Published online Apr 24, 2026. doi: 10.5306/wjco.v17.i4.118070
Revised: January 25, 2026
Accepted: March 5, 2026
Published online: April 24, 2026
Processing time: 119 Days and 20.1 Hours
There have been no comparative studies on doxorubicin (DOX), oxaliplatin (OXA) and gemcitabine (GEM) drug-eluting bead transarterial chemoembolisation (DEB-TACE) in treating patients with unresectable non-small cell lung cancer (NSCLC).
To compare the efficacy and safety of different loading drugs in the treatment of these patients.
A total of 123 patients with unresectable NSCLC were enrolled, including the DOX-eluting DEB-TACE group (DOX group, n = 47), OXA-eluting DEB-TACE group (OXA group, n = 29) and GEM-eluting DEB-TACE (GEM group, n = 47). Treatment response, overall survival (OS), progression-free survival (PFS) and adverse events were evaluated.
The 1- and 3-month tumor response showed no significant change among the th
DOX, OXA and GEM eluting DEB-TACE may be an effective and safe treatment approach for unresectable NSCLC, with patients in the OXA group potentially exhibiting a prolonged OS. Randomized controlled trials are required to further clarify the efficacy of different drug-eluting DEB-TACE in unresectable NSCLC.
Core Tip: This is a retrospective study to investigate the safety, effectiveness and cost-effectiveness of doxorubicin (DOX), oxaliplatin (OXA) and gemcitabine (GEM) drug-eluting bead transarterial chemoembolisation (DEB-TACE) in treating patients with unresectable non-small cell lung cancer (NSCLC). Our study indicates that DOX, OXA and GEM-eluting DEB-TACE may be an effective and safe treatment approach for unresectable NSCLC, with patients in the OXA group pot
- Citation: Yuan HF, Kuang DL, Jiao DC, Bi YH. Three kinds of drug-eluting bead transarterial chemoembolisation for treating patients with unresectable non-small cell lung cancer. World J Clin Oncol 2026; 17(4): 118070
- URL: https://www.wjgnet.com/2218-4333/full/v17/i4/118070.htm
- DOI: https://dx.doi.org/10.5306/wjco.v17.i4.118070
Simultaneous radiotherapy and chemotherapy are recommended for patients with advanced lung cancer. As a promising treatment, platinum-based chemotherapy has been widely used to treat patients with non-small cell lung cancer (NSCLC), and oxaliplatin (OXA) has demonstrated excellent inhibitory efficacy against cancers[1,2]. Moreover, pira
Compared with cTACE, DEB-TACE can slowly release the eluting anti-tumour drug into the tumour mass and thus theoretically yield better efficacy and safety[8]. CalliSpheres beads have been used for eluting different types of che
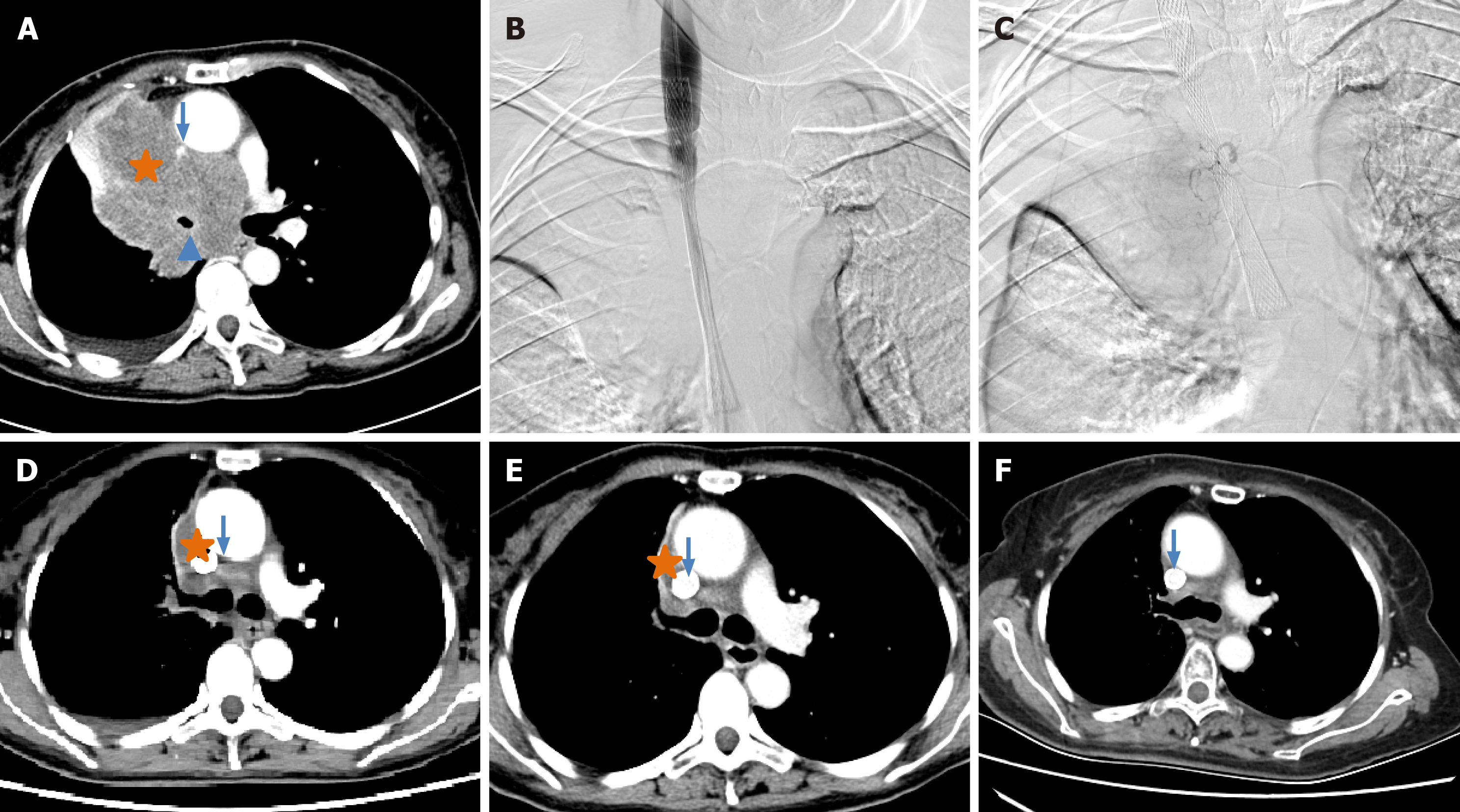
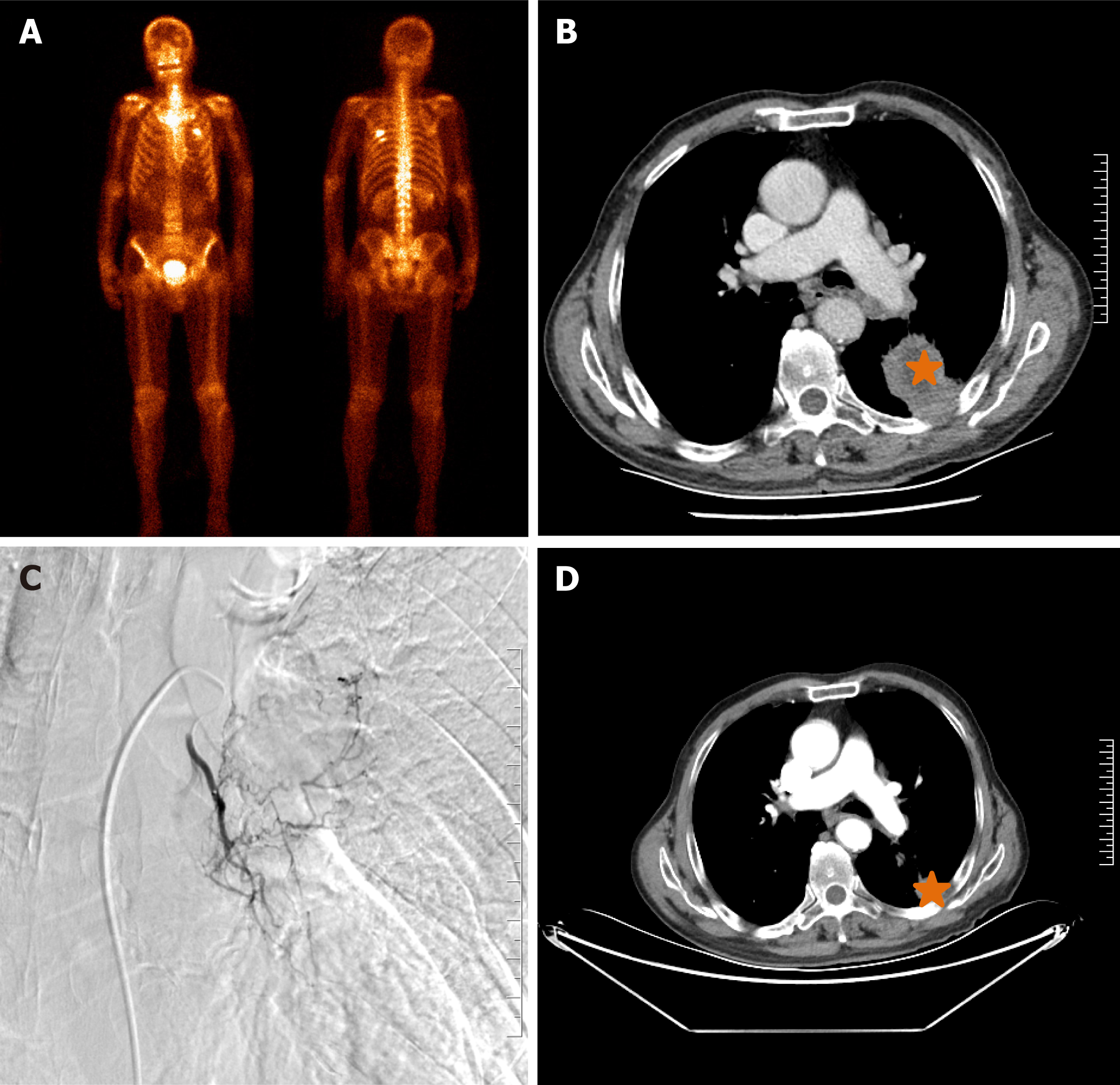
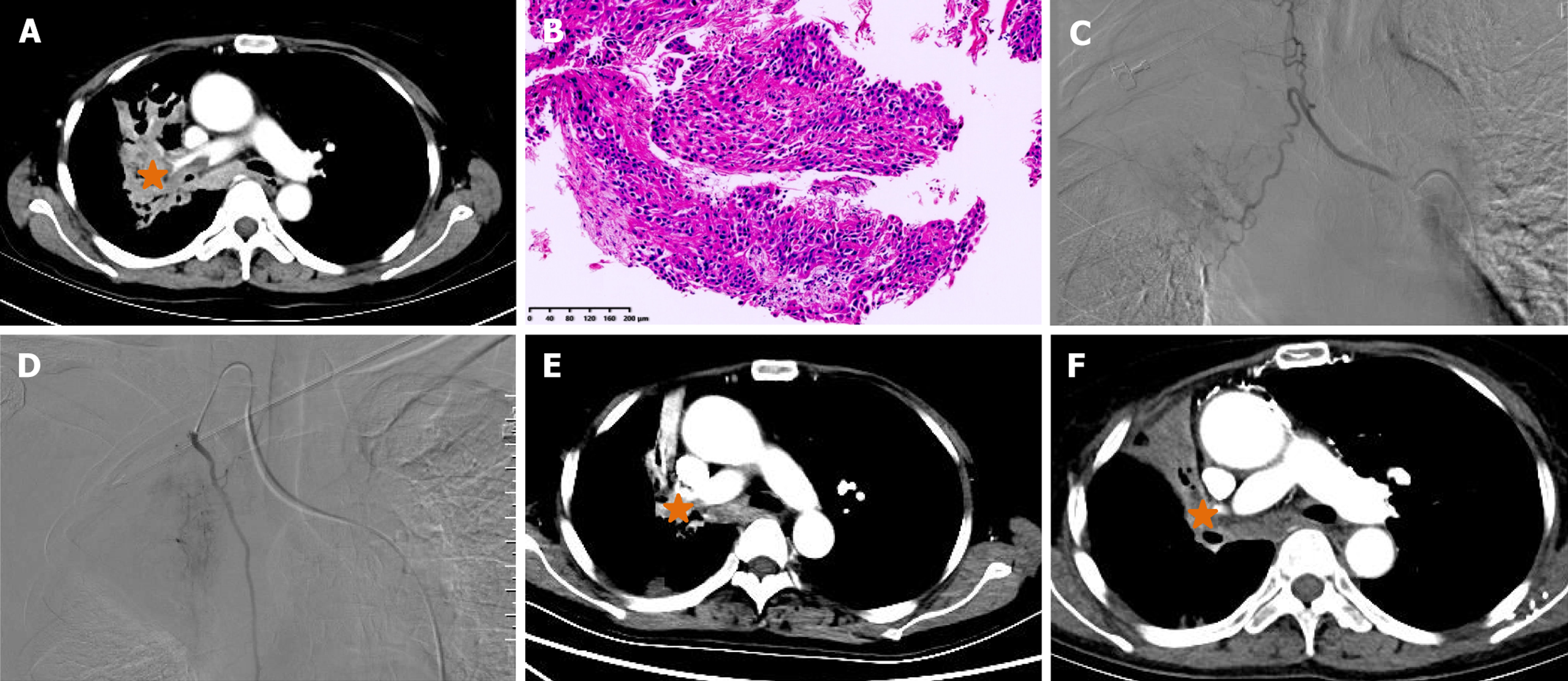
This retrospective study was approved by the Institutional Review Board of Zhengzhou university committee on human investigation, with all methods conducted in compliance with the relevant guidelines and regulations. As this study was designed as a retrospective study with clinical data analysed retrospectively and anonymously, the need for informed consent was waived by the Institutional Review Board of the First Affiliated Hospital of Zhengzhou University (No. 2023-KY-1002-002). All records were de-identified and stored in encrypted computer after data collection. A total of 123 patients with unresectable NSCLC who were treated in our department from July 2016 to July 2023 were retrospectively analysed. The inclusion criteria: (1) Biopsy proven NSCLC; (2) TNM stage II-IV according to the American Joint Committee on Cancer (AJCC) 7th Edition Cancer Staging System (Figures 1A, 2A, 2B, 3A and 3B); (3) Unresectable NSCLC or refused surgical resection due to severe comorbidities, advanced age and financial difficulties; and (4) Age 18 years to 85 years. The exclusion criteria: (1) Child-Pugh stage C or D; (2) Severe lung or heart dysfunction; (3) Severe infection; (4) Lactation or pregnancy; (5) Allergic to chemotherapeutic drugs or contrast agent; (6) Eastern Cooperative Oncology Group (ECOG) score > 2; (7) White blood cell < 3000 cells/mm3; (8) Total bilirubin > 2 mg/dL; and (9) Creatinine > 2 mg/dL.
Patients’ baseline data were recorded, including age, sex, course of disease, clinical symptom, pathology, ECOG score, tumour location, number of lung tumours, tumour size, lymph node metastasis status, TNM stage, obstructive atelectasis or pneumonia and previous treatment details.
DOX, OXA or GEM was loaded using CalliSpheres beads (Jiangsu Hengrui Medicine Co. Ltd., Jiangsu Province, China) for 30 minutes, and then mixed with iodixanol contrast developer in a 1:1 ratio before embolisation. DEB-TACE was performed under fluoroscopic guidance. After successfully puncturing the femoral artery, a 5-F sheath was inserted, and a 5-F Cobra catheter (Terumo, Japan) was inserted into the bronchial artery, intercostal artery or internal thoracic artery for angiography to identify the location of the tumour-feeding artery[14]. Next, a 2.6-F microcatheter (Asahi, Japan) was inserted into these arteries for advanced superselective catheterization. Chemotherapeutic drugs were infused into the tumour mass via the microcatheter. The total dosage of the chemotherapeutic agent was allocated according to the extent of tumour staining observed in each artery if there were three or more feeding arteries feeding the tumour[15]. Then, 1 g of drug-eluting CalliSpheres beads was used for chemoembolisation until blood flow stopped (Figures 1B, 1C, 2C, 3C and 3D).
According to the treatment plan in this study, the aim was to compare the following three different treatment groups based on DEB-TACE: DOX group (n = 47), OXA group (n = 29) and GEM group (n = 47). GEM and OXA/cisplatin were used in principle as first-line treatment regimens, whereas DOX was administered instead of cisplatin for patients with renal dysfunction. In the DOX group, OXA or cisplatin was intra-arterially infused if platinum-based chemotherapy had not been previously used, and then DOX (20-60 mg) drug-eluting beads were used for embolisation. In the OXA group, paclitaxel (100-300 mg) or docetaxel (20-40 mg) was intra-arterially infused, and then embolisation was performed using OXA (100 mg) drug-eluting beads. In the GEM group, docetaxel was intra-arterially infused for patients receiving a non-platinum-containing regimen and then GEM (0.4-1.0 g) drug-eluting beads were used for chemoembolisation.
Considering that TACE theoretically reduces the incidence of adverse events and increases local drug concentration than conventional chemotherapy, the doses of the other chemotherapeutic agents were limited to about half of the dose used in systemic chemotherapy. The total doses of GEM (Jiangsu Haosen Pharmaceutical Co. Ltd., Jiangsu Province, China) and cisplatin (Jiangsu Haosen Pharmaceutical Co. Ltd., Jiangsu Province, China) were 1 g/m2 and 65 mg/m2, respectively. The doses limit for cisplatin is < 50 mg, and for OXA, it is 100 mg. The treatment was generally applied at least twice at an interval of 4-6 weeks. DEB-TACE cycles with the same regimen were repeated when residual tumor lesions were observed on contrast-enhanced computed tomography (CT).
All patients were followed up until the study endpoint or death. At approximately 1-, 3- and 6-month follow-up after treatment initiation, the treatment response was evaluated via chest CT according to the response evaluation criteria in solid tumors 1.1 criteria[16] (Figures 1D-F, 2D, 3E and 3F). Progression-free survival (PFS) was defined as the time interval from DEB-TACE treatment to disease progression or death, whichever occurred first. Overall survival (OS) was defined as the time interval from the DEB-TACE treatment to death. Objective response rate (ORR) was defined as the sum of complete response and partial response, and disease control rate (DCR) was calculated as the sum of complete response, partial response and stable disease. Adverse events were monitored and graded according to the National Cancer Institute Common Toxicity Criteria for Adverse Events version 4.03[17].
Prism 5.0 (GraphPad Software, Inc., SanDiego, CA, United States) was used for data analysis and creating graphics. Data were expressed as mean ± SD or n (%). Comparisons among the three groups were performed using χ2 tests followed by Tukey’s post-hoc multiple comparisons or one-way analysis of variance. OS and PFS were described using Kaplan-Meier curves, and the log-rank (Mantel-Cox) test was used for comparisons. P < 0.05 was considered as statistically significant.
The mean ages were 65.9 ± 10.3 years, 62.9 ± 13.4 years, and 60.5 ± 9.6 years in the GEM, OXA and DOX groups, respectively. The number of men and women was 38 (80.9%) and 9 (19.1%) in the GEM group, 19 (65.5%) and 10 (34.5%) in the OXA group and 38 (80.9%) and 9 (19.1%) in the DOX group, respectively. Regarding pathological diagnosis, the number of squamous cell carcinoma and adenocarcinoma cases were 33 (70.2%) and 11 (23.4%) in the GEM group, 11 (37.9%) and 12 (41.4%) in the OXA group and 24 (51.1%) and 14 (29.8%) in the DOX group, respectively. The numbers of patients in TNM stages III and IV were 19 (40.4%) and 24 (51.1%) in the GEM group, 9 (31.0%) and 16 (55.2%) in the OXA group and 19 (40.4%) and 27 (57.4%) in the DOX group, respectively. Patients’ baseline data, including tumours types and stages of the different groups, were similar or comparable (all P > 0.05, Table 1).
| | DOX group | OXA group | GEM group | P value |
| Total number of patients | 47 | 29 | 47 | |
| Gender (male) | 38 (80.9) | 19 (65.5) | 38 (80.9) | 0.23 |
| Gender (female) | 9 (19.1) | 10 (34.5) | 9 (19.1) | 0.23 |
| Mean age, years | 60.5 ± 9.6 | 62.9 ± 13.4 | 65.9 ± 10.3 | 0.06 |
| Course of disease (months) | 3.5 (1.0-12.0) | 3.0 (1.0-6.8) | 3.0 (0.5-8.0) | 0.57 |
| Chemotherapy | 15 (31.9) | 10 (34.5) | 20 (42.6) | 0.54 |
| Radiotherapy or chemoradiotherapy | 6 (12.8) | 3 (10.3) | 2 (4.3) | 0.34 |
| Genetic mutations | 4 (8.5) | 6 (20.7) | 8 (17.0) | 0.29 |
| Targeted therapy | 7 (14.9) | 8 (27.6) | 6 (12.8) | 0.22 |
| Immunotherapy | 5 (10.6) | 2 (6.9) | 11 (23.4) | 0.09 |
| Targeted and Immunotherapy | 2 (4.3) | 4 (13.8) | 4 (8.5) | 0.33 |
| Tyrosine kinase inhibitors | 8 (17.0) | 10 (34.5) | 8 (17.0) | 0.24 |
| Bevacizumab | 1 (2.1) | 2 (6.9) | 2 (4.3) | 0.59 |
| Only one tumor | 34 (72.3) | 20 (69.0) | 34 (72.3) | |
| Two tumors | 2 (4.3) | 0 (0.0) | 6 (12.8) | 0.12 |
| Not less than three tumors | 11 (23.4) | 9 (31.0) | 7 (14.9) | |
| Diameter of tumor (mm) | 67.0 (48.0-88.9) | 49.9 (37.4-74.5) | 66.0 (49.0-84.0) | 0.20 |
| Lymph node metastasis | 14 (29.8) | 8 (27.6) | 18 (38.3) | 0.55 |
| Distant organ metastasis | 10 (21.3) | 10 (34.5) | 12 (25.5) | 0.47 |
| Squamous carcinoma | 24 (51.1) | 11 (37.9) | 33 (70.2) | |
| Adenocarcinoma | 14 (29.8) | 12 (41.4) | 11 (23.4) | 0.06 |
| Other pathological types | 9 (19.1) | 6 (20.7) | 3 (6.4) | |
| II stage | 1 (2.2) | 4 (13.8) | 4 (8.5) | |
| III stage | 19 (40.4) | 9 (31.0) | 19 (40.4) | 0.38 |
| IV stage | 27 (57.4) | 16 (55.2) | 24 (51.1) | - |
| Central type | 25 (53.2) | 19 (65.5) | 29 (61.7) | 0.38 |
| Peripheral type | 22 (46.8) | 10 (34.5) | 18 (38.3) | 0.38 |
| Location, right lung | 30 (63.8) | 18 (62.1) | 29 (61.7) | 0.98 |
| Location, left lung | 14 (29.8) | 11 (37.9) | 16 (34.0) | 0.76 |
| Hemoptysis | 18 (38.3) | 7 (24.1) | 13 (27.7) | 0.36 |
| Obstructive atelectasis or pneumonia | 12 (25.5) | 13 (44.8) | 17 (36.2) | 0.21 |
The incidence rates of adverse events were comparable between the DOX and OXA groups but were lower in the GEM group; however, there was no statistically significant difference (P = 0.63). The incidence rates of fever, chest pain, pain of access site and nausea or vomiting showed no differences among the three groups (all P > 0.05) (Table 2). No severe complications or treatment-related deaths occurred, with most adverse events being grade I or II, indicating that the DEB-TACE treatment was well-tolerated in all groups.
| | DOX group (n = 64) | OXA group (n = 44) | GEM group (n = 61) | P value |
| Initial technical success | 63 (98.4) | 42 (95.5) | 59 (96.7) | 0.66 |
| Procedure time (minute) | 85.0 (70.0-120.0) | 90.0 (60.8-111.3) | 93.0 (60.0-118.0) | 1.00 |
| Hospital day | 14.5 (10.8-20.0) | 11.0 (8.0-17.0) | 14.0 (10.0-19.5) | 0.12 |
| Total cost of hospitalization, × 104 (¥) | 6.6 ± 2.2 | 6.2 ± 2.1 | 6.9 ± 1.9 | 0. 35 |
| Session of DEB-TACE | 1.4 ± 0.6 | 1.6 ± 1.0 | 1.3 ± 0.6 | 0.22 |
| I125 seed implantation | 15 (31.9) | 2 (6.9) | 15 (31.9) | < 0.01 |
| Thermal ablation | 5 (10.6) | 0 (0.0) | 0 (0.0) | 0.01 |
| cTACE | 9 (19.1) | 5 (17.2) | 7 (14.9) | 0.86 |
| TAE for hemoptysis | 6 (12.8) | 1 (3.4) | 4 (8.5) | 0.38 |
| DEB-TACE for liver or renal metastasis | 1 (2.1) | 5 (17.2) | 1 (2.1) | < 0.01 |
| Esophageal or airway stenting | 6 (12.8) | 1 (3.4) | 4 (8.5) | 0.38 |
| Vascular stent implantation | 1 (2.1) | 0 (0.0) | 2 (4.3) | 0.50 |
| Complications1 | 19 (40.4) | 10 (34.5) | 15 (31.9) | 0.63 |
| Fever | 3 (6.4) | 2 (6.9) | 3 (6.4) | 1.00 |
| Nausea or vomiting | 8 (17.0) | 4 (13.8) | 5 (10.6) | 0.67 |
| Chest pain | 8 (17.0) | 5 (17.2) | 6 (12.8) | 0.81 |
| Pain of access site | 2 (4.3) | 0 (0.0) | 2 (4.3) | 0.53 |
| Others | 1 (2.1) | 1 (3.4) | 3 (6.4) | 0.57 |
Two patients were lost to follow-up. At 1-month post-DEB-TACE, the ORR and DCR were 19.1% and 76.6% in the GEM group, 20.7% and 72.4% in the OXA group and 14.9% and 89.4% in the DOX group, respectively (Table 3). No difference was observed in ORR or DCR among the three groups at 1-, 3- and 6-month follow-up (all P > 0.05). Furthermore, subsequent two-group analyses revealed no difference in ORR or DCR among the three groups (all P > 0.05). In general, all groups showed with a stable trend of ORR and a downward trend of DCR over time. Regarding the treatment response, the DOX group exhibited the best ORR and DCR, followed by the OXA and GEM groups.
| | DOX group | OXA group | GEM group | P value |
| Objective response rates | ||||
| 1 month | 7 (14.9) | 6 (20.7) | 9 (19.1) | 0.78 |
| 3 months | 8 (21.1) | 6 (30.0) | 10 (23.8) | 0.75 |
| 6 months | 6 (17.6) | 7 (33.3) | 6 (17.1) | 0.29 |
| Disease control rates | ||||
| 1 month | 42 (89.4) | 21 (72.4) | 36 (76.6) | 0.13 |
| 3 months | 26 (68.4) | 14 (70.0) | 29 (69.0) | 0.99 |
| 6 months | 14 (41.2) | 12 (57.1) | 15 (42.9) | 0.47 |
| Loss of follow up | 0 (0.0) | 1 (3.4) | 1 (2.1) | 1.00 |
| Deaths | 28 (59.6) | 10 (34.5) | 26 (55.3) | 0.09 |
| Median OS (months) | 10.2 | 29.6a | 10.7 | 0.05 |
| Median PFS (months) | 6.8 | 9.9 | 7.2 | 0.31 |
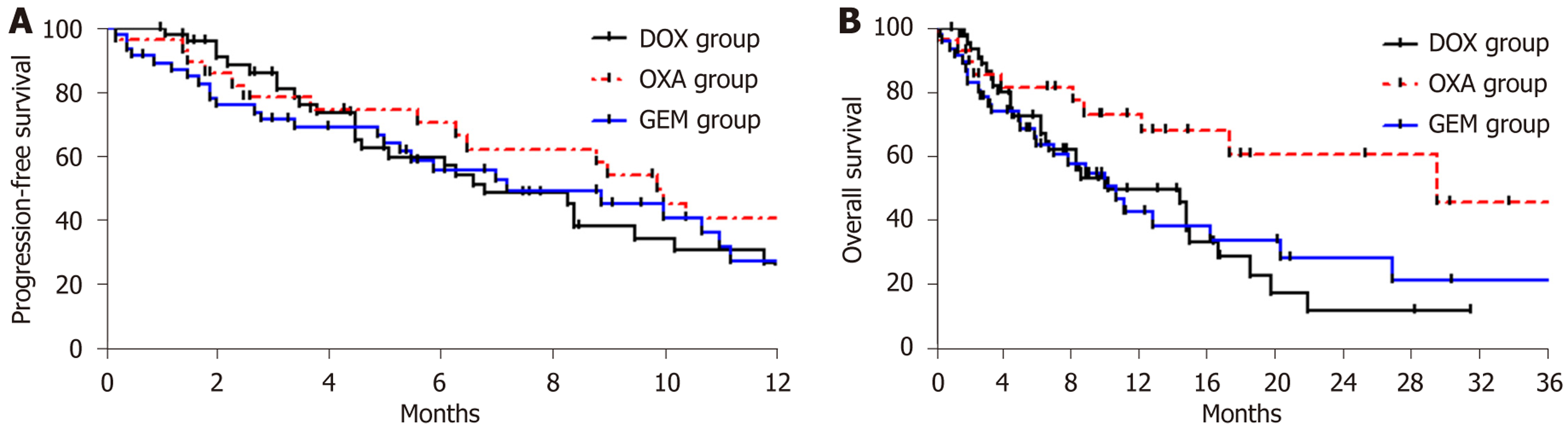
The highest PFS was observed in the OXA group [median: 9.9 months, interquartile range (IQR): 2.5-13.4 months], followed by the GEM (median: 7.2 months, IQR: 2.0-9.2 months) and DOX (median: 6.8 months, IQR: 2.7-8.4 months) groups (P = 0.31, χ2 = 2.363). Subsequent two-group comparisons also showed that the OXA group had prolonged PFS compared with the DOX (P = 0.10, χ2 = 2.720) and GEM (P = 0.30, χ2 = 1.095) groups, although there was no statistically significant difference (Figure 4).
Three-group comparisons revealed the longest OS in the OXA group (median: 29.6 months, IQR: 4.6-18.5 months), followed by the DOX (median: 10.2 months, IQR: 3.8-14.5 months) and GEM (median: 10.7 months, IQR: 3.1-11.2 months) groups (P = 0.05, χ2 = 5.818). Subsequent two-group comparisons showed that the DOX group had a similar OS compared with the GEM group (P = 0.94, χ2 = 0.005); furthermore, the OXA group showed increased OS compared with the GEM (P = 0.04, χ2 = 4.357) and DOX (P = 0.02, χ2 = 5.875) groups.
Platinum-based chemotherapy[18] or non-platinum-containing regimens (GEM + docetaxel)[19] have been recommended for treating patients with advanced NSCLC. Cisplatin-based chemotherapy, such as cisplatin + paclitaxel, cisplatin + GEM or cisplatin + docetaxel, may improve survival[20]; however, its clinical application is limited because of cumulative neurotoxicity, nephrotoxicity and embryogenesis[21]. As a less toxic platinum analogue, OXA exhibits less gast
Conventional chemotherapy may cause unanticipated adverse events because of the high doses of the chemotherapeutic drug[23]. As a novel palliative treatment, DEB-TACE can slowly release the eluting chemotherapeutic drug into the tumour mass and thus theoretically exhibit better efficacy and safety[8]. CalliSpheres beads are the first Chinese drug-eluting bead product and have been widely used to treat several different types of malignant tumours, such as hepatocelluar carcinoma[10] and cervical cancer[24].
Although DEB-TACE is not a firmly established technique for lung cancer itself, DEB-TACE with CalliSpheres beads has also been used for loading and releasing different types of chemotherapeutic drugs for advanced NSCLC patients[25]. For instance, Shang et al[9] reported that DEB-TACE with adriamycin-eluting beads is effective in patients with stage II-IV lung cancer. Bie et al[12] also reported that DEB-TACE with GEM-eluting CalliSpheres beads is feasible in six NSCLC patients. Furthermore, DEB-TACE with pirarubicin-eluting beads has been used for treating patients with advanced NSCLC[9,14,26]; however, pirarubicin is not a standard regimen for these patients[18]. Considering that there have been no comparative studies on different loading drugs in the treatment of patients with NSCLC, the present study aimed to compare the efficacy and safety of DOX, OXA or GEM-eluting DEB-TACE in patients with unresectable NSCLC.
Regarding safety, no severe adverse events or procedure-related deaths occurred. The incidence rates of adverse events were similar between the DOX and OXA groups but were lower in the GEM group, although no significant difference was observed. Moreover, all adverse events in the three groups were well tolerated, demonstrating the safety of DEB-TACE in advanced NSCLC patients. Such safety was also supported by other studies, where there were no severe com
Regarding efficacy, a median OS of 11.3 months with chemotherapy alone has been reported in patients with metastatic NSCLC[28]. Another study on DEB-TACE with GEM-eluting beads reported median OS of 8.0 months and 16.5 months[12], which is similar to that in GEM group of our study. In the OXA group of our study, the median OS was 29.6 months, which was longer than that in the GEM and DOX groups and better than that achieved by DEB-TACE with GEM-eluting beads in the previous study[12]. Previous studies have reported a median PFS of 8.0-8.8 months with DEB-TACE for lung cancer[9,12], which is similar to that in the present study. Shang et al[9] conducted a study on 30 patients with stage II-IV lung cancer who received CalliSpheres beads loaded with adriamycin for chemoembolisation and reported 6-month DCR and ORR of 93.3% and 86.7%, respectively. Although their results appear to be better than ours, to our confusion, their median PFS was not longer than our result, and they did not report the OS. In the present study, ORR and DCR were higher in the OXA group than in the GEM and DOX groups at the 6-month follow-up, although there was no difference among the three groups. Our results suggest that OXA eluting DEB-TACE is better in terms of disease response and OS in the treatment of patients with unresectable NSCLC.
This study has some limitations. First, it was a retrospective study conducted at in a single centre. The sample size of the OXA group was relatively smaller than that of the DOX and GEM groups, which may have introduced bias during comparisons. Additionally, approximately half doses of chemotherapeutic agents were transarterially injected in our study to decrease the incidence of adverse event, a practice lacking evidence. However, this approach may not impair treatment efficacy considering the high local drug concentrations in the tumour, which can lead to tumour necrosis after transarterial embolisation.
DOX, OXA and GEM-eluting DEB-TACE may be an effective and safe treatment approach for unresectable NSCLC, with patients in the OXA group potentially exhibiting a prolonged OS. Randomized controlled trials are required to further clarify the efficacy of different drug-eluting DEB-TACE in unresectable NSCLC.
| 1. | Ibrahim A, Hirschfeld S, Cohen MH, Griebel DJ, Williams GA, Pazdur R. FDA drug approval summaries: oxaliplatin. Oncologist. 2004;9:8-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 125] [Article Influence: 5.7] [Reference Citation Analysis (3)] |
| 2. | Nobili S, Checcacci D, Filippelli F, Del Buono S, Mazzocchi V, Mazzei T, Mini E. Bimonthly chemotherapy with oxaliplatin, irinotecan, infusional 5-fluorouracil/folinic acid in patients with metastatic colorectal cancer pretreated with irinotecan- or oxaliplatin-based chemotherapy. J Chemother. 2008;20:622-631. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 3. | Drings P, Günther IU, Gatzemeier U, Berdel W, Stahl M, Salewski E, Edler L. Pirarubicin in advanced non-small cell lung cancer. A trial of the Phase I/II Study Group of the Association for Medical Oncology of the German Cancer Society. Onkologie. 1990;13:180-184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 4. | Pandit B, Royzen M. Recent Development of Prodrugs of Gemcitabine. Genes (Basel). 2022;13:466. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 81] [Cited by in RCA: 57] [Article Influence: 14.3] [Reference Citation Analysis (1)] |
| 5. | Bullinger KL, Nardelli P, Wang Q, Rich MM, Cope TC. Oxaliplatin neurotoxicity of sensory transduction in rat proprioceptors. J Neurophysiol. 2011;106:704-709. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 6. | Gridelli C, Rossi A, Carbone DP, Guarize J, Karachaliou N, Mok T, Petrella F, Spaggiari L, Rosell R. Non-small-cell lung cancer. Nat Rev Dis Primers. 2015;1:15009. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 404] [Cited by in RCA: 791] [Article Influence: 71.9] [Reference Citation Analysis (1)] |
| 7. | Bi Y, Shi X, Ren J, Yi M, Han X, Song M. Transarterial chemoembolization with doxorubicin-loaded beads for inoperable or recurrent colorectal cancer. Abdom Radiol (NY). 2021;46:2833-2838. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 8. | Bi Y, Shi X, Ren J, Yi M, Han X, Song M. Clinical outcomes of doxorubicin-eluting CalliSpheres® beads-transarterial chemoembolization for unresectable or recurrent esophageal carcinoma. BMC Gastroenterol. 2021;21:231. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 9. | Shang B, Li J, Wang X, Li D, Liang B, Wang Y, Han X, Dou W, Chen G, Shang J, Jiang S. Clinical effect of bronchial arterial infusion chemotherapy and CalliSpheres drug-eluting beads in patients with stage II-IV lung cancer: A prospective cohort study. Thorac Cancer. 2020;11:2155-2162. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 33] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 10. | Bi Y, Ren K, Ren J, Ma J, Han X. Oxaliplatin Eluting CalliSpheres Microspheres for the Treatment of Unresectable or Recurrent Hepatocellular Carcinoma. Front Pharmacol. 2022;13:923585. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 11. | Han X, Chen Q, Sun Y, Han L, Sha X. Morphology, Loadability, and Releasing Profiles of CalliSpheres Microspheres in Delivering Oxaliplatin: An In Vitro Study. Technol Cancer Res Treat. 2019;18:1533033819877989. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 21] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 12. | Bie Z, Li Y, Li B, Wang D, Li L, Li X. The efficacy of drug-eluting beads bronchial arterial chemoembolization loaded with gemcitabine for treatment of non-small cell lung cancer. Thorac Cancer. 2019;10:1770-1778. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 56] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 13. | Bi Y, Zhang B, Ren J, Han X, Wu W. Clinical outcomes of gemcitabine-loaded callispheres drug-eluting beads for patients with advanced and inoperable lung cancer: A case series study. Front Pharmacol. 2022;13:992526. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 14. | Bi Y, Shi X, Yi M, Han X, Ren J. Pirarubicin-loaded CalliSpheres® drug-eluting beads for the treatment of patients with stage III-IV lung cancer. Acta Radiol. 2022;63:311-318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 27] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 15. | Nakanishi M, Demura Y, Umeda Y, Mizuno S, Ameshima S, Chiba Y, Ishizaki T. Multi-arterial infusion chemotherapy for non-small cell lung carcinoma--significance of detecting feeding arteries and tumor staining. Lung Cancer. 2008;61:227-234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 38] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 16. | Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S, Mooney M, Rubinstein L, Shankar L, Dodd L, Kaplan R, Lacombe D, Verweij J. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45:228-247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24191] [Cited by in RCA: 22959] [Article Influence: 1350.5] [Reference Citation Analysis (5)] |
| 17. | Nation Cancer Institute. Cancer Therapy Evaluation Program (CTEP). [cited 28 February 2026]. Available from: http://ctep.cancer.gov/protocol Development/electronic_applications/ctc.htm. |
| 18. | Schiller JH, Harrington D, Belani CP, Langer C, Sandler A, Krook J, Zhu J, Johnson DH; Eastern Cooperative Oncology Group. Comparison of four chemotherapy regimens for advanced non-small-cell lung cancer. N Engl J Med. 2002;346:92-98. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4239] [Cited by in RCA: 3979] [Article Influence: 165.8] [Reference Citation Analysis (5)] |
| 19. | Pujol JL, Breton JL, Gervais R, Rebattu P, Depierre A, Morère JF, Milleron B, Debieuvre D, Castéra D, Souquet PJ, Moro-Sibilot D, Lemarié E, Kessler R, Janicot H, Braun D, Spaeth D, Quantin X, Clary C. Gemcitabine-docetaxel versus cisplatin-vinorelbine in advanced or metastatic non-small-cell lung cancer: a phase III study addressing the case for cisplatin. Ann Oncol. 2005;16:602-610. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 95] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 20. | Franciosi V, Barbieri R, Aitini E, Vasini G, Cacciani GC, Capra R, Camisa R, Cascinu S. Gemcitabine and oxaliplatin: a safe and active regimen in poor prognosis advanced non-small cell lung cancer patients. Lung Cancer. 2003;41:101-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 28] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 21. | Wang D, Lippard SJ. Cellular processing of platinum anticancer drugs. Nat Rev Drug Discov. 2005;4:307-320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2728] [Cited by in RCA: 2783] [Article Influence: 132.5] [Reference Citation Analysis (3)] |
| 22. | Monnet I, Brienza S, Hugret F, Voisin S, Gastiaburu J, Saltiel JC, Soulié P, Armand JP, Cvitkovic E, de Cremoux H. Phase II study of oxaliplatin in poor-prognosis non-small cell lung cancer (NSCLC). ATTIT. Association pour le Traitement des Tumeurs Intra Thoraciques. Eur J Cancer. 1998;34:1124-1127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 53] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 23. | Jain A, Jain SK, Ganesh N, Barve J, Beg AM. Design and development of ligand-appended polysaccharidic nanoparticles for the delivery of oxaliplatin in colorectal cancer. Nanomedicine. 2010;6:179-190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 142] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 24. | Bi Y, Wang Y, Zhang J, Shi X, Wang Y, Xu M, Han X, Ren J. Clinical outcomes of uterine arterial chemoembolization with drug-eluting beads for advanced-stage or recurrent cervical cancer. Abdom Radiol (NY). 2021;46:5715-5722. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 25. | Bi Y, Li F, Ren J, Han X. The safety and efficacy of oxaliplatin-loaded drug-eluting beads transarterial chemoembolization for the treatment of unresectable or advanced lung cancer. Front Pharmacol. 2022;13:1079707. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 26. | Lin H, Wang Q, Tian F, Zhang R, Mu M, Zhao W, Bao P. Drug-Eluting Beads Bronchial Arterial Chemoembolization in Treating Relapsed/Refractory Small Cell Lung Cancer Patients: Results from a Pilot Study. Cancer Manag Res. 2021;13:6239-6248. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 27. | Kennoki N, Hori S, Yuki T, Sueyoshi S, Hori A. [Transcatheter Arterial Chemoembolization with Super absorbent Polymer Microspheres for a Large Lung Cystic Adenocarcinoma in the Left Pulmonary Cavity]. Gan To Kagaku Ryoho. 2015;42:1407-1410. [PubMed] |
| 28. | Paz-Ares L, Luft A, Vicente D, Tafreshi A, Gümüş M, Mazières J, Hermes B, Çay Şenler F, Csőszi T, Fülöp A, Rodríguez-Cid J, Wilson J, Sugawara S, Kato T, Lee KH, Cheng Y, Novello S, Halmos B, Li X, Lubiniecki GM, Piperdi B, Kowalski DM; KEYNOTE-407 Investigators. Pembrolizumab plus Chemotherapy for Squamous Non-Small-Cell Lung Cancer. N Engl J Med. 2018;379:2040-2051. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1941] [Cited by in RCA: 2854] [Article Influence: 356.8] [Reference Citation Analysis (3)] |