Published online Mar 9, 2026. doi: 10.5409/wjcp.v15.i1.109301
Revised: June 22, 2025
Accepted: November 5, 2025
Published online: March 9, 2026
Processing time: 303 Days and 18.6 Hours
Cow’s milk protein allergy (CMPA) is the most common food allergy in infants, with significant clinical, nutritional, and economic consequences. In Jordan, the Ministry of Health (MoH) provides specialized formulas for affected infants; however, local data on clinical presentation, treatment duration, costs, and breastfeeding barriers are scarce.
To evaluate the clinical manifestations and outcomes of infants diagnosed with CMPA in a public hospital in Jordan and to assess treatment costs and maternal breastfeeding barriers.
This retrospective cross-sectional study included 38 infants aged < 1 year diagnosed with CMPA at a MoH tertiary hospital in Salt, Jordan, between 2020 and 2023. Data were collected from medical records and caregiver phone interviews. Infants who were exclusively breastfed or received mixed formulas (extensively hydrolyzed formula and amino acid-based formula) were excluded. Statistical analysis included descriptive statistics and Fisher’s exact test to determine associations between breastfeeding barriers and maternal factors.
Constipation (47.4%) and diarrhea (44.7%) were among the most frequent symptoms. One-third of the infants had coexisting food allergies, and elevated liver enzyme levels or isolated jaundice was noted in 21%. The average treatment duration was 13.89 months, with a mean cost of JD 1802 ($2523) per child. Maternal perception of milk insufficiency (36.8%) was the most reported breastfeeding barrier. Maternal employment, but not education (P = 0.22), was significantly associated with breastfeeding challenges (OR = 7.20, P = 0.03).
CMPA in Jordanian infants was associated with delayed diagnosis, high treatment costs, and significant breastfeeding barriers, particularly among employed mothers. These findings highlight the need for improved awareness, earlier diagnosis, and supportive policies to promote breastfeeding and reduce the disease burden.
Core Tip: This study provides the first comprehensive analysis of cow’s milk protein allergy (CMPA) in Jordanian infants. It highlights less common clinical presentations, such as constipation and elevated liver enzymes, and evaluates treatment duration, costs, and coexisting food allergies. A novel aspect of the study is its investigation of breastfeeding barriers, revealing a significant association with maternal employment. The findings emphasize the clinical, social, and economic burden of CMPA in a publicly funded healthcare system and underscore the need for targeted education, early diagnosis, and breastfeeding support to improve outcomes and reduce costs.
- Citation: Khdair Ahmad F, Alkayid MR, Ahmad TM, Hadidi RH, Rahhal RM. Cow’s milk protein allergy in infants: Clinical presentations and outcomes. World J Clin Pediatr 2026; 15(1): 109301
- URL: https://www.wjgnet.com/2219-2808/full/v15/i1/109301.htm
- DOI: https://dx.doi.org/10.5409/wjcp.v15.i1.109301
Cow’s milk protein allergy (CMPA) is the most common food allergy in infants and young children[1,2], and it has significant effects on infants and their families, posing challenges to nutrition, growth, and overall quality of life (QoL)[3,4]. The need for dietary restrictions, financial burden associated with treatment, and risk of severe allergic reactions contribute to increased parental anxiety and use of health care services[5].
CMPA can be classified based on immunological mechanisms into immunoglobulin E (IgE)-mediated, non-IgE-mediated, and mixed-type forms[3,5-7]. IgE-mediated CMPA typically involves immediate hypersensitivity reactions, such as urticaria, vomiting, or wheezing, and is confirmed by positive serum-specific IgE or skin-prick test results[3,5]. In contrast, non-IgE-mediated CMPA is associated with delayed symptoms, such as persistent diarrhea, blood-streaked stools, or failure to thrive, and lacks reliable confirmatory biomarkers, often requiring an elimination diet followed by an oral food challenge for diagnosis[3,5,6,7]. Mixed-type CMPA presents with overlapping features of both mechanisms and may occur in other conditions, such as atopic dermatitis or eosinophilic gastrointestinal disorders[3]. Differentiating between these types is essential for developing appropriate diagnostic and management strategies[5].
Several risk factors are associated with the development of CMPA. A family history of allergic diseases, such as asthma, eczema, or seasonal allergies, particularly in first-degree relatives, is considered a major risk factor[8]. Other contributing factors include cesarean delivery, formula feeding, and antibiotic exposure during or after pregnancy[9]. These factors may interfere with the natural development of the infant gut microbiome, resulting in altered microbial composition that can predispose individuals to CMPA[10].
CMPA management depends on the infant’s feeding method. For breastfed infants, the mother is recommended to eliminate all sources of cow’s milk protein from her diet, as these proteins can be passed through breast milk to infants[1]. For formula-fed infants, extensively hydrolyzed formulas (eHFs), including amino acid-based formulas (AAFs) in some cases, are recommended[1]. However, maternal and social barriers may limit breastfeeding and increase dependence on specialized formulas, leading to higher management costs and caregiver strain in infants with CMPA[9].
The primary objective of this study was to examine the clinical manifestations and outcomes of Jordanian children with CMPA. The secondary objective was to evaluate treatment cost and explore breastfeeding barriers among Jordanian women.
This retrospective cross-sectional study was conducted at the Al-Hussein New Hospital (HUH) in Salt City, Jordan, from July 2020 to April 2023. HUH is a 200-bed public hospital operated by the Ministry of Health (MoH) and affiliated with the School of Medicine at Balqa Applied University (BAU). The hospital’s outpatient clinics handle approximately 50000 visits annually. The study was conducted during the sabbatical year of the primary investigator (FKA) at BAU. The study received ethical approval from the institutional review boards of the University of Jordan School of Medicine and the MoH. Written informed consent was obtained during the clinical visit from one parent of each infant included in the study.
Infants diagnosed with CMPA and referred to the Pediatric Gastroenterology (Peds GI) clinic at the HUH were included in the study. This clinic provides non breastfed infants diagnosed with CMPA access to AAF and eHF. These formulas are fully covered by MoH insurance. According to national policy, all Jordanian children aged < 6 years are insured by the MoH. The market price of a 400-g (14.1 oz) can of AAF and eHF is approximately 20 and 15 Jordanian dinars ($28 and $21), respectively. According to the MoH protocols followed across all Peds GI clinics, including HUH, formula-fed infants aged ≤ 6 months diagnosed with CMPA are entitled to eight 400-g cans of AAF/eHF per month and those aged > 6 months receive four cans monthly.
The inclusion criteria were as follows: Infants aged < 1 year with a clinical diagnosis of CMPA made by the referring physician (RP), who were either mixed-fed (breast milk and formula) or exclusively formula-fed and who were referred to the HUH Peds GI clinic for CMPA treatment. The exclusion criteria were as follows: Mixed-fed infants whose mothers did not adhere to a cow’s milk protein and dairy–free diet, infants who received both eHF and AAF (mixed treatment), and those who were exclusively breastfed. The diagnosis of CMPA made by the RP was reassessed by the primary investigator (FKA) using the definition set by the European Society for Pediatric Gastroenterology, Hepatology, and Nutrition (ESPGHAN)[1]. A food challenge was not always performed, particularly when the likelihood of CMPA was high or when rechallenging was considered too risky[1]. Infants who did not meet the ESPGHAN diagnostic criteria were excluded from the study.
Data collected from medical records included epidemiological information, age at CMPA diagnosis, symptoms at presentation, treatment duration, nutritional history and diagnostic process, parental education and employment status, presence of additional food allergies, family history of atopy, and reasons for not initiating or continuing breastfeeding. The risk factors associated with CMPA development were also recorded. Relevant laboratory tests and imaging studies were reviewed and documented.
To address missing data in the medical records, families were contacted via telephone. During the call, the mothers were provided with a list of potential reasons for not breastfeeding and were asked to select those that applied. They were also allowed to provide additional responses. Infants for whom data remained incomplete were excluded from the study. To reduce potential recall bias during phone interviews, calls were made within a short period after the child’s clinical visit. To minimize subjectivity and variability in responses, caregivers were interviewed using standardized closed-ended questions with predefined response options, particularly for items related to breastfeeding barriers. When possible, information obtained from the caregivers was cross-checked with the child’s medical records to verify key details and enhance data accuracy.
All data were entered into a Microsoft Excel® spreadsheet, which was securely stored on a password-protected desktop computer. Fisher’s exact test was used to evaluate the associations between breastfeeding barriers and maternal characteristics because of the categorical nature of the data and small subgroup sizes. Breastfeeding barriers were classified as “Common” or “Less Common”, whereas maternal education was categorized as “Higher” (bachelor’s degree or PhD) or “Lower” (diploma, high school, or less), and work status was categorized as “Employed” or “Nonemployed”. Fisher’s exact test was used separately to evaluate the association of breastfeeding barriers with maternal work status and educational level. Results were expressed as odds ratios (ORs) with P values, and a significance threshold of P < 0.05 was used.
Descriptive statistics, including means, medians, interquartile ranges (Q1–Q3), and percentages, were used to summarize demographic and clinical characteristics. Categorical variables are presented as frequencies and percentages, whereas continuous variables are expressed as means ± SD. Associations between categorical variables were assessed using Fisher’s exact test. A P value < 0.05 was considered statistically significant. SPSS version 25 (IBM SPSS Statistics for Windows, IBM Corp. Armonk, NY, United States) was used to perform all statistical analyses. The study was conducted in accordance with the STROBE guidelines for reporting cross-sectional studies.
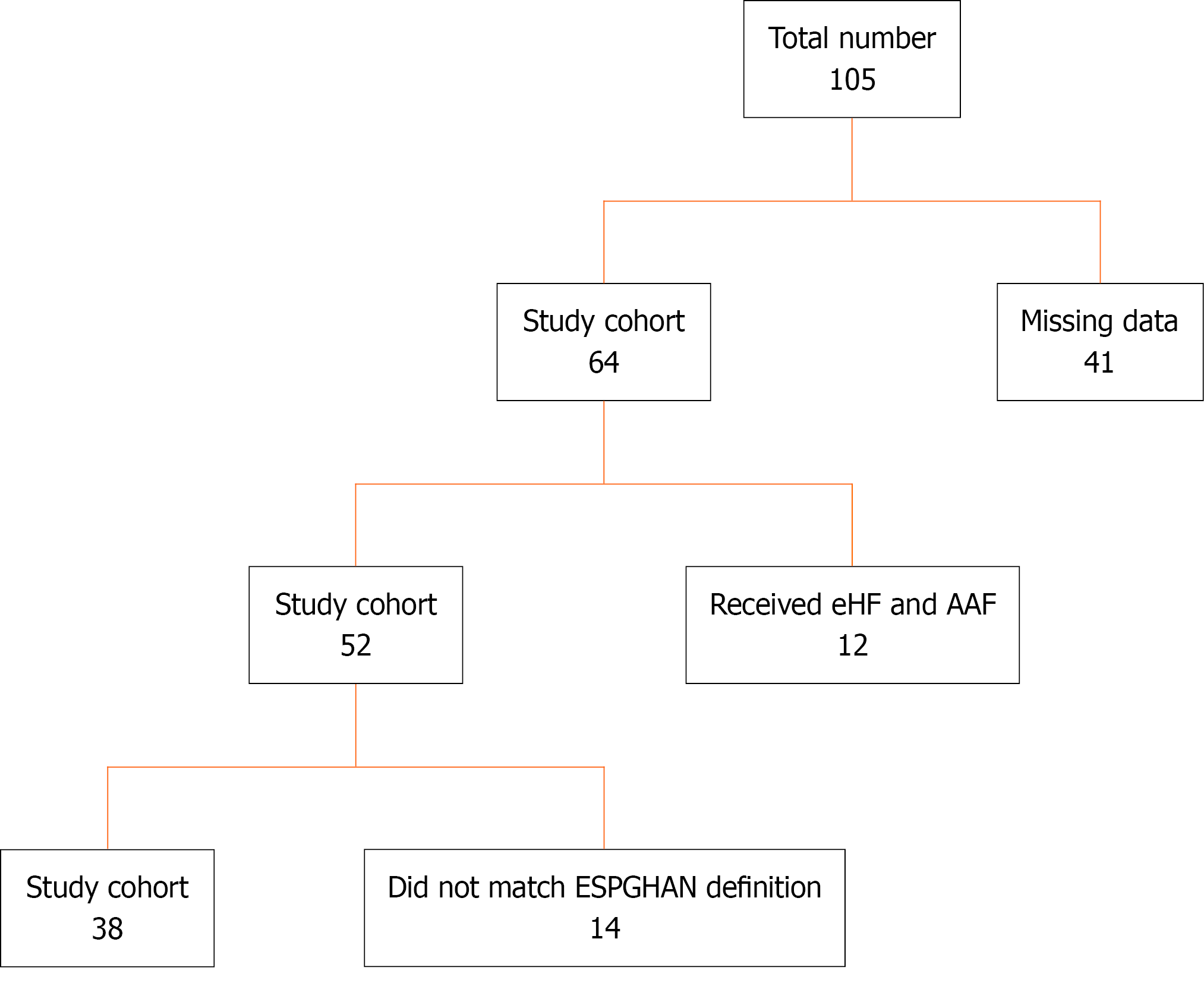
A total of 105 infants initially met the inclusion criteria for this study. Subsequently, 41 infants were excluded because of missing data in their medical records or lack of valid contact information. During the study period, eHF was available at the HUH Peds GI clinic on an intermittent basis. Consequently, infants were provided with eHF only when it was in stock; otherwise, the AAF was provided. Because of this inconsistency in treatment, children who received eHF were excluded from the final analysis.
As shown in Figure 1, 14 of the remaining 64 children did not meet the ESPGHAN diagnostic definition and were excluded. The final study cohort comprised 38 children. Table 1 summarizes the clinical characteristics of the study population.
| Category | Number | Percentage (%) |
| Total study cohort number | 38 | |
| Mean age at diagnosis (months, Q1-Q3) | 3.0 (0.5–5) | |
| Sex: Male | 24 | 63.2 |
| Symptoms at presentation: | ||
| Flatulence | 35 | 92.1 |
| Abdominal distension | 33 | 86.8 |
| Constipation | 18 | 47.4 |
| Recurrent vomiting | 18 | 47.4 |
| Diarrhea | 17 | 44.7 |
| Eczema (face and body) | 17 | 44.7 |
| Poor growth | 16 | 42.1 |
| Mucus in the stool | 15 | 39.5 |
| Eczema (face only) | 15 | 39.5 |
| Difficulty breathing and/or cough | 14 | 36.8 |
| Wheezes | 14 | 36.8 |
| Multiple food allergy | 13 | 34.2 |
| Edema or urticaria | 11 | 28.9 |
| Blood in the stool | 8 | 21.1 |
| Jaundice - indirect hyperbilirubinemia | 6 | 15.8 |
| Severe crying and/or irritability | 6 | 15.8 |
| Anemia | 5 | 13.2 |
| Increased liver enzyme levels (ALT and AST) | 3 | 7.9 |
| Allergy to medication(s) | 2 | 5.3 |
| Fever | 2 | 5.3 |
| Vomiting blood | 1 | 2.6 |
| Dehydration | 1 | 2.6 |
| Family history | ||
| Average number of children in the family (Q1-Q3) | 2.3 (2-3) | |
| Average child order in the family | 2.1 | |
| Other cases of CMPA in the family | 13 | 34.2 |
| Brother | 8 | |
| Sister | 3 | |
| Cousin | 1 | |
| Aunt | 1 | |
| Asthma, eczema, or any type of allergy in the family | 22 | 57.9 |
| Mother’s work status | ||
| Housewife | 21 | 55.3 |
| Employed | 17 | 44.7 |
| Father’s work status | ||
| Employed | 37 | 97.4 |
| Unemployed | 1 | 2.6 |
| Mother’s education level | ||
| Bachelor’s | 26 | 68.4 |
| High school | 9 | 23.7 |
| PhD | 1 | 2.6 |
| Diploma | 1 | 2.6 |
| Elementary school | 1 | 2.6 |
| Place of CMPA diagnosis: | ||
| Private clinic | 20 | 52.6 |
| MoH hospital/MoH health center | 10 | 26.3 |
| University of Jordan Hospital | 4 | 10.5 |
| Royal medical services hospital | 3 | 7.9 |
| Another country | 1 | 2.6 |
| CMPA diagnosis made by | ||
| Pediatric gastroenterologist | 27 | 71.1 |
| General pediatrician | 11 | 28.9 |
| Pregnancy: | ||
| Use of antibiotics during pregnancy–Yes | 11 | 28.9 |
| Use of antibiotics during pregnancy–No | 27 | 71.1 |
| Mode of delivery: | ||
| Cesarean section | 23 | 60.5 |
| Normal vaginal | 15 | 39.5 |
| Feeding: | ||
| Mixed (breast milk and formula) | 23 | 60.5 |
| Formula only | 15 | 39.5 |
| Duration of AA-based formula (months, Q1–Q3) | 13.89 (9-17.5) | |
| > 1 year | 19 | 50 |
| 6 months to 1 year | 14 | 36.8 |
| < 6 months | 5 | 13.2 |
Following the onset of CMPA symptoms, the average time taken by RPs to establish the diagnosis was 1.58 months (Q1-Q3: 0.5-2 months), with 31.6% of cases diagnosed within 1 month. In 28.9% of patients, the diagnosis was based solely on history and physical examination. In the remaining patients, additional diagnostic tools, such as stool studies, blood tests, and a combination of both in 13.2%, 21.1%, and 36.8%, respectively, were used.
Stool studies requested by the RPs included microscopic stool analysis for infection or inflammation and fecal calprotectin (FC) testing to evaluate intestinal inflammation. Blood tests included complete blood count, C-reactive protein, and immunoglobulin assays, including IgE. Radioallergosorbent tests were also performed in some children, particularly those with suspected multiple food allergies. On average, each infant’s formula was changed 3.1 times (range: 0-10) before the final diagnosis of CMPA was established, reflecting multiple switches between different formula types and brands. These changes were often initiated by parents, sometimes in consultation with their pediatricians, due to persistent symptoms or uncertainty about the diagnosis.
In addition to CMPA, approximately one-third (34.2%) of the children in this cohort had documented allergies to at least one other food. Specifically, four children were allergic to the following additional food items: One child to three items, three children each to two items, and five children each to one item. The most frequently reported allergens were egg (38%), fish (23%), nuts (23%), wheat (15%), and peanut (15%). The less commonly reported allergens were berries (5.3%), carrot, cinnamon, apple, sesame, avocado, and kiwi (each 2.6%).
Thirty-one children in the study were diagnosed with CMPA at ≤ 6 months of age, and seven were diagnosed at > 6 months of age. The average treatment duration until symptom resolution was 13.89 months (Q1-Q3: 9-17.5 months). Throughout the treatment period, the children received an AAF according to the MoH distribution protocol: Eight cans per month for children ≤ 6 months of age and four cans per month for those > 6 months of age. Some children received additional cans if medically necessary, particularly children with multiple food allergies or limited solid food options. Table 2 summarizes the treatment costs, with the average cost for CMPA management using AAF being approximately JD1802 ($2523) per child or JD130 ($183) per child per treatment month.
| Child age during treatment | Number of AAF cans dispensed during treatment | Treatment cost, JD ($) |
| < 6 months | 1150 | 23000 (32200) |
| 6 months to 1 year | 2275 | 45500 (63700) |
| Total | 3425 | 68500 (95900) |
Mothers were also surveyed regarding breastfeeding barriers. The most commonly reported reason was the maternal perception of insufficient milk supply (36.8%), followed by a personal preference not to initiate or continue breastfeeding (34.2%). Table 3 presents the full range of reported reasons for not breastfeeding.
| Reasons for not initiating/continuing breastfeeding | Number | Percentage (%) |
| Mother’s perception of inadequate milk supply | 14 | 36.8 |
| Personal choice: No specific reason | 13 | 34.2 |
| Work limitations | 4 | 10.5 |
| Baby-related causes: Latching difficulties and NICU admission | 3 | 7.9 |
| Maternal causes: Health issues and depression | 2 | 5.3 |
| Twin babies | 2 | 5.3 |
| Total | 38 | 100 |
To explore the relationship between breastfeeding barriers and maternal characteristics, Fisher’s exact test was performed. These barriers were classified into two categories: “common”, which included perceived inadequate milk supply and personal choice, and “less common”, which included infant-related issues, maternal illness, work-related limitations, and caring for twins. Maternal educational level was categorized into “higher education” (bachelor’s degree or PhD) and “lower education” (high school, elementary school, or diploma), whereas maternal work status was classified as “employed” and “nonemployed” (including housewives and those not working).
Fisher’s exact test was performed separately to determine breastfeeding barriers in relation to work status and educational level. The results revealed a significant association between breastfeeding barriers and maternal work status [OR = 7.20, 95% confidence interval (CI): 1.21-42.82, P = 0.03] (Table 4), indicating that employed mothers were 7.2 times more likely to encounter breastfeeding challenges than nonemployed mothers. In contrast, no significant association was observed between educational level and breastfeeding barriers (OR = 0.19, 95%CI: 0.01-2.70, P = 0.22), indicating that maternal education had a minimal effect on breastfeeding difficulties.
This study is the largest and most comprehensive investigation of CMPA among Jordanian infants, with an in-depth analysis of various factors associated with the condition, including treatment cost and breastfeeding barriers.
Although national prevalence data on CMPA in Jordan are limited, the condition has been well recognized and reported in several neighboring Middle Eastern countries with comparable prevalence rates. In Egypt, a cross-sectional study conducted among children under two years of age found a CMPA prevalence of 3.4%[11]. Similarly, in Saudi Arabia, estimates range between 2% and 3% among infants, based on clinical and immunologic criteria[12]. A Lebanese pediatric survey reported a self-reported prevalence of 3.8% and a physician-diagnosed rate of 1.9% for CMPA[13]. These regional data indicate that CMPA is a relatively common pediatric condition across the Middle East, underscoring the need for increased awareness and standardized diagnostic practices within the region.
Our findings show that the average age at diagnosis was 3 months (Q1-Q3: 0.5-5 months), with a diagnostic interval of 1.58 months from symptom onset (Q1-Q3: 0.5-2 months). These results reflect an earlier diagnosis and shorter time to diagnosis than those in a previous Jordanian study (average diagnostic age of 4.7 months; interval of 3.2 months)[14]. Globally, the diagnostic timing ranges from 2.8 months to 3.5 months[15,16]. The improved timing in Jordan may be attributed to increased awareness among parents and health care providers about CMPA symptoms and diagnostic criteria.
In our cohort, most patients with CMPA were diagnosed and referred by Peds GI rather than primary care physicians (PCPs) at a rate of 71.1% vs 28.9%. Diagnostic investigations were performed in most cases despite the ESPGHAN guidelines advocating for a clinical diagnosis without extensive reliance on laboratory tests[1,17]. Notably, FC testing shows high variability among affected individuals and healthy controls, limiting its diagnostic reliability[17].
Increased awareness among PCPs and Peds GI specialists could help reduce unnecessary investigations and further shorten diagnostic time. The issue of limited awareness of factors associated with CMPA, including treatment cost and breastfeeding barriers, is not unique to our setting. The diagnostic process for CMPA varies globally, and delays and multiple formula changes have been consistently reported. For example, a primary care-based practical guide in the United Kingdom highlighted frequent caregiver dissatisfaction due to prolonged diagnostic processes and inconsistent approaches[7]. Similarly, Canadian and European retrospective studies have described the substantial clinical and economic burdens associated with delayed CMPA diagnosis, including repeated formula switching and reduced QoL[5]. These findings align with our results and emphasize the universal need for standardized, timely diagnostic pathways, as advocated in international guidelines, such as those issued by ESPGHAN[1,16].
Most children in this study exhibited non-IgE-mediated symptoms of CMPA, often presenting as delayed gas
Elevated liver enzyme levels, specifically those of alanine transaminase and aspartate transaminase, were observed in three children, two of whom also had indirect hyperbilirubinemia. Although the mechanism linking CMPA to elevated liver enzymes is poorly understood, similar cases have been reported previously[14,24-27]. One hypothesis is that increased intestinal permeability due to allergic inflammation enables macromolecules to reach Kupffer cells in the liver[23,28]. These antigen-presenting cells may activate cytotoxic T-lymphocytes and trigger hepatic inflammation[29]. Notably, in our cohort, liver abnormalities resolved after CMPA treatment.
Four children also presented with isolated indirect hyperbilirubinemia, without elevated liver enzyme levels or signs of hemolysis or anemia. Their median age was 2 weeks, and their jaundice resolved with CMPA treatment. Although jaundice is an uncommon manifestation of CMPA, previous studies have indicated that neonatal hyperbilirubinemia may increase the risk of food allergies, including CMPA[30-33], possibly due to increased gut permeability and enhanced uptake of allergenic macromolecules[33]. Because our sample was too small to confirm this association, further research is warranted.
One-third of the children in our cohort had coexisting food allergies. Managing multiple food allergies poses significant challenges, including dietary restrictions that may lead to nutritional deficiencies, impaired growth, and reduced QoL for children and their families[3]. The need for special food preparation and avoidance of certain social situations can further increase anxiety and stress, thereby worsening QoL[34]. Considering the complex dietary res
The average treatment duration for CMPA in our study was 13.89 months (Q1-Q3: 9-17.5 months). The ESPGHAN guidelines recommend the use of eHF for 6-12 months after diagnosis before assessing for resolution[1]. Although 50% of children are expected to outgrow CMPA by the age of 1 year, > 75% do so by the age of 3 years[1]. Our cohort received AAF instead of eHF; however, the treatment duration was not shorter than the typical duration associated with eHF. This finding may reflect referral bias or a higher prevalence of multiple food allergies, resulting in a more complex clinical course. Prolonged treatment duration and dietary restrictions have important implications for health care providers and policymakers, including effects on family QoL, child nutrition, and economic burden[3,5].
The financial burden of CMPA is substantial. The average cost of AAF in our cohort was JD130 ($183) per child per month, which is substantially higher than previous estimates from the United States ($77.60) and Colombia ($73.40)[36,37]. This discrepancy may be partly attributed to formula prices, which range from $20-25 per can in Jordan compared with $12.23-$18.35 in Colombia. Additionally, the exclusive use of AAF rather than eHF may have increased costs. This financial pressure can cause stress and reduce treatment adherence, with potentially negative consequences for child health and family well-being.
Jordan’s CMPA treatment model is unique because the government, through the MoH, provides free formula to non breastfed infants with CMPA. However, families still bear the cost of medications for associated conditions (e.g., asthma and eczema), allergy investigations, epinephrine auto-injectors, and specialized food items[38,39]. National campaigns to promote breastfeeding and increase public awareness of CMPA could help reduce disease incidence and significantly lower treatment costs.
Several risk factors contribute to the development of CMPA, including the replacement of breastfeeding with formula feeding[8,40]. In our study, the primary barrier to breastfeeding was the maternal perception of inadequate milk supply (36.8%), consistent with findings from other Jordanian studies[41,42]. Additionally, approximately one-third of mothers cited “personal choice” as their reason for not breastfeeding. Together, these two barriers accounted for > 70% of the decisions to use formula, highlighting the need for targeted health education. With exclusive breastfeeding rates in Jordan remaining low (25%)[43], national efforts by the MoH and health care providers are urgently needed to promote breastfeeding and mitigate the CMPA burden.
Our findings also underscore the role of maternal employment in challenges associated with breastfeeding. Although mothers with higher education levels were less likely to report barriers, this trend was not statistically significant. In contrast, employed mothers were significantly more likely to experience difficulties in breastfeeding. Creating a supportive work environment, including longer maternity leave, flexible working hours, and designated breastfeeding spaces, can help mothers continue breastfeeding. These workplace policies can increase breastfeeding rates, reduce CMPA incidence, and benefit families and society at large. Notably, although the association between maternal employment and breastfeeding barriers was significant, the confidence interval was wide (95%CI: 1.21-42.82), indicating that the limited precision was due to the relatively small sample size.
The findings of this study may help guide regional clinical practice by highlighting the need to improve the awareness of CMPA among PCPs and general pediatricians. Notably, symptoms such as chronic constipation and elevated liver enzyme levels observed in our cohort are often overlooked as possible presentations of CMPA. Increased recognition of these atypical features, along with enhanced support for breastfeeding and earlier referrals, may help reduce unnecessary investigations, shorten the diagnostic process, and lower overall health care costs. Insights from this middle-income country setting may also help inform the adaptation of the ESPGHAN guidelines to better match the resources and practices available in the region.
In recent years, oral immunotherapy (OIT) has emerged as a promising approach for desensitization in children with CMPA. Several studies have reported that OIT can induce clinical tolerance in approximately 60%-80% of affected children, although the risk of adverse reactions remains significant and the long-term durability of tolerance is variable[44-46]. These findings suggest that OIT may become a useful therapeutic option for selected patients under close specialist supervision, complementing conventional dietary elimination strategies.
Our study has several limitations that should be acknowledged. First, the sample size was relatively small, which affected some statistical estimates and may limit the generalizability of the findings to a broader population. The small sample size was largely due to the application of strict inclusion criteria and the exclusion of cases with incomplete data; however, this approach ensured the inclusion of well-documented cases with high data integrity.
Second, the study was conducted at the HUH, which is a single center, which may not reflect the full spectrum of CMPA cases across Jordan. Nonetheless, our inclusion of patients referred from multiple health care centers adds some degree of representativeness and broadens the applicability of the findings. However, as a tertiary referral center, HUH may have received more severe or complex cases than nonreferral facilities, which could have introduced selection bias and limited the generalizability of the results to patients with milder disease often observed in primary care.
Third, exclusively breastfed infants were excluded from this study, which prevented the evaluation of CMPA manifestations in this subgroup. This decision was deliberate because the primary focus was on formula-fed infants in the context of treatment costs and breastfeeding barriers. In addition, infants who received both eHF and AAF were excluded to maintain consistency in treatment exposure; however, this may further limit the generalizability of our findings to real-world settings in which mixed regimens are used. These exclusions may have inadvertently introduced a selection bias toward patients with more complex or treatment-resistant disease who were referred to tertiary care, potentially overestimating the clinical and economic burden of CMPA.
Fourth, some data, particularly regarding breastfeeding barriers, were collected through phone interviews, which may have been subject to recall bias. Although this method can reduce accuracy, it was necessary to complete the missing information and better understand the factors influencing the mothers’ feeding decisions. Finally, the absence of a control group limited our ability to directly compare outcomes, such as treatment cost or growth parameters, with those of unaffected infants, which should be addressed in future prospective studies.
Despite these limitations, this study offers valuable insights into the clinical presentation, treatment cost, and breastfeeding challenges associated with CMPA in Jordanian infants. Future studies with larger sample sizes and more diverse populations are essential for validating and expanding these findings.
CMPA is an increasingly recognized condition with significant implications for child health, family QoL, and health care systems. In addition to its clinical impact, CMPA exerts emotional, social, and financial burdens on affected families. Identifying and addressing risk factors, such as reduced breastfeeding rates, and promoting public awareness and early intervention are critical for reducing the prevalence and severity of CMPA. Targeted education, supportive health care policies, and interventions at both the family and national levels can contribute to better outcomes and improved QoL for children and caregivers. These findings provide a foundation for the implementation of targeted interventions aimed at reducing the burden of CMPA and improving the health and well-being of children in Jordan and other comparable contexts. The study’s main limitations include its single-center, retrospective design and relatively small sample size, which may limit the generalizability of the findings. Future prospective, multicenter studies with larger cohorts are recommended to confirm these observations and explore additional predictors of clinical outcomes and breastfeeding barriers.
The authors thank Sara Khaled, BPharm, MS, biomedical statistician at the University of Jordan, for her support in reviewing and confirming the statistical analysis used in this study. We also thank Khetam Hawamdeh, BPharm, Head Pharmacist at Al-Hussein New Hospital, for her assistance in preparing the patient lists used for data collection.
| 1. | Koletzko S, Niggemann B, Arato A, Dias JA, Heuschkel R, Husby S, Mearin ML, Papadopoulou A, Ruemmele FM, Staiano A, Schäppi MG, Vandenplas Y; European Society of Pediatric Gastroenterology, Hepatology, and Nutrition. Diagnostic approach and management of cow's-milk protein allergy in infants and children: ESPGHAN GI Committee practical guidelines. J Pediatr Gastroenterol Nutr. 2012;55:221-229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 611] [Cited by in RCA: 491] [Article Influence: 35.1] [Reference Citation Analysis (6)] |
| 2. | Rona RJ, Keil T, Summers C, Gislason D, Zuidmeer L, Sodergren E, Sigurdardottir ST, Lindner T, Goldhahn K, Dahlstrom J, McBride D, Madsen C. The prevalence of food allergy: a meta-analysis. J Allergy Clin Immunol. 2007;120:638-646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1017] [Cited by in RCA: 828] [Article Influence: 43.6] [Reference Citation Analysis (3)] |
| 3. | Morou Z, Tatsioni A, Dimoliatis ID, Papadopoulos NG. Health-related quality of life in children with food allergy and their parents: a systematic review of the literature. J Investig Allergol Clin Immunol. 2014;24:382-395. [PubMed] |
| 4. | Sathya P, Fenton TR. Cow's milk protein allergy in infants and children. Paediatr Child Health. 2024;29:382-396. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (2)] |
| 5. | Sorensen K, Meyer R, Grimshaw KE, Cawood AL, Acosta-Mena D, Stratton RJ. The clinical burden of cow's milk allergy in early childhood: A retrospective cohort study. Immun Inflamm Dis. 2022;10:e572. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 6. | Nocerino R, Coppola S, Carucci L, Paparo L, De Giovanni Di Santa Severina AF, Berni Canani R. Body growth assessment in children with IgE-mediated cow's milk protein allergy fed with a new amino acid-based formula. Front Allergy. 2022;3:977589. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 7. | Venter C, Brown T, Shah N, Walsh J, Fox AT. Diagnosis and management of non-IgE-mediated cow's milk allergy in infancy - a UK primary care practical guide. Clin Transl Allergy. 2013;3:23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 118] [Cited by in RCA: 96] [Article Influence: 7.4] [Reference Citation Analysis (4)] |
| 8. | Sardecka I, Łoś-Rycharska E, Ludwig H, Gawryjołek J, Krogulska A. Early risk factors for cow's milk allergy in children in the first year of life. Allergy Asthma Proc. 2018;39:e44-e54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 23] [Article Influence: 2.9] [Reference Citation Analysis (3)] |
| 9. | Yang X, Zhou C, Guo C, Wang J, Chen I, Wen SW, Krewski D, Yue L, Xie RH. The prevalence of food allergy in cesarean-born children aged 0-3 years: A systematic review and meta-analysis of cohort studies. Front Pediatr. 2022;10:1044954. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 20] [Reference Citation Analysis (0)] |
| 10. | Jeong S. Factors influencing development of the infant microbiota: from prenatal period to early infancy. Clin Exp Pediatr. 2022;65:439-447. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 51] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 11. | Elmahdy EA, Maksoud HM, Al Seheimy LA, Hassan KA, Salem M. Frequency of cow milk protein allergy in children during the first 2 years of life in Damietta Governorate. Al-Azhar Assiut Med J. 2019;17:86. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 0.6] [Reference Citation Analysis (2)] |
| 12. | Baghlaf MA, Eid NMS. Prevalence, Risk Factors, Clinical Manifestation, Diagnosis Aspects and Nutrition Therapy in Relation to both IgE and IgG Cow’s Milk Protein Allergies among a Population of Saudi Arabia: A Literature Review. Curr Res Nutr Food Sci. 2021;9:375-389. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 13. | Irani C, Maalouly G. Prevalence of Self-Reported Food Allergy in Lebanon: A Middle-Eastern Taste. Int Sch Res Notices. 2015;2015:639796. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 17] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 14. | Altamimi E. Cow’s Milk Protein Allergy in South Jordan. J Med J. 2014;48:151-157. [DOI] [Full Text] |
| 15. | Vandenplas Y, Koletzko S, Isolauri E, Hill D, Oranje AP, Brueton M, Staiano A, Dupont C. Guidelines for the diagnosis and management of cow's milk protein allergy in infants. Arch Dis Child. 2007;92:902-908. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 302] [Cited by in RCA: 238] [Article Influence: 12.5] [Reference Citation Analysis (7)] |
| 16. | Luyt D, Ball H, Makwana N, Green MR, Bravin K, Nasser SM, Clark AT; Standards of Care Committee (SOCC) of the British Society for Allergy and Clinical Immunology (BSACI). BSACI guideline for the diagnosis and management of cow's milk allergy. Clin Exp Allergy. 2014;44:642-672. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 319] [Cited by in RCA: 253] [Article Influence: 21.1] [Reference Citation Analysis (3)] |
| 17. | Koninckx CR, Donat E, Benninga MA, Broekaert IJ, Gottrand F, Kolho KL, Lionetti P, Miele E, Orel R, Papadopoulou A, Pienar C, Schäppi MG, Wilschanski M, Thapar N. The Use of Fecal Calprotectin Testing in Paediatric Disorders: A Position Paper of the European Society for Paediatric Gastroenterology and Nutrition Gastroenterology Committee. J Pediatr Gastroenterol Nutr. 2021;72:617-640. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 65] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 18. | Connor F, Salvatore S, D'Auria E, Baldassarre ME, Acunzo M, Di Bella G, Farella I, Sestito S, Pensabene L. Cows' Milk Allergy-Associated Constipation: When to Look for It? A Narrative Review. Nutrients. 2022;14:1317. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 19. | 19 Meyer R, Vandenplas Y, Lozinsky AC, Vieira MC, Berni Canani R, du Toit G, Dupont C, Giovannini M, Uysal P, Cavkaytar O, Knibb R, Fleischer DM, Nowak-Wegrzyn A, Venter C. Diagnosis and management of food allergy-induced constipation in young children-An EAACI position paper. Pediatr Allergy Immunol. 2024;35:e14163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 20. | Mohammadi Bourkheili A, Mehrabani S, Esmaeili Dooki M, Haji Ahmadi M, Moslemi L. Effect of Cow's-milk-free diet on chronic constipation in children; A randomized clinical trial. Caspian J Intern Med. 2021;12:91-96. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 21. | Carroccio A, Scalici C, Maresi E, Di Prima L, Cavataio F, Noto D, Porcasi R, Averna MR, Iacono G. Chronic constipation and food intolerance: a model of proctitis causing constipation. Scand J Gastroenterol. 2005;40:33-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 38] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 22. | Zhang S, Wang R, Li D, Zhao L, Zhu L. Role of gut microbiota in functional constipation. Gastroenterol Rep (Oxf). 2021;9:392-401. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 128] [Cited by in RCA: 109] [Article Influence: 21.8] [Reference Citation Analysis (1)] |
| 23. | Carroccio A, Montalto G, Custro N, Notarbartolo A, Cavataio F, D'Amico D, Alabrese D, Iacono G. Evidence of very delayed clinical reactions to cow's milk in cow's milk-intolerant patients. Allergy. 2000;55:574-579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 33] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 24. | Saito M, Obi M, Kimura M. Infantile hepatic dysfunction improved by elimination of cows' milk formulas. Pediatr Allergy Immunol. 2005;16:445-448. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 25. | Tuna Kirsaclioglu C, Hizal G, Kocabas CN. Transient hypertransaminasemia related to cow's milk protein antigen induced proctocolitis in infants. Allergol Immunopathol (Madr). 2016;44:280-281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 26. | Yada K, Yoshida K, Sakurai Y, Kimura M, Yasuhara H, Tanaka I, Yoshioka A. Casein hydrolysate formula-induced liver dysfunction in a neonate with non-immunoglobulin E-mediated cow's milk allergy. J Investig Allergol Clin Immunol. 2008;18:67-70. [PubMed] |
| 27. | Palanisamy S, Srinivasan R, Perumal T. Unusual Presentation of Cow's Milk Protein Allergy in a Neonate. J Clin Neonatol. 2021;10:239-241. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 28. | Heyman M. Gut barrier dysfunction in food allergy. Eur J Gastroenterol Hepatol. 2005;17:1279-1285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 57] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 29. | Mehal WZ, Azzaroli F, Crispe IN. Antigen presentation by liver cells controls intrahepatic T cell trapping, whereas bone marrow-derived cells preferentially promote intrahepatic T cell apoptosis. J Immunol. 2001;167:667-673. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 76] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 30. | Wei CC, Lin CL, Shen TC, Kao CH. Neonatal jaundice and risks of childhood allergic diseases: a population-based cohort study. Pediatr Res. 2015;78:223-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 29] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 31. | Kuniyoshi Y, Tsujimoto Y, Banno M, Taito S, Ariie T. Neonatal jaundice, phototherapy and childhood allergic diseases: An updated systematic review and meta-analysis. Pediatr Allergy Immunol. 2021;32:690-701. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 32. | Sun HL, Lue KH, Ku MS. Neonatal jaundice is a risk factor for childhood allergic rhinitis: a retrospective cohort study. Am J Rhinol Allergy. 2013;27:192-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 33. | Raimondi F, Indrio F, Crivaro V, Araimo G, Capasso L, Paludetto R. Neonatal hyperbilirubinemia increases intestinal protein permeability and the prevalence of cow's milk protein intolerance. Acta Paediatr. 2008;97:751-753. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 34. | Ciaccio C, Dunne J, Bever A, Johnston K, Kowal S, Seetasith A, Lloyd A, Mickle A. Living with and Caring for People with Multiple Food Allergies: A Qualitative Study. Patient Prefer Adherence. 2024;18:1949-1960. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 35. | Larson K, McLaughlin J, Stonehouse M, Young B, Haglund K. Introducing Allergenic Food into Infants' Diets: Systematic Review. MCN Am J Matern Child Nurs. 2017;42:72-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 18] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 36. | Fakih-Botero I, Cerchiaro-Mejia MC, Rusinque MJ, Palacios-Ariza MA, Plata-Garcia CE, Peña-Hernández S. Out-of-pocket expenses and parent reported quality of life in children with cow's milk protein allergy in Bogotá, Colombia. J Pediatr Gastroenterol Nutr. 2024;79:18-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 37. | Gupta R, Holdford D, Bilaver L, Dyer A, Holl JL, Meltzer D. The economic impact of childhood food allergy in the United States. JAMA Pediatr. 2013;167:1026-1031. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 289] [Cited by in RCA: 381] [Article Influence: 29.3] [Reference Citation Analysis (3)] |
| 38. | Cawood AL, Meyer R, Grimshaw KE, Sorensen K, Acosta-Mena D, Stratton RJ. The health economic impact of cow's milk allergy in childhood: A retrospective cohort study. Clin Transl Allergy. 2022;12:e12187. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 39. | Fong AT, Ahlstedt S, Golding MA, Protudjer JLP. The Economic Burden of Food Allergy: What We Know and What We Need to Learn. Curr Treat Options Allergy. 2022;9:169-186. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 51] [Article Influence: 12.8] [Reference Citation Analysis (8)] |
| 40. | Food Allergy in Children and Young People: Diagnosis and Assessment of Food Allergy in Children and Young People in Primary Care and Community Settings. London: National Institute for Health and Clinical Excellence (NICE); 2011 Feb- . [PubMed] |
| 41. | Khasawneh W, Khasawneh AA. Predictors and barriers to breastfeeding in north of Jordan: could we do better? Int Breastfeed J. 2017;12:49. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 30] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 42. | Abuidhail J, Al-Modallal H, Yousif R, Almresi N. Exclusive breast feeding (EBF) in Jordan: prevalence, duration, practices, and barriers. Midwifery. 2014;30:331-337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 43. | UNICEF. Factsheet on the Situation of Children in Jordan. 2024. [ cite 2 May 2025]. Available from: https://www.unicef.org/mena/media/26706/file. |
| 44. | Wang Y, Liu S, Lu M, Guo J, Lv C, Huang L. Oral immunotherapy for cow's milk allergy in children: a systematic review and meta-analysis. Front Immunol. 2025;16:1570050. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 45. | Tang L, Yu Y, Pu X, Chen J. Oral immunotherapy for Immunoglobulin E-mediated cow's milk allergy in children: A systematic review and meta analysis. Immun Inflamm Dis. 2022;10:e704. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 46. | Tosca MA, Olcese R, Marinelli G, Schiavetti I, Ciprandi G. Oral Immunotherapy for Children with Cow's Milk Allergy: A Practical Approach. Children (Basel). 2022;9:1872. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/