag kept as per allocation sequence, and initiated as per the study protocol. The treating physician, the patient, and the outcome assessor; all were blinded. The study was approved by the Institute Ethics Committee. The study is reported as per the declaration of Helsinki.
Inclusion criteria included children of 5-14 years of age with a diagnosis of DKA presenting to the emergency department. DKA was defined as per the ISPAD 2022 criteria[2]. The criteria includes-hyperglycemia (blood glucose about 200 mg/dL or > 11 mmol/L), metabolic acidosis (arterial pH < 7.3 or bicarbonate < 18 mmol/L), and presence of ketonemia or moderate to large ketonuria. Children with pre-existing renal insufficiency, significant cardiac disease, liver disease, obvious features of cerebral edema, hyponatremia (Na < 135 meq/L) or hyperkalemia (K > 5.5 meq/L) at admission, or contraindications to study fluids, or received prior treatment were excluded. Written informed consents were taken from the parents or care givers, and assents were taken from children > 7 years age.
After a quick history and focused clinical examinations, blood samples were drawn for analysis of blood glucose, serum osmolality, complete blood count, liver and renal function tests, and serum electrolytes. Blood gas analysis was done to look for pH, bicarbonate (HCO3), and PaCO2. A urine dipstick test was used to test for ketonuria. If the child presented with shock, the trial fluid was given as a bolus of 20 mL/kg. If the child was not in shock, the trial fluid volume to be given over next 48 hours was calculated as a sum of deficit and maintenance[12-14]. Fluid deficit was calculated based on the dehydration status, and the later was classified based on severity of DKA; 5 to 7% for moderate DKA, and 7% to 10% for severe DKA. After the first hour of fluid therapy, regular insulin infusion a rate of 0.05 U/kg/hour was started. The fluid was changed to N2 5% dextrose (N/2 D5) once the blood glucose decreased to ≤ 250 mg/dL. Any decrease in blood glucose below 150 mg/dL was corrected with the addition of D5 to maintain the blood glucose between 150 to 250 mg/dL to enable insulin infusion in case acidosis persisted. In case of persistent DKA, dose of insulin was hiked to 0.1 U/kg/hour.
Blood glucose (capillary) was measured hourly by “Glucocheck Glucose Meter” (Aktivmed GmbH, Germany). Blood gas analysis was done every 4 hours, as was urine ketone by dipstick. Renal function test and serum electrolytes were measured twice or thrice daily till the end points were achieved. Clinical parameters (heart rate, respiratory rate, capillary refill time, blood pressure, and neurological status), intake and output monitoring was done every 1-2 hours.
All the data pertaining to history, demography, clinical and laboratory parameters, management, and outcome details were recorded in the case record forms. AKI was defined as per the serum creatinine (as urine output is not reliable in DKA) based on Kidney Disease Improving Global Outcomes staging.
The sample size calculation was based on previously published studies from India[7-9]. Considering the mean difference in the time to resolution of acidosis to be of 4 hours with α of 0.05, power of 90%, and allocation ratio of 1:1, the total sample size was calculated to be 54. Assuming 10% attrition, the final sample size was calculated at 60 (30 in each group).
The data were entered into Microsoft excel sheet. IBM SPSS Statistics for Windows, version 21 (IBM Corp., Armonk, NY, United States) was used for analysis. Continuous data were presented either as mean (SD) or median [inter-quartile range (IQR)] depending on the type of data. Categorical data were presented as frequencies and percentages (%). Categorical data (nominal) were analyzed by using the χ2 test or Fisher’s exact test. Continuous data were analyzed by using the student t-test, or Mann-Whitney U test. A P-value of < 0.05 was taken as statistically significant. Hazard ratio was estimated using Cox proportional hazard model. A P-value of < 0.05 was considered as statistically significant. The effect sizes were reported along with 95%CI.
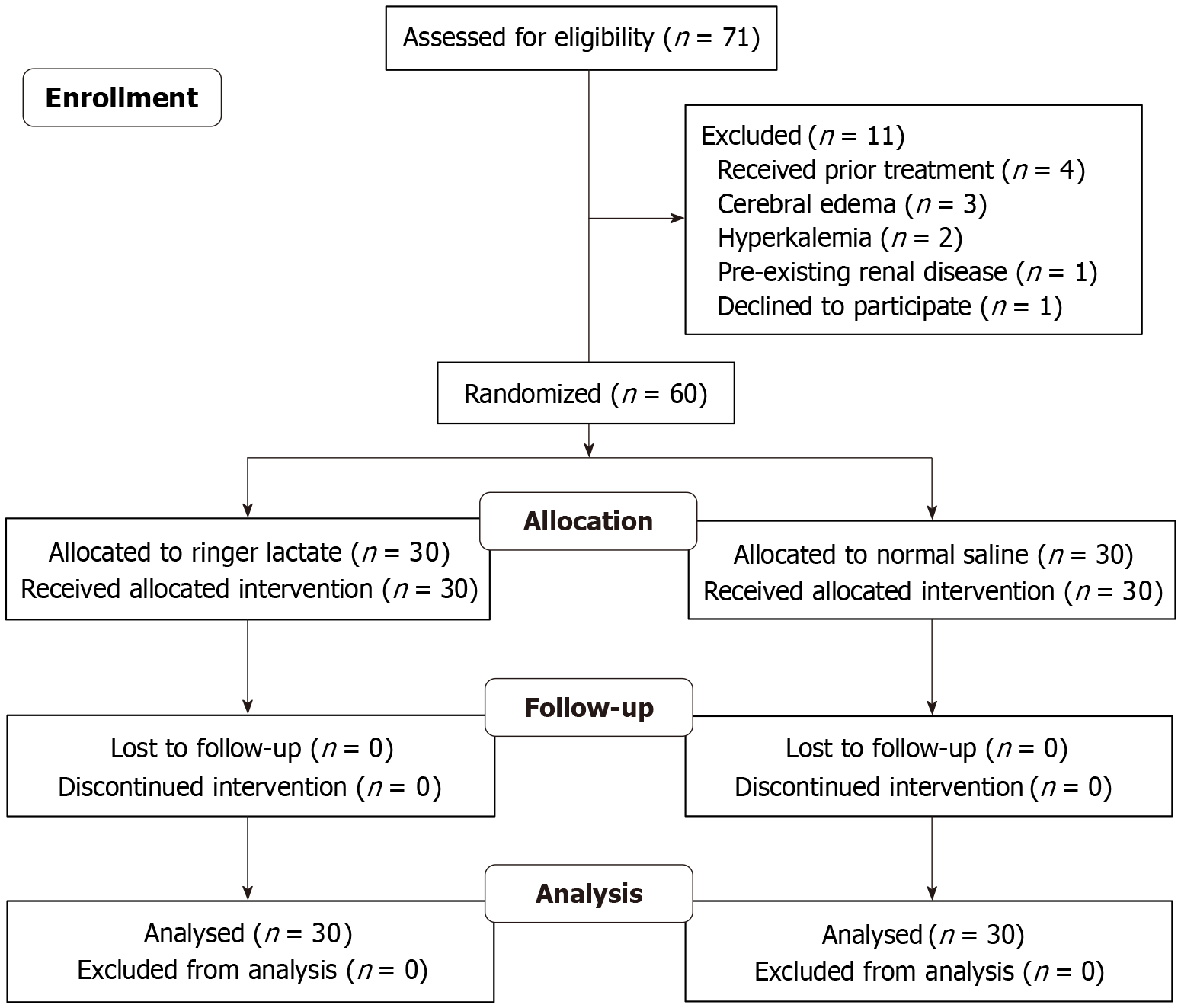
A total of 71 children with DKA were screened, and 11 were excluded. The details of the study flow have been mentioned in Figure 1. The two groups were comparable as shown in Table 1. The mean (SD) age of the children was 10.7 (1.42) years, and the majority were female (73.3%). The mean Twenty-one children were known case of diabetes, and 5 had family history of diabetes. The mean age of diabetes onset in the cohort was 5 years. Previous history of DKA was present in 33 (55%) children. Pain abdomen (41%), fever (40%), vomiting (15%), and breathing problems (8%) were the common complaints noted at presentation. The mean GCS at presentation was 13, and majority (70%) had altered sensorium (irritability, or lethargy) at presentation.
| Characteristics | BES (n = 30) | NS (n = 30) | P value |
| Age (years) | 10.77 ± 1.36 | 10.63 ± 1.5 | 0.72 |
| Female | 23 (76.7) | 21 (70) | 0.56 |
| Anthropometric parameters | |||
| Weight (kg) | 19.2 ± 6.5 | 19.5 ± 6.4 | 0.84 |
| Height (cm) | 131.2 ± 24.1 | 130.9 ± 24.3 | 0.77 |
| BMI (kg/m2) | 19.6 ± 3.4 | 19.4 ± 3.5 | 0.92 |
| Socio-economic status (poor) | 6 (20) | 5 (16.7) | 0.63 |
| Family history of diabetes | 3 (10) | 2 (6.7) | 0.44 |
| Known case of diabetes | 10 (33.3) | 11 (36.7) | 0.59 |
| Age (year) at onset of diabetes | 5 ± 2 | 5 ± 2 | 0.99 |
| Previous history of DKA | 16 (53.3) | 17 (56.7) | 0.73 |
| Precipitating event present | 15 (50) | 17 (56.7) | 0.42 |
| Infection present | 9 (30) | 7 (23.3) | 0.39 |
| Poor compliance | 17 (56.7) | 18 (60) | 0.65 |
| Severity of DKA at presentation | |||
| Mild | 2 (3.3) | 2 (6.7) | |
| Moderate | 7 (6.9) | 8 (3.3) | 0.39 |
| Severe | 21 (70) | 20 (66.7) | |
| Shock at presentation | 2 (6.7) | 1 (3.3) | 0.17 |
| GCS at admission | 13 ± 2 | 13 ± 2 | 0.97 |
| Blood glucose (mg/dL) | 452 (364-548) | 448 (354-542) | 0.48 |
| Hb1Ac (%) | 9.8 (8.8-12) | 9.6 (8.6-11.5) | 0.76 |
| Laboratory parameters | |||
| Blood pH | 7.01 (6.9-7.18) | 7.02 (6.9-7.2) | 0.59 |
| PCO2 | 20 (15-23) | 19 (16-22) | 0.71 |
| HCO3 (mmol/L) | 7.2 (5.6-10.1) | 7 (5.5-9.5) | 0.81 |
| Lactate (mmol/L) | 2 (1-3) | 2 (1-3) | 0.93 |
| Anion gap (mmol/L) | 16 (14-20) | 17 (14-19) | 0.77 |
| Serum osmolality (mmol/kg) | 296 (291-310) | 298 (293-310) | 0.56 |
| Sodium (corrected) (meq/L) | 136 (132-139) | 138 (134-141) | 0.84 |
| Potassium (meq/L) | 3.9 (3.4-4.2) | 3.7 (3.4-4.1) | 0.51 |
| Phosphate (mg/dL) | 3.4 (3.1-4) | 3.8 (3.2-4) | 0.48 |
| Chloride (meq/L) | 108 (104-112) | 106 (101-110) | 0.59 |
| Renal function test | |||
| Urea (mg/dL) | 35 (28-61) | 32 (25-54) | 0.23 |
| Creatinine (mg/dL) | 0.7 (0.4-1.2) | 0.7 (0.5-1.3) | 0.65 |
The distribution of different types of DKA was as follows: Mild (n = 4, 6.7%), moderate (n = 15, 25%), and severe (n = 41, 68.3%). Three (10%), children had shock. The median blood glucose was 450 mg/dL, and the median Hb1Ac was 9.7%. The blood gas, electrolytes, anion gap, and lactate are described in Table 1. Renal function at presentation was deranged in few (n = 6, 10%).
These are described in Table 2. The median estimated fluid deficit was 7.5%, and the median total fluid administered was 1.25 L with study fluid being 0.42 L. The urine output (median) was maintained at the rate of 2.55 mL/kg/hour at the end of treatment. A hike in the dose of insulin was required in 16 children.
| Characteristics | BES (n = 30) | NS (n = 30) | P value |
| Estimated fluid deficit (%) | 7.5 (6.5-8.5) | 7.5 (6.5-8.5) | 0.91 |
| Total fluid administered (L) | 1.12 (0.9-1.79) | 1.38 (1-1.81) | 0.29 |
| Study fluid administered (L) | 0.4 (0.3-0.62) | 0.44 (0.3-0.58) | 0.36 |
| Duration of study fluid (hour) | 5 (3-6) | 5 (3, 6) | 0.77 |
| Need of insulin hike | 9 (30) | 7 (23.3) | 0.39 |
| Insulin dose (maximum), unit/kg | 0.05 (0.05-0.05) | 0.05 (0.05-1) | 0.21 |
| Urine output (mL/kg/hour) till end-point | 2.6 (2-4) | 2.5 (2-4) | 0.63 |
| Total fluid balance (mL/kg) | 34 (14-52) | 36 (16-55) | 0.54 |
These are described in Table 3. The median time (IQR) taken for resolution of DKA in BES group was 12 hours (8, 18), and in NS group was 16 hours (10, 22). Although the time taken in BES group was shorter, the difference was not statistically significant (P = 0.16). The NS group had a higher incidence of hyperchloremia (P = 0.09), AKI (P = 0.07), and prolonged hospitalization (P = 0.23), but all these were not statistically significant. Other outcomes were comparable in both the groups (Table 3).
| Characteristics | BES (n = 30) | NS (n = 30) | P value |
| Primary outcome | |||
| Time taken for resolution of acidosis (hour) | 12 (8-18) | 16 (10-22) | 0.16 |
| Secondary outcomes | |||
| Duration of hospital stay (day) | 4 (3-5) | 5 (3-6) | 0.23 |
| Complications | |||
| Acute kidney injury1 | 0 (0) | 3 (10) | 0.07 |
| Cerebral edema | 0 (0) | 1 (3.3) | 0.21 |
| Required mechanical ventilation | 1 (3.3) | 1 (3.3) | 0.86 |
| Required inotropes | 1 (3.3) | 0 (0) | 0.21 |
| Hypernatremia | 1 (3.3) | 1 (3.3) | 0.86 |
| Hyponatremia | 5 (16.7) | 4 (13.3) | 0.42 |
| Hyperchloremia | 1 (3.3) | 3 (10) | 0.09 |
| Hypokalemia | 3 (10) | 5 (16.7) | 0.08 |
This RCT included 60 children with DKA, and compared the two fluids (NS, and BES) that are commonly used in the initial resuscitation. The time taken for resolution of DKA in both the groups was not significantly different (P = 0.16). The secondary outcomes including the incidence of electrolyte imbalances, AKI (P = 0.07), and duration of hospitalization, cerebral edema were not significantly different between the two groups.
The present trial findings are in agreement with previously published trials from India and abroad[7-9,15]. Agarwal et al[9], in their RCT included 67 children with DKA and compared NS with RL. The mean time to resolution of DKA was shorter in RL (12.9 hours) group compared to the NS group (16.8 hours), without any significant inter-group difference (P = 0.06). The authors explained that this difference is significant on the notion that the 95%CI of the mean difference (0.3 to 8 hours) did not include 0. On subgroup analysis, the time to severe DKA resolution was significantly shorter in the RL group (P = 0.01). This trial result is similar to a trial from Australia that included 77 children, and compared NS vs HS (Hartmann’s solution)[15]. In this trial, although no difference was found between the time taken for DKA resolution, on sub-group analysis, the time was significantly shorter in the severe DKA group (P = 0.0024). Singhal et al[8], in their RCT included 50 children, and compared NS vs RL. The children in this trial were bit older (till 18 years age). The time taken for resolution of DKA was not significantly different between the two groups, although a shorter duration was noted with RL. Williams et al[7], in their RCT (SPinK Trial) included 66 children, and compared NS vs Plasma-Lyte (PL)[7]. Again, they did not find any difference between the two groups in the time to resolution of DKA. In addition, no significant difference was found for other outcomes (mortality, AKI, duration of hospital stay). The finding of no difference between BES and NS has also been vetted by studies on adults with DKA[16,17]. Chua et al[15], included 23 adults with DKA, and compared NS vs PL[16]. No significant difference was found between the two groups, although a shorter time was noted with PL. Van Zyl et al[16], included 57 adults with DKA, and compared NS vs RL, but there was no inter-group difference in the time to resolution of DKA[17].
In recent years, there has been an intense debate going on the choice of fluid (BES or NS) in the initial resuscitation of critically ill children. Many pediatric adult trials have shown unequivocal results with some showing BES being better than NS, and some showing no difference between the two[5,6,18]. However, a consistent trend has been found between NS and two complications across majority of these trials-hyperchloremia, and AKI (new onset or progressive)[5,6,18]. In the trial mentioned above including the present trial on DKA, these two complications were commoner with NS, although the difference was not statistically significant. However, the delayed resolution of DKA in NS group could explain this as the hyperchloremia and underlying AKI prolong resolution of acidosis. The same is mitigated in the BES group as bicarbonate is present in addition to a lower chloride concentration. The later was shown in the trial by Agarwal et al[9].
We could not find any significant difference in the normalization of blood glucose or anion gap, rate of insulin administration, total fluid volume, cerebral edema or duration of hospital stay. Similar findings were noted in other pediatric and adult studies on DKA[3,7-9,15,16].
The limitations of present study include the following: (1) Small sample size, and single center study; (2) The trial was not registered; (3) The study findings may not be generalized to primary and secondary health care centers; and (4) Inability to do blood ketone level to correlate the resolution of DKA with these values.
This trial did not find any significant difference between BES and NS (0.9%) in the initial resuscitation of children with DKA. A trend towards higher incidences of hyperchloremia, AKI, and prolonged hospitalization in then NS group indicates than BES might be more suitable for DKA management in children. Large, multi-center trials are needed to explore these findings and provide recommendation on their use in children with DKA.
| 1. | Kostopoulou E, Sinopidis X, Fouzas S, Gkentzi D, Dassios T, Roupakias S, Dimitriou G. Diabetic Ketoacidosis in Children and Adolescents; Diagnostic and Therapeutic Pitfalls. Diagnostics (Basel). 2023;13:2602. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 22] [Reference Citation Analysis (3)] |
| 2. | Glaser N, Fritsch M, Priyambada L, Rewers A, Cherubini V, Estrada S, Wolfsdorf JI, Codner E. ISPAD clinical practice consensus guidelines 2022: Diabetic ketoacidosis and hyperglycemic hyperosmolar state. Pediatr Diabetes. 2022;23:835-856. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 185] [Article Influence: 46.3] [Reference Citation Analysis (0)] |
| 3. | Kuppermann N, Ghetti S, Schunk JE, Stoner MJ, Rewers A, McManemy JK, Myers SR, Nigrovic LE, Garro A, Brown KM, Quayle KS, Trainor JL, Tzimenatos L, Bennett JE, DePiero AD, Kwok MY, Perry CS 3rd, Olsen CS, Casper TC, Dean JM, Glaser NS; PECARN DKA FLUID Study Group. Clinical Trial of Fluid Infusion Rates for Pediatric Diabetic Ketoacidosis. N Engl J Med. 2018;378:2275-2287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 187] [Cited by in RCA: 147] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 4. | Fernández-Sarmiento J, Casas-Certain C, Ferro-Jackaman S, Solano-Vargas FH, Domínguez-Rojas JÁ, Pilar-Orive FJ. A brief history of crystalloids: the origin of the controversy. Front Pediatr. 2023;11:1202805. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 5. | Sankar J, Muralidharan J, Lalitha AV, Rameshkumar R, Pathak M, Das RR, Nadkarni VM, Ismail J, Subramanian M, Nallasamy K, Dev N, Kumar UV, Kumar K, Sharma T, Jaravta K, Thakur N, Aggarwal P, Jat KR, Kabra SK, Lodha R. Multiple Electrolytes Solution Versus Saline as Bolus Fluid for Resuscitation in Pediatric Septic Shock: A Multicenter Randomized Clinical Trial. Crit Care Med. 2023;51:1449-1460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 26] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 6. | Sankar J, Das RR, Banothu KK. Fluid resuscitation in children with severe infection and septic shock: a systematic review and meta-analysis. Eur J Pediatr. 2024;183:3925-3932. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 7. | Williams V, Jayashree M, Nallasamy K, Dayal D, Rawat A. 0.9% saline versus Plasma-Lyte as initial fluid in children with diabetic ketoacidosis (SPinK trial): a double-blind randomized controlled trial. Crit Care. 2020;24:1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 174] [Cited by in RCA: 286] [Article Influence: 47.7] [Reference Citation Analysis (0)] |
| 8. | Singhal D, Gupta S, Kumar V. Normal Saline Versus Ringer's Lactate for Intravenous Fluid Therapy in Children with Diabetic Ketoacidosis: A Randomized Controlled Trial. Indian J Pediatr. 2024. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 9. | Agarwal A, Jayashree M, Nallasamy K, Dayal D, Attri SV. 0.9% Saline versus Ringer's lactate as initial fluid in children with diabetic ketoacidosis: a double-blind randomized controlled trial. BMJ Open Diabetes Res Care. 2025;13:e004623. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 10. | Liu Y, Zhang J, Xu X, Zou X. Comparison of balanced crystalloids versus normal saline in patients with diabetic ketoacidosis: a meta-analysis of randomized controlled trials. Front Endocrinol (Lausanne). 2024;15:1367916. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 11. | Patino-Galarza D, Duque-Lopez A, Cabra-Bautista G, Calvache JA, Florez ID. Fluids in the treatment of diabetic ketoacidosis in children: A systematic review. J Evid Based Med. 2024;17:317-328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 12. | Holliday MA, Segar WE. The maintenance need for water in parenteral fluid therapy. Pediatrics. 1957;19:823-832. [PubMed] |
| 13. | Kellum JA, Lameire N; KDIGO AKI Guideline Work Group. Diagnosis, evaluation, and management of acute kidney injury: a KDIGO summary (Part 1). Crit Care. 2013;17:204. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2260] [Cited by in RCA: 2054] [Article Influence: 158.0] [Reference Citation Analysis (1)] |
| 14. | Yung M, Letton G, Keeley S. Controlled trial of Hartmann's solution versus 0.9% saline for diabetic ketoacidosis. J Paediatr Child Health. 2017;53:12-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 32] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 15. | Chua HR, Venkatesh B, Stachowski E, Schneider AG, Perkins K, Ladanyi S, Kruger P, Bellomo R. Plasma-Lyte 148 vs 0.9% saline for fluid resuscitation in diabetic ketoacidosis. J Crit Care. 2012;27:138-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 90] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 16. | Van Zyl DG, Rheeder P, Delport E. Fluid management in diabetic-acidosis--Ringer's lactate versus normal saline: a randomized controlled trial. QJM. 2012;105:337-343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 92] [Article Influence: 6.6] [Reference Citation Analysis (1)] |
| 17. | Hammond DA, Lam SW, Rech MA, Smith MN, Westrick J, Trivedi AP, Balk RA. Balanced Crystalloids Versus Saline in Critically Ill Adults: A Systematic Review and Meta-analysis. Ann Pharmacother. 2020;54:5-13. [PubMed] [DOI] [Full Text] |
| 18. | Bergmann KR, Abuzzahab MJ, Nowak J, Arms J, Cutler G, Christensen E, Finch M, Kharbanda A. Resuscitation With Ringer's Lactate Compared With Normal Saline for Pediatric Diabetic Ketoacidosis. Pediatr Emerg Care. 2021;37:e236-e242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/