This editorial refers to "Palpable vs non-palpable breast cancers in screened populations: Clinicopathological features and prognostic implications" by Improta et al, 2026; https://doi.org/10.5306/wjco.v17.i2.115245.
INTRODUCTION
Breast cancer is one of the most common malignant tumors among women worldwide. In recent years, approximately 2.3 million new cases have been reported each year globally, and the incidence of breast cancer is continuously increasing, becoming a significant public health issue threatening women’s health[1]. Studies have shown that breast cancer can be prevented and its mortality rate can be reduced through early screening and diagnosis[2]. In clinical diagnosis and treatment, palpable tumors can mostly be detected by palpation. Such tumors are usually larger in size with prominent clinical manifestations that cannot be ignored, and the patients generally have a relatively poor prognosis. In contrast, non-palpable tumors cannot be identified by palpation and are thus more prone to be overlooked in clinical practice. In low-income areas, due to the scarcity of medical resources, the mortality rate of tumors remains high. In contrast, in high-income areas, the reduction in the tumor mortality rate is mainly attributed to the establishment of standardized population screening systems and the regular implementation of early diagnosis[3]. This study, based on relevant literature, systematically reviewed and compared the clinical application advantages and limitations of mammography, ultrasound, and breast magnetic resonance imaging (MRI). Among them, mammography, as an important imaging method for early screening and diagnosis of breast cancer, provides a reliable basis for the precise diagnosis of breast tumors that are non-palpable to the naked eye in clinical practice[4]. The differences in biological behavior and clinical pathological characteristics between palpable and non-palpable tumors are significant, and these differences directly affect the treatment options and prognosis of patients. The large-scale retrospective cohort study conducted by Improta et al[5], published in the recent issue of the World Journal of Clinical Oncology, filled this gap. This study focused on breast cancer patients who were screened as eligible and had negative clinical lymph nodes. It systematically compared the clinical and pathological characteristics as well as long-term prognosis of palpable and non-palpable tumors, providing high-quality evidence to answer the aforementioned core questions. Based on the existing clinical research results, this article systematically analyzes the prognostic differences between palpable and non-palpable tumors in breast cancer screening, as well as the screening and prevention aspects, providing a reference for clinical practice.
PALPABLE TUMORS EXHIBIT A HIGHER MALIGNANT POTENTIAL THAN NON-PALPABLE TUMORS
Definition and current clinical cognitive status
The palpable tumor refers to a solid mass in the breast that can be clearly felt through standard physical examinations (including self-examination by the patient or clinical examination by the doctor), and it is an important indicator of the onset of clinical symptoms of the disease. The non-palpable tumor, on the other hand, refers to an abnormal lesion that cannot be detected through a comprehensive and meticulous physical examination but can only be identified through imaging examinations (such as mammography, ultrasound, or breast MRI). It is a manifestation of the disease being in a subclinical state. Most patients have insufficient knowledge about the symptoms and screening methods of breast cancer. They often only consider “a palpable lump” as the sole sign of the disease, while ignoring the characteristic that early-stage tumors usually have no symptoms. Some patients even develop an avoidance mentality due to fear of the disease and refuse to seek medical examination, thus causing the tumor to progress from the “non-palpable” early stage to the “palpable” middle and advanced stages[6], missing the best treatment opportunity.
Biological characteristic differences
Improta et al’s study[5], compared with the non-palpable tumors identified through imaging screening, the volume of the tumors that could palpable was larger (the average diameter of the tumors in the palpable group was 17.5 ± 8.6 mm, while that in the non-palpable group was 11.0 ± 6.7 mm). This was not merely due to the difference in size. It can be regarded as an external manifestation of the unique biological characteristics. From the perspective of core characteristics, palpable tumors tend to have a faster growth rate. This feature not only shortens the subclinical latency period, allowing the tumors to progress to the clinically palpable lesion stage before they can be detected through routine screening, but also often accompanies a series of highly invasive-related manifestations, such as a higher histological grade [palpable (G3): 33% vs non-palpable (G3): 16.3%], a significantly increased Ki-67 proliferation index (palpable: 24.7% ± 11.9% vs non-palpable: 15.1% ± 9.4%), and a higher proportion of triple-negative malignant subtypes (palpable: 37.1% vs non-palpable: 20.6%). In terms of clinical outcome-related indicators, the risk of lymph node metastasis by palpable tumors is significantly higher (palpable: 27.6% vs non-palpable: 16.7%), and there is a greater likelihood of pathological manifestations such as lymphatic and vascular invasion. These pathological features are closely associated with a lower disease-specific survival rate[7-9].
Differences in treatment strategies
In terms of treatment strategies, due to the larger palpable tumor lesions and later stage of the disease, the usage rates of total mastectomy and axillary lymph node dissection are higher, and the demand for adjuvant chemotherapy is more urgent. The application rates of breast-conserving surgery and hormone therapy are lower than those for non-palpable tumors[10]. This comprehensive difference from biological characteristics, clinical pathological manifestations to treatment strategies and prognosis highlights the clinical value of palpable tumors as an independent risk stratification indicator, and also suggests that more targeted individualized treatment plans should be developed in diagnosis and treatment. Therefore, for such breast tumors, clinical screening and assessment strategies should be more proactive to achieve early detection and diagnosis.
ANALYSIS OF THE IMPACT ON BREAST CANCER PROGNOSIS
The influence of tumor pathological characteristics
The core difference in breast cancer lies in invasive cancer and non-invasive cancer. Invasive cancer has the biological ability to invade surrounding tissues and undergo distant metastasis, and different molecular subtypes of invasive cancer show significant differences in clinical characteristics and prognosis. Among them, triple-negative invasive breast cancer is particularly prominent: Due to the fact that the tumor cells of this subtype are negative for estrogen receptors, progesterone receptors, and human epidermal growth factor receptor 2 (HER2), patients usually cannot benefit from endocrine therapy or anti-HER2 targeted therapy. This subtype of tumor typically exhibits higher invasive characteristics and is associated with a higher risk of recurrence and metastasis as well as poor prognosis. In contrast, non-triple-negative invasive breast cancer (including hormone receptor positive type and HER2 overexpression type) has clear therapeutic targets and can achieve better treatment outcomes through endocrine therapy or anti-HER2 targeted therapy. This tumor subtypes tend to exhibit lower invasiveness and are associated with relatively favorable prognosis[11,12].
The influence of the patient’s age
Age is an important factor influencing the prognosis of breast cancer patients, and the prognostic characteristics of young and elderly patients show significant differences. Young breast cancer patients (≤ 35 years old) have a lower incidence of comorbidities (such as diabetes, etc.), but their tumor cell proliferation activity is stronger, the tumor stage at diagnosis is later, and they are also affected by factors such as hormonal fluctuations in the body. As a result, the risk of tumor recurrence is significantly increased, and the overall prognosis is relatively poor[13]. The situation for elderly breast cancer patients (aged 65 and above) is the opposite. Although these patients have more comorbidities (such as diabetes, hypertension, coronary heart disease, etc.) and more severe underlying diseases, the tumor stage at diagnosis is generally earlier, and the proportion of high expression of hormone receptors is higher. Therefore, their prognosis is more favorable compared to younger patients. It is worth noting that although there are significant differences in recurrence risk and disease characteristics between young and elderly patients, the research results show that there are no statistically significant differences in overall survival and mortality rates between the two groups[14].
BREAST CANCER SCREENING METHODS AND SCREENING STRATEGIES DURING PREGNANCY
With the continuous advancement of technology and the increasingly refined screening techniques, early screening, early diagnosis, and early treatment of breast cancer have become key means to effectively reduce its incidence and mortality rate. Currently, the commonly used breast screening methods in clinical practice include Mammography, ultrasound, breast MRI, etc. Each of these techniques, based on its unique imaging principles and clinical characteristics, has its own advantages in breast lesion screening and can specifically meet the screening needs of different scenarios and populations.
Mammography is a widely used screening technique for breast cancer. It cannot only effectively detect benign and malignant breast lesions, but also can precisely identify calcification foci. For early-stage lesions such as ductal carcinoma in situ, which are mainly characterized by calcification, its identification efficiency is significantly superior to that of other single techniques. This technology has the advantages of simple operation, short examination time, low cost, high patient acceptance, and is suitable for large-scale population screening[15].
Ultrasound, as a supplementary technique for breast cancer screening, has the characteristics of “no radiation, high adaptability, and multi-dimensionality”. It can penetrate dense breast tissues to detect hidden lesions, compensating for the insufficient penetration of mammography in dense breast tissue. At the same time, by evaluating the lesions from multiple dimensions, when combined with mammography, it can further enhance the screening ability and reduce the risk of missed diagnosis[16].
Breast MRI, as a high-resolution imaging examination technique, can clearly display the location, size, infiltration range of the lesion, as well as its anatomical relationship with surrounding tissues. It has a prominent display advantage for small lesions and soft tissue abnormalities, providing important reference for tumor staging assessment and the formulation of treatment plans[17]. However, the use of breast MRI alone for diagnosing breast lesions still has certain limitations. For breast cancer patients with breast cancer gene mutations and a clear genetic background who are at high risk, breast MRI should be the preferred examination method, and combined with mammography for screening[18]. This combined approach can effectively enhance the sensitivity of lesion detection and reduce the risk of missed diagnoses; it not only enables precise monitoring of high-risk individuals but also takes into account the practicality and ease of operation in clinical applications, providing a reliable basis for early intervention in breast cancer. The breast tissue of women undergoes periodic changes during the menstrual cycle, characterized by glandular hyperplasia and thickening, accompanied by enhanced blood flow signals and significant background enhancement. These physiological changes can interfere with the signal contrast between the lesion and normal tissue, easily causing benign hyperplasia to be mistaken for suspicious lesions, thereby resulting in false positive results. Therefore, selecting the appropriate scanning time can help minimize such interference and enhance diagnostic accuracy[19].
The Multimodal Artificial Intelligence Germline Genetic Testing model has shown outstanding performance in breast cancer screening. Its core lies in integrating the histopathological microenvironment features of the entire section images with the clinical phenotypic data from electronic health records. Through the cross-modal latent representation unification mechanism, it mines complementary biological information and its prediction performance is significantly superior to that of single-modal models. It can be applied to various sample types such as surgical resection specimens, biopsy samples, etc. It has a good predictive effect for different tumor sizes and most molecular subtypes. Moreover, it adopts weak supervision learning and clustering sampling strategies, and does not require expensive manual annotation. It can be carried out solely based on conventional pathological sections and electronic medical records, with controllable costs and strong scalability, making it suitable for promotion in grassroots medical institutions. In addition, the model focuses on predicting germline breast cancer gene 1/2 mutations. The results can directly guide the selection of polyadenosine-diphosphate-ribose polymerase inhibitor therapy and high-risk family cascade testing, providing strong support for personalized treatment and precise risk assessment. This significantly enhances the accessibility and clinical translational value of genetic testing[20].
The physiological changes during pregnancy can increase the difficulty of breast cancer screening and diagnosis, posing a significant clinical challenge. For this specific group, it is necessary to select safe and appropriate imaging techniques based on age, risk level, and clinical symptoms. The core principle is to prioritize the use of non-radiative or low-radiative methods to minimize the risk of fetal radiation exposure. Ultrasound does not cause ionizing radiation and is safe and reliable for the mother and fetus[21]. Moreover, its sensitivity is higher than that of mammography. It is the preferred initial imaging method for screening breast cancer during pregnancy. Although mammography involves a slight level of radiation, The American College of Radiology Appropriateness Criteria[22] indicate that it is safe for screening and diagnosis in pregnant women, and the fetus only receives an extremely low dose of indirect scattered radiation, with no adverse effects. For women aged 40 and above with any risk level, and those aged 25 and above with high-risk pregnancy, breast mammography (including digital breast tomosynthesis) screening can be prioritized; for women with dense breasts, ultrasound can be combined for supplementary assessment to improve the detection rate of lesions. For pregnant women under 30 years old and those with new clinical symptoms, the initial screening is mainly based on ultrasound; when ultrasound indicates suspicious lesions, breast mammography is combined for further diagnosis to enhance the assessment accuracy. Contrast-enhanced MRI is prohibited during pregnancy because the gadolinium contrast agent can pass through the placental barrier and enter the fetal blood circulation, dissociating into toxic free gadolinium ions[21]. The study suggests that non-enhanced breast MRI can be used as a supplementary diagnostic method for breast cancer during pregnancy. However, the current clinical research is not sufficient, and the relevant conclusions still need to be further verified[23,24].
OVERDIAGNOSIS AND FALSE POSITIVES IN BREAST SCREENING
Overdiagnosis is the core pain point in the field of breast screening. The research conducted by Welch et al[25] indicates that the abnormal lesions detected during screening often receive excessive medical treatment despite not requiring it. This issue is underpinned by significant methodological challenges, including the lack of a unified criterion for determining “benign lesions”, as well as methodological flaws in risk stratification of the lesions. These factors lead to frequent overdiagnosis. At the same time, conventional screening techniques such as mammography, ultrasound, and MRI cannot avoid false positives. The core root cause lies in the inherent balance contradiction between sensitivity and specificity. Coupled with inherent deficiencies in methodology (defects in the technical characteristics of different methods, inconsistent positive determination standards), and the clinical excessive pursuit of sensitivity while neglecting the decline in specificity, this ultimately results in a persistently high false positive rate, creating a dual diagnostic dilemma with excessive diagnosis, which hinders the improvement of screening quality.
The combined harm of overdiagnosis and false positives imposes a double burden on individuals and the medical system: In overdiagnosis, unnecessary treatments such as surgery and radiotherapy do not bring significant survival benefits[26]. Instead, it will harm the patient’s body, cause long-term psychological distress, waste medical resources and increase the burden on patients for follow-up visits; false positives will exacerbate the patient’s psychological stress, reduce the compliance with screening, and increase the economic burden on low-income groups by triggering unnecessary medical treatments[27]. Moreover, invasive procedures such as excessive needle biopsy not only increase the risk of complications but also further waste medical resources and impose a heavier economic burden[28].
Regarding the issues of overdiagnosis and false positives, the two major strategies proposed by the clinical field are quite targeted, but they have obvious limitations and face significant methodological challenges: The integration of artificial intelligence-assisted screening into the double-reading process theoretically can enhance detection efficiency, reduce the recall rate, and alleviate the burden on doctors. Zhou et al[29] conducted research indicating: It has methodological flaws such as training data selection bias, insufficient generalization ability of the model, poor interpretability of the diagnostic results, difficulty in meeting the needs of different levels of medical institutions, and even the possibility of increasing diagnostic uncertainty. Although standardized diagnostic criteria and operations can reduce diagnostic heterogeneity and avoid false positives caused by subjective errors, they lack quantitative and repeatable methodological support. Moreover, they are affected by the precision of medical equipment in medical institutions and the professional level of doctors. Additionally, there is a lack of an effective supervision and assessment mechanism. Therefore, the implementation of the norms is difficult and it is impossible to fundamentally solve the dual predicament.
In conclusion, the issues of overdiagnosis and false positives in breast cancer screening are the result of the combined effects of inherent technical limitations, imperfect methodology systems, imbalanced diagnostic and therapeutic concepts, and insufficient implementation of response strategies. The current strategies have failed to break through the bottleneck of balancing sensitivity and specificity, nor have they addressed the core methodological challenges. In the future, we need to optimize the screening system with critical thinking, taking into account technological innovation, unified determination standards, addressing data bias, standard implementation, and concept guidance, in order to reduce the harm of overdiagnosis and false positives, and achieve a two-way improvement in the accuracy and practicality of screening.
STAGE MIGRATION AND SCREENING BIAS FOR PALPABLE TUMORS AND NON-PALPABLE TUMORS
Previous studies have limitations in their understanding of the differences in prognosis between the two types of tumors. The differences are not caused by a single factor but are the result of the combined effects of tumor stage progression and screening bias. The methodological flaws in the existing studies have further exacerbated the conclusion bias, making it difficult to reveal the essential differences. Tumors that are non-palpable have an early advantage due to “stage migration”. Previous studies have confirmed that the disease-free survival period of breast cancer patients diagnosed through screening is longer than that of those diagnosed based on clinical symptoms[30]. However, most of these conclusions are based on observational studies that fail to adequately control for confounding factors, and the studies often mistakenly equate staged migration with clinical benefits, resulting in significant cognitive biases. Screening bias and insufficient correction are the core methodological challenges in the prognosis assessment of these two types of tumors. These biases have been underestimated in previous studies and have exaggerated the illusion of prognosis disparity. Self-selection bias is difficult to avoid[31], the health advantages of the screened population may mislead the attribution of prognosis. The lead-time bias[32] causes studies to mistakenly interpret the earlier detection time as an extension of survival period, and the lack of standardization in bias correction methods results in the incompatibility of research results.
The comparative study of the prognosis of these two types of tumors also faces multiple methodological challenges: The existing studies solely rely on clinical staging to assess tumor progression, without integrating biomarker information, which leads to attribution bias[31]. Most studies rely on data from single-center small samples, and the incomplete recording of tumor detection methods further exacerbates the bias[30]. Previous studies were mostly of retrospective design and overly selected samples, resulting in conclusions lacking representativeness and generalizability[33].
The previous studies’ deficiencies in terms of cognition and methodology prevented them from objectively reflecting the essential prognostic differences between the two types of tumors and the true benefits of screening. Future research needs to overcome three major challenges: Adopting the method proposed by Buschmann et al[32] to establish a standardized bias correction system, integrating clinical staging and biomarker information[31], and prioritizing the use of large-scale population-level registration data and standardizing the recording[30]. Only in this way can reliable evidence-based basis be provided for the optimization and precise management of tumor screening.
THE ADVANTAGES AND CHALLENGES OF INTEGRATING ARTIFICIAL INTELLIGENCE IN BREAST CANCER SCREENING
The integrated application of artificial intelligence, as an innovative technological approach in the field of breast cancer screening, not only effectively overcomes the inherent limitations of the traditional single-screening method, but also opens up a brand-new direction for the precise and efficient early screening and diagnosis of breast cancer.
The integrated application of artificial intelligence and imaging technology provides a crucial support for alleviating the shortage of human resources in the field of imaging. Compared with the traditional double-doctor review and reading of mammograms, artificial intelligence can independently undertake the initial screening work. Without reducing the overall diagnostic accuracy rate, it effectively reduces the workload of radiologists, enabling limited medical resources to focus on the diagnosis of difficult cases, precise diagnosis and decision-making, and other high-value core aspects[34].
Liquid biopsy can achieve non-invasive, real-time and dynamic monitoring of tumors by analyzing biomarkers such as circulating tumor DNA and circulating tumor cells in body fluids like blood and urine[35]. However, in early-stage breast cancer patients, the abundance of circulating tumor DNA in the peripheral blood is extremely low (typically less than 0.1%), and it is easily interfered with by normal free cell DNA, making it difficult for traditional detection methods to effectively identify it[36]. Artificial intelligence technology can precisely identify the characteristics of methylation signals, significantly breaking through this detection bottleneck[37]. Gao et al[38] proposed that by combining microdroplet digital polymerase chain reaction technologies with machine learning (ML) algorithms, it is possible to achieve ultra-sensitive detection of 0.001% low-concentration methylation signals. Zhang et al[39] developed a 26-marker panel of free cell DNA methylation signatures combined with the ML algorithm. When used in conjunction with mammography, the detection specificity was improved by 3.89% compared to the individual methylation model, effectively avoiding overdiagnosis and unnecessary invasive examinations. The THEMIS model integrates four modal data including DNA methylation, fragmentomics, copy number variation and terminal motifs through ensemble learning. Its overall cancer detection area under the curve reaches 0.966, demonstrating significantly superior performance compared to single-modal analysis (the area under the curve of methylation single-modal analysis is 0.947)[40]. The ML classifier based on methylation features can accurately identify the origin of tumor tissues[41], solving the core problem of “only knowing there is cancer but not knowing the primary site” in multi-cancer early detection, and providing a clear direction for the subsequent formulation of treatment and diagnosis plans[42].
Although artificial intelligence has provided new ideas for breast cancer diagnosis and treatment through multi-technology integration, its clinical application faces many limitations and reflects methodological flaws in related research, making it difficult to integrate into the standard diagnosis and treatment system. The clinical value of this technology needs to be carefully evaluated.
Lukac et al[43] have shown that current research in artificial intelligence is mainly conducted with small samples and a single-center retrospective approach, which has obvious methodological shortcomings. The selection bias in retrospective studies leads to insufficient sample representativeness and poor extrapolation of results, the lack of support from large-scale real-world data hampers the generalization ability of the algorithm. The absence of long-term follow-up makes it impossible to verify the stability of the algorithm and the cumulative harm of false positives. Moreover, the efficacy calculation does not take into account the actual clinical issues, resulting in an overestimation of the results. Therefore, multi-center prospective studies are urgently needed to validate these findings.
Freeman et al[44] have confirmed through their research that there is significant heterogeneity in the detection performance of different artificial intelligence systems. Among the 36 evaluated systems, 94% of their accuracy rates were lower than those of a single radiologist. Moreover, due to the inconsistent validation indicators and the absence of a quality assessment system, the research results are unreliable, making it impossible to conduct horizontal comparisons between the systems and thus difficult to gain clinical trust. Bao et al’s research[45] indicates that the core technologies such as high-throughput next-generation sequencing and microfluidic circulating tumor cell capture are costly, and the research has not conducted cost optimization and grassroots adaptation design. Therefore, they are difficult to be implemented at the grassroots level[35]. Kan et al’s research[46] has confirmed that there is currently no unified standard for key steps in liquid biopsy. The consistency of test results is poor, and they cannot be recognized among different laboratories[47]. In addition, medical insurance coverage is not available, and there is a lack of cost-benefit analysis. The accessibility of the technology and its large-scale promotion are severely restricted[48].
The current guidelines clearly support the clinical application potential of artificial intelligence and liquid biopsy technology in the combined screening of multiple types of malignant tumors. The synergy of the two can break through the limitations of traditional single-cancer screening and provide a key direction for non-invasive and efficient early screening of tumors. This technology needs to meet four core conditions - regulatory compliance, mature technology, sufficient evidence, and complete supporting facilities - to be included in clinical routine applications. Currently, it is in a critical stage of transitioning from clinical research to clinical application. To promote the standardized clinical transformation of this technology, it is necessary to address the limitations of artificial intelligence in clinical applications of breast cancer, establish unified research norms, conduct multi-center prospective studies, optimize scenario adaptation and costs, in order to drive the standardization of clinical transformation, empower breast cancer diagnosis and treatment, achieve early detection and intervention of malignant tumors, and reduce the mortality rate related to malignant tumors[49].
THE INFLUENCE OF INTERNAL AND EXTERNAL FACTORS ON TUMOR DEVELOPMENT
The quality of tumor screening is significantly influenced by external factors. During the coronavirus disease 2019 (COVID-19) pandemic, the accessibility of routine screening decreased, and public attention to preventive medical care also declined. As a result, tumors that could have been detected through early screening failed to be identified in a timely manner until they became palpable tumors and were diagnosed. Gligorov et al[50] point out this issue. During the pandemic, a significant number of medical resources were diverted to treating COVID-19. Due to isolation and travel restrictions, cancer patients were unable to complete follow-up and monitoring, resulting in the failure to intervene in tumor progression in a timely manner. Eventually, the tumors were only discovered at an advanced stage. Overall, the COVID-19 pandemic did not directly promote tumor growth. Instead, it interfered with the screening and diagnostic processes, reducing early intervention for tumors, thus causing tumors to be detected at a more advanced, less accessible stage.
The external factor of the COVID-19 pandemic has significantly impacted the screening work for breast cancer patients, and internal factors also play a crucial role in the occurrence and progression of breast cancer. Breast cancer patients, on the one hand, are affected by the pandemic, with their access to medical treatment being restricted and screening plans being forced to be suspended or delayed; on the other hand, the disease itself and its related symptoms can easily cause them to fall into negative emotions such as anxiety and depression. Moreover, the high recurrence rate of the disease and the potential risk of death further trigger continuous panic and psychological stress, resulting in a dual problem of physical and mental distress[51]. In addition, the physical pain and discomfort caused by the tumor further trigger or exacerbate their emotional abnormalities. Li et al[52] reported that anxiety and depressive emotions can cause the hypothalamic-pituitary-adrenal axis to over-activate, leading to a series of abnormal reactions in the endocrine system, thereby disrupting the balance between estrogen and progesterone and further exacerbating the disease. What is even more worthy of attention is that such negative emotions may also cause some patients to be deeply afraid of the disease, develop a strong avoidance mentality, and be reluctant to seek medical treatment and undergo examinations actively. As a result, they miss the best window period for early intervention of the tumor, and the disease cannot be effectively controlled in a timely manner[6].
BREAST CANCER RISK STRATIFICATION AND EARLY PREVENTION AND CONTROL STRATEGIES
According to relevant research, the research subjects can be classified into three levels based on the risk grade of breast diseases: Those without any high-risk factors are classified as the low-risk group; those diagnosed with dense breast tissue but without other high-risk factors are classified as the medium-risk group; those with any one of the high-risk factors (such as genetic susceptibility, breast cancer gene mutation, etc.) are defined as the high-risk group, and stratified management strategies are implemented for different risk levels of the population[53]. At the same time, the core concept of “prevention before disease onset, and prevention of disease progression after onset” is deeply integrated into the screening and prevention system for risk populations, thereby enhancing the overall disease prevention and control efficiency.
Low-risk individuals should implement scientific daily health management. By adopting a light diet, engaging in moderate exercise[54], at the same time, combined with the traditional Chinese exercise form “Baduanjin”, this intervention method can improve people’s physical and mental conditions, enhance their quality of life, and also help alleviate physical discomfort symptoms. This exercise form can achieve this by unblocking meridians, regulating qi and blood, strengthening the body, and regulating emotions[55]. It is also recommended that regular screenings be conducted once every two years[53].
For the medium-risk group, based on the patient’s menopausal status and individual differences, conservative treatment options such as endocrine therapy or breast-conserving surgery can be chosen. At the same time, traditional Chinese medicine intervention can be supplemented. Gui et al[56] have confirmed that the Taohong Siwu decoction can effectively inhibit the proliferation, invasion and metastasis of breast cancer cells by targeting and regulating multiple signaling pathways such as Kirsten rat sarcoma viral oncogene homologue and phosphatidylinositol 3-kinase (PI3K)/protein kinase B (AKT), as well as key molecular targets such as myelocytomatosis oncogene and epidermal growth factor, and can also induce apoptosis of tumor cells. Li et al’s research[57] indicates that Xihuang pill can inhibit tumor growth and metastasis, delay the development of breast hyperplasia, and enhance the immune function of the body during the treatment of breast cancer. Its pharmacological effects are closely related to the multi-target regulatory mechanism of nuclear factor kappa B, PI3K/AKT signaling pathway, and Th1/Th2 cell balance. The above research has confirmed that the traditional Chinese medicine compound, by virtue of its multi-target and multi-pathway intervention advantages, demonstrates excellent potential as an auxiliary treatment in the clinical diagnosis and treatment of breast tumors[58]. Herbal formulas such as Xiangbei Yangrong decoction[59] and Taohong Siwu decoction[58] can regulate the overall function of the body through multiple approaches including invigorating the spleen and replenishing qi, and promoting blood circulation and removing blood stasis. These interventions improve the circulation of qi and blood, enhance the body’s immune function, and thereby boost the body’s disease resistance. This group of people suggests conducting screening and follow-up every one to two years[60].
High-risk individuals should undergo a biopsy first to determine the benign or malignant nature of the tumor: If it is a benign lesion, they should be directly transferred to the management process for medium-risk individuals; if it is a malignant tumor, an individualized treatment plan should be formulated based on the biopsy results, and the subsequent precise diagnosis and treatment can be laid a foundation by comprehensively considering core clinical indicators such as tumor molecular typing and lymph node metastasis status[53]. Clinically, tumors can be controlled through a combination of surgical, radiotherapy, chemotherapy, endocrine therapy, and targeted therapy, etc. Regular follow-ups are conducted to dynamically assess the condition[60]; Song et al[61] have conducted research confirming that traditional Chinese medicine combined with chemotherapy, radiotherapy, targeted therapy, etc. for treating breast cancer can enhance the anti-tumor effect by regulating the tumor cell cycle, inducing cell apoptosis, and modulating the immune microenvironment. At the same time, it can alleviate the adverse reactions caused by Western medicine treatment, such as nausea and vomiting, bone marrow suppression, skin damage, and cardiac toxicity, improve the quality of life of patients, and enhance treatment tolerance. For example, Sanhuang Decoction upregulates nuclear factor erythroid-2-related factor 2 through the PI3K/AKT/mammalian target of rapamycin pathway, regulates inflammation and oxidative stress, and induces apoptosis and necrosis of breast cancer cells, reducing their invasiveness[62]. The Xiaoyao san regulates the Janus kinase 2/signal transducer and activator of transcription 3 and epithelial-mesenchymal transition signaling pathways to induce apoptosis of breast cancer cells[63], inhibit cell proliferation and metastasis, and alleviate side effects such as chemotherapy-induced bone marrow suppression[64] and cancer-related fatigue[65], thereby achieving the effect of “enhancing efficacy and reducing toxicity”[66].
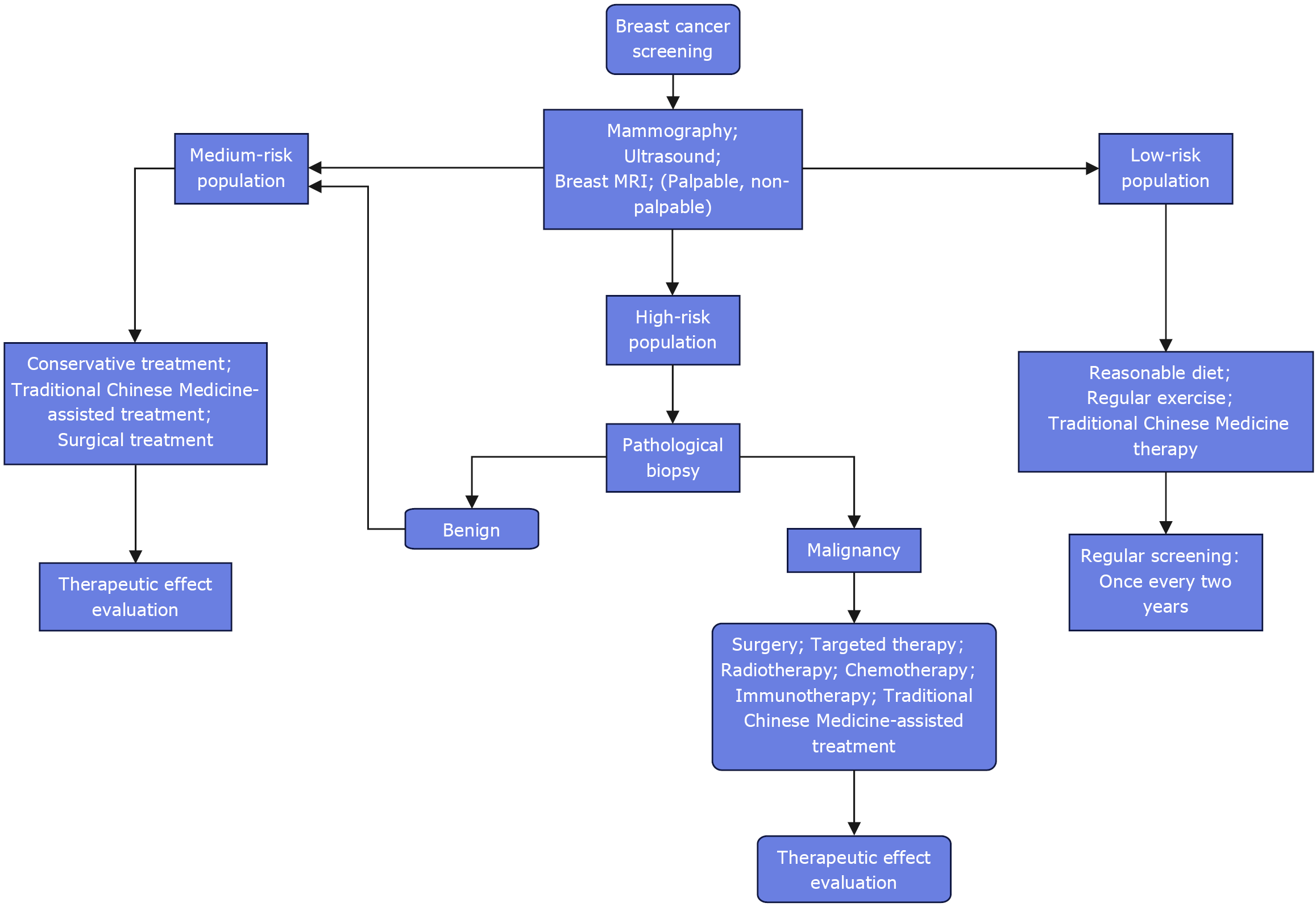
This stratified intervention model can not only reasonably allocate medical resources, balance the benefits and costs of screening, but also effectively delay tumor progression and significantly improve the quality of life of patients. It provides important support for the entire process management and treatment of breast cancer (Figure 1).
Figure 1 Flowchart of breast screening.
Conduct breast cancer screening in accordance with relevant guidelines to stratify the population into low, medium, and high-risk groups. Then, implement differentiated intervention measures and corresponding regular follow-up plans for people in different risk levels. Among them, high-risk individuals need to further determine the benign or malignant nature of breast lesions through pathological tests. Those with benign lesions are included in the medium-risk group for unified management, while those with malignant lesions will initiate subsequent targeted treatment interventions and simultaneously carry out regular follow-ups and efficacy evaluations. MRI: Magnetic resonance imaging.
FUTURE AND OUTLOOK
The core of future research is to establish a standardized bias adjustment system, carry out multicenter, large-sample prospective cohort studies, and integrate clinical staging with biomarker information including Ki-67, molecular subtypes and methylation signatures, so as to objectively evaluate the intrinsic prognostic differences between the two types of tumors, clarify the actual clinical benefits of different screening strategies, and provide a standardized basis for clinical risk stratification and intervention. Meanwhile, the recording standards of tumor detection methods should be unified, and population-based registry data should be prioritized to improve the representativeness and generalizability of research conclusions.
For non-palpable tumors, it is necessary to optimize multi-modal combined early screening techniques to achieve early detection and intervention, and reduce the probability of progression to palpable tumors. A dynamic follow-up system for high-risk populations should be established to adjust screening and intervention plans according to follow-up results, so as to realize stratified screening and dynamic management.
In view of the shortage of medical resources, low screening rates and high tumor mortality in low-income regions, low-cost and easy-to-operate screening techniques such as ultrasound combined with artificial intelligence tools adapted for primary care should be promoted. Training for primary care physicians in breast cancer screening and diagnosis should be strengthened, and a screening and referral system linking tertiary hospitals and primary medical institutions should be established to achieve seamless connection between primary screening and diagnosis and treatment in higher-level hospitals. This will improve the early screening rate of breast cancer in low-income areas, reduce tumor progression caused by insufficient screening, and lower the overall mortality rate.
Ultimately, breast cancer screening should be transformed from universal screening to precise risk-stratified screening, so as to realize the individualized identification and management of the two types of tumors and improve the overall efficiency of prevention and control. Relying on the integrated prevention and control system of traditional Chinese and Western medicine and the widely applied primary screening model, regional medical disparities will be narrowed, providing a scheme for global early screening and diagnosis of breast cancer, and achieving the core goal of reducing the incidence and mortality of breast cancer.
CONCLUSION
Palpable tumors possess significantly higher malignant potential than non-palpable tumors, and both types deserve close attention throughout the entire process of screening, prevention, treatment and prognosis. Clinical breast screening techniques have their own advantages and disadvantages, and combined screening is the core approach to improving the efficacy of early screening. Artificial intelligence has provided a new direction for the clinical screening of breast tumors. However, relevant studies have obvious methodological flaws: Most are single-center, small-sample retrospective designs with large sample bias and insufficient representativeness of conclusions. In addition, there is a lack of standardized bias adjustment systems, with inadequate correction for lead-time bias and self-selection bias. Tumor progression is only evaluated by clinical staging without integrating biomarker information, which makes it impossible to distinguish stage migration from real clinical benefits and difficult to objectively reflect the intrinsic prognostic differences between the two types of tumors. Meanwhile, the development of artificial intelligence technology is still immature, with problems such as inconsistent detection standards, high costs, poor adaptability to primary care settings and lack of medical insurance coverage, making large-scale clinical promotion difficult.
Peer review: Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Oncology
Country of origin: China
Peer-review report’s classification
Scientific quality: Grade B, Grade B
Novelty: Grade B, Grade C
Creativity or innovation: Grade B, Grade C
Scientific significance: Grade B, Grade B
P-Reviewer: Ke QH, PhD, Chief Physician, China S-Editor: Zuo Q L-Editor: A P-Editor: Wang CH