Published online Mar 9, 2026. doi: 10.5409/wjcp.v15.i1.111066
Revised: August 8, 2025
Accepted: October 30, 2025
Published online: March 9, 2026
Processing time: 257 Days and 14.3 Hours
Infantile colic (IC) is a common, self-limiting condition marked by excessive, un
To systematically review and meta-analyze the effectiveness of eHFs in managing symptoms of IC.
A comprehensive literature search of MEDLINE, Cochrane and Web of Science databases (1980-2025) identified randomized controlled trials and crossover stu
Six studies met the inclusion criteria, with a total of 340 infants across intervention and comparator arms. In all studies, eHF significantly reduced crying duration and colic symptoms compared to regular formula or dicy
eHF appear significantly more effective than standard formulas and pharmacologic comparators in reducing colic-related symptoms. These findings support eHF as a viable dietary intervention for IC, though larger, well-controlled trials are warranted to confirm long-term outcomes.
Core Tip: Infantile colic is a distressing yet common condition in early infancy. This systematic review and meta-analysis is the first to specifically evaluate the role of extensively hydrolyzed formulas (eHF) in its dietary management. Synthesizing data from six randomized controlled trials, we found that eHF significantly reduces crying duration and improves colic-related symptoms compared to standard formula or pharmacologic interventions. These findings support eHF as an effective and safe dietary option for formula-fed infants with colic, especially in cases where cow's milk protein sensitivity is suspected.
- Citation: Dodos K, Kalamara TV, Xinias S, Mavroudi A, Kapoukranidou D, Sarantaki A, Diamanti A, Georgakopoulou VE. Effectiveness of the extensively hydrolyzed formulas in the management of infantile colic: A systematic review and meta-analysis. World J Clin Pediatr 2026; 15(1): 111066
- URL: https://www.wjgnet.com/2219-2808/full/v15/i1/111066.htm
- DOI: https://dx.doi.org/10.5409/wjcp.v15.i1.111066
Infantile colic (IC) is the term used to describe a group of signs and behaviors in which protracted crying, discomfort, and fussiness are prevalent. During these episodes, infants may also exhibit leg raising, gas passing, hip bending, and fist gripping. Another distinguishing feature of all these symptoms is that they affect infants who are typically healthy and well-fed, with the crying being excessive, unexplained, and occurring in the late afternoon to early evening[1-6]. It is generally recognized as a functional gastrointestinal condition with a benign prognosis, but it can have substantial and long-lasting implications on the relationship between the infant and parents as well as the standard of living in the home[7].
After undergoing a chemical, enzymatic, or thermal process that attempts to hydrolyze caseins and whey proteins, extensively hydrolyzed milk formula (eHF) is created from regular formula[8]. Due to the rising incidence of cow milk allergies in the western world, the use of eHF has become widely accepted in contemporary pediatrics[9].
Considering that IC represents a common clinical condition affecting a substantial proportion of infants, the evaluation of therapeutic strategies such as eHF holds significant clinical relevance. In recent years, numerous studies have investigated the role of eHF in managing colic symptoms, consistently demonstrating a reduction in crying duration and overall distress compared to standard cow’s milk formula or other interventions[10-15]. Other approaches, such as probiotics, soymilk formulas, or partially hydrolyzed and hypoallergenic formulas, have also been assessed in meta-analyses, but their efficacy remains controversial and inconclusive[5,6].
To date, no systematic reviews or meta-analyses have been conducted that specifically address the potential role of hydrolyzed milk formulas in the dietary management of IC. The main objective of this systematic review and meta-analysis is to assess whether the use of eHF in infants with symptoms of colic is effective in reducing the severity and frequency of the symptoms (crying, fussiness, discomfort, etc.) that are associated with IC when compared to normal formula or placebo.
The study was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines[15]. This systematic review has been registered in the International Prospective Register of Systematic Reviews with ID number CRD420251078293.
The research question was developed using the Population, Intervention, Comparator, Outcome framework, which provides a structured and systematic method for framing clinical inquiries. The population includes infants diagnosed with IC. The intervention being assessed is the use of eHF. The comparators involve placebo, standard milk formulas, exclusive maternal milk, dicyclomine, or any other relevant alternative. The outcome focuses on the reduction in the severity and frequency of symptoms associated with IC, including excessive crying, fussiness, and discomfort.
Accordingly, the full research question can be formulated as follows: Does the use of extensively hydrolyzed milk formulas in colicky infants reduce the severity of symptoms associated with the condition, such as crying, fussiness, and discomfort, when compared to placebo, standard milk formulas, or other therapeutic interventions?
A comprehensive systematic search was conducted in the primary electronic databases MEDLINE (via PubMed), the Cochrane Central Register of Controlled Trials and Web of Science due to their extensive coverage of biomedical and clinical research, covering the publication period from 1980 to January 2025. Grey literature sources were also included to ensure a more exhaustive evidence base. Additionally, the reference lists of all eligible studies were manually screened to identify any further relevant trials. The PubMed search strategy combined both Medical Subject Headings (MeSH) and free-text terms, utilizing Boolean operators and a validated randomized controlled trial (RCT) filter to optimize both sensitivity and precision.
The IC component of the search included the terms: [“infant* colic*” (MeSH Terms) OR “infant* colic*” OR “baby colic*” OR “newborn colic*”]. For the intervention, the search included the terms: “extensively hydrolyzed formula” OR “eHF” OR “extensively hydrolyzed milk formula” OR “whey hydrolysate” OR “casein hydrolysate” OR “hypoallergic milk formula” OR “infant formula” OR “dietary management” OR “dietary intervention”.
The RCT filter incorporated a combination of publication types and title/abstract keywords as follows: Randomized controlled trial (pt), controlled clinical trial (pt), randomized (tiab), placebo (tiab), clinical trials as topic (mesh: Noexp), randomly (tiab), and trial (ti). These terms were combined using the OR operator and then filtered to exclude animal-only studies [animals (mh) NOT humans (mh)]. The final search strategy combined the IC search terms, the hydrolyzed formula search terms, and the refined RCT filter.
All records retrieved through this search strategy were imported into reference management software (Mendeley) for screening, deduplication, and further evaluation.
Eligible studies included either RCTs or randomized crossover trials. To be considered for inclusion, studies had to meet predefined criteria regarding population, intervention, comparator, and outcome. The population of interest comprised infants aged six months or younger, with no prior diagnosis of gastrointestinal or neurological disorders, who were either exclusively or partially formula-fed. Furthermore, included infants were required to have a formal diagnosis of IC based on Wessel’s criteria, modified Wessel’s criteria, or the ROME III diagnostic framework.
The intervention under investigation was the administration of eHF, which could be either casein-based or whey protein-based. Studies assessing additional interventions were eligible only if they incorporated a clearly delineated arm in which eHF was the primary intervention.
Acceptable comparators included standard cow’s milk-based infant formulas, exclusive maternal breastfeeding, pharmacological agents such as dicyclomine, or any other relevant comparator groups. The primary outcome was the change in average daily crying duration. Secondary outcomes included changes in other IC-associated symptoms such as fussiness and apparent discomfort.
Studies were excluded if they enrolled infants who were not formally diagnosed using the specified diagnostic criteria (e.g., were only described as “colicky”), or involved infants with unclear or undefined comorbid health conditions. No sample size restrictions were applied. Additional exclusion criteria were applied to studies involving infants older than six months or studies published in languages other than English.
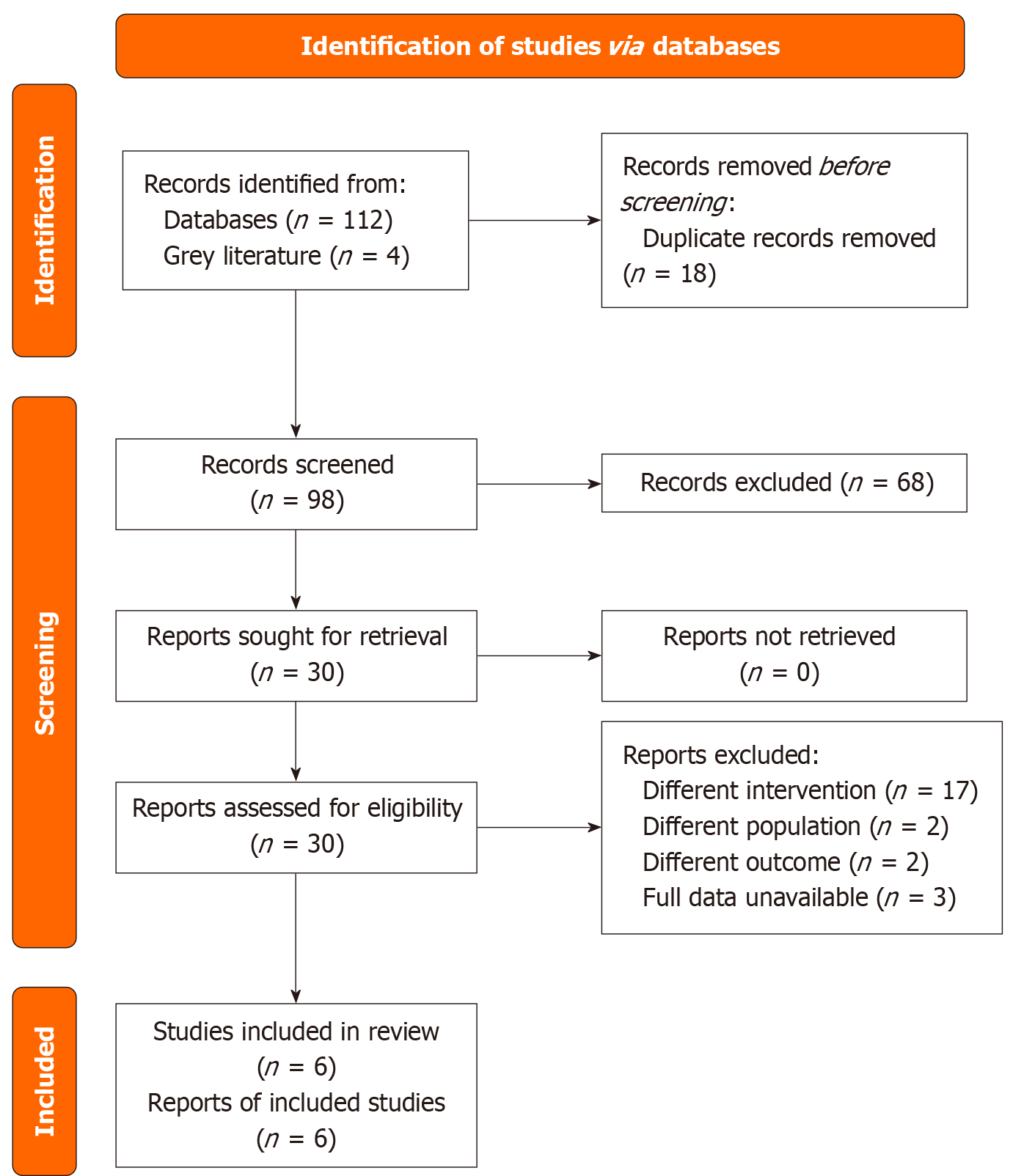
We performed a search for articles published in the electronic databases PubMed, Cochrane Library, Web of Science from their inception up to January 2025. There were no restrictions regarding sample size or study setting. We applied MeSH terms for both the intervention (eHF) and the diagnosis (IC), along with free-text keywords. Boolean operators “OR” and “AND” were utilized as well. We also used an RCT filter to ensure that all available RCTs are detected. As shown in the corresponding PRISMA flow diagram (Figure 1), our search strategy retrieved 116 results in total. After deduplication, we initially screened 98 records at the title and abstract level. Of these, 68 records were excluded. The remaining 30 records underwent a detailed full-text assessment. At this point, 24 studies were excluded due to different intervention, different population or outcome mismatch. Six studies were eligible for inclusion in the qualitative synthesis and meta-analysis[10-14,16].
All the included studies were critically assessed in terms of their quality according to the Cochrane’s Collaboration Risk of Bias Tool for randomized trials[17]. Two independent researchers used standardized forms in order to extract key study characteristics regarding study design, sample size, population characteristics, intervention, comparator and outcome. Discrepancies between the reviewers were solved via discussion. The quality assessment results can be summarized in Figure 2. Assessment of methodological quality revealed variability across studies. Adequate random sequence generation was reported in the studies conducted by Hill et al[10], Lucassen et al[11], and Xinias et al[12], while Forsyth[16], Jakobsson et al[14], and Oggero et al[13] provided insufficient detail to permit judgment. Allocation concealment was inadequately described in all trials, resulting in an unclear risk for selection bias. Blinding of participants and personnel was adequate in four studies (Forsyth[16], Hill et al[10], Oggero et al[13], Lucassen et al[11]) but absent or insufficiently described in Jakobsson et al[14] and Xinias et al[12]. Blinding of outcome assessors was achieved in Hill et al[10], Lucassen et al[11], and Xinias et al[12], whereas the remaining trials lacked this safeguard. Incomplete outcome data and selective reporting were generally unclearly reported, with insufficient information on attrition per group and reasons for exclusion. All studies were rated as unclear for “other bias”, as no additional risks were explicitly described but insufficient information was available to rule them out.
Data was independently extracted by two reviewers using a standardized extraction form. The information collected included study characteristics (authors, publication year, country, and study design), participant details (sample size, age range, and diagnostic criteria for IC), intervention specifics (type of eHF and duration), comparator details, and outcome measures. Primary outcomes focused on changes in crying duration, while secondary outcomes included fussiness, discomfort, and parental quality of life. Any discrepancies between reviewers were resolved through discussion or consultation with a third reviewer. Extracted data were recorded in an electronic database for subsequent analysis.
Both qualitative and quantitative synthesis methods were applied. Where possible, data from eligible studies were pooled in a meta-analysis using a random-effects model to account for heterogeneity. Dichotomous outcomes were reported as odds ratios (ORs) with 95% confidence intervals (CIs), and continuous outcomes (e.g., crying duration) were analyzed using mean differences or standardized mean differences. Studies with crossover designs were analyzed with attention to phase sequence effects, as their lack of independent controls limits direct comparability to parallel trials. Statistical heterogeneity was assessed using the I2 statistic, with values of 0%-25%, 25%-50%, and 50%-75% representing low, moderate, and high heterogeneity, respectively. In cases where meta-analysis was not feasible due to data limitations or significant clinical heterogeneity, a narrative synthesis was provided. Sensitivity analyses were also performed, where applicable, by excluding studies at high risk of bias.
All statistical analyses were conducted using Review Manager (RevMan) version 5.4, developed by the Cochrane Collaboration.
A total of six trials were included in this systematic review[10-14,16], each evaluating the effectiveness of eHFs in the management of IC in otherwise healthy, term infants diagnosed using standardized criteria such as Wessel’s or ROME III. Despite variations in design, sample size, intervention duration, and comparator arms, the studies consistently de
| Ref. | Age | Intervention | Comparator | Outcome | Main results |
| Hill et al[10], 1995 | 4-16 weeks | 19 infants -hydrolysate formula (casein based) | 19 infants -regular cow's milk formula | Crying time reduction | 61% positive in intervention (12/19)- 43% in comparator (8/19) |
| Lucassen et al[11], 2000 | < 6 months | 20 infants -hydrolysate formula (whey based) | 18 infants - regular cow's milk formula | Crying time reduction | 13/20 positive in the intervention group - 6/18 in the comparator |
| Xinias et al[12], 2017 | 20-60 days | 40 infants - hydrolysate formula (whey based) | 20 infants -regular cow's milk formula | Crying time reduction | 40/40 positive in intervention group - 13/20 in the comparator |
| Oggero et al[13], 1994 | 3-12 weeks | 15 infants -Hydrolysate formula | 60 infants - dicyclomine | Crying time reduction | 13/15 positive in intervention group - 34/60 in comparator group |
| Jakobsson et al[14], 2000 | 2-8 weeks | 14 infants -hydrolysate formula (CH1-CH2) | Same infants (13) (another time period) - Regular cow's milk formula | Decrease in crying time | 11/14 positive in intervention group - 2/13 in comparator group |
| Forsyth[16], 1989 | 0-8 weeks | 17 infants -Hydrolysate formula | 17 infants - regular cow's milk formula | Decrease in crying time | 20/51 in intervention group (out of a total of 51 formula changes from regular cow milk formula to eHF, 20 had a favorable result) - 7/51 in comparator group |
Hill et al[10] studied formula-fed infants aged 4 to 16 weeks and observed a 117-minute reduction in crying time in the eHF group, compared to a 46-minute reduction in the standard formula group. A general improvement in distress was observed in 61% of the intervention group vs 43% in controls, with an OR of 2.12 (95%CI: 1.01-4.46). Moreover, a ≥ 25% reduction in distress occurred more frequently in the eHF group (39% vs 16%; P = 0.012). Age under six weeks was identified as a significant predictor of treatment response (OR: 2.36; 95%CI: 1.01-5.5).
In the trial by Lucassen et al[11], 20 infants receiving whey hydrolysate formula experienced a 63-minute greater reduction in crying time than the 18 infants receiving standard formula (95%CI: 1-127 minute/day). Multivariate analysis revealed that baseline crying time and gender were significant predictors of response. Although the study was limited by a small sample size and limited parental blinding, the findings supported eHF efficacy.
Xinias et al[12] enrolled 60 infants aged 20-60 days and found a mean crying time reduction of 2.7 hours in the intervention group, compared to 1.2 hours in the control group (P < 0.001). A significant 1.5-hour average difference in favor of eHF was observed. While stool composition became waterier in the eHF group, urination frequency remained unchanged. Quality of life parameters, including sleep, work productivity, and parent-child interaction—improved more significantly in the intervention group.
Oggero et al[13] compared eHF to dicyclomine in a cohort of 75 infants and reported that 86.6% (13/15) of infants in the eHF group achieved a reduction in crying time to less than one hour per day, vs 56% (34/60) in the dicyclomine group (P = 0.01). The intervention period lasted 30 days, and a hospital-based 48-hour pre-evaluation phase ensured standardized baseline data.
Jakobsson et al[14] assessed the effect of two different eHF formulations (CH1 and CH2) in a crossover design involving 22 infants aged 2-8 weeks. At least a 30% reduction in crying time was achieved in 15 infants (P = 0.01), with average crying time falling from 6.04-8.68 hours at baseline to 1.81-3.63 hours post-intervention. The nutritional impact of both formulas was deemed equivalent, though stool composition became waterier.
Forsyth et al[16] employed a multiple crossover design with alternating periods of eHF and standard formula. Among 51 formula changes, 20 resulted in positive outcomes when infants were switched to eHF, whereas only 7 positive responses were observed when switching to standard formula (P = 0.05). Notably, crying increased during standard formula periods and decreased with eHF, reinforcing its therapeutic benefit.
Overall, this qualitative synthesis indicates that eHF consistently outperformed standard formulas and even pharmacological agents like dicyclomine in reducing crying duration and improving related symptoms such as fussiness and discomfort. Improvements in stool characteristics and caregiver-reported quality of life were also more favorable in the eHF groups. While methodological limitations such as small sample sizes and variable comparator arms were noted, the cumulative evidence strongly supports the use of eHF in the dietary management of IC.
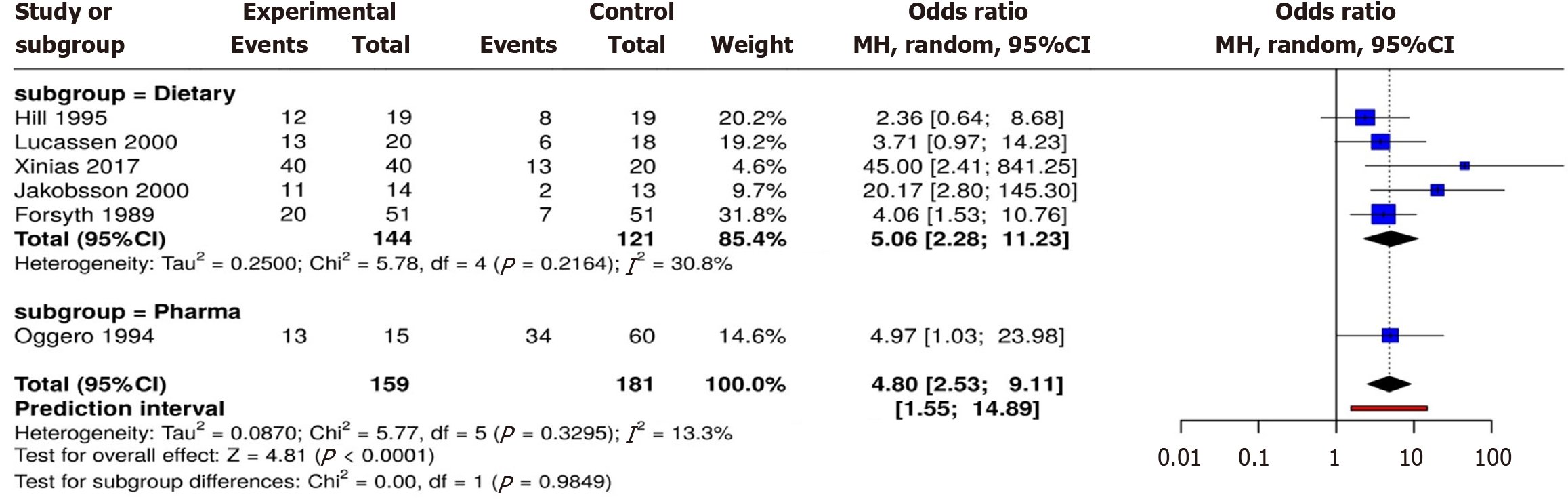
All six research projects were included in the meta-analysis. As indicated by each study individually in comparison to a comparator (regular milk formula, dicyclomine), we sought to summarize the findings of the studies in this part and compute the OR of eHF in successfully minimizing the symptoms connected to IC. According to Hill et al[10], 12/19 participants in the intervention group and 8/19 in the comparator group had successful outcomes. According to Luccasen et al[11], 13/20 samples from the intervention group and 6/18 from the comparator showed good results. According to Oggero et al[13], 13/15 participants in the intervention group and 34/60 in the control group achieved positive results. According to Jakobsson et al[14], 11/14 of the infants in the intervention group and 2/13 of the newborns in the comparison group had successful outcomes. According to Forsyth[16], out of a total of 51 formula changes from regular cow milk formula to eHF, 20 had a favorable result, while out of 51 formula changes from eHF to standard cow formula, there were 7. Finally, Xinias et al[12], employed an intervention group of 40 infants, all of whom showed a decrease in crying time, and a comparator group of 20 infants, only 13 of whom showed a favorable outcome.
Subgroup analysis was conducted regarding the type of comparator. The first subgroup contained five studies that utilized dietary type comparator (standard milk formula) and the second subgroup contained one study that used pharmacological type comparator (dicyclomine). A meta-analysis of six studies, comprising 159 subjects in the experimental cohort (eHF) and 181 in control (comparator) cohort, revealed a statistically significant difference between the two groups. Using a random effects model with the Mantel-Haenszel method, the overall OR was 4.8 (95%CI: 2.53-9.11, P < 0.05), indicating a strong association in favor of the experimental cohort. No significant heterogeneity was detected, suggesting consistent effect sizes across studies in both magnitude and direction. These findings support a robust and uniform treatment effect. Lastly, the test for subgroup differences was negative (χ2 = 0, P = 0.9). Additionally, a sensitivity analysis was conducted to assess the robustness of the primary findings by excluding the Forsyth[16] study from the pooled analysis. The rationale for this exclusion was that this study employed a multiple-crossover design, yielding repeated observations from the same infants, which may violate the independence assumption in meta-analysis and potentially inflate precision and effect size estimates. In this adjusted model, five studies were analyzed, comprising a total of 108 participants in the experimental cohort and 130 participants in the comparison cohort. Using a random-effects model with the Mantel-Haenszel method, the overall OR was 5.57 (95%CI: 2.29-13.55), indicating a statistically significant difference between the two groups (P < 0.05). Importantly, no significant heterogeneity was observed, suggesting that the effect sizes were consistent in both magnitude and direction across the included studies. These results demonstrate that the exclusion of the study by Forsyth[16] did not materially alter the conclusions of the meta-analysis, supporting the stability and reliability of the overall effect estimate (Figure 3).
This systematic review and meta-analysis evaluated the therapeutic efficacy of eHF in infants formally diagnosed with IC. Across six studies, consistent evidence emerged showing that eHF significantly reduced crying duration and improved associated symptoms such as fussiness and discomfort. The meta-analysis demonstrated a pooled odds ratio of 4.8 (95%CI: 2.53-9.11, P < 0.05), indicating a markedly greater likelihood of clinical improvement in infants receiving eHF compared to those receiving standard formula or dicyclomine. Heterogeneity across studies was low (I2 = 13.3%). The inclusion of crossover trials, while valuable for within-participant comparisons, precludes definitive conclusions about absolute efficacy due to the absence of independent control groups. Overall, the findings support the superiority of eHF over conventional feeding approaches in the dietary management of IC.
The proposed mechanisms by which eHF formulas exert their effects include the presence of peptides that are more easily digestible, potentially reducing gastrointestinal irritation. The hydrolysis process diminishes the allergenic potential of proteins, which is particularly relevant for infants with milk protein sensitivity[16,18]. Furthermore, eHF may positively influence gut microbiota composition, fostering a healthier intestinal environment that mitigates symptoms associated with IC[19].
From a clinical perspective, the consistent improvement in crying time and reduction in distress observed across studies suggest that eHF may represent a significant and helpful intervention for formula-fed infants with IC. This is particularly relevant in cases where cow’s milk protein sensitivity is suspected but not definitively diagnosed. Compared to pharmacologic treatments such as dicyclomine, which have been associated with potential adverse effects—including serious anticholinergic reactions, eHF offers a non-pharmacologic alternative with a favorable safety and tolerability profile[13,16]. Thus, current evidence demonstrates short-term efficacy (≤ 30 days) of eHF for infant colic, though long-term safety data remain unavailable. Clinical decisions should weigh this temporary benefit against cost and availability. eHFs are specialized, higher-cost products typically reserved for cow’s milk allergy, but their demonstrated benefit in reducing colic symptoms may justify use in severe or refractory cases. Clinicians should weigh this against accessibility and family resources. Additionally, no serious adverse events were reported with eHF use. While one study[12] noted waterier stools in the eHF group, a known effect of hydrolysates due to their rapid absorption[13], this was not clinically harmful. Overall, eHFs appear well-tolerated in infants with colic.
While eHFs represent a premium-priced therapeutic option primarily indicated for cow's milk protein allergy, their demonstrated efficacy in alleviating severe infant colic symptoms may warrant consideration in refractory cases. Clinicians should carefully evaluate the risk-benefit ratio for individual patients, taking into account symptom severity and impact on family wellbeing, local formulary availability and reimbursement policies, and the family's socioeconomic capacity to maintain this intervention if continued use proves necessary. Particularly in cases where standard interventions have failed and parental distress is significant, a time-limited eHF trial (2-4 weeks) with predefined response criteria may represent a clinically justified option despite the cost premium.
No serious adverse events were reported with eHF use. While one study noted waterier stools in the eHF group, a known effect of hydrolysates due to their rapid absorption, this was not clinically harmful. Overall, eHFs appear well-tolerated in infants with colic.
These findings are consistent with and build upon prior literature. Previous systematic reviews and meta-analyses examining alternative dietary interventions—such as probiotics, soy-based formulas, or partially hydrolyzed formulas—have reported inconsistent or modest benefits in the management of IC[5,6,19,20]. In contrast, the present review emphasizes the more robust and consistent clinical efficacy of extensively hydrolyzed formulas. Notably, studies that directly compared eHF with dicyclomine[13] or with standard formulas[10-12,14,16] demonstrated the clear superiority of eHF in reducing crying episodes and overall distress in colicky infants.
While the meta-analysis presents compelling evidence supporting the efficacy of eHFs, several limitations warrant consideration. The methodological quality of the included studies varied significantly, with some studies exhibiting small sample sizes and inadequate blinding, potentially introducing bias into the results. Most of the included studies assessed short-term outcomes, leaving a gap in knowledge regarding the long-term effects of eHF formulas on growth, development, and subsequent gastrointestinal health. The studies encompassed a diverse range of infant populations with varying degrees of severity in IC symptoms. This heterogeneity can limit the generalizability of the findings, as the effectiveness of eHFs may be context-dependent. Variations in the definition of IC and the criteria used for diagnosis across studies may have impacted the consistency of results. A standardized approach to defining and diagnosing IC is essential for future research.
Moreover, due to the limited number of studies (< 10), we did not perform formal publication bias analyses such as funnel plots or Egger’s test, as these methods are underpowered in small datasets. Nevertheless, the possibility of undetected publication bias, particularly the non-publication of small negative trials, cannot be excluded and should be considered when interpreting our findings.
An important limitation of our meta-analysis relates to the definition of “successful reduction of IC symptoms”. Each included study applied its own criteria to define treatment success. For example, Hill et al[10] defined improvement as a ≥ 25% reduction in distress episodes, Oggero et al[13] required crying time to fall below 1 hour per day, while Xinias et al[12] classified any measurable decrease in daily crying duration as a positive response. These varying thresholds introduce clinical heterogeneity into the pooled binary outcome, as “success” in one study may represent a substantially different magnitude of symptom change than in another.
While our analysis treated all definitions as equivalent for the purposes of pooling odds ratios, this approach assumes comparability across definitions, which may not fully reflect the underlying variability in effect size or patient-perceived benefit. Such heterogeneity could lead to either underestimation or overestimation of the true treatment effect.
Continuous outcomes, such as mean change in daily crying duration (minutes), would allow for more direct cross-trial comparison. Indeed, several included trials reported these data. However, due to inconsistent reporting formats (e.g., medians, ranges, non-parametric summaries) and missing variance estimates, a pooled analysis of continuous outcomes could not be conducted for all studies. Where available, continuous outcome data indicated a reduction in crying time consistent in direction with our primary binary outcome findings, suggesting that the overall effect of eHF is robust across outcome metrics.
Nevertheless, we caution that the variability in “treatment success” definitions across studies should be taken into account when interpreting the pooled OR. This limitation underscores the need for future RCTs to adopt standardized, validated criteria for assessing IC symptom improvement, ideally reporting both binary responder rates and continuous measures of crying time reduction.
Looking forward, several avenues warrant further investigation. Future research should prioritize larger, high-quality RCTs that employ standardized diagnostic criteria and consistent outcome measures. Such studies are essential for confirming the observed benefits of eHF and for evaluating its long-term impact on infant growth, development, and gastrointestinal health. In addition, exploring the underlying mechanisms of action—such as the influence of eHF on gut microbiota composition, immune modulation, and reduction of allergenic potential—could provide valuable insights into its efficacy. Investigations into the synergistic effects of combining eHF with complementary interventions, such as probiotics, also hold promise for enhancing treatment outcomes. Addressing these research gaps will ultimately contribute to the development of comprehensive, evidence-based clinical guidelines for the management of IC, thereby improving outcomes for affected infants and their families.
In conclusion, this systematic review and meta-analysis demonstrates that eHFs significantly reduce the severity and frequency of IC symptoms compared to standard formulas and other interventions. The evidence indicates that eHF is associated with considerable improvements in crying duration and overall infant comfort, thereby lessening the distress experienced by both infants and their caregivers. These findings support the therapeutic potential of eHF as an effective dietary management option for IC.
| 1. | Zhang D, Zhang Y, Sang Y, Zheng N, Liu X. The Relationship between Infant Colic and Migraine as well as Tension-Type Headache: A Meta-Analysis. Pain Res Manag. 2019;2019:8307982. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 2. | Icke S, Genc R. Effect of Reflexology on Infantile Colic. J Altern Complement Med. 2018;24:584-588. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 15] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 3. | Mai T, Fatheree NY, Gleason W, Liu Y, Rhoads JM. Infantile Colic: New Insights into an Old Problem. Gastroenterol Clin North Am. 2018;47:829-844. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 39] [Article Influence: 4.9] [Reference Citation Analysis (4)] |
| 4. | Gelfand AA, Buse DC, Cabana MD, Grimes B, Goadsby PJ, Allen IE. The Association Between Parental Migraine and Infant Colic: A Cross-Sectional, Web-Based, U.S. Survey Study. Headache. 2019;59:988-1001. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 5. | Sung V, D'Amico F, Cabana MD, Chau K, Koren G, Savino F, Szajewska H, Deshpande G, Dupont C, Indrio F, Mentula S, Partty A, Tancredi D. Lactobacillus reuteri to Treat Infant Colic: A Meta-analysis. Pediatrics. 2018;141:e20171811. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 183] [Cited by in RCA: 148] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 6. | Rivas-Fernández M, Diez Izquierdo A, Cassanello P, Balaguer A. Do probiotics help babies with infantile colic? Arch Dis Child. 2019;104:919-923. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 7. | Botha E, Joronen K, Kaunonen M. The consequences of having an excessively crying infant in the family: an integrative literature review. Scand J Caring Sci. 2019;33:779-790. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 20] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 8. | Inuo C, Tanaka K, Nakajima Y, Yamawaki K, Matsubara T, Iwamoto H, Tsuge I, Urisu A, Kondo Y. Tolerability of partially and extensively hydrolysed milk formulas in children with cow's milk allergy. Asia Pac J Clin Nutr. 2019;28:49-56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 9. | Ahern GJ, Hennessy AA, Ryan CA, Ross RP, Stanton C. Advances in Infant Formula Science. Annu Rev Food Sci Technol. 2019;10:75-102. [PubMed] [DOI] [Full Text] |
| 10. | Hill DJ, Hudson IL, Sheffield LJ, Shelton MJ, Menahem S, Hosking CS. A low allergen diet is a significant intervention in infantile colic: results of a community-based study. J Allergy Clin Immunol. 1995;96:886-892. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 70] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 11. | Lucassen PL, Assendelft WJ, Gubbels JW, van Eijk JT, Douwes AC. Infantile colic: crying time reduction with a whey hydrolysate: A double-blind, randomized, placebo-controlled trial. Pediatrics. 2000;106:1349-1354. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 84] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 12. | Xinias I, Analitis A, Mavroudi A, Roilides I, Lykogeorgou M, Delivoria V, Milingos V, Mylonopoulou M, Vandenplas Y. Innovative Dietary Intervention Answers to Baby Colic. Pediatr Gastroenterol Hepatol Nutr. 2017;20:100-106. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 13. | Oggero R, Garbo G, Savino F, Mostert M. Dietary modifications versus dicyclomine hydrochloride in the treatment of severe infantile colics. Acta Paediatr. 1994;83:222-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 25] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 14. | Jakobsson I, Lothe L, Ley D, Borschel MW. Effectiveness of casein hydrolysate feedings in infants with colic. Acta Paediatr. 2000;89:18-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 16] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 15. | Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, Moher D. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9803] [Reference Citation Analysis (0)] |
| 16. | Forsyth BW. Colic and the effect of changing formulas: a double-blind, multiple-crossover study. J Pediatr. 1989;115:521-526. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 67] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 17. | Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, Cates CJ, Cheng HY, Corbett MS, Eldridge SM, Emberson JR, Hernán MA, Hopewell S, Hróbjartsson A, Junqueira DR, Jüni P, Kirkham JJ, Lasserson T, Li T, McAleenan A, Reeves BC, Shepperd S, Shrier I, Stewart LA, Tilling K, White IR, Whiting PF, Higgins JPT. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22257] [Cited by in RCA: 20410] [Article Influence: 2915.7] [Reference Citation Analysis (7)] |
| 18. | Wilsey MJ, Florio J, Beacker J, Lamos L, Baran JV, Oliveros L, Sriaroon P, Brown JM, Vanderhoof JA. Extensively Hydrolyzed Formula Improves Allergic Symptoms in the Short Term in Infants with Suspected Cow's Milk Protein Allergy. Nutrients. 2023;15:1677. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 18] [Reference Citation Analysis (0)] |
| 19. | Beacker J, Brown JM, Florio J, Baran JV, Lamos L, Oliveros L, Vanderhoof JA, Sriaroon P, Wilsey MJ. Clinician Experience with Using Hypoallergenic Formulas to Treat Infants with Suspected Cow's Milk Protein Allergy: A Secondary Analysis of a Prospective Survey Cohort. Pediatr Gastroenterol Hepatol Nutr. 2023;26:277-283. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 20. | Kansu A, Urganci N, Bukulmez A, Kutluk G, Gulcu Taskin D, Sahin Keskin L, Igde M, Molon L, Dogan Y, Sekerel BE, Yuksek M, Bostanci I, Gerenli N, Polat E, Dalgic B, Ayyildiz H, Usta M, Basturk A, Yuce Kirmemis O, Tuna Kirsaclioglu C, Gulerman HF, Alptekin Sarioglu A, Erdogan S. Growth, tolerance and safety outcomes with use of an extensively hydrolyzed casein-based formula in infants with cow's milk protein allergy. Front Pediatr. 2023;11:1230905. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/