Published online Mar 9, 2026. doi: 10.5409/wjcp.v15.i1.109864
Revised: June 21, 2025
Accepted: October 23, 2025
Published online: March 9, 2026
Processing time: 286 Days and 19.8 Hours
The current 2020 surviving sepsis guideline recommends fluid boluses of 10-20 mL/kg/bolus up to 40-60 mL/kg, but recent studies showed better outcomes when fluid boluses were restricted.
To analyze the outcome of initial resuscitation of septic shock without giving fluid bolus and early initiation of inotropes.
In this open label, randomized controlled clinical trial conducted at the Pediatric Emergency Medicine unit of a tertiary care hospital in central India, patients aged 1 month to 14 years who were hospitalized for septic shock were randomly al
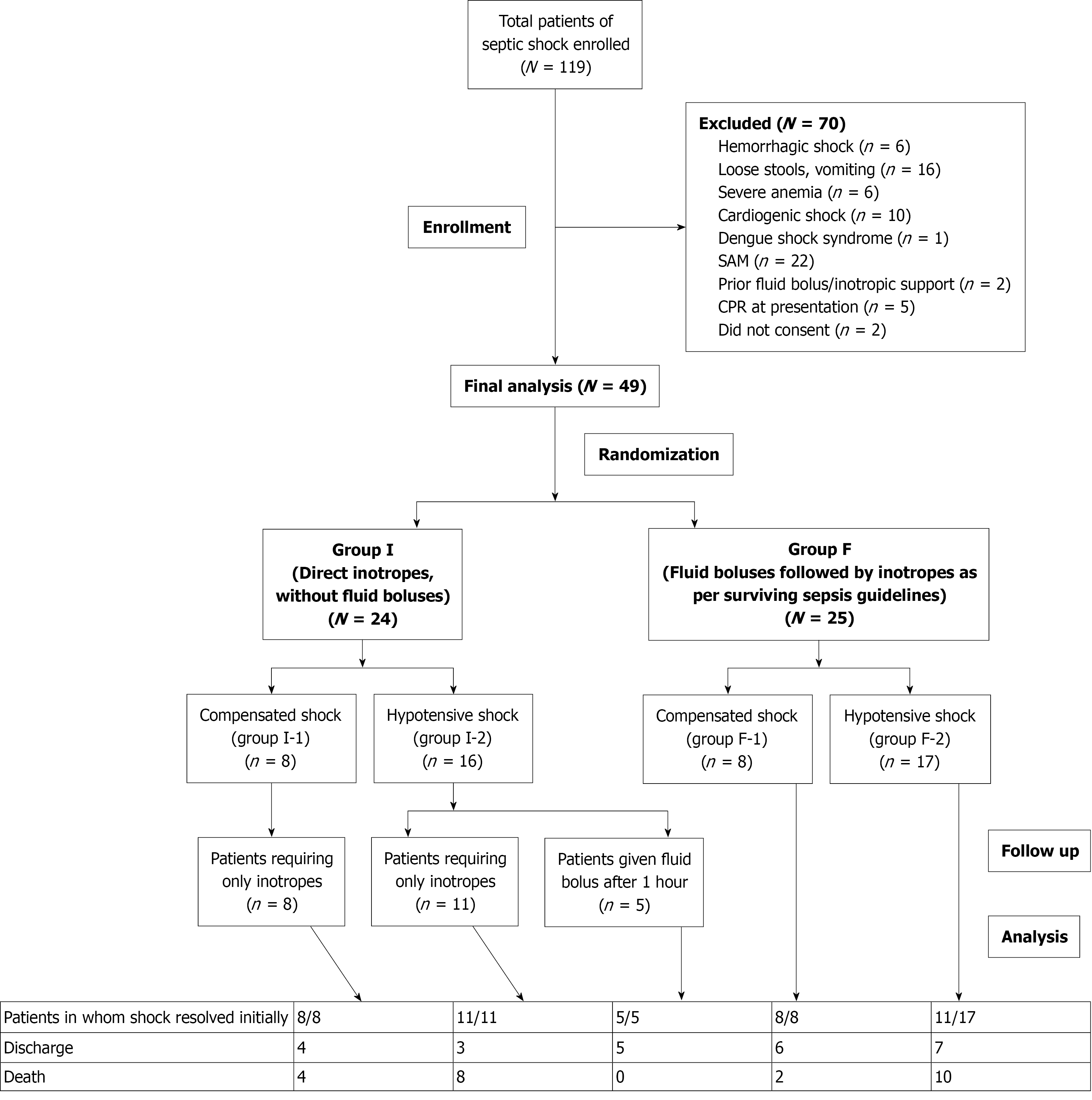
Out of 119 patients with septic shock, 49 were included in the final analysis (24 patients in Group I and 25 patients in Group F. 16/49 (32.65%) patients had compensated shock, and 33/49 (67.35%) had hypotensive shock. During the initial resuscitation phase, shock resolved in all (100%) of the patients in Group I. In Group F only in 19/25 (76.00%) patients experienced resolution of shock. The mean time of resolution of shock was significantly shorter in Group I (83.98 + 42.02 min) compared with Group F (116.72 + 41.67 min, P = 0.015). Invasive mechanical ventilation was required in 34/49 (69.40%) and was comparable across the groups. In the comparator Group F, 16/25 (65.00%) of the patients had some features of fluid overload. There was no statistical difference in the initial vasoactive inotropic support score, the total duration of ventilation, total length of hospital stay, mortality at 48 h, and overall mortality across the two groups. In a subgroup analysis of patients with hypotensive shock, mortality at 48 h was significantly higher in patients of Group F (9/17, 52.94%) than in patients of Group I (3/16, 18.75%, P = 0.041). The overall mortality rate in the study subjects was 24/49 (49.00%), and it was positively correlated with the Pediatric Sequential Organ Failure Assessment- Lactate score and maximum vasoactive inotropic support scores.
The use of fluid boluses in pediatric septic shock is dubious and contributes to fluid overload. The management of septic shock in children involves the use of adjuncts such as bedside ultrasound, early inotropes, and restricted and well-regulated fluid boluses.
Core Tip: Recent surviving sepsis guidelines showed better outcomes when fluid boluses were restricted. This open-label study in children aged 1 month to 14 years who were hospitalized for septic shock were randomized into two groups: Group I receiving early inotropes without fluid bolus; and Group F receiving fluid bolus. There were no statistically significant differences in the initial inotropic support (vasoactive inotropic support score), the total duration of ventilation, total length of hospital stay, mortality at 48 h, or overall mortality between the two groups. Patients with hypotensive shock who received fluid boluses had significantly higher 48-h mortality.
- Citation: Keshri S, Goel AK, Garg AK, Anand V, Rathia SK, Yusuf S. Early inotropic support without fluid bolus vs standard resuscitation in pediatric septic shock: A randomized controlled trial. World J Clin Pediatr 2026; 15(1): 109864
- URL: https://www.wjgnet.com/2219-2808/full/v15/i1/109864.htm
- DOI: https://dx.doi.org/10.5409/wjcp.v15.i1.109864
In the absence of ongoing losses like acute gastroenteritis, the optimal amount of fluid for initial resuscitation of septic shock is an important unanswered question. Current surviving sepsis guidelines 2020 recommend fluid boluses of 10-20 mL/kg/bolus up to 40-60 mL/kg[1]. Fluid bolus is not recommended for patients with sepsis without hypotension in the absence of intensive care settings, and maintenance fluids should be started[1]. The American College of Critical Care Medicine (ACCM) recommends a bolus push of 20 mL/kg up to 60 mL/kg if there is no hepatomegaly or crackles until perfusion improves, and subsequently inotropes are started[2].
Studies show that excessive fluid administration is associated with worse outcomes[3-7]. The FEAST trial compared bolus of 20-40 mL/kg vs no bolus in 3141 children with septic shock and reported 35% higher mortality associated with fluid bolus resuscitation[8]. However, the study was conducted in patients without severe hypotension, and nearly half the patients had severe anemia requiring blood transfusion, a potentially confounding factor. Other studies also had better outcomes with restricted fluid boluses and early initiation of inotropes in terms of reversal of shock, pulmonary edema, number of days of ventilation, number of days of hospital stay, and mortality as compared with liberal fluid boluses[9-13].
In a developing nation like ours, a large number of patients seek treatment for sepsis in primary or secondary healthcare facilities before being sent to a tertiary care hospital. This frequently results in delays, which causes mortality and morbidity rates to rise significantly. We aimed to analyze the outcome of initial resuscitation of septic shock without giving fluid bolus in this study.
This was an open-label, single-center randomized controlled trial conducted at the Pediatric Emergency Medicine unit of a tertiary care hospital in central India. We hypothesized that fluid bolus was not required in the initial resuscitation of septic shock in children. We aimed to analyze the outcome of initial resuscitation of septic shock without giving fluid bolus with early inotropic support to children aged 1 month to 14 years. All were evaluated for efficacy and safety. The Institute’s ethical approval was provided by AIIMS Raipur, Chhattisgarh, India (AIIMSRPR/IEC/20e21/722 dated 19 Dec 2020).
We enrolled patients aged 1 month to 14 years (adjusted for premature birth) who were hospitalized for presenting to the Pediatric Emergency Medicine unit between April 27, 2021 and December 20, 2022 and fulfilling the case definition of septic shock defined by the surviving sepsis guidelines. Children presenting with hypovolemic shock (hemorrhagic shock, gastroenteritis, burns, trauma, diabetic ketoacidosis), conditions in which volume expansion was guarded (severe anemia due to sickle cell anemia, malaria, cardiogenic shock with congestive heart failure, acute kidney injury, dengue shock), or severe acute malnutrition or who already received treatment in the form of fluid boluses or inotropes from another hospital, were terminally ill, or required cardiopulmonary resuscitation at presentation were excluded from the study. Written informed consent was obtained from the legal guardians of all participants. Basic vital parameters were monitored and recorded. All data were recorded in the case record sheet (Supplementary material). The patient in
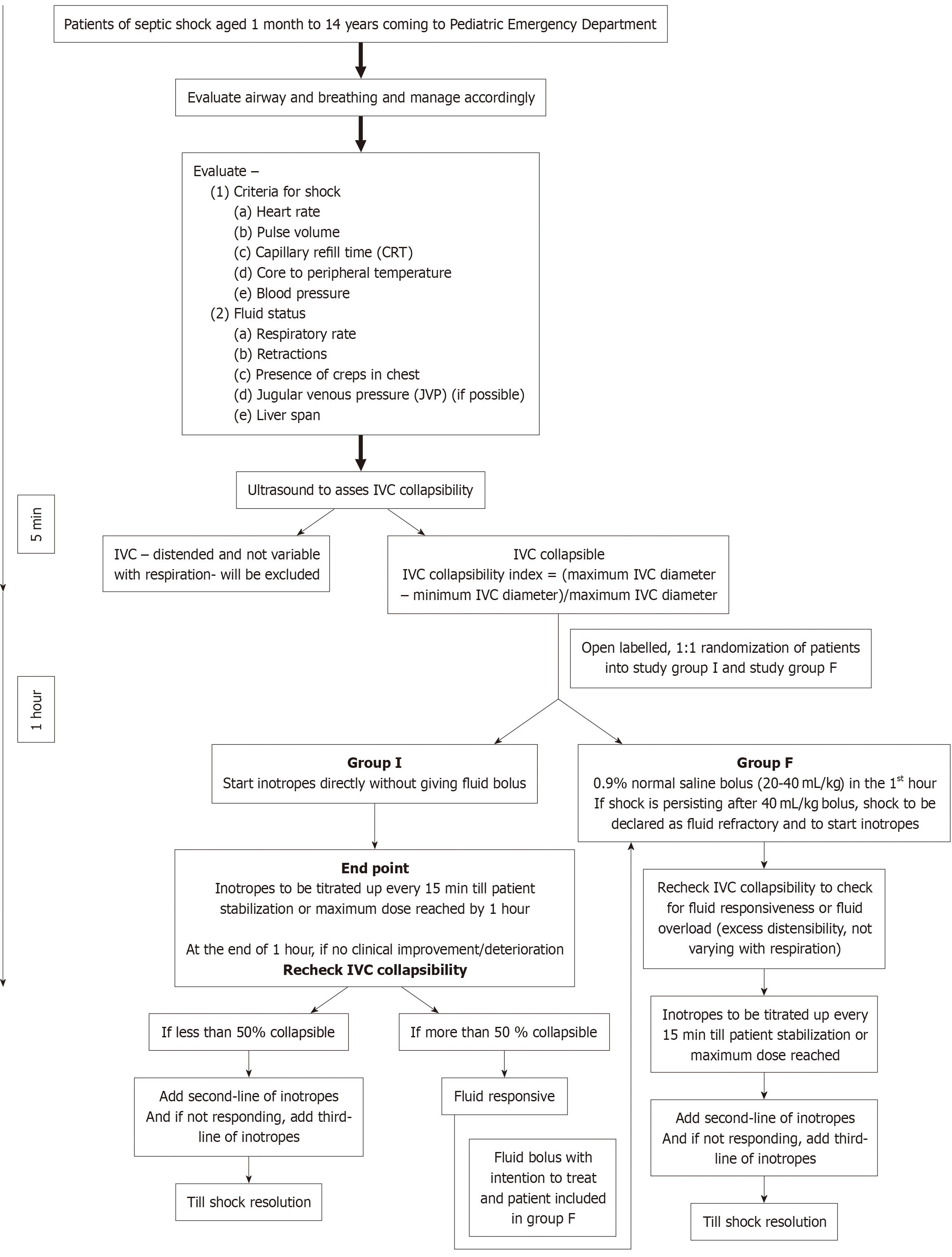
After informed consent from the parent/guardian, patients were allocated to two groups by computer-generated block random allocation of four and kept in sealed envelopes. Patients were randomized to the intervention group (Group I receiving early inotropes without giving fluid bolus) or the comparator group (Group F receiving fluid resuscitation) with an allocation ratio of 1:1 (Figure 1). Since this was an open-label randomized controlled clinical trial, there was no blinding at any level, and the authors were directly involved in the enrollment of participants and assigned them to the trial groups.
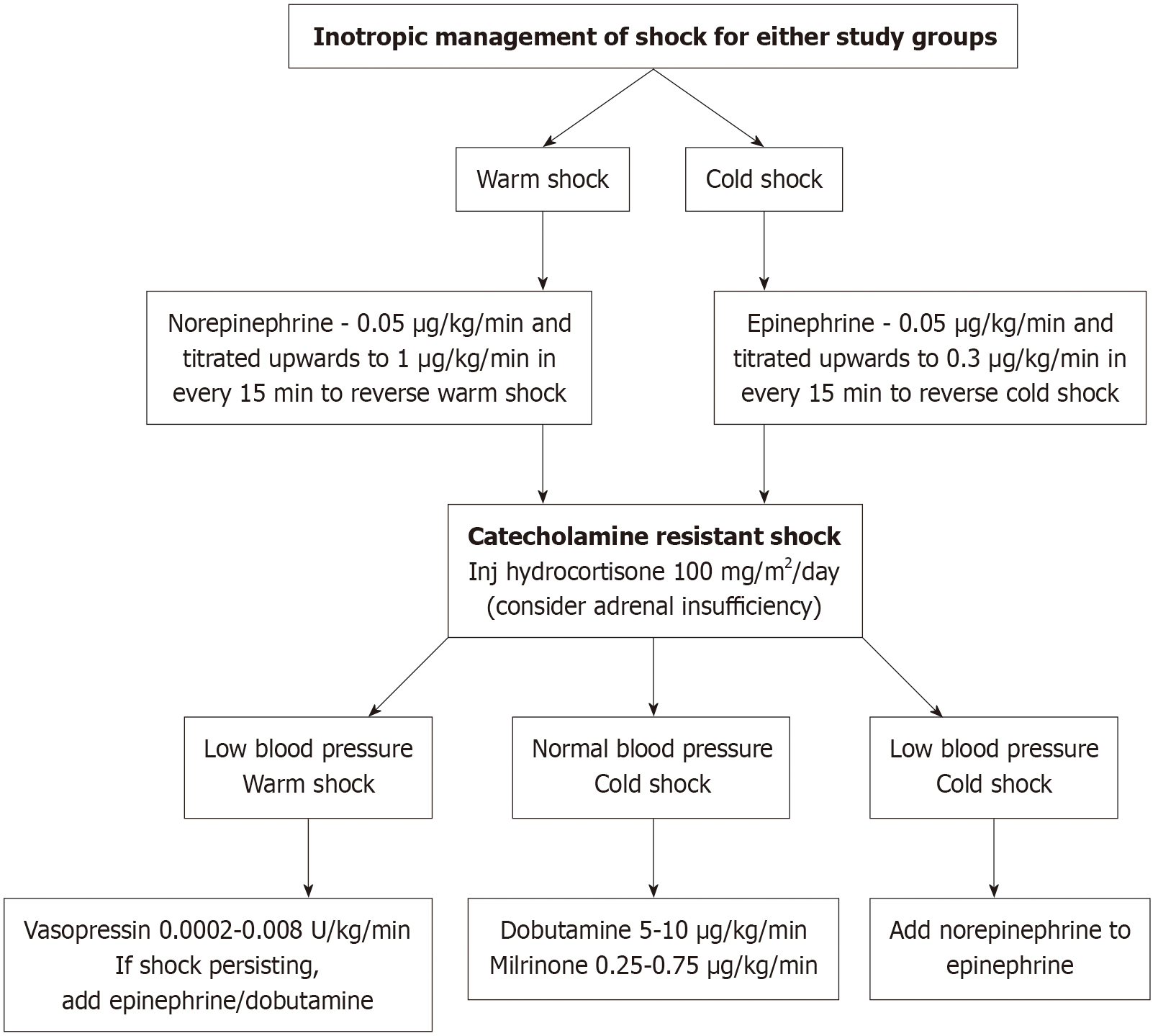
Procedure: In addition to the clinical examination, bedside ultrasonography was performed to look for inferior vena cava (IVC) collapsibility, which was performed by Keshri S, Yusuf S, Goel AK in all subjects. The ultrasound model number was Mindray Mobile Trolley Model: UMT-200 (December 10, 2018). Classic septic shock results in hyperdynamic cardiac activity and low systemic vascular resistance (SVR), manifesting as warm shock. Cold septic shock exhibits low cardiac output and elevated SVR.
Group I received inotropes as per the requirement and type of shock (warm or cold) for managing shock without receiving any fluid bolus (Figure 2). Group F received fluid bolus per the current surviving sepsis guidelines (2020). Vitals were monitored, and inotropes were escalated every 15 min or until the resolution of shock. The endpoint was either resolution of shock or the patient’s clinical status at 1 h from the initiation of resuscitation, whichever occurred first. Initial therapeutic end points of resuscitation of septic shock: Capillary refill of < 2 s; blood pressure (BP) more than the 5th percentile for age; tachycardia resolved with no differential between peripheral and central pulses; warm extremities; urine output of 1 mL/kg/hour; and normal mentation.
At the end of 1 hour if shock persisted, a repeat IVC collapsibility was performed. If the IVC was not collapsible (i.e. non-fluid responsive), second-line inotropes and third-line inotropes were added subsequently per the ACCM guidelines (Figure 2). But if the IVC collapsibility index was > 50%, it represented fluid responsiveness[14,15]. The patient was switched to study Group F, and fluid boluses were given under monitoring, followed by inotropic management per the ACCM guidelines. The resolution of shock was monitored up to 3 h from the time of its presentation. Surrogate parameters for tissue perfusion were venous lactate and hourly urine output monitoring. Parameters for organ dysfunction, such as renal function test, liver function test, prothrombin time, international normalized ratio, and arterial blood gas, were monitored and noted.
All patients in both groups received other standard care for sepsis management per the requirement, including oxygen by suitable interface, antibiotics per Institute protocol, ventilation (if required), symptomatic medication, and in
Outcomes: The primary endpoint was resolution of shock. If there was any clinical deterioration/no improvement at the end of 1 hour and the IVC was collapsible, Group I subjects were shifted to Group F in which standard treatment care per the recent guidelines was given. The secondary endpoints were non-resolution of shock at 1 h and death.
Based on the FEAST trial[8] and calculating the relative risk of mortality amongst the bolus group vs no bolus group (221/2097 vs 76/1044) using a 95% confidence interval, the sample size estimation was 36 patients (18 in each group). It was agreed to enroll patients until the completion of the study period because the researcher was an academic resident and an interim analysis was completed with the Institute’s safety review committee at the end of the proposed enrollment of the study subjects. A convenient sampling method was adopted.
The data procured was entered into MS Excel 16.68 and analyzed using SPSS software version (25.0). Descriptive statistics like mean ± SD, median (interquartile range), range and frequency (percentages) were used. Tests of normality including Shapiro-Wilk were applied to the data. Data following a normal distribution were evaluated as mean (± SD), and a test of significance between the two groups was done by an unpaired t-test. Data not following normal distribution were evaluated as median (interquartile range), and a test of significance between the two groups was done by the Mann-Whitney U test. For binary data in unpaired samples, cross tables were used to compare the outcomes using the χ2 test. Kaplan-Meier graphs were used for comparison of mortality over time. A P < 0.05 was considered statistically significant.
Out of 119 patients with septic shock, 49 were included in the final analysis (24 patients in Group I and 25 patients in Group F) (Figure 3). In Group I 8/24 (33.00%) patients had compensated shock (Group I-1), and 16/24 (67.00%) patients had hypotensive shock (Group I-2). Septic shock in all 8 (100%) patients in Group I-1 was reversed by inotropes without requiring any fluid boluses. All patients in Group I-2 had shock reversal: 11/16 (68.70%) reversed with inotropic support; and fluid boluses were required in 5 (31.30%) patients at the end of 1 hour. In Group F, 8/25 (32.00%) patients had compensated shock (Group F-1), and 17/25 (68.00%) patients had hypotensive shock (Group F-2). All (100%) of the patients in Group F required some form of inotropic support following fluid boluses. None responded to fluid boluses. In Group F-1 all 8 (100%) patients had reversal of shock. In Group F-2 6/17 (35.30%) patients had persistent fluid-refractory and catecholamine-refractory septic shock and did not have reversal of shock and ultimately succumbed.
The two groups had no significant differences in baseline characteristics and laboratory parameters (Tables 1 and 2). Warm shock constituted 21/49 (42.90%) of the patients. Overall, 16/49 (32.65%) patients had compensated shock, and 33/49 (67.35%) had hypotensive shock with no statistical difference between the two groups (P = 1.000). Both groups had a similar severity of illness with a mean pSOFA-L score of I-12.0 (± 3.89) and F-11.56 (± 3.71) (P = 0.687). Organisms were identified in 26/49 (53.00%) patients with methicillin-sensitive Staphylococcus aureus being the most common (Table 3).
| Group I (n = 24) | Group F (n = 25) | P value | |
| mean (± SD), median (IQR) | mean (± SD), median (IQR) | ||
| Age, years | 6.5 (1.0-11.0) | 6.0 (1.13-11.0) | 0.936 |
| Sex, males | 15 (62.5) | 18 (72.0) | 0.551 |
| Duration of illness, days | 6.5 (4.0-14.5) | 5.0 (4.0-10.0) | 0.402 |
| Weight, kg | 17.4 (12.1) | 15.9 (9.8) | 0.795 |
| Height, cm | 101.7 (40.7) | 102.3 (29.7) | 0.826 |
| HR, per min | 139 (27.6) | 154 (30.8) | 0.076 |
| RR, per min | 45 (19.4) | 48 (20.9) | 0.837 |
| SBP | NR-3; 83.71 (16.9) | NR-6; 78.90 (14.7) | 0.346 |
| DBP | NR-3; 38.30 (12.4) | NR-7; 38.00 (15.4) | 0.941 |
| MAP | NR-3; 53.50 (11.1) | NR-7; 51.60 (13.6) | 0.642 |
| Systolic shock index = HR/SBP | n = 20; 1.8 (0.58) | n = 20; 1.8 (0.50) | 0.599 |
| Diastolic shock index = HR/DBP | n = 21; 3.976 (1.543) | n = 18; 4.612 (2.066) | 0.278 |
| IVC collapsibility index | 31.70 (22.30-39.75) | 34.00 (25.95-56.28) | 0.190 |
| Fever | 19/24 (79.2) | 20/25 (80.0) | 1.000 |
| GCS | 14.50 (4.25-15.00) | 13.00 (7.00-15.00) | 0.916 |
| Underlying comorbidity | 12/24 (50.0) | 11/25 (44.0) | 0.778 |
| Received prior intravenous antibiotics | 13/24 (54.2) | 10/25 (40.0) | 0.396 |
| Warm shock; cold shock | 13/24 (54.2); 11/24 (45.8) | 8/25 (32.0); 17/25 (68.0) | 0.154 |
| Compensated shock | 8/24 (33.3) | 8/25 (32.0) | 1.000 |
| Hypotensive shock | 16/24 (66.7) | 17/25 (68.0) | |
| Hypoglycemia | 3/24 (12.5) | 2/25 (8.0) | 0.667 |
| Focus of infection | 0.111 | ||
| Blood stream | 0/24 (0) | 2/25 (8.0) | |
| Respiratory | 5/24 (20.8) | 9/25 (36.0) | |
| Abdomen | 3/24 (12.5) | 3/25 (12.0) | |
| CNS | 5/24 (20.8) | 6/25 (24.0) | |
| Joints/bones | 2/24 (8.3) | 0/25 (0) | |
| Renal | 0/24 (0) | 3/25 (12.0) | |
| Skin | 4/24 (16.7) | 1/25 (4.0) | |
| Disseminated | 5/24 (20.8) | 1/25 (4.0) | |
| pSOFA-L score | 12.0 (3.9) | 11.56 (3.7) | 0.687 |
| Group I (n = 24) | Group F (n = 25) | P value | |
| mean (SD), median (IQR) | mean (SD), median (IQR) | ||
| pH | 7.33 (7.17-7.43) | 7.26 (7.13-7.37) | 0.211 |
| pCO2, mm Hg | 28.3 (25.9-34.4) | 26.3 (21.7-31.6) | 0.140 |
| Bicarbonate, mEq/L | 15.67 (6.44) | 13.47 (6.44) | 0.238 |
| Base deficit, mEq/L | 10.5 (6.2-13.3) | 12.7 (8.3-17.7) | 0.052 |
| Lactate, mmol/L | 3.83 (2.53-6.42) | 4.38 (2.55-9.33) | 0.696 |
| Hemoglobin, gm/dL | 9.32 (1.66) | 9.04 (2.45) | 0.647 |
| Platelet, counts/mL | 94000 (42250-261500) | 140000 (32500-242000) | 0.984 |
| TLC, counts/mL | 20900 (16004) | 14867 (11514) | 0.135 |
| ANC, counts/mL | 9750 (4425-23975) | 8400 (3240-14670) | 0.250 |
| Sodium, mEq/L | 137 (133-148) | 136 (133-143) | 0.920 |
| Potassium, mEq/L | 4.20 (3.80-5.53) | 4.10 (3.60-5.05) | 0.589 |
| Chloride, mEq/L | 103.5 (97.0-111.8) | 103.0 (100.5-108.0) | 0.802 |
| Urea, mg/dL | 49.00 (24.25-92.00) | 44.00 (26.50-97.50) | 0.968 |
| Creatinine, mg/dL | 0.78 (0.46-1.43) | 0.82 (0.59-2.19) | 0.490 |
| Uric acid, mg/dL | 5.55 (3.47-11.87) | 6.10 (4.05-10.00) | 0.904 |
| PT, seconds | 13.50 (11.82-25.00) | 13.20 (12.20-17.50) | 0.849 |
| INR | 1.30 (1.10-2.28) | 1.20 (1.10-1.60) | 0.672 |
| aPTT, seconds | 34.20 (31.40-60.15) | 39.00 (29.75-50.50) | 0.936 |
| Total bilirubin, mg/dL | 0.94 (0.42-1.98) | 0.74 (0.52-1.28) | 0.379 |
| Direct bilirubin, mg/dL | 0.36 (0.14-0.81) | 0.21 (0.10-0.62) | 0.267 |
| Indirect bilirubin, mg/dL | 0.43 (0.30-1.47) | 0.45 (0.32-0.75) | 0.749 |
| AST, U/L | 97.00 (45.25-455.00) | 69.00 (38.00-134.00) | 0.254 |
| ALT, U/L | 79.00 (29.25-151.00) | 43.00 (16.50-105.00) | 0.144 |
| ALP, U/L | 187.5 (116.5- 272.5) | 158.0 (96.0-275.0) | 0.368 |
| GGT, U/L | 66.5 (33.5-142.5) | 43.0 (17.0-89.5) | 0.093 |
| Total protein, g/dL | 5.30 (1.18) | 5.29 (1.18) | 0.976 |
| Albumin, g/dL | 2.50 (0.63) | 2.79 (0.79) | 0.277 |
| Globulin, g/dL | 2.76 (0.98) | 2.42 (0.73) | 0.242 |
| CRP, mg/dL | 80.00 (33.48-159.97) | 80.00 (37.06-154.36) | 1.000 |
| Organism | Number of patients |
| Methicillin sensitive Staphylococcus aureus | 6 |
| Scrub typhus | 5 |
| Escherichia coli | 5 |
| Methicillin resistant Staphylococcus aureus | 2 |
| Coagulase-negative Staphylococci | 2 |
| Plasmodium falciparum | 1 |
| Pseudomonas aeruginosa | 1 |
| Klebsiella pneumoniae | 1 |
| Proteus mirabilis | 1 |
| Morganella morganii | 1 |
| Candida albicans | 1 |
| Total | 26 |
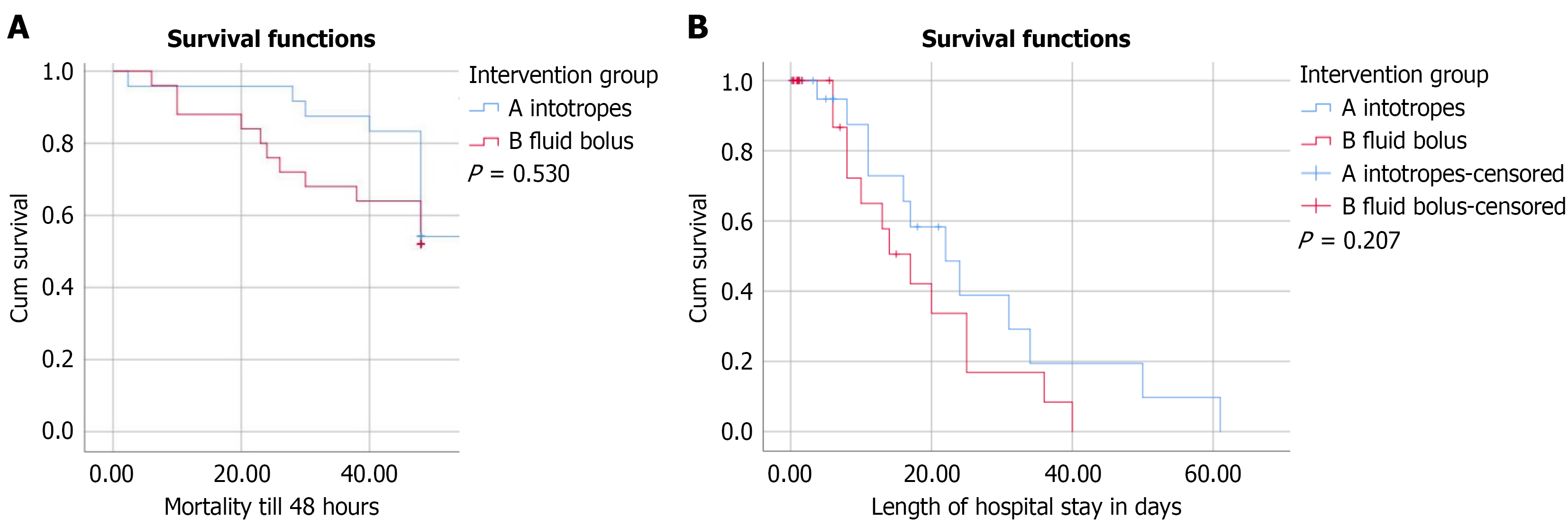
During the initial resuscitation phase, shock resolved in all (100%) of the patients in Group I, but only in 19/25 (76.00%) patients in Group F. The mean time of resolution of shock was significantly shorter in Group I [83.98 (± 42.02) min] compared with Group F [116.72 (± 41.67) min] (P = 0.015). Invasive mechanical ventilation was required in 34/49 (69.40%) patients and was comparable across the groups. There was no statistical difference in the initial VIS score, the total duration of ventilation, the length of hospital stay, mortality at 48 h, and overall mortality across the two groups (Figure 4). The overall mortality rate in the study subjects was 24/49 (49.00%), and it positively correlated to the pSOFA-L and maximum VIS scores (Table 4).
| Unstandardized B | t | Sig | |
| Constant | 1.609 | 2.775 | 0.009 |
| Intervention group | -0.790 | -0.509 | 0.614 |
| Underlying comorbidity | -0.207 | -1.375 | 0.180 |
| Diastolic shock index | -0.065 | -1.636 | 0.112 |
| Ventilatory requirement | 0.082 | 0.813 | 0.423 |
| Compensated vs hypotensive shock | -0.150 | -0.981 | 0.335 |
| Signs of fluid overload | -0.130 | -0.840 | 0.408 |
| Maximum VIS score | 0.003 | 2.074 | 0.047 |
| pSOFA-L score | 0.047 | 2.349 | 0.026 |
In a subgroup analysis of patients with hypotensive shock, initial shock reversal occurred in all (100%) patients in Group I, but only in 11/17 (64.70%) patients in Group F (P = 0.018) (Table 5). The mean time of resolution of shock and inotropic support required was similar across the two groups. Patients in Group I required a significantly longer total duration of inotropic support. Mortality at 48 h was significantly higher in patients in Group F (9/17, 52.94%) than in patients in Group I (3/16, 18.75%, P = 0.041).
| Parameters | Patients with compensated shock (n = 16) | Patients with hypotensive shock (n = 33) | |||||
| Group I-1 (n = 8) | Group F-1 (n = 8) | P value | Group I-2 (n = 16) | Group F-2 (n = 17) | P value | ||
| Patients who had resolution of shock | 8/8 (100) | 8/8 (100) | 1 | 16/16 (100) | 11/17 (64.70) | 0.018 | |
| Time of resolution of shock, minutes | 80.63 (25.05) | 125.63 (54.34) | 0.052 | 85.66 (49.04) | 110.23 (30.81), n = 11 | 0.154 | |
| VIS for resolution of shock | 30.00 (13.09) | 21.25 (9.90) | 0.161 | 31.56 (15.02) | 42.73 (32.59), n = 11 | 0.24 | |
| Mean fluid required within 6 h, mL/kg | 0 (0), n = 0 | 27.50 (8.86) | 28.00 (11.80), n = 5 | 31.76 (13.80) | |||
| Signs of fluid overload | 0/8 (0) | 4/8 (50.00) | 1/16 (6.25) | 12/17 (70.00) | 0 | ||
| Duration of inotropes, hours | 68.00 (36.00-111.75) | 70.50 (17.36-102.00) | 0.574 | 113.52 (88.70) | 37.82 (34.62) | 0.003 | |
| Duration of ventilation, hours | 109.00 (37.25-138.00), n = 4 | 50.00 (13.00-222.00), n = 5 | 0.556 | 76.00 (18.00-144.00), n = 11 | 22.50 (10.00-85.50), n = 14 | 0.222 | |
| Length of hospital stay, days | 11.00 (4.06-20.00) | 12.00 (7.25-22.50) | 0.574 | 9.50 (3.88-23.50) | 1.58 (0.89-15.00) | 0.094 | |
| Maximum VIS during hospital stay | 48.75 (36.82) | 42.50 (37.70) | 0.742 | 85.31 (48.35) | 103.53 (70.71) | 0.397 | |
| Mortality at 48 h | 1/8 (12.50) | 0/8 (0) | 3/16 (18.75) | 9/17 (52.94) | 0.041 | ||
| Discharge; death | 4 (50.00); 4 (50.00) | 6 (75.00); 2 (25.00) | 0.608 | 8 (50.00); 8 (50.00) | 7 (41.20); 10 (58.80) | 0.732 | |
More than half (16/25, 65.00%) of the patients had some features of fluid overload in Group F. All (100%) presented with subtle signs like increase in heart rate and respiratory rate from baseline with 12/16 (75.00%), 7/16 (43.80%), 7/16 (43.80%), 1/16 (6.25%), 3/16 (18.80%), and 15/16 (93.80%) developing puffiness of eyes, increase in liver size, crepitations, gallop rhythm, overt pulmonary edema, and full and distended IVC on bedside ultrasound, respectively. None of the patients had serious adverse effects to the inotropes like arrhythmia, limb ischemia, amputation, or abdominal compartment syndrome. Other adverse effects like hypertension and hyperglycemia were commonly observed 24-48 h after the resolution of shock, requiring tapering of inotropic support. An interim analysis was done in front of a safety review committee.
We aimed to analyze the outcome of initial resuscitation of septic shock without giving fluid boluses with early inotropic support. A majority (67.35%, 33/49) of the patients in our study had hypotensive shock. This finding highlights a stark contrast compared with earlier studies like the FEAST trial and the FiSH trial, which reported only 6% and 5% of patients with moderate hypotension, respectively[8,10]. The study done in India had a lower mean systolic BP, diastolic BP, and mean arterial pressure (MAP)[11].
The Shock Index, Pediatric Age-Adjusted (SIPA), which is the ratio of the heart rate to systolic BP with a cutoff value of > 1.22 (ages 4-6), > 1.0 (7-12), and > 0.9 (13-16), can help predict the mortality in pediatric patients[18,19]. The mean SIPA score in our study was 1.84 (+ 0.54), indicating a high severity of illness and risk of mortality. The mean diastolic shock index was as high as 4.26 (+ 1.8) in our study. A high diastolic shock index > 2.2 is associated with higher risks of organ failure and mortality[20].
In our study the mean pSOFA-L score was 11.78 (± 3.77), implying high severity of illness. The FEAST trial did not objectively use any scoring system[8]. The FiSH trial incorporated the Pediatric Index of Mortality-2 score with median values of 2.1 and 2.0 in the two groups, representing a very low severity of illness[10]. The PRISM III score was a commonly used scoring system[11-13] with comparable high severity of illness in some studies[12,13].
The spectrum of infections contributing to septic shock in the participants was diverse, predominantly affecting the respiratory system (28.50%) and the central nervous system (22.45%). This aligned with findings from Ranjit et al[12]. The majority (57%) of the study subjects in the landmark FEAST trial were positive for malaria. This was a confounding factor as they presented with hemolysis and severe anemia, making fluid boluses put them at risk for increased fluid overload and mortality[8,21]. Another study done in India had 50% of the patients test as dengue positive[11], making it yet another confounding factor as the pathophysiology of dengue involves the third spacing of fluid into the extracellular space due to increase in capillary permeability with a definitive role of fluid resuscitation[22]. The heterogeneity of the focus of infection and exclusion of patients with dengue and severe anemia in our study was imperative to the general applicability of the questionable role of fluid boluses in cases of septic shock.
The primary outcome focused on the resolution of shock during the resuscitation phase with findings suggesting improved outcomes in patients receiving direct inotropes without prior fluid bolus administration. The results indicated that early inotropic support may not only enhance contractility of the sepsis-affected myocardium but also improve vascular tone, thereby reversing vasoplegia commonly associated with septic shock[23]. About a quarter of the patients in Group F did not achieve initial shock reversal, leading to mortality. This may be a result of septic shock converting from a reversible compensated stage to an irreversible decompensated stage, emphasizing the necessity of early addition of vasoactive support[11,24].
The majority of patients (34/49, 69.40%) required invasive mechanical ventilation due primarily to the disease or catecholamine refractory septic shock. It was due to fluid overload in 6/34 (17.64%) patients. There was no significant difference in the duration of ventilation across the two groups, possibly due to the high severity of illness and multiorgan involvement. The ventilatory requirement in other studies ranged from zero (no facility for a ventilator in resource-limited settings) in African countries[8] to 17.64% in resourceful countries with a low severity of illness[10] to 95.58% where strict ventilatory criteria were kept for advanced hemodynamic monitoring of the patients[12]. Studies have demonstrated a significant increased need and duration of ventilation in those receiving more fluid boluses[11,13].
Fluid overload emerged as a significant concern (65.00%) although it did not correlate with the volume of fluid boluses given. Other studies have reported a positive correlation between the amount of fluid boluses given and signs of fluid overload[1,12]. The FEAST trial did not report any significant difference in pulmonary edema or raised intracranial pressure[8]. This implies that the underlying pathophysiology of each patient in septic shock is unique and may be attributed to multiple simultaneously underlying processes, like vasoplegia, loss of arterial tone, third spacing of fluids, and different extent of myocardial involvement. Patients can develop fluid overload even at small bolus volumes of 10-20 mL/kg due to depressed myocardial function[25,26].
We noticed yet another adverse event of fluid boluses: Either no response (increase in MAP < 5.0) or a fall in blood pressure in as many as 12/25 (48%) of patients in Group F. This effect may be attributed to fluid resuscitation-induced vasodilation by mechanisms like recruitment of previously closed blood vessels or shear injury of the vascular endothelium resulting in nitric oxide-induced vasodilation or atrial natriuretic peptides due to increased cardiac filling pressures[27].
A similar paradoxical fall in MAP or no response to the addition of inotropes was also observed as an initial response in 6/24 (25.00%) patients in group I. There is usually an underlying sepsis-induced myocarditis[28], peripheral va
While some studies have noted comparable lengths of hospital stay as in our study[10,30], some studies have a significantly longer hospital stay in the patients receiving liberal fluid boluses[11-13]. The length of hospital stay depends on a number of factors like multiorgan involvement, duration of ventilation, duration of antibiotic treatment, hospital-acquired infection etc.
The overall mortality in the study subjects was 24/49 (49.00%) and was comparable across the two groups [12/24 (50.00%) in Group I and 12/25 (48.00%) in Group F]. However, in patients who experienced hypotensive shock, the mortality at 48 h was significantly higher in patients in Group F [9/17 (52.94%) vs 3/16 (18.75%), P = 0.041]. The FEAST trial noted increased mortality (relative risk: 1.45) at 48 h and 4 weeks in the patients who received boluses. The mortality rate in patients of the FEAST trial with severe hypotension was 62%, and it was comparable to our subgroup of patients with hypotensive shock (54.50%)[8]. Other studies have noted similar overall mortality rates between the two groups[11-13]. The high mortality was attributed to the severity of illness and multiorgan dysfunction, objectified by pSOFA-L score and the severity of shock during pediatric intensive care unit stay (high maximum VIS score)[16,17]. Because of limited resources, we could measure only noninvasive BP. The systolic BP and diastolic BP were not recordable in 10 patients by noninvasive BP monitoring due to nonpalpable pulses and the absence of Korotkoff sounds. Myocarditis, though we were unable to establish the diagnosis clinically apart from electrocardiogram changes, may be a confounding factor, especially for those with a poor outcome.
All patients in septic shock managed according to the surviving sepsis guidelines subsequently required inotropic support following fluid boluses and required more time for resolution of shock than those patients who initially received inotropes without fluid boluses. Furthermore, we observed a conversion to refractory septic shock in one-quarter of patients, fluid overload and mortality at 48 h in patients in Group F, emphasizing the paramount importance of the early addition of inotropes in shock reversal. Though our study favors the intervention group, recommendations of no bolus in pediatric septic shock is still a matter of debate and needs a large and robust study to justify it. Hence, we conclude that in the absence of ongoing fluid losses, the role of fluid boluses in pediatric septic shock is questionable and contributes to serious adverse effects. Restricted and closely monitored fluid boluses with early addition of inotropes, the use of adjuncts like bedside ultrasound, and individualization of cases is the key to the management of septic shock in children.
We thank all the patients and families for their support of this clinical trial and all academic residents and staff of the Division of Pediatric Emergency Medicine, Department of Pediatrics, AIIMS Raipur, India for their dedication and contributions to the study.
| 1. | Weiss SL, Peters MJ, Alhazzani W, Agus MSD, Flori HR, Inwald DP, Nadel S, Schlapbach LJ, Tasker RC, Argent AC, Brierley J, Carcillo J, Carrol ED, Carroll CL, Cheifetz IM, Choong K, Cies JJ, Cruz AT, De Luca D, Deep A, Faust SN, De Oliveira CF, Hall MW, Ishimine P, Javouhey E, Joosten KFM, Joshi P, Karam O, Kneyber MCJ, Lemson J, MacLaren G, Mehta NM, Møller MH, Newth CJL, Nguyen TC, Nishisaki A, Nunnally ME, Parker MM, Paul RM, Randolph AG, Ranjit S, Romer LH, Scott HF, Tume LN, Verger JT, Williams EA, Wolf J, Wong HR, Zimmerman JJ, Kissoon N, Tissieres P. Surviving Sepsis Campaign International Guidelines for the Management of Septic Shock and Sepsis-Associated Organ Dysfunction in Children. Pediatr Crit Care Med. 2020;21:e52-e106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 375] [Cited by in RCA: 699] [Article Influence: 116.5] [Reference Citation Analysis (0)] |
| 2. | Davis AL, Carcillo JA, Aneja RK, Deymann AJ, Lin JC, Nguyen TC, Okhuysen-Cawley RS, Relvas MS, Rozenfeld RA, Skippen PW, Stojadinovic BJ, Williams EA, Yeh TS, Balamuth F, Brierley J, de Caen AR, Cheifetz IM, Choong K, Conway E Jr, Cornell T, Doctor A, Dugas MA, Feldman JD, Fitzgerald JC, Flori HR, Fortenberry JD, Graciano AL, Greenwald BM, Hall MW, Han YY, Hernan LJ, Irazuzta JE, Iselin E, van der Jagt EW, Jeffries HE, Kache S, Katyal C, Kissoon N, Kon AA, Kutko MC, MacLaren G, Maul T, Mehta R, Odetola F, Parbuoni K, Paul R, Peters MJ, Ranjit S, Reuter-Rice KE, Schnitzler EJ, Scott HF, Torres A Jr, Weingarten-Arams J, Weiss SL, Zimmerman JJ, Zuckerberg AL. American College of Critical Care Medicine Clinical Practice Parameters for Hemodynamic Support of Pediatric and Neonatal Septic Shock. Crit Care Med. 2017;45:1061-1093. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 474] [Cited by in RCA: 444] [Article Influence: 49.3] [Reference Citation Analysis (0)] |
| 3. | Inwald DP, Butt W, Tasker RC. Fluid resuscitation of shock in children: what, whence and whither? Intensive Care Med. 2015;41:1457-1459. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 4. | Flori HR, Church G, Liu KD, Gildengorin G, Matthay MA. Positive fluid balance is associated with higher mortality and prolonged mechanical ventilation in pediatric patients with acute lung injury. Crit Care Res Pract. 2011;2011:854142. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 66] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 5. | Valentine SL, Sapru A, Higgerson RA, Spinella PC, Flori HR, Graham DA, Brett M, Convery M, Christie LM, Karamessinis L, Randolph AG; Pediatric Acute Lung Injury and Sepsis Investigator's (PALISI) Network; Acute Respiratory Distress Syndrome Clinical Research Network (ARDSNet). Fluid balance in critically ill children with acute lung injury. Crit Care Med. 2012;40:2883-2889. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 153] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 6. | Sinitsky L, Walls D, Nadel S, Inwald DP. Fluid overload at 48 hours is associated with respiratory morbidity but not mortality in a general PICU: retrospective cohort study. Pediatr Crit Care Med. 2015;16:205-209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 89] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 7. | Bhaskar P, Dhar AV, Thompson M, Quigley R, Modem V. Early fluid accumulation in children with shock and ICU mortality: a matched case-control study. Intensive Care Med. 2015;41:1445-1453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 59] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 8. | Maitland K, Kiguli S, Opoka RO, Engoru C, Olupot-Olupot P, Akech SO, Nyeko R, Mtove G, Reyburn H, Lang T, Brent B, Evans JA, Tibenderana JK, Crawley J, Russell EC, Levin M, Babiker AG, Gibb DM; FEAST Trial Group. Mortality after fluid bolus in African children with severe infection. N Engl J Med. 2011;364:2483-2495. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1347] [Cited by in RCA: 1105] [Article Influence: 73.7] [Reference Citation Analysis (0)] |
| 9. | Santhanam I, Sangareddi S, Venkataraman S, Kissoon N, Thiruvengadamudayan V, Kasthuri RK. A prospective randomized controlled study of two fluid regimens in the initial management of septic shock in the emergency department. Pediatr Emerg Care. 2008;24:647-655. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 68] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 10. | Inwald DP, Canter R, Woolfall K, Mouncey P, Zenasni Z, O'Hara C, Carter A, Jones N, Lyttle MD, Nadel S, Peters MJ, Harrison DA, Rowan KM; PERUKI (Paediatric Emergency Research in the UK and Ireland) and PICS SG (Paediatric Intensive Care Society Study Group). Restricted fluid bolus volume in early septic shock: results of the Fluids in Shock pilot trial. Arch Dis Child. 2019;104:426-431. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 62] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 11. | Karanvir, Gupta S, Kumar V. Practices of Initiation of Vasoactive Drugs in Relation to Resuscitation Fluids in Children with Septic Shock: A Prospective Observational Study. Indian J Crit Care Med. 2021;25:928-933. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 12. | Ranjit S, Natraj R, Kandath SK, Kissoon N, Ramakrishnan B, Marik PE. Early norepinephrine decreases fluid and ventilatory requirements in pediatric vasodilatory septic shock. Indian J Crit Care Med. 2016;20:561-569. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 35] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 13. | van Paridon BM, Sheppard C, G GG, Joffe AR; Alberta Sepsis Network. Timing of antibiotics, volume, and vasoactive infusions in children with sepsis admitted to intensive care. Crit Care. 2015;19:293. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 62] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 14. | Ciozda W, Kedan I, Kehl DW, Zimmer R, Khandwalla R, Kimchi A. The efficacy of sonographic measurement of inferior vena cava diameter as an estimate of central venous pressure. Cardiovasc Ultrasound. 2016;14:33. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 83] [Cited by in RCA: 124] [Article Influence: 12.4] [Reference Citation Analysis (1)] |
| 15. | Porter TR, Shillcutt SK, Adams MS, Desjardins G, Glas KE, Olson JJ, Troughton RW. Guidelines for the use of echocardiography as a monitor for therapeutic intervention in adults: a report from the American Society of Echocardiography. J Am Soc Echocardiogr. 2015;28:40-56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 264] [Cited by in RCA: 329] [Article Influence: 29.9] [Reference Citation Analysis (1)] |
| 16. | Kumbar SC. Assessment of pSOFA-L score in predicting the clinical outcome of critically ill children. Int J Contemp Pediatr. 2020;7:925. [DOI] [Full Text] |
| 17. | McIntosh AM, Tong S, Deakyne SJ, Davidson JA, Scott HF. Validation of the Vasoactive-Inotropic Score in Pediatric Sepsis. Pediatr Crit Care Med. 2017;18:750-757. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 236] [Cited by in RCA: 208] [Article Influence: 23.1] [Reference Citation Analysis (0)] |
| 18. | Huang KC, Yang Y, Li CJ, Cheng FJ, Huang YH, Chuang PC, Chiu IM. Shock Index, Pediatric Age-Adjusted Predicts Morbidity and Mortality in Children Admitted to the Intensive Care Unit. Front Pediatr. 2021;9:727466. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 19. | Acker SN, Bredbeck B, Partrick DA, Kulungowski AM, Barnett CC, Bensard DD. Shock index, pediatric age-adjusted (SIPA) is more accurate than age-adjusted hypotension for trauma team activation. Surgery. 2017;161:803-807. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 60] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 20. | Ospina-Tascón GA, Hernandez G, Alvarez I, Calderón-Tapia LE, Manzano-Nunez R, Sánchez-Ortiz AI, Quiñones E, Ruiz-Yucuma JE, Aldana JL, Teboul JL, Cavalcanti AB, De Backer D, Bakker J. Effects of very early start of norepinephrine in patients with septic shock: a propensity score-based analysis. Crit Care. 2020;24:52. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 80] [Cited by in RCA: 119] [Article Influence: 19.8] [Reference Citation Analysis (1)] |
| 21. | Houston KA, George EC, Maitland K. Implications for paediatric shock management in resource-limited settings: a perspective from the FEAST trial. Crit Care. 2018;22:119. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 18] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 22. | Hung NT. Fluid management for dengue in children. Paediatr Int Child Health. 2012;32 Suppl 1:39-42. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 22] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 23. | Shi R, Hamzaoui O, De Vita N, Monnet X, Teboul JL. Vasopressors in septic shock: which, when, and how much? Ann Transl Med. 2020;8:794. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 64] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 24. | Bonanno FG. Clinical pathology of the shock syndromes. J Emerg Trauma Shock. 2011;4:233-243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 41] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 25. | Ford N, Hargreaves S, Shanks L. Mortality after fluid bolus in children with shock due to sepsis or severe infection: a systematic review and meta-analysis. PLoS One. 2012;7:e43953. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 40] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 26. | Gelbart B. Fluid Bolus Therapy in Pediatric Sepsis: Current Knowledge and Future Direction. Front Pediatr. 2018;6:308. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 27. | Marik PE, Byrne L, van Haren F. Fluid resuscitation in sepsis: the great 30 mL per kg hoax. J Thorac Dis. 2020;12:S37-S47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 65] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 28. | Vieillard-Baron A. Septic cardiomyopathy. Ann Intensive Care. 2011;1:6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 193] [Cited by in RCA: 167] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 29. | Gelbart B, Glassford NJ, Bellomo R. Fluid Bolus Therapy-Based Resuscitation for Severe Sepsis in Hospitalized Children: A Systematic Review. Pediatr Crit Care Med. 2015;16:e297-e307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 27] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 30. | Eisenberg MA, Riggs R, Paul R, Balamuth F, Richardson T, DeSouza HG, Abbadesa MK, DeMartini TKM, Frizzola M, Lane R, Lloyd J, Melendez E, Patankar N, Rutman L, Sebring A, Timmons Z, Scott HF; IPSO collaborative investigators. Association Between the First-Hour Intravenous Fluid Volume and Mortality in Pediatric Septic Shock. Ann Emerg Med. 2022;80:213-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/