Published online Mar 9, 2026. doi: 10.5409/wjcp.v15.i1.111093
Revised: July 27, 2025
Accepted: November 7, 2025
Published online: March 9, 2026
Processing time: 256 Days and 20.2 Hours
There is a limited number of studies on the efficacy and safety of dual-targeted therapy (DTT) in children with inflammatory bowel diseases (IBD); the latest reports focus on the combination of biological drugs with Janus kinase inhibitors (JAKI).
To evaluate the efficacy and safety of DTT in children with IBD.
This retrospective cohort study included 18 IBD patients, aged 2-17 years, with 13 (72%) having Crohn's disease (CD) and 5 (28%) having ulcerative colitis, who were treated with DTT. In two children, two different combinations of DTT were used sequentially. The data on clinical and laboratory changes were assessed at the beginning of DTT and at three months (M3) and six months (M6). Fecal cal
A total of 20 courses of DTT in 18 patients were included in the analysis: (1) 12 (60%) were treated with JAKI and vedolizumab; (2) 6 (30%) with JAKI and us
A DTT may be considered for children with refractory IBD. This strategy was shown to be most effective among children with a very early onset of the disease.
Core Tip: We conducted a single-center retrospective cohort study on 18 children with inflammatory bowel disease who received dual-targeted therapy (DTT) with biologics and Janus kinase inhibitors. Clinical remission was achieved in 9 out of 20 (45%) and 13 out of 20 (65%) DTT courses at three months and six months (M6), respectively. In contrast, laboratory and endoscopic remission were achieved in 8 out of 20 (40%) and 9 out of 17 (53%) treatment courses, respectively, within M6 of treatment initiation. Predictors of poor response to DTT were perianal lesions and upper gastrointestinal tract involvement in Crohn’s disease. Early onset of inflammatory bowel diseases was associated with higher remission rates.
- Citation: Gabrusskaya T, Ulanova N, Shilova E, Volkova N, Kilina S, Kornienko E, Kostik M. Efficacy and safety of dual-targeted therapy in children with inflammatory bowel disease: Retrospective cohort study. World J Clin Pediatr 2026; 15(1): 111093
- URL: https://www.wjgnet.com/2219-2808/full/v15/i1/111093.htm
- DOI: https://dx.doi.org/10.5409/wjcp.v15.i1.111093
Biological drugs (BD) are one of the main treatment strategies for inflammatory bowel diseases (IBD), also in children. Currently, tumor necrosis factor-alpha (TNF-α) inhibitors are a single class of BD approved for use in pediatric patients. However, the high rate of primary or secondary inefficacy of anti-TNF-α has prompted pediatricians worldwide to utilize other classes of biologics, including vedolizumab (VD), ustekinumab (UST), and synthetic target drugs, such as Janus kinase inhibitors (JAKI).
However, some patients fail to achieve sustained remission[1,2]. Patients with poor response to more than one BD with different mechanisms of action are currently considered patients with difficult-to-treat IBD[3]. Dual-targeted therapy (DTT) - a combination of BD or a combination of BD with JAKI is one of the possible management strategies for this group.
In the first randomized trial confirming DTT efficacy and safety, natalizumab or placebo was added to patients with an incomplete response to infliximab (INFL). The co-administration of natalizumab and INFL showed superiority over INFL monotherapy[4].
As new types of BD and small molecules become available, the range of potential treatment combinations is expanding. Currently, there are no well-defined regimens or data on the comparative efficacy of different DTTs. Notably, the least reported combination is BD with JAKI. Our study aimed to evaluate the efficacy and safety of DTT in children with IBD.
The retrospective cohort study included 18 patients with IBD aged 2-17 years treated in Gastroenterology Department of Saint-Petersburg State Pediatric Medical University between 2022 and 2024. The diagnosis of Crohn's disease (CD) and ulcerative colitis (UC) was established based on the Porto criteria[5]. All patients had an active course of the disease despite standard therapy, which required the administration of DTT. Inclusion and exclusion criteria are shown in Table 1.
| Inclusion criteria | Exclusion criteria |
| Clinical or laboratory activity of IBD | Monogenic form of IBD requiring specific therapy (such as bone marrow transplantation, etс.) |
| Endoscopic activity | |
| History of ineffectiveness or intolerance of tumor necrosis factor-alpha blockers as monotherapy or in combination with immunosuppressors, and at least one more biological drugs with a different mechanism |
All drugs were prescribed with an induction course, including JAKI. The doses of anti-TNF drugs were administered according to the instructions. The dose of VD for children weighing more than 30 kg was 300 mg, and for children weighing less than 30 kg, it was 10 mg/kg. The administration regimen for UST in children weighing more than 30 kg was similar to that for adults. All children who received UST weighed more than 30 kg. The most challenging aspect for pediatricians is administering JAKI in children. Children weighing more than 30 kg received JAKI at the recommended adult dose. For children weighing less than 30 kg, the dose was calculated individually. The doses for each child are listed in Table 2.
| ID | Gender | Disease | Location of CD | Location of UC | Perianal disease | Extraintestinal involvement | Previous therapy | Current combination | Dose | Age of onset (years) | Age of beginning dual-targeted therapy (years) |
| 1 | M | CD | L3 | - | No | No | 5-ASA, GCS, AZA, INFL, UP | UP + VD | UP: 30 mg/day 12 weeks, then 15 mg/day. VD: 10 mg/kg/8 weeks | 2 | 5 |
| 2 | F | CD | L2 | - | Yes | Yes | GCS, AZA, MTX, INFL, VD | VD + TOF/UP + UST | VD: 300 mg/6 weeks. TOF: 20 mg/day 8 weeks, then 10 mg/day. UP: 45 mg/day 12 weeks, then 30 mg/day. UST: 90 mg/4 weeks | 11 | 12 |
| 3 | M | UC | - | Pancolitis | - | Yes | 5-ASA, GCS, AZA, MTX, INFL, ADA, VD | VD + TOF | VD: 300 mg/6 weeks. TOF: 20 mg/day 8 weeks, then 10 mg/day | 7 | 10 |
| 4 | M | UC | - | Pancolitis | - | No | 5-ASA, GCS, AZA, INFL, UP | UP + VD | UP: 45 mg/day 8 weeks, then 30 mg/day. VD: 300 mg/8 weeks | 11 | 13 |
| 5 | M | CD | L3 | - | Yes | No | 5-ASA, GCS, AZA, MTX, INFL, ADA, TOF | TOF + VD | TOF: 20 mg/day 8 weeks, then 10 mg/day. VD: 300 mg/6 weeks | 13 | 15 |
| 6 | F | CD | L3+L4 | - | No | No | Enteral nutrition, 5-ASA, GCS, TAC, SRL, INFL, ADA, VD, TOF | VD + UP | VD: 300 mg/6 weeks. UP: 45 mg/day 12 weeks, then 15 mg/day | 2 | 12 |
| 7 | M | UC | - | Left-sided | - | No | 5-ASA, GCS, AZA, INFL, VD, TOF | TOF + ADA | TOF: 5 mg/day. ADA induction (40-80 mg), then 40 mg/2 weeks | 2 | 2 |
| 8 | M | CD | L2 | - | Yes | Yes | GCS, AZA, TAC, INFL, ADA, UST | UST + UP | UST: 90 mg/4 weeks. UP: 45 mg/day 12 weeks, then 30 mg/day | 8 | 15 |
| 9 | F | UC | - | Pancolitis | - | Yes | 5-ASA, GCS, AZA, INFL, VD | VD + UP | VD: 300 mg/6 weeks. UP: 45 mg/day 8 weeks, then 15 mg/day | 6 | 13 |
| 10 | F | UC | - | Left-sided | - | No | 5-ASA, GCS, AZA, TAC, INFL, ADA | ADA + UP | ADA: 40 mg/2 weeks. UP: 30 mg/day 8 weeks, then 15 mg/day | 2 | 6 |
| 11 | M | CD | L3 | - | No | No | GCS, AZA, INFL, ADA, UP | UP + UST | UP: 45 mg/day 12 weeks, then 30 mg/day. UST: 90 mg/8 weeks | 14 | 16 |
| 12 | M | CD | L2 | - | No | Yes | GCS, AZA, MTX, INFL, VD, TOF | VD + UP | VD: 300 mg/4 weeks. UP: 45 mg/day 12 weeks, then 15 mg/day | 13 | 15 |
| 13 | F | CD | L3 + L4 | - | No | No | 5-ASA, GCS, AZA, MTX, INFL, ADA, golimumab, TAC, VD, TOF | VD + TOF/TOF + UST | VD: 10 mg/kg/6 weeks. TOF: 20 mg/day 8 weeks, then 5 mg/day. UST: 90 mg/8 weeks | 2 | 11 |
| 14 | F | CD | L3 + L4 | - | No | No | 5-ASA, GCS, INFL, AZA, TAC, SRL, VD | VD + TOF | VD: 10 mg/kg/6 weeks. TOF: 15 mg/day 8 weeks, then 10 mg/day | 1 | 6 |
| 15 | M | CD | L2 | - | No | Yes | 5-ASA, GCS, AZA, MTX, TAC, mycophenolate mofetil, INFL, ADA, UST | UST + UP | UST: 90 mg/4 weeks. UP: 45 mg/day 12 weeks, then 15 mg/day | 5 | 17 |
| 16 | M | CD | L3 | - | No | No | 5-ASA, GCS, AZA, MTX, TAC, INFL, ADA, VD | VD +UP | VD: 300 mg/4 weeks. UP: 45 mg/day 12 weeks, then 15 mg/day | 5 | 12 |
| 17 | F | CD | L2 + L4 | - | Yes | No | GCS, AZA, MTX, INFL, ADA, UST | UST + UP | UST: 90 mg/8 weeks. UP: 45 mg/day 12 weeks, 15 mg/day | 3 | 17 |
| 18 | M | CD | L3 | - | No | No | GCS, AZA, MTX, INFL, ADA, VD | VD + TOF | VD: 10 mg/kg/6 weeks. TOF: 10 mg/day | 1 | 8 |
In every patient, the following parameters were assessed thrice: Before the DTT initiation (baseline, BL), at three months (M3), and six months (M6): Determined at BL and M6.
Clinical parameters include the Pediatric Crohn’s Disease Activity Index (PCDAI) and the Pediatric Ulcerative Colitis Activity Index (PUCAI)[6,7].
Clinical remission (CR) was defined as a decrease in PCDAI to 10 points or less in children with CD and a PUCAI score of less than 10 in patients with UC.
Endoscopic examination (colonoscopy and esophagogastroduodenoscopy) was performed before the start of therapy and at M6. We used the simple endoscopic index of CD (SES-CD) in patients with CD and the Mayo endoscopic index of UC activity in patients with UC[8,9].
Adverse events (AEs) that occurred during DTT were recorded. An adverse event was considered serious if it required withdrawal of therapy and/or hospitalization. The schematic design of the study is presented in Table 3.
| Baseline | At three months | At six months | |
| Pediatric Crohn’s Disease Activity Index (in Crohn’s disease) | + | + | + |
| Pediatric Ulcerative Colitis Activity Index (in ulcerative colitis) | + | + | + |
| Endoscopy | + | - | + |
| Hemoglobin | + | + | + |
| Erythrocyte sedimentation rate | + | + | + |
| C-reactive protein | + | + | + |
| Fecal calprotectin | + | - | + |
The sample was not calculated initially. Statistical analysis was performed using the StatTech v. 4.8.0 program (developer: StatTech LLC, Russia). Quantitative indicators were evaluated for conformity to normal distribution using the Shapiro-Wilk criterion. Quantitative indicators whose sample distribution conformed to a normal distribution were described using the arithmetic mean and SD. Quantitative data were described using the median and lower and upper quartiles (Q1 to Q3) if there was no normal distribution. Categorical data were described with absolute values and percentages. The Wilcoxon matched-pairs test was used to compare the quantitative indicators whose distributions differed from usual in two related groups.
Student's t-criterion was used for the comparison of two groups of the quantitative indicators, if the distribution of which corresponded to normal; otherwise, the Mann-Whitney U-criterion was applied. Comparison of percentages in the analysis of four-field contingency tables was performed using the χ2 test or Fisher's exact test (in cases where the expected frequency was less than 5). Differences were considered statistically significant at P < 0.05.
Eighteen children, with an average age at diagnosis of five years, participated in the study. The group consisted of 13 children (72%) with CD, five children (28%) with UC, and included seven girls (39%) and 11 boys (61%). Most patients with CD (n = 13; 79%) had an inflammatory form of the disease. Eight patients (61.5%) had ileocolitis, and five patients (38.5%) had colitis. Endoscopic examination was performed in all patients at BL, and erosions or ulcers were found in 100% of cases. Upper gastrointestinal tract (UGT) involvement was found in 4/13 (31%) of children with CD, and 4/13 (31%) had perianal involvement. In the UC group, there was a slight predominance of children with pancolitis (60%) compared to left-sided colitis (40%). Extraintestinal manifestations (EIMs) were observed in 6/18 (33%) children, with the most frequent being arthritis (n = 4; 22%), fever (n = 5; 28%), lung lesions (n = 3; 17%), and growth delay (n = 3; 17%). Surgical treatment was previously required in 4/13 (31%) CD patients; three out of four patients had surgery related to perianal lesions (PL), and one had colonic stenosis resection.
A relatively large group of children with a very early onset (VEO) of the disease (younger than 6 years of age) accounted for 61% of our study. The proportion of children with infantile-onset was 38.9%. Patients’ characteristics are presented in Table 2.
All patients had a history of ineffectiveness of 16/18 (89%) or intolerance of 2/18 (11%) of TNF-α blockers. The in
Besides, there was a group with severe steroid dependence in the study: (1) Five patients received GCS for more than M6; and (2) The average duration of their intake in this group was 4.60 ± 1.14 years.
All patients received DTT with BD and JAKI. Twelve (60%) received a combination of JAKI and VD, 6 (30%) received JAKI and UST, and 2 (10%) received JAKI and adalimumab (ADA). Two children received two different DTT combinations sequentially, resulting in twenty courses of DTT being evaluated in eighteen children. Specifically, 12 children (60%) received upadacitinib (UP), and 8 (40%) received tofacitinib (TOF).
The overall rate of CR of DTT at M3 and M6 was 45% (9/20) and 68% (13/20), respectively. CR was observed in 71% (10/14) of CD patients and 60% (3/5) of UC patients at M6. A significant decrease in the mean PCDAI was observed in the CD group under DTT: From 15 points at BL to 10 points at M3 and 5 points at M6 (P < 0.001).
There was no significant reduction in the average PUCAI in the UC group during follow-up, which is likely due to the small sample size (n = 5). By M6 of treatment in 2 patients (10.5%), DTT was discontinued due to the development of a serious adverse event in one case and an increase in disease activity that necessitated a change in therapy in the other patient.
During the study, GCS was discontinued in 4/7 (57%) cases at M3 and in 7/7 (100%) patients (100%) who were taking GCS at BL by M6.
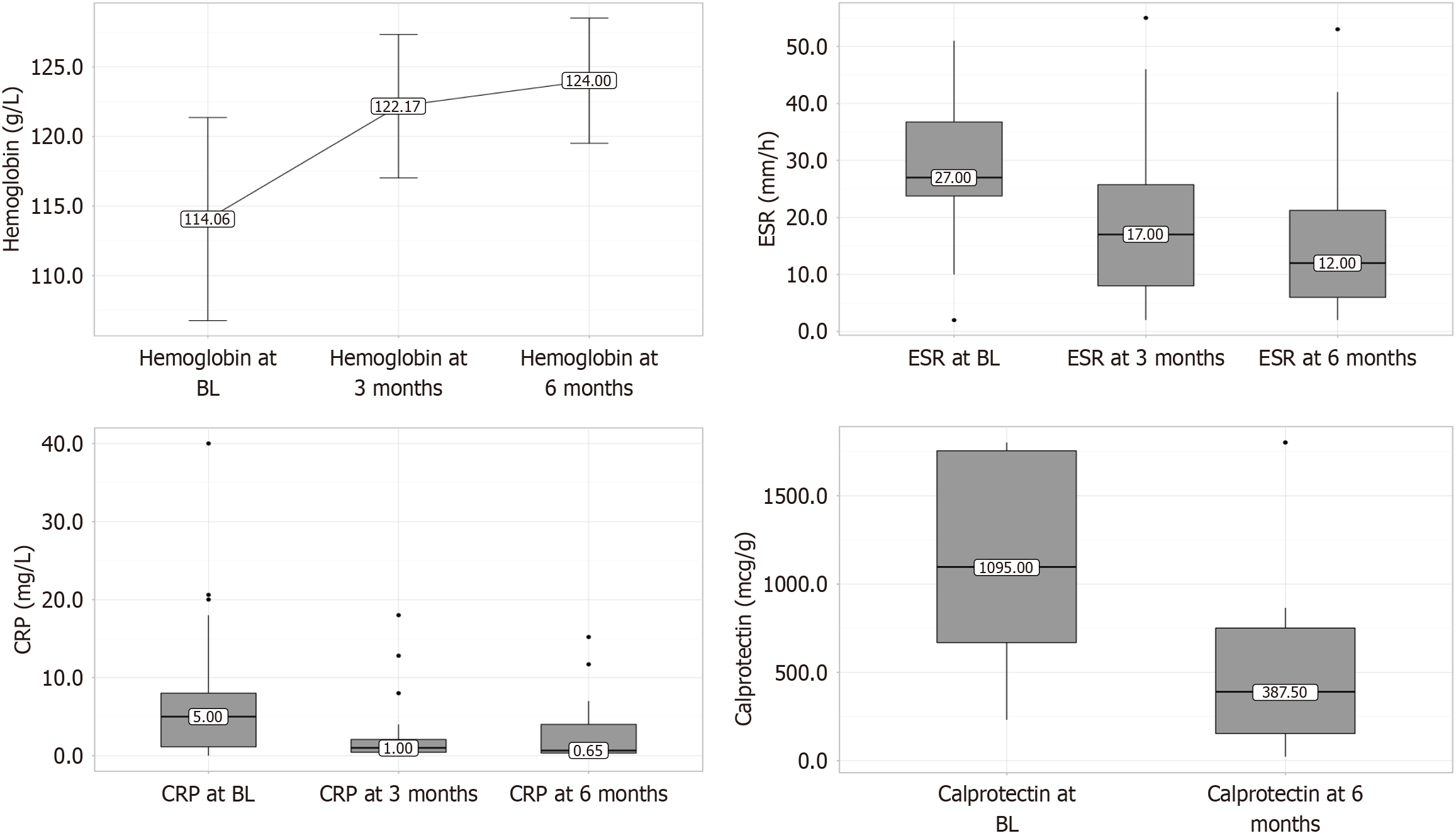
The markers of inflammation significantly decreased during the study from BL to the M3 and M6 of the study: Ery
Laboratory remission, which was considered as normalization of the three leading indices (CRP, ESR, hemoglobin) and decrease in the level of calprotectin below 200 microgram/g in M6 after the start of DTT was achieved in 45% of patients, while normalization of CRP, ERS, and hemoglobin at M6 - in 67% of patients.
Follow-up endoscopy control at M6 was performed in 17/20 patients (85%), and three children did not undergo control endoscopic examination: (1) Two patients had discontinued therapy earlier; and (2) The parents of one patient refused the procedure. Endoscopic remission (ER), interpreted as the complete absence of erosive and ulcerative elements, was achieved in 9/17 (53%) of patients at M6: In 6/14 (42.9%) of CD patients and 3/3 (100%) of UC patients who underwent control endoscopy after M6.
A significant decrease in mean SES-CD levels was observed in CD patients M6 after DTT initiation compared to BL: 8 vs 2, respectively (P = 0.005).
The decrease in the values of the Mayo endoscopic activity index was not statistically significant, which is most likely due to the small sample size of UC patients in the study (n = 5); the endoscopic examination was performed at M6 in only three children with UC.
Boys were significantly more likely (n = 10/11; 90.9%) to achieve CR on DTT compared to girls (n = 3/8; 37.5%) at M6 (P = 0.024), without differences at the M3 time point.
No significant disparities were detected in laboratory parameters, including ESR, CRP, and FC levels at the six-month follow-up.
There were no differences in the rate of clinical and laboratory remission at M3 or M6, as well as no differences in CRP, ESR, and FC levels between CD and UC groups were observed.
PL and UGT involvement were negatively associated with the efficacy of the therapy in CD patients. Thus, patients with PL had higher CRP levels at both M3 and M6 compared to patients without PL: (1) 1.4 mg/L vs 0.65 mg/L (P = 0.035); and (2) 0.4 mg/L vs 1.0 mg/L (P = 0.027), respectively.
Higher FC levels at M6 were also found in patients with PL compared to patients without PL: 600 microgram/g vs 158 microgram/g (P = 0.254). Only 1/5 (20%) patients with PL achieved ER at endoscopy at M6, compared to 5/9 (55.6%) without PL (P = 0.301).
CR at M6 was diagnosed in 1/5 (20%) patients with UGT involvement compared to 9/9 (100%) without UGT lesions (P = 0.006). The mean PIACD at M3 and M6 was significantly higher in patients with UGT involvement compared to children without it: (1) 14.2 vs 6.6 (P = 0.040); and (2) 11.5 vs 3.6 (P = 0.016), respectively.
Children with UGT involvement had higher mean ESR levels at M3 and M6 months and a trend towards higher FC at M6 compared to children without such lesions, similar to a lower rate of ER [1/5 (20%) vs 5/9 (55.6%); P = 0.301], respectively (Table 4). The presence of UGT lesions was negatively associated with SES-CD values at M6 (Table 4).
| Parameter | Upper gastrointestinal tract lesions | P value | |
| No | Yes | ||
| Нb at M3, median (IQR) | 127.0 (122.0-134.0) | 120.5 (117.8-121.0) | 0.020a |
| ESR at M3, median (SD) | 10.8 (6.8) | 28.5 (12.6) | 0.004a |
| CRP at M3, median (IQR) | 1.0 (0.8-1.4) | 0.8 (0.3-9.9) | 0.905 |
| Hb at M6, median (IQR) | 129.0 (121.0-134.0) | 121.0 (116.0-124.0) | 0.109 |
| ESR at M6, median (IQR) | 6.0 (6.0-11.0) | 19.0 (16.0-42.0) | 0.019a |
| CRP at M6, median (IQR) | 0.6 (0.20-1.0) | 1.0 (0.5-11.7) | 0.283 |
| Fecal calprotectin at M6, median (IQR) | 175.0 (95.0-600.0) | 600.0 (155.0-800.0) | 0.421 |
| simple endoscopic index of CD at M6, median (IQR) | 0.0 (0.0-2.0) | 10.0 (6.0-10.0) | 0.042a |
| Pediatric Crohn’s Disease Activity Index at M6, mean (SD) | 3.6 (3.3) | 11.5 (7.4) | 0.016a |
The most significant differences were observed in analyzing the response to therapy based on the age of disease onset. In children with VEO of IBD (up to 6 years of age), CRP (0.45 mg/L vs 3.0 mg/L; P = 0.020) at M3, ESR (9 mm/hour vs 25.62 mm/hour (P = 0.022), and FC (152 microgram/g vs 799 microgram/g; P = 0.006) at M6 were significantly lower than in children with disease onset older than 6 years, respectively. The rate of laboratory remission not including FC (90% vs 37.5%; P = 0.043), laboratory remission including FC (80% vs 0%; P = 0.001) was significantly higher in patients with VEO IBD compared to children with onset older than 6 years, respectively, while ER was more often achieved in children with early onset of disease (before 10 years of age) compared with later onset (88.9% vs 62.5%; P = 0.050). Surprisingly, there were no differences in the rates of laboratory and ER tests depending on the age of DTT initiation or the time between diagnosis and DTT onset. Other variables, such as the form of the disease, location, and severe attacks in UC patients, disease duration, or EIMs, were not associated with clinical, laboratory, or ER.
AEs were reported in 6/20 (30%) of courses, with only 1 case (5%) involving a serious AE. This child developed a septic condition, and DTT was discontinued. In five patients, AEs were considered not serious: (1) Four patients had moderate cholesterol elevation; and (2) One child experienced an increased frequency of acute respiratory illnesses, requiring oral antibacterial therapy. There were no cases of activation of herpes virus infections, varicella zoster, Clostridial, or other opportunistic infections during this study.
Our study is one of the first studies to evaluate the safety and efficacy of the combined use of BD and JAKI in children with IBD. We demonstrated a relatively high level of clinical and ER at M6, with rates of 68% and 53%, respectively, and a relatively low rate of side effects, 30%. We have found that PL and UGT involvement may negatively affect DTT efficacy in CD patients. We have also shown for the first time that children with early onset of the disease are more likely to achieve clinical and ER compared with children with later onset.
The majority of published studies[10-13] have evaluated the effectiveness of combined biological therapy, and observations of BD with a JAKI combination are scarce. In all studies, the number of children receiving BD and JAKI was limited to 3-12 participants[14-16]. The most significant number of children treated with this scheme was reported in the Cohen et al’s study[11], where 75% (12/16) received a combination of BD and JAKI. All participants in this study received TOF as a JAKI[11], whereas in our work, TOF was administered in 8 out of 20 (40%) courses and UP in 12 out of 20 (60%) courses of DTT treatment.
All children who participated in our study were classified as difficult-to-treat patients at the time of inclusion, and all of them had failed more than two BD, which allowed us to categorize them as such.
Colectomy was a possible therapeutic strategy in some of them. In addition, seven (35%) patients were receiving GCS at the beginning of DTT, with five patients failing to discontinue GCS for more than a year.
CR was achieved in 45% of cases at M3 and 68% at M6 of DTT. By M6, in all patients, tapering GCS CR coincided with steroid-free remission. Our data on the clinical efficacy of combination therapy with JAKI and BD are very similar to those from the study by Dolinger et al[15], In which most children received a combination of JAKI and BD, achieving steroid-free CR at M6 in 75% of cases. The results of these two studies are slightly superior to those demonstrated in the most extensive multicenter study to date on DTT in children with IBD[14]. In the DOUBLE-PIBD study[14], the rates of CR were 35% at M3 and 50% at M6. However, it should be emphasized that in this study, the proportion of children on the combination of BD and JAKI was small (only three patients, 4.8%); the leading group consisted of patients on the combination of two BD[14].
A decrease of FC below 200 microgram/g is an important predictor of possible ER. After M6 of DTT, a reduction was observed in 50% of patients, which is slightly lower than the similar rate in the DOUBLE-PIBD study[14], where the frequency of such a decrease at M6 was 59.6%.
Of particular importance to our study is the fact that in 17 out of 20 (85%) cases, endoscopic follow-up has been performed M6 after DTT initiation. Mucosal healing is one of the most important goals in the therapy of patients of any age with IBD. The majority of participants in our observation (78%) had never achieved ER before DTT, dictating the need to change or intensify therapy. ER at M6 (complete absence of erosive and ulcerative elements on colonoscopy and upper endoscopy) was observed in 53% of patients. In all cases, it was accompanied by steroid-free CR.
The rate of ER in our observation was significantly higher than in the majority of adult DTT studies[17-19]. According to data from a meta-analysis evaluating the efficacy of DTT in adult patients, which included 279 patients, ER was found in only 34% of patients[20]. Data on ER in the pediatric DTT cohort are scarce. In the DOUBLE-PIBD study[14], endo
Out of 20 patients in our study, two children required a change of therapy due to an increase in clinical activity of the disease. In one case, a girl treated with TOF and UST developed a significant increase in clinical and laboratory activity. Therefore, DTT was canceled after M3. GCS was prescribed, followed by another DTT combination, including TOF and VD. In another child, DTT with TOF and VD was discontinued after M6 due to worsening of the disease, and the current DTT was replaced with UST and UP, with clinical improvement in M3. None of our patients on DTT required colectomy.
The identification of predictors of response to therapy is a topic of special interest. So far, there is currently no clear evidence in the literature about which patient group DTT will be most effective in. According to our data, while children with CD more often achieved CR (10/14), ER was found more often in UC patients on DTT (3/3). However, these differences were not statistically significant in both cases, which is most likely due to the small sample size of patients with UC (n = 5), and only 3 out of 5 people with UC underwent colonoscopy after M6. One patient with 1 UC had discontinued therapy earlier due to an adverse event, and the parents of one patient refused the procedure. Among patients with CD, the main risk factors of poor response to therapy were the presence of PL as well as UGT involvement. These groups of patients were less likely to achieve both clinical and ER and had higher laboratory indices of inflammation on DTT. The presence of both PL and UGT involvement is generally recognized as a predictor of an unfavorable course of CD[21]. In this group, the response to therapy with all known BD is significantly worse[22-26]. The impact of these factors on the effectiveness of JAKI therapy has been less extensively investigated. Multicenter international study by Cohen et al[11] revealed a trend towards lower rates of CR at 8 weeks of UP monotherapy in the presence of PL, although the difference did not reach statistical significance (P = 0.245). However, the presence of UGT involvement in this study did not affect the achievement of CR. There were no differences in response to therapy depending on the form of the disease, including the presence of UGT and PL, in the DOUBLE-PIBD study[14].
In our study, no differences in clinical and/or endoscopic response were observed between patients with colitis and those with ileocolitis. However, a better response to JAKI has been reported in patients with colitis compared to those with ileocolitis in previous studies[16]. Furthermore, no effect of EIMs on the efficacy of DTT has been demonstrated. However, in a meta-analysis of studies on dual biological therapy in adults, patients with EIMs achieved remission more frequently[20]. This difference is most likely because a significant number of adult patients (12%) had EIMs or concomitant rheumatologic disease as an indication for DTT[20], in contrast to our study, in which all patients had IBD activity as the indication for DTT.
Another significant feature of our observation was the presence of a large number of children with a VEO of IBD; their share in our study amounted to 61% (mean age of IBD onset was 5 years), as well as prescription of DTT in a large number of children younger than 10 years (n = 5; 25%). We showed that VEO of IBD (younger than 6 years) was the main predictor of CR, while children with early onset (younger than 10 years) were more likely to achieve ER: 89% of children with ER had early onset. Interestingly, no differences were observed in achieving clinical, laboratory, and endoscopic outcomes, regardless of the age at which DTT initiation occurred or the time elapsed between diagnosis and initiation of DTT. It is the first description of the association between age of onset and response to DTT in children. The most important predictor of therapy response is the disease phenotype, which differs in children with onset at different age intervals rather than the age and timing of DTT initiation.
For example, the prevalence of colitis in children with VEO, compared to onset at other age intervals, the prevalence of the inflammatory form, and the absence of complications such as stenosis and PL may contribute to a better response to DTT. Additionally, specific pathophysiological mechanisms may contribute to the early onset of IBD. The phenomenon of a good response to DTT in children with early onset, but not the time of initiation of DTT, requires further confirmation in a larger number of patients and an investigation of the factors influencing this phenomenon.
Our study showed acceptable safety of DTT in children with difficult-to-treat IBD. Side effects were recorded in 30% of treatment courses, but in the majority of cases, they were non-serious and did not require treatment withdrawal. Moderate hypercholesterolemia was detected in 5 of 20 treatment courses, with the highest expression occurring during the induction dose period, followed by a reduction that did not return to the BL cholesterol level. Dietary fibers and regular laboratory monitoring were carried out on these patients. One child had septic process development during DTT, which required a prescription of antibacterial therapy and de-escalation of immunosuppression. It should be noted that this was a boy with UC who received corticosteroids in addition to TOF and ADA and had a central venous catheter due to infusion therapy and partial parenteral nutrition requirement. ADA therapy was discontinued, and treatment with TOF was continued, along with antibacterial therapy. The catheter was also replaced, resulting in stabilization of the condition and further development of CR. No episodes of CMV, Clostridial infection, or herpes virus infection activation were recorded in our study.
There are several limitations to our study. Firstly, it is a retrospective single-center study. The major limitation of our observation is a relatively small sample size, particularly in the UC group (n = 5), which does not allow for extrapolation of all the data and correlations obtained to the general child population with IBD. Second, the M6 is a short period to fully evaluate the long-term efficacy and safety of the DTT. Larger studies with more extended periods of observation are needed to confirm the findings. Special attention should be paid to the reasonable duration of DTT. Thirdly, a significant number of children were taking GCS at the beginning of DTT. Although in all cases, the GCS dose was stable at the time of initiation, and none of the patients required dose escalation of GCS during the study, we still suspect that concomitant steroids in 35% of patients may have influenced the efficacy of DTT in our study.
A combination of JAKI and BD may be considered in the treatment of children with difficult-to-treat IBD. This therapy was shown to be effective and relatively safe in this patient group. This strategy was shown to be most effective among children with a VEO of the disease.
| 1. | Hyams J, Crandall W, Kugathasan S, Griffiths A, Olson A, Johanns J, Liu G, Travers S, Heuschkel R, Markowitz J, Cohen S, Winter H, Veereman-Wauters G, Ferry G, Baldassano R; REACH Study Group. Induction and maintenance infliximab therapy for the treatment of moderate-to-severe Crohn's disease in children. Gastroenterology. 2007;132:863-73; quiz 1165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 722] [Cited by in RCA: 642] [Article Influence: 33.8] [Reference Citation Analysis (4)] |
| 2. | Hyams JS, Griffiths A, Markowitz J, Baldassano RN, Faubion WA Jr, Colletti RB, Dubinsky M, Kierkus J, Rosh J, Wang Y, Huang B, Bittle B, Marshall M, Lazar A. Safety and efficacy of adalimumab for moderate to severe Crohn's disease in children. Gastroenterology. 2012;143:365-74.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 254] [Cited by in RCA: 227] [Article Influence: 16.2] [Reference Citation Analysis (3)] |
| 3. | Parigi TL, D'Amico F, Abreu MT, Dignass A, Dotan I, Magro F, Griffiths AM, Jairath V, Iacucci M, Mantzaris GJ, O'Morain C, Reinisch W, Sachar DB, Turner D, Yamamoto T, Rubin DT, Peyrin-Biroulet L, Ghosh S, Danese S. Difficult-to-treat inflammatory bowel disease: results from an international consensus meeting. Lancet Gastroenterol Hepatol. 2023;8:853-859. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 77] [Article Influence: 25.7] [Reference Citation Analysis (0)] |
| 4. | Sands BE, Kozarek R, Spainhour J, Barish CF, Becker S, Goldberg L, Katz S, Goldblum R, Harrigan R, Hilton D, Hanauer SB. Safety and tolerability of concurrent natalizumab treatment for patients with Crohn's disease not in remission while receiving infliximab. Inflamm Bowel Dis. 2007;13:2-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 163] [Article Influence: 8.6] [Reference Citation Analysis (3)] |
| 5. | Levine A, Koletzko S, Turner D, Escher JC, Cucchiara S, de Ridder L, Kolho KL, Veres G, Russell RK, Paerregaard A, Buderus S, Greer ML, Dias JA, Veereman-Wauters G, Lionetti P, Sladek M, Martin de Carpi J, Staiano A, Ruemmele FM, Wilson DC; European Society of Pediatric Gastroenterology, Hepatology, and Nutrition. ESPGHAN revised porto criteria for the diagnosis of inflammatory bowel disease in children and adolescents. J Pediatr Gastroenterol Nutr. 2014;58:795-806. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1195] [Cited by in RCA: 1085] [Article Influence: 90.4] [Reference Citation Analysis (4)] |
| 6. | Turner D, Griffiths AM, Walters TD, Seah T, Markowitz J, Pfefferkorn M, Keljo D, Waxman J, Otley A, LeLeiko NS, Mack D, Hyams J, Levine A. Mathematical weighting of the pediatric Crohn's disease activity index (PCDAI) and comparison with its other short versions. Inflamm Bowel Dis. 2012;18:55-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 246] [Article Influence: 17.6] [Reference Citation Analysis (3)] |
| 7. | Turner D, Ruemmele FM, Orlanski-Meyer E, Griffiths AM, de Carpi JM, Bronsky J, Veres G, Aloi M, Strisciuglio C, Braegger CP, Assa A, Romano C, Hussey S, Stanton M, Pakarinen M, de Ridder L, Katsanos KH, Croft N, Navas-López VM, Wilson DC, Lawrence S, Russell RK. Management of Paediatric Ulcerative Colitis, Part 2: Acute Severe Colitis-An Evidence-based Consensus Guideline From the European Crohn's and Colitis Organization and the European Society of Paediatric Gastroenterology, Hepatology and Nutrition. J Pediatr Gastroenterol Nutr. 2018;67:292-310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 221] [Cited by in RCA: 182] [Article Influence: 22.8] [Reference Citation Analysis (0)] |
| 8. | Daperno M, D'Haens G, Van Assche G, Baert F, Bulois P, Maunoury V, Sostegni R, Rocca R, Pera A, Gevers A, Mary JY, Colombel JF, Rutgeerts P. Development and validation of a new, simplified endoscopic activity score for Crohn's disease: the SES-CD. Gastrointest Endosc. 2004;60:505-512. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1538] [Cited by in RCA: 1408] [Article Influence: 64.0] [Reference Citation Analysis (3)] |
| 9. | Schroeder KW, Tremaine WJ, Ilstrup DM. Coated oral 5-aminosalicylic acid therapy for mildly to moderately active ulcerative colitis. A randomized study. N Engl J Med. 1987;317:1625-1629. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2583] [Cited by in RCA: 2342] [Article Influence: 60.1] [Reference Citation Analysis (9)] |
| 10. | Guo C, Zhou J, Wang G, Wu J. Dual Biologic or Small Molecule Therapy in Pediatric Inflammatory Bowel Disease: A Single Center Experience. Children (Basel). 2025;12:75. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 11. | Cohen S, Spencer EA, Dolinger MT, Suskind DL, Mitrova K, Hradsky O, Conrad MA, Kelsen JR, Uhlig HH, Tzivinikos C, Henderson P, Wlazlo M, Hackl L, Shouval DS, Bramuzzo M, Urlep D, Olbjørn C, D'Arcangelo G, Pujol-Muncunill G, Yogev D, Kang B, Gasparetto M, Rungoe C, Kolho KL, Hojsak I, Norsa L, Rinawi F, Sansotta N, Rimon RM, Granot M, Scarallo L, Trindade E, Rodríguez-Belvís MV, Turner D, Yerushalmy-Feler A. Upadacitinib for Induction of Remission in Paediatric Crohn's Disease: An International Multicentre Retrospective Study. Aliment Pharmacol Ther. 2025;61:1372-1380. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 17] [Article Influence: 17.0] [Reference Citation Analysis (1)] |
| 12. | Runde J, Ryan K, Hirst J, Lebowitz J, Chen W, Brown J, Strople J. Upadacitinib is associated with clinical response and steroid-free remission for children and adolescents with inflammatory bowel disease. J Pediatr Gastroenterol Nutr. 2025;80:133-140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 13. | Yerushalmy-Feler A, Spencer EA, Dolinger MT, Suskind DL, Mitrova K, Hradsky O, Conrad MA, Kelsen JR, Uhlig HH, Tzivinikos C, Ancona S, Wlazlo M, Hackl L, Shouval DS, Bramuzzo M, Urlep D, Olbjorn C, D'Arcangelo G, Pujol-Muncunill G, Yogev D, Kang B, Gasparetto M, Rungø C, Kolho KL, Hojsak I, Norsa L, Rinawi F, Sansotta N, Magen Rimon R, Granot M, Scarallo L, Trindade E, Velasco Rodríguez-Belvís M, Turner D, Cohen S. Upadacitinib for Induction of Remission in Pediatric Ulcerative Colitis: An International Multicenter Study. J Crohns Colitis. 2025;19:jjae182. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 14. | Yerushalmy-Feler A, Olbjorn C, Kolho KL, Aloi M, Musto F, Martin-de-Carpi J, Lozano-Ruf A, Yogev D, Matar M, Scarallo L, Bramuzzo M, de Ridder L, Kang B, Norden C, Wilson DC, Tzivinikos C, Turner D, Cohen S. Dual Biologic or Small Molecule Therapy in Refractory Pediatric Inflammatory Bowel Disease (DOUBLE-PIBD): A Multicenter Study from the Pediatric IBD Porto Group of ESPGHAN. Inflamm Bowel Dis. 2024;30:159-166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 32] [Article Influence: 16.0] [Reference Citation Analysis (1)] |
| 15. | Dolinger MT, Spencer EA, Lai J, Dunkin D, Dubinsky MC. Dual Biologic and Small Molecule Therapy for the Treatment of Refractory Pediatric Inflammatory Bowel Disease. Inflamm Bowel Dis. 2021;27:1210-1214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 71] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 16. | Spencer EA, Bergstein S, Dolinger M, Pittman N, Kellar A, Dunkin D, Dubinsky MC. Single-center Experience With Upadacitinib for Adolescents With Refractory Inflammatory Bowel Disease. Inflamm Bowel Dis. 2024;30:2057-2063. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 33] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 17. | Yang E, Panaccione N, Whitmire N, Dulai PS, Vande Casteele N, Singh S, Boland BS, Collins A, Sandborn WJ, Panaccione R, Battat R. Efficacy and safety of simultaneous treatment with two biologic medications in refractory Crohn's disease. Aliment Pharmacol Ther. 2020;51:1031-1038. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 126] [Cited by in RCA: 121] [Article Influence: 20.2] [Reference Citation Analysis (5)] |
| 18. | Colombel JF, Ungaro RC, Sands BE, Siegel CA, Wolf DC, Valentine JF, Feagan BG, Neustifter B, Kadali H, Nazarey P, James A, Jairath V, Qasim Khan RM. Vedolizumab, Adalimumab, and Methotrexate Combination Therapy in Crohn's Disease (EXPLORER). Clin Gastroenterol Hepatol. 2024;22:1487-1496.e12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 76] [Article Influence: 38.0] [Reference Citation Analysis (3)] |
| 19. | Glassner K, Oglat A, Duran A, Koduru P, Perry C, Wilhite A, Abraham BP. The use of combination biological or small molecule therapy in inflammatory bowel disease: A retrospective cohort study. J Dig Dis. 2020;21:264-271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 63] [Article Influence: 10.5] [Reference Citation Analysis (3)] |
| 20. | Ahmed W, Galati J, Kumar A, Christos PJ, Longman R, Lukin DJ, Scherl E, Battat R. Dual Biologic or Small Molecule Therapy for Treatment of Inflammatory Bowel Disease: A Systematic Review and Meta-analysis. Clin Gastroenterol Hepatol. 2022;20:e361-e379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 142] [Article Influence: 35.5] [Reference Citation Analysis (3)] |
| 21. | van Rheenen PF, Aloi M, Assa A, Bronsky J, Escher JC, Fagerberg UL, Gasparetto M, Gerasimidis K, Griffiths A, Henderson P, Koletzko S, Kolho KL, Levine A, van Limbergen J, Martin de Carpi FJ, Navas-López VM, Oliva S, de Ridder L, Russell RK, Shouval D, Spinelli A, Turner D, Wilson D, Wine E, Ruemmele FM. The Medical Management of Paediatric Crohn's Disease: an ECCO-ESPGHAN Guideline Update. J Crohns Colitis. 2021;15:jjaa161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 472] [Cited by in RCA: 439] [Article Influence: 87.8] [Reference Citation Analysis (3)] |
| 22. | Gu B, Venkatesh K, Williams AJ, Ng W, Corte C, Gholamrezaei A, Ghaly S, Xuan W, Paramsothy S, Connor S. Higher infliximab and adalimumab trough levels are associated with fistula healing in patients with fistulising perianal Crohn's disease. World J Gastroenterol. 2022;28:2597-2608. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 6] [Cited by in RCA: 28] [Article Influence: 7.0] [Reference Citation Analysis (2)] |
| 23. | Zulqarnain M, Deepak P, Yarur AJ. Therapeutic Drug Monitoring in Perianal Fistulizing Crohn's Disease. J Clin Med. 2022;11:1813. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 24. | Yarur A, Mantzaris GJ, Wang S, Adsul S, Kamble P, Cook E, Sajeev G, Guerin A, Bressler B. Stratified Patient Profiling for Vedolizumab Effectiveness in Crohn's Disease: Identifying Optimal Subgroups for Enhanced Treatment Response in the EVOLVE Study. Adv Ther. 2024;41:2324-2341. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 25. | Takenaka K, Hisamatsu T, Kobayashi T, Yamamoto H, Ohmiya N, Hayashi R, Matsuoka K, Saruta M, Andoh A, Fukata N, Watanabe K, Imaeda H, Hirai F, Matsumoto T, Hirakawa A, Yano T, Matsuura M, Okamoto R, Ohtsuka K, Watanabe M. Inadequate Efficacy of Biologics for Treating Proximal Ileal Lesions in Crohn's Disease: A Prospective Multicenter Study. Clin Gastroenterol Hepatol. 2025;23:1991-2000. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 10] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 26. | Yu WL, Hua ZC. Efficacy and prognosis of adalimumab for Crohn's disease at different disease locations: a systematic review and meta-analysis. Eur Rev Med Pharmacol Sci. 2022;26:2036-2048. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/