Copyright: ©Author(s) 2026.
World J Clin Oncol. Apr 24, 2026; 17(4): 118606
Published online Apr 24, 2026. doi: 10.5306/wjco.v17.i4.118606
Published online Apr 24, 2026. doi: 10.5306/wjco.v17.i4.118606
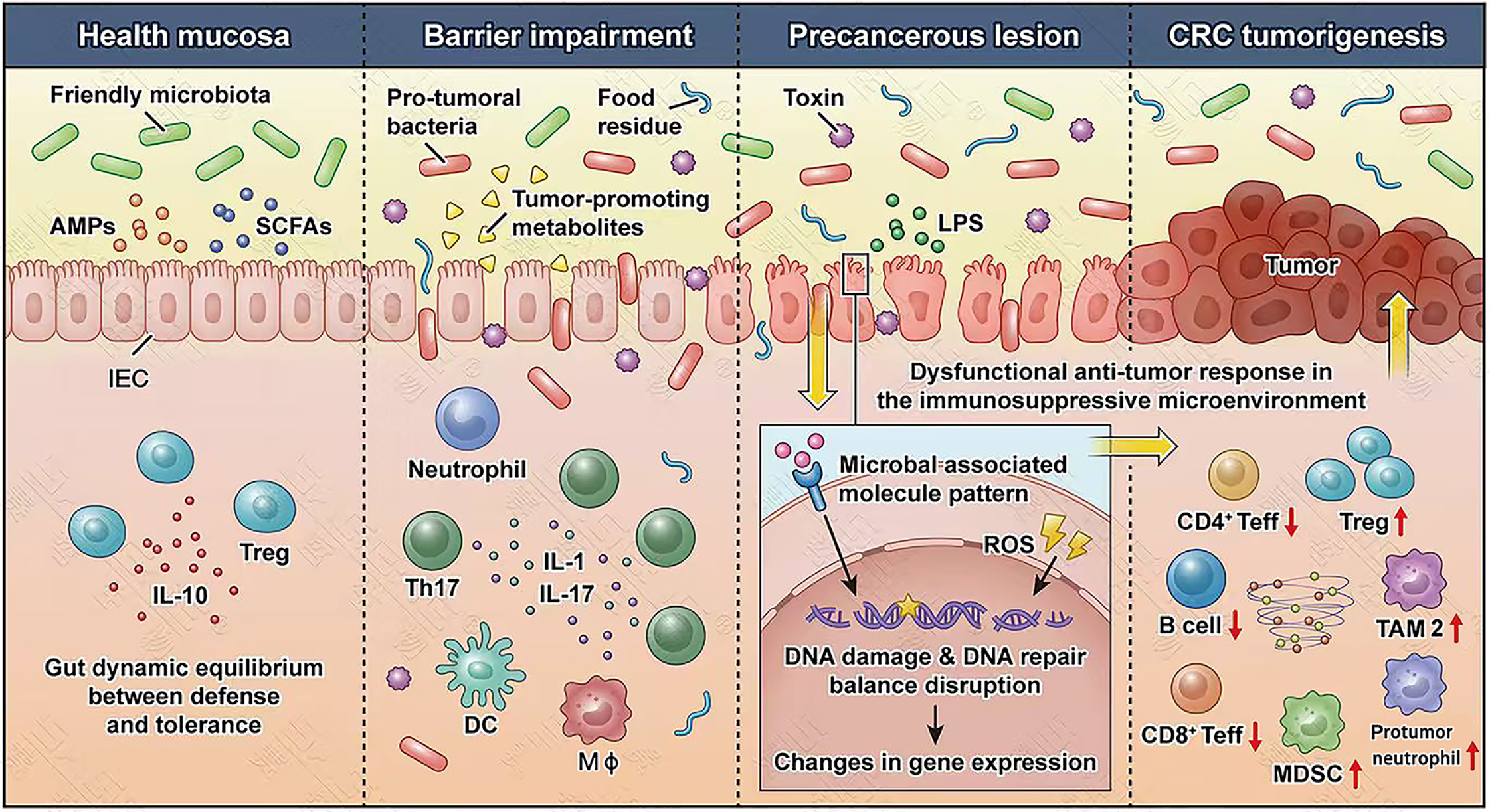
Figure 1 Schematic diagram of gut microbiome dysbiosis attribute to colorectal cancer initiation and development.
In healthy individuals, the symbiotic gut microbiome educates the host immune system to induce tolerance and re-establish immune homeostasis for survival and replication in host cells. In colorectal cancer development and progression, various oncogenic stimuli derived from gut microbiome dysbiosis increase the genotoxicity of intestinal epithelial cells and damage barrier permeability, which subsequently triggers a multistep protumoral inflammatory cascade, marked by regulatory cell recruitment (e.g., myeloid-derived suppressor cell, tumor-associated macrophage type 2) and effector cell loss (e.g., CD8+ effector T-cell, B cell), allowing tumor immune evasion. SCFAs: Short-chain fatty acids; AMPs: Antimicrobial peptides; IEC: Intestinal epithelial cell; Treg: Regulatory T cell; IL: Interleukin; Th17: T helper 17 cell; Mφ: Macrophage; DC: Dendritic cell; LPS: Lipopolysaccharide; ROS: Reactive oxygen species; CRC: Colorectal cancer; Teff: Effector T-cell; TAM2: Tumor-associated macrophage type 2; MDSC: Myeloid-derived suppressor cell.
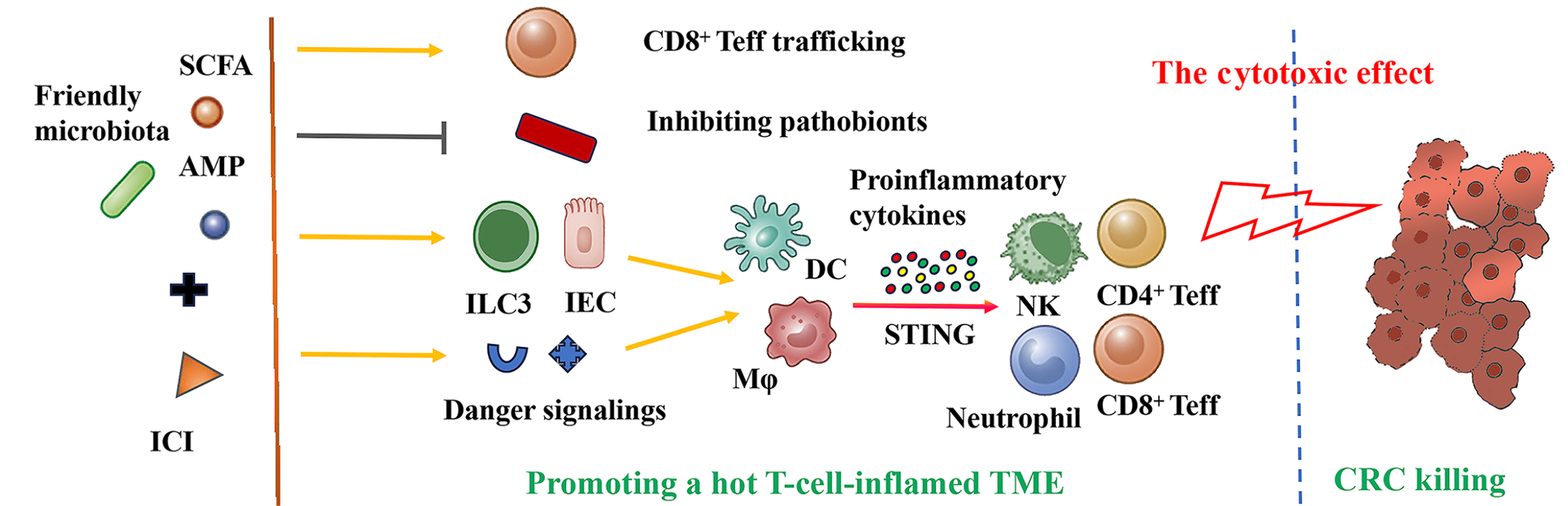
Figure 2 Key mechanisms by which the gut microbiome reinforces immune checkpoint inhibitor’ antitumor efficacy.
When responding to immune cells, friendly microbiota and metabolites facilitate host innate and adaptive immune responses, stimulating “cold-warm-hot” tumor microenvironment transition. They inhibit pathobionts and promote CD8+ effector T-cell infiltration. As “danger” signals, they are cross-presented by antigen-presenting cells (e.g., macrophage, dendritic cell) to activate natural killer cell, neutrophil, and CD8+ and CD4+ effector T-cell via the stimulator of interferon genes signaling and facilitate immune-mediated tumor clearance. AMP: Antimicrobial peptide; SCFA: Short-chain fatty acid; ICI: Immune checkpoint inhibitor; Teff: Effector T-cell; ILC3: Innate lymphoid cell 3; IEC: Intestinal epithelial cell; DC: Dendritic cell; Mφ: Macrophage; STING: Stimulator of interferon genes; NK: Natural killer cell; CRC: Colorectal cancer; TME: Tumor microenvironment.
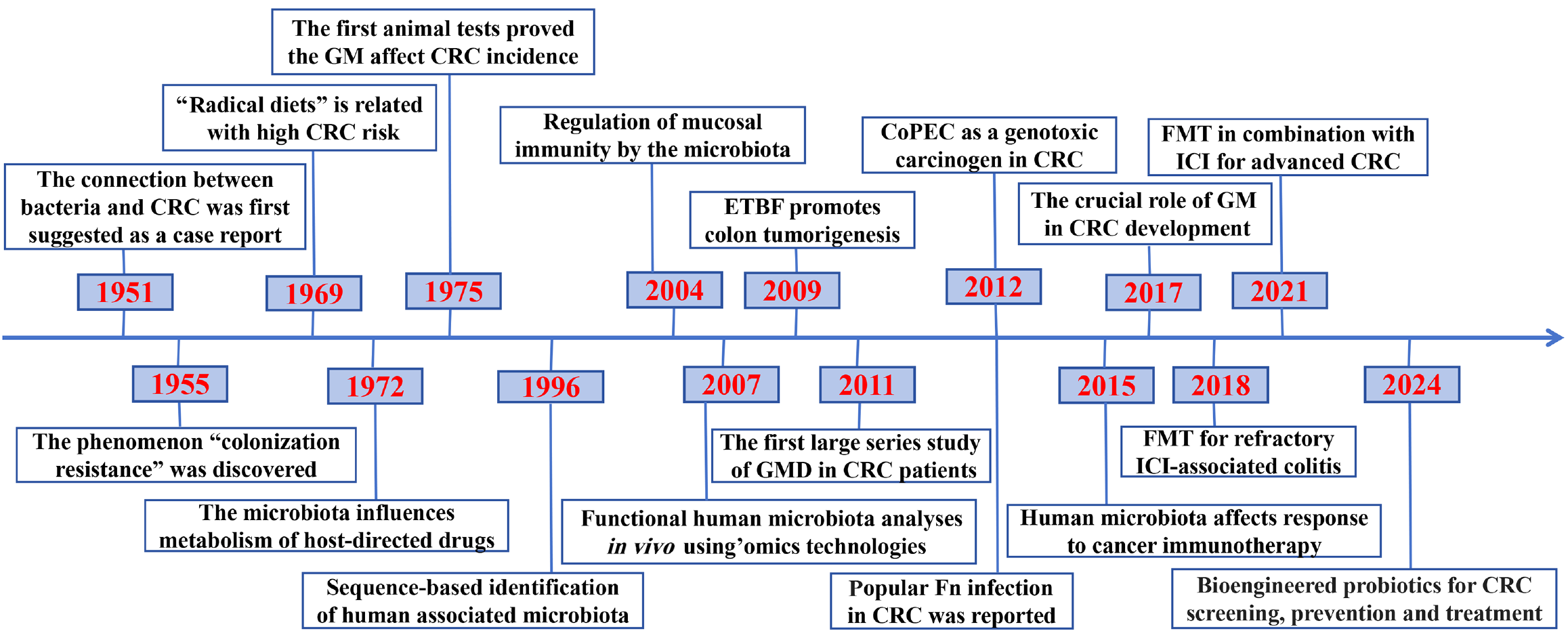
Figure 3 History of core research achievements in the microbiota-colorectal cancer crosstalk.
These key achievements, from a mere case report to detailed molecular and immunological mechanism explorations, mark significant advancements in the field of host-gut microbiome mutualism under physiological conditions and cancer development, tracing back from 1951 to the current era. With the deepening understanding that suggests remodeling the microbial community represents a potential method to reinforce the antitumor efficacy of immune checkpoint inhibitors, microbe-based intervention should translate into an indispensable component of clinical therapy in the future. CRC: Colorectal cancer; GM: Gut microbiome; ETBF: Enterotoxigenic Bacteroides fragilis; GMD: Gut microbiome dysbiosis; CoPEC: Colibactin-producing Escherichia coli; FMT: Fecal microbiota transplantation; ICI: Immune checkpoint inhibitor; Fn: Fusobacterium nucleatum.
- Citation: Ti DD, Liu P, Wu CY, Shi ZM, Guo SM, Gao ZC. Host and gut microbiota crosstalk: A new paradigm for colorectal cancer immunotherapy. World J Clin Oncol 2026; 17(4): 118606
- URL: https://www.wjgnet.com/2218-4333/full/v17/i4/118606.htm
- DOI: https://dx.doi.org/10.5306/wjco.v17.i4.118606