Published online Mar 22, 2026. doi: 10.4291/wjgp.v17.i1.113971
Revised: October 6, 2025
Accepted: December 18, 2025
Published online: March 22, 2026
Processing time: 192 Days and 16.6 Hours
Functional abdominal pain syndrome (FAPS) is a subset of functional gastroin
To determine the frequency of fructose malabsorption (FM) in patients with FAPS and to evaluate the occurrence of resultant symptoms.
This was a prospective, case-control study conducted over 22 months, from Dece
FM was detected in 26% of patients with FAPS vs 3% of controls (P = 0.009). Sym
This study demonstrated a high prevalence of FM and intolerance in patients with FAPS, with younger age and male sex as predictors.
Core Tip: This case-control study was performed to evaluate the frequency of fructose malabsorption (FM) in adult patients with functional abdominal pain syndrome (FAPS) after systematically ruling out small intestinal bacterial overgrowth. We found that FM was significantly more common in patients with FAPS compared to healthy controls. Notably, some patients with FAPS reported fructose-related symptoms even without biochemical evidence of FM, which supports the role of visceral hypersensitivity. These findings highlight the potential advantages of dietary strategies, such as fermentable oligo-, di-, and monosaccharides and polyols restriction, in managing FAPS.
- Citation: Agarwal M, Vaithiyam V, Reddy RT, Aneesh PSR, Paliwal P, Jain SR, Dalal A, Kumar A, Puri AS, Sachdeva S. Frequency and predictors of fructose malabsorption in patients with functional abdominal pain syndrome: A case-control study. World J Gastrointest Pathophysiol 2026; 17(1): 113971
- URL: https://www.wjgnet.com/2150-5330/full/v17/i1/113971.htm
- DOI: https://dx.doi.org/10.4291/wjgp.v17.i1.113971
Functional abdominal pain syndrome (FAPS) is a subset of functional gastrointestinal disorders (FGIDs) characterized by persistent or nearly continuous abdominal pain without any identifiable structural, biochemical, or inflammatory cause[1]. FAPS is described as centrally mediated pain, typically not related to physiological events such as eating or defeca
The pathophysiology of FAPS is complicated and involves many factors. While central mechanisms, such as dysregulation of the gut-brain axis, altered pain perception, and psychological comorbidities, play a pivotal role, increasing attention is being directed toward the role of peripheral factors, including dietary components and gastrointestinal motility abnormalities[4]. Recent research shows that in some patients, certain poorly absorbed fermentable carbohy
Fructose, a six-carbon monosaccharide and a key component of fermentable oligo-, di-, monosaccharides, and polyols, is commonly present in fruits, sweeteners (such as high-fructose corn syrup), honey, and other processed foods. It is absorbed in the small intestine through a carrier-mediated, facilitated diffusion process via the GLUT5 transporter, which can become saturated at relatively low concentrations, especially when fructose is not co-ingested with glucose[9]. Fructose malabsorption (FM) occurs when absorption capacity is exceeded, allowing unabsorbed fructose to reach the colon, where bacteria ferment it. This process produces hydrogen, methane, carbon dioxide, and short-chain fatty acids. The increased osmotic content draws water into the intestinal lumen by osmosis, leading to symptoms such as bloating, flatulence, diarrhea, and abdominal discomfort[10,11].
FM is typically diagnosed through fructose hydrogen breath testing (FHBT), where an increase in exhaled hydrogen after consuming fructose indicates malabsorption[12]. The reported prevalence of FM varies widely depending on the administered fructose dose, diagnostic criteria, patient population, and coexisting conditions. In healthy individuals, FM rates range from 2% to 40%, with higher doses (≥ 50 g fructose) often resulting in false positives[13-16]. The prevalence of FM in studies comprising children with chronic or recurrent abdominal pain, including patients with irritable bowel syndrome (IBS), functional dyspepsia, or FAPS, varies in previous studies[17-20]. However, most studies focus on Western populations or pediatric groups, and the role of FM in adult Indian patients with FAPS remains uncertain.
Moreover, many previous studies are limited by their methodology, such as the absence of standardized fructose dosing, failure to exclude small intestinal bacterial overgrowth (SIBO), inconsistent symptom reporting, and lack of healthy control groups for comparison. SIBO produces falsely abnormal sugar breath tests, and eradication of SIBO has been shown to result in the normalization of sugar breath tests in most patients[21-23]. There is also confusion between FM (biochemically detected) and intolerance (a symptomatic response to fructose intake, with or without objective evidence of malabsorption). This distinction is vital, as it affects treatment approaches, such as dietary modifications.
Therefore, this study was performed to address these significant gaps. We aimed to determine the frequency of FM in adult patients with FAPS using standardized hydrogen methane breath testing after administering glucose and fructose. We included a well-matched healthy control group and monitored their symptoms to assess fructose intolerance. Additionally, we aimed to identify the factors that predict FM among patients with FAPS. Using this case-control design, we sought to gain a deeper understanding of the role of FM in the development of FAPS.
This prospective, case-control study was conducted over 22 months (from December 2016 to September 2018) at the Department of Gastroenterology, Govind Ballabh Pant Institute of Postgraduate Medical Education and Research, New Delhi, India. The Institutional Ethics Committee of Maulana Azad Medical College and associated hospitals granted ethical approval (No. F.No./11/IEC/MAMC/2016/09) and approved this study, which was conducted in accordance with the ethical guidelines of the 1975 Declaration of Helsinki. All participants provided written informed consent before enrollment, and parents provided consent for participants under 18 years old.
Patients aged 12-60 years attending the outpatient department with abdominal pain who fulfilled the Rome III criteria for FAPS were screened for inclusion[24]. The diagnosis of FAPS was established based on the presence of continuous or nearly continuous abdominal pain lasting at least 6 months, with no relation to physiological events such as defecation or eating, causing significant interference with daily activities, and without meeting criteria for other FGIDs. Age-matched and sex-matched healthy individuals without any gastrointestinal or other complaints were recruited as controls. Controls were screened through a comprehensive history and physical examination to exclude FGIDs or organic diseases.
Participants were excluded from the study if they had suspected or confirmed hereditary fructose intolerance, diarrhea or constipation, diabetes mellitus, thyroid dysfunction, or connective tissue disorders. Additional exclusion criteria included a history of major gastrointestinal surgery, alcohol or substance abuse, and significant systemic illness involving cardiac, hepatic, renal, or respiratory failure. Pregnant or lactating individuals and those with organic gastrointestinal diseases, such as inflammatory bowel disease, gastrointestinal neoplasms, or human immunodeficiency virus infection, were also excluded. Furthermore, subjects who had taken proton pump inhibitors, antibiotics, promotility agents, or antimotility agents within the previous 4 weeks were excluded. Participants showing red flag signs, such as unintended weight loss, gastrointestinal bleeding, fever, or a family history of inflammatory bowel disease or colorectal cancer, were excluded. Finally, individuals who did not provide informed consent were excluded.
A comprehensive baseline assessment, including a detailed clinical history and physical examination, was conducted for both cases and controls. Complete hemogram, liver and kidney function tests, and serum tissue transglutaminase immunoglobulin A were performed only in cases. Routine stool examination and microscopy were also conducted.
Pre-test preparation: Participants were advised to avoid slow-absorbing carbohydrates and fibers 48 hours prior to the test. Tobacco chewing, any form of exercise, and smoking were to be avoided for 2 hours before and throughout the procedure. The participants were instructed to brush their teeth and use a chlorhexidine mouthwash before the test.
Evaluation of SIBO: Both cases and controls initially underwent assessment for SIBO using the glucose hydrogen breath test (GHBT). An SC Microlyser (QuinTron Instrument Company, Inc., Milwaukee, WI, United States) was used for breath test analysis. GHBT was conducted with 100 g glucose dissolved in 200 mL water following overnight fasting. Subse
Evaluation of FM: The FHBT was conducted using 25 g of fructose dissolved in 250 mL of water in participants who tested negative for SIBO[13,26]. Samples were collected every 15 minutes over 3 hours following ingestion. FM was diagnosed if there was a rise of > 20 ppm in hydrogen or methane above the baseline in two or more consecutive samples[13,26]. Subjects with a fasting breath methane concentration of ≥ 3 ppm were categorized as methane producers (MPs), and this subgroup was analyzed separately[27].
Symptom assessment during FHBT: All participants were monitored for the development of gastrointestinal symptoms during the FHBT procedure. Symptoms assessed included nausea, vomiting, bloating, flatulence, abdominal pain, abdominal distension, and diarrhea. These were graded using a 4-point Likert scale (0: None, 1: Mild, 2: Moderate, and 3: Severe).
The sample size was calculated using the OpenEpi (Open-Source Epidemiologic Statistics for Public Health) software. Based on previous literature estimating the prevalence of FM in FGID populations (54.5%) vs healthy controls (16%), with an alpha of 0.05 and a power of 80%, the required sample size was 75 subjects (50 cases and 25 controls). Ultimately, 68 patients and 31 controls were recruited and analyzed. Data were analyzed using Statistical Package for the Social Sciences version 25.0 (SPSS Inc., Chicago, IL, United States). Continuous variables were presented as medians and interquartile ranges and compared using the Mann-Whitney U test. Categorical variables were expressed as n (%) and compared using the Fisher’s exact test. Univariate logistic regression was used to identify the clinical and demographic factors associated with FM. Variables with P < 0.05 in univariate analysis were included in a multivariate logistic regression model to determine independent predictors. P < 0.05 was considered statistically significant.
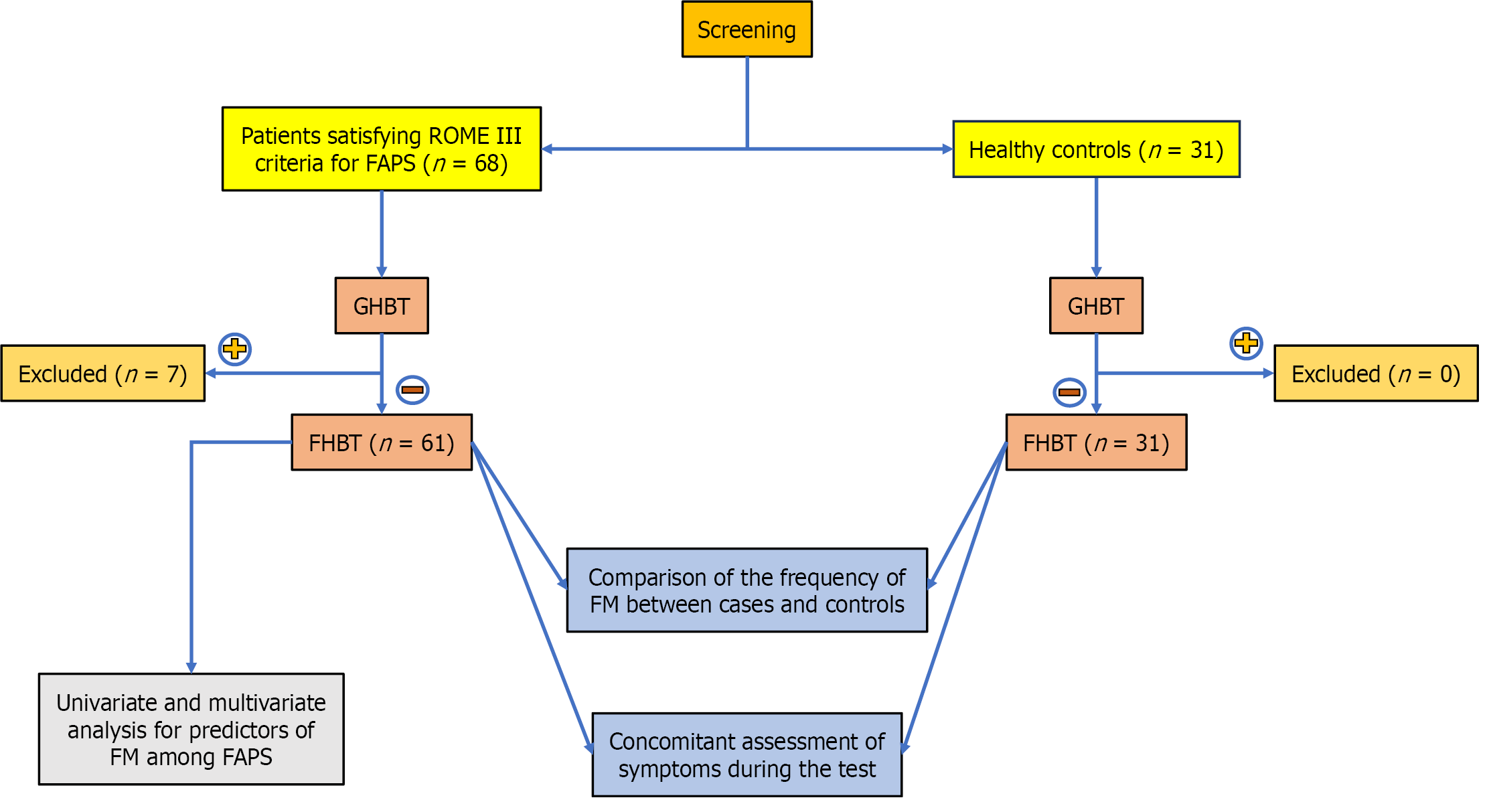
During the study period, 68 patients with FAPS and 31 healthy controls were recruited. Seven patients tested positive for SIBO using the GHBT. They were excluded from further analysis, and none of the control subjects tested positive for SIBO. Consequently, 92 participants, comprising 61 patients with FAPS and 31 healthy controls, were included in the final analysis (Figure 1).
Baseline demographic, dietary characteristics, and body mass index (BMI) were comparable between patients with FAPS and healthy controls. The median age of both groups was 30 years (cases: 15-58 years; controls: 18-48 years; P = 0.61). The sex distribution was 47.5% males among cases and 51.6% among controls (P = 0.71). The dietary pattern (vegetarian vs mixed) was also similar between the groups (P = 0.21), indicating a well-matched control population (Table 1). All patients with FAPS had normal hemogram, normal stool routine microscopy, and were negative for tissue transglutaminase immunoglobulin A antibody.
| Characteristics | Cases (n = 61) | Controls (n = 31) | P value |
| Age (years) | 30 (15, 58) | 30 (18, 48) | 0.61 |
| Gender | Males: 29 (47.5) | Males: 16 (51.6) | 0.71 |
| Body mass index (kg/m2) | 20.83 | 21.8 | 0.96 |
| Dietary pattern | Mixed: 17 | Mixed: 5 | 0.21 |
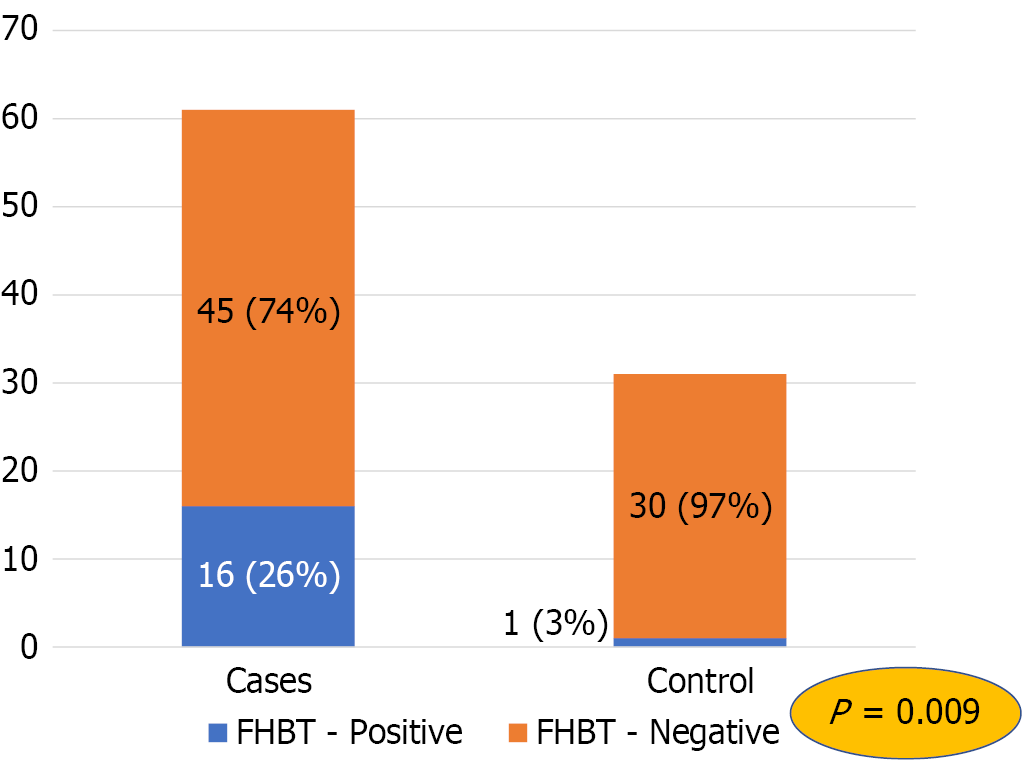
Among the 61 FAPS patients, 16 (26%) tested positive for FHBT, indicating FM, whereas 45 (74%) tested negative. Of the 31 healthy controls, only one individual (3%) had a positive FHBT result, whereas 30 (97%) tested negative (Figure 2). This difference was statistically significant (P = 0.009), indicating a significantly higher prevalence of FM among patients with FAPS than among healthy controls.
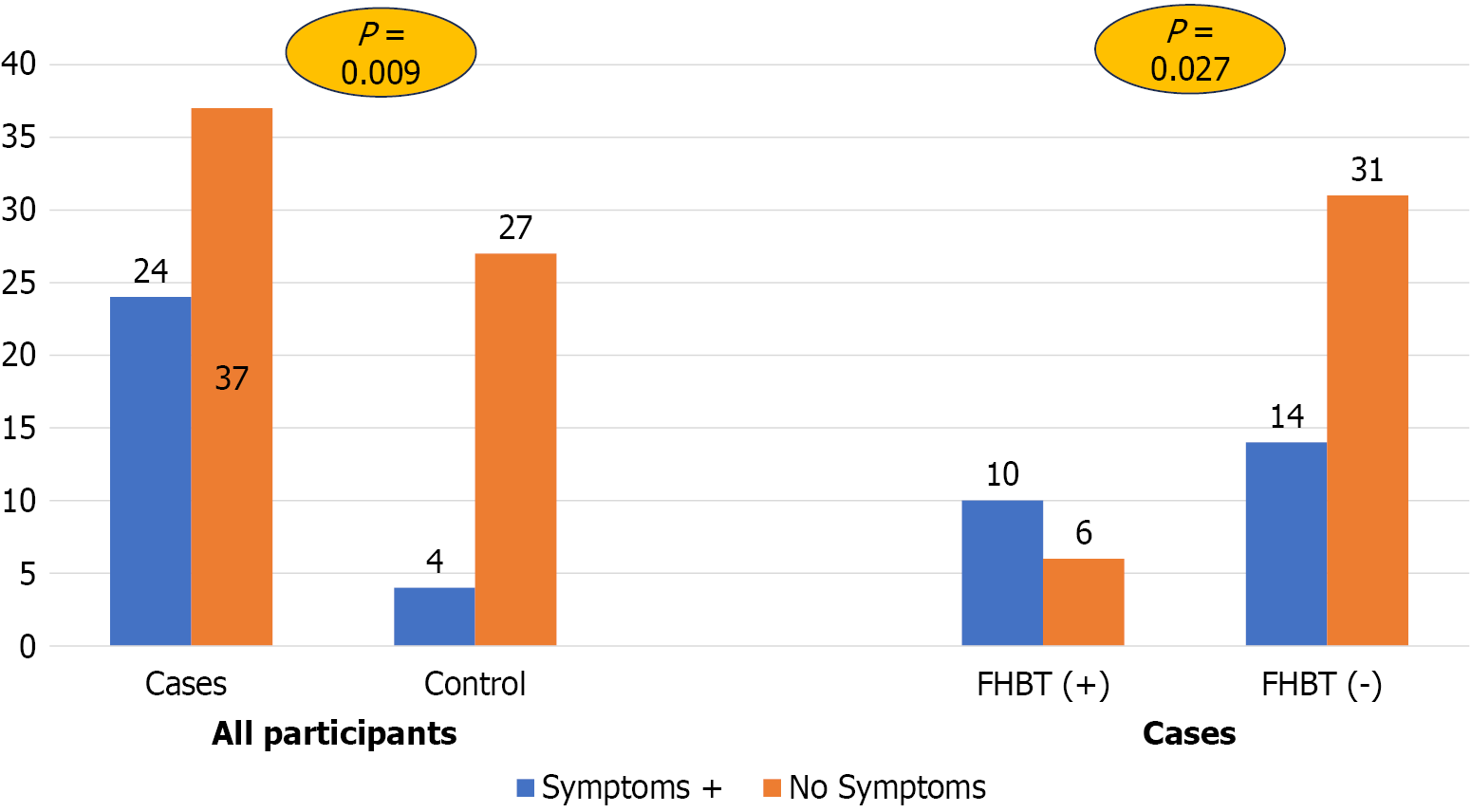
Overall, 28 of the 92 (30.4%) participants experienced gastrointestinal symptoms during the FHBT. A significantly higher proportion of FAPS patients (24/61; 39.3%) reported symptoms during testing than controls (4/31; 12.9%) (P = 0.009; Figure 3).
Among FAPS cases, symptom development was significantly higher among FHBT-positive cases than FHBT-negative cases [62.5% (10/16) vs 31% (14/45), P = 0.027; Figure 3]. Among controls, symptom development did not differ between FHBT-positive controls and FHBT-negative controls [0% (0/1) vs 15.3% (4/26), P = 1.000].
Baseline hydrogen and methane levels were similar in FHBT-positive and FHBT-negative subjects. As expected, peak hydrogen was significantly higher in the FHBT-positive group (< 0.001). The total number of MPs (basal breath methane ≥ 3) in the FHBT-positive group was 6/16 (37.5%), and in the FHBT-negative group was 17/45 (37.7%). The difference between the two groups was not statistically significant (P = 0.98; Table 2).
| Number | Parameters | FHBT positive (n = 16) | FHBT negative (n = 45) | P value |
| 1 | FHBT: Baseline H2 | 4 (1, 13) | 5 (1, 10) | 0.81 |
| 2 | FHBT: Peak H2 | 34.5 (21, 163) | 8 (2, 22) | < 0.001 |
| 3 | Methane: Baseline | 2 (0, 8) | 2 (0, 11) | 0.72 |
| 4 | Methane: Peak | 5.5 (3, 17) | 4 (1, 22) | 0.27 |
| 5 | Predominant methane producers (CH4 ≥ 3) | 6 (37.5) | 17 (37.7) | 0.98 |
Univariate analysis revealed that patients with FHBT-positive FAPS were significantly younger (median age: 20 years vs 31 years; P = 0.001) and more likely to be male (69% vs 40%; P = 0.048). A higher proportion of FHBT-positive patients followed a mixed diet (50%) than FHBT-negative patients (20%), a difference that was also statistically significant (P = 0.022; Table 3). However, symptom duration, Bristol scores, BMI, PPI and anti-cholinergic usage and abdominal symp
| Characteristics | FHBT positive (n = 16) | FHBT negative (n = 45) | P value |
| Age (years) | 20 | 31 | 0.001 |
| Gender | Males: 11 (69) | Males: 18 (40) | 0.048 |
| Duration of symptoms (months) | 8 | 8 | 0.27 |
| Body mass index (kg/m2) | 19.7 | 22.5 | 0.404 |
| Dietary pattern | Mixed: 8 (50) | Mixed: 9 (20) | 0.022 |
| Bristol score (> 4) | 3 (19) | 3 (7) | 0.17 |
| Severity of pain (severe) | 3 (19) | 15 (33) | 0.35 |
| Proton pump inhibitor usage | 16 (100) | 45 (100) | 1.0 |
| Anti-cholinergic usage | 5 (31) | 13 (29) | 1.0 |
| Abdominal distension | 0 | 1 | 1.0 |
| Belching | 2 | 3 | 0.60 |
| Flatulence | 0 | 1 | 1.0 |
| Nausea | 2 | 3 | 0.62 |
| Incomplete evacuation | 2 | 6 | 1.0 |
| Mucus in stools | 3 | 2 | 0.11 |
| Constipation | 2 | 11 | 0.48 |
| Loose stools | 3 | 3 | 0.17 |
| Variable | Odds ratio (95%CI) | P value |
| Age | 1.093 (1.010-1.182) | 0.027 |
| Gender | 0.269 (0.070-1.027) | 0.055 |
| Diet | 3.140 (0.801-12.307) | 0.101 |
This is the first adequately powered case-control study from India to evaluate the frequency of FM in adult patients with FAPS. The present study showed that FM was significantly more frequent in the FAPS group [16/61 (26.2%)] vs in controls group [1/31 (3.2%)] (P = 0.009). Multivariate analysis revealed that younger age was an independent predictor, while male sex trended toward significance. One important finding of this study is the symptom generation among cases without FM (31%), which highlights the pathophysiological pathway other than FAPS in symptom generation other than FM.
This is clinically meaningful because it is derived from a protocol that reduces false positives, utilizes physiological dosing, excludes SIBO, and measures both hydrogen and methane. Among patients with functional gastrointestinal symptoms, particularly IBS and functional dyspepsia, the prevalence of FM is significantly higher, often exceeding 30%-60%[17-20]. Research in children with recurrent abdominal pain has shown a prevalence of FM as high as 30%-45%[28,29].
The higher frequency of FM in the studies described above could be attributed to the use of a higher fructose load in the breath test and the lack of exclusion of SIBO in some studies. The North American consensus has standardized the indications, preparation, sampling intervals, and positivity thresholds for FHBT[26]. Based on the consensus, we used a 25 g test load that aligns with healthy absorption capacity, measured both hydrogen and methane, and documented symptoms with timing relative to the increase in gas. The pathophysiological link between FM and abdominal pain likely involves several mechanisms, including rapid bacterial fermentation of unabsorbed fructose in the colon, which produces hydrogen, methane, carbon dioxide, and short-chain fatty acids. This process generates gas and increases osmotic load, drawing water into the intestinal lumen. The resulting luminal distension stimulates mechanoreceptors and activates visceral hypersensitivity pathways, which are heightened in patients with FAPS. Additionally, colonic fermentation pro
In the study by Yuce et al[30], which excluded SIBO and used a 25 g fructose loading dose, the FHBT positivity rate in the subset of 17 patients with FAPS was 17.6% among Turkish children, consistent with our study. However, the study by Sharma et al[14] on FM in patients with IBS from India revealed a rate of FM of only 14.9%. Hence, there is likely ethnic variation in fructose absorption in various FGIDs that requires further exploration.
Patients with FAPS had a significantly higher prevalence of symptoms during FHBT than healthy controls (P = 0.009). Among these cases, those with a positive FHBT result had a higher prevalence of symptoms than those with a negative FHBT result (P = 0.03). Symptoms of fructose intolerance without malabsorption were observed in 31% (14/45) of patients with FAPS in our study. A similar study by Wilder-Smith et al[31] demonstrated that symptoms of fructose intolerance occur independently of fructose metabolites; however, the symptoms are strongly linked to breath hydrogen and breath methane. This suggests that a component of visceral hypersensitivity in patients with FAPS contributes to symptom development regardless of measurable FM. This finding has therapeutic potential, as a FAPS patient with a positive fructose breath test or reproducible symptoms during a supervised challenge may benefit from a stepwise fructose reduction trial or a low-FODMAP program with structured reintroduction, offering a biologically plausible opportunity for intervention. Fructose intolerance in the absence of malabsorption has been reported in 18%-40% of subjects, as per previously published literature[32,33].
Younger age (P = 0.012) was an independent predictor of a positive FHBT, and male sex showed a trend towards significance (P = 0.055) in our study. In their study of FM in patients with IBS, Melchior et al[23] also found that younger age and male sex were significantly associated with FM compared to patients without FM. However, this difference has not been reported in other studies on FM in IBS and other FGIDs. Female sex has been identified as a predictive factor for SIBO in IBS[34,35]. Hormones are suggested to have a role in bloating and various FGIDs[36,37]. Further research is needed to determine if hormones contribute to FM.
Baseline hydrogen and methane levels were not different between those with and without a positive hydrogen breath test. Total 23/61(37%) patients with FAPS were MPs. In their study of FM in patients with unexplained gastrointestinal symptoms, Choi et al[38] found an 11% prevalence of methanogenic flora. In their study of FM in healthy individuals, Rao et al[13] found that 30% of study participants were MPs. In this study, the presence of MPs did not differ between individuals with and without FM. The higher prevalence of MPs in our study signifies the importance of testing both hydrogen and methane during the evaluation of FM.
The strengths of our study include its case-control design with a well-matched healthy control group, which enabled robust comparison and reduced bias. We systematically ruled out SIBO using GHBT, thereby addressing a significant confounder in FM research. The use of a standardized 25 g fructose load enhanced diagnostic accuracy by reducing false positives. Additionally, we assessed gastrointestinal symptoms during testing with a structured Likert scale, which helped differentiate between biochemical FM and fructose intolerance. Finally, this is among the first adequately powered studies from India to provide essential data from a non-Western population with unique dietary patterns and genetic predispositions. The implications of the study include recommendations regarding testing FM in patients with FAPS and restricting/reducing fructose consumption in those having FM or FM.
Our study had several limitations. First, the total number of participants (92, including 61 patients and 31 controls) was modest for an epidemiological study, which reduced the statistical power and limited the generalizability of our findings. The imbalance between cases and controls (nearly 2:1) can reduce statistical power compared to a balanced design. Second, the study was conducted at a single tertiary hospital in New Delhi; therefore, the results may not be directly extrapolated to populations with different ethnic, cultural, or dietary characteristics. Dietary patterns specific to the Indian population may influence the prevalence of FM, potentially reducing the external validity of the studies. Third, we did not perform precise dietary monitoring or quantify fructose/FODMAP intake. Without such an assessment, it is not possible to establish a direct correlation between measured malabsorption and clinically significant food intolerance in real life. Fourth, diagnosis was based solely on the hydrogen breath test, which, although widely used, has known limitations in terms of sensitivity and specificity, particularly in individuals with methane-producing bacteria (37% in our cohort). Fructose was administered alone rather than with glucose, which may not reflect typical dietary patterns and could underestimate absorption in real-life situations. Fifth, the diagnosis of FAPS was based on the Rome III criteria rather than the updated Rome IV criteria. Although symptoms during breath testing were carefully evaluated with the Likert Scale, subjective reports may have introduced recall or response bias. FAPS overlaps with other FGIDs, potentially introducing diagnostic heterogeneity. Sixth, no longitudinal follow-up was performed to evaluate the impact of dietary modifications in patients with positive results. Finally, although age and sex were identified as predictors of FM, our multivariate model did not adjust for other potentially relevant factors, such as BMI, dietary history, comorbidities, or socioeconomic status, which leaves scope for residual confounding. Future research should address these limitations through larger, multicenter studies that incorporate dietary assessment, Rome IV-based diagnostic criteria, standardized symptom measurement, and interventional dietary arms.
In summary, our study showed that more than one-quarter of patients with FAPS had evidence of FM, and many of these patients also displayed symptoms consistent with fructose intolerance. Younger age was identified as an independent predictor of FM, with a trend towards significance for male sex. These findings suggest that dietary fructose restriction or structured low-FODMAP interventions may help manage selected patients with FAPS. Future multicenter studies with larger sample sizes, comprehensive dietary monitoring, validated symptom assessment tools, and interventional arms are necessary to confirm these results and better define the therapeutic role of fructose restriction in FGIDs.
| 1. | Grover M, Drossman DA. Functional abdominal pain. Curr Gastroenterol Rep. 2010;12:391-398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 2. | Keefer L, Drossman DA, Guthrie E, Simrén M, Tillisch K, Olden K, Whorwell PJ. Centrally Mediated Disorders of Gastrointestinal Pain. Gastroenterology. 2016;S0016-5085(16)00225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 88] [Article Influence: 8.8] [Reference Citation Analysis (2)] |
| 3. | Drossman DA. Functional Gastrointestinal Disorders: History, Pathophysiology, Clinical Features and Rome IV. Gastroenterology. 2016;S0016-5085(16)00223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1904] [Cited by in RCA: 1544] [Article Influence: 154.4] [Reference Citation Analysis (7)] |
| 4. | Farmer AD, Aziz Q. Mechanisms and management of functional abdominal pain. J R Soc Med. 2014;107:347-354. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 32] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 5. | Whelan K, Martin LD, Staudacher HM, Lomer MCE. The low FODMAP diet in the management of irritable bowel syndrome: an evidence-based review of FODMAP restriction, reintroduction and personalisation in clinical practice. J Hum Nutr Diet. 2018;31:239-255. [PubMed] [DOI] [Full Text] |
| 6. | Ong DK, Mitchell SB, Barrett JS, Shepherd SJ, Irving PM, Biesiekierski JR, Smith S, Gibson PR, Muir JG. Manipulation of dietary short chain carbohydrates alters the pattern of gas production and genesis of symptoms in irritable bowel syndrome. J Gastroenterol Hepatol. 2010;25:1366-1373. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 474] [Cited by in RCA: 402] [Article Influence: 25.1] [Reference Citation Analysis (4)] |
| 7. | Tuck CJ, Muir JG, Barrett JS, Gibson PR. Fermentable oligosaccharides, disaccharides, monosaccharides and polyols: role in irritable bowel syndrome. Expert Rev Gastroenterol Hepatol. 2014;8:819-834. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 92] [Article Influence: 7.7] [Reference Citation Analysis (1)] |
| 8. | Wilder-Smith CH, Olesen SS, Materna A, Drewes AM. Predictors of response to a low-FODMAP diet in patients with functional gastrointestinal disorders and lactose or fructose intolerance. Aliment Pharmacol Ther. 2017;45:1094-1106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 55] [Article Influence: 6.1] [Reference Citation Analysis (1)] |
| 9. | Ferraris RP, Choe JY, Patel CR. Intestinal Absorption of Fructose. Annu Rev Nutr. 2018;38:41-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 132] [Article Influence: 16.5] [Reference Citation Analysis (1)] |
| 10. | Cash BD, Patel D, Scarlata K. Demystifying Carbohydrate Maldigestion: A Clinical Review. Am J Gastroenterol. 2025;120:1-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 11. | Zingone F, Bertin L, Maniero D, Palo M, Lorenzon G, Barberio B, Ciacci C, Savarino EV. Myths and Facts about Food Intolerance: A Narrative Review. Nutrients. 2023;15:4969. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 28] [Reference Citation Analysis (1)] |
| 12. | Helwig U, Koch AK, Koppka N, Holtmann S, Langhorst J. The Predictive Value of the Hydrogen Breath Test in the Diagnosis of Fructose Malabsorption. Digestion. 2019;99:140-147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 13. | Rao SS, Attaluri A, Anderson L, Stumbo P. Ability of the normal human small intestine to absorb fructose: evaluation by breath testing. Clin Gastroenterol Hepatol. 2007;5:959-963. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 111] [Cited by in RCA: 105] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 14. | Sharma A, Srivastava D, Verma A, Misra A, Ghoshal UC. Fructose malabsorption is not uncommon among patients with irritable bowel syndrome in India: a case-control study. Indian J Gastroenterol. 2014;33:466-470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 1.2] [Reference Citation Analysis (2)] |
| 15. | Ladas SD, Grammenos I, Tassios PS, Raptis SA. Coincidental malabsorption of lactose, fructose, and sorbitol ingested at low doses is not common in normal adults. Dig Dis Sci. 2000;45:2357-2362. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 16. | Densupsoontorn N, Jirapinyo P, Thamonsiri N, Wongarn R. Fructose malabsorption in Thai adult. Asia Pac J Clin Nutr. 2007;16:209-212. [PubMed] |
| 17. | Jung KW, Seo M, Cho YH, Park YO, Yoon SY, Lee J, Yang DH, Yoon IJ, Seo SY, Lee HJ, Park SH, Kim KJ, Ye BD, Byeon JS, Jung HY, Yang SK, Kim JH, Myung SJ. Prevalence of Fructose Malabsorption in Patients With Irritable Bowel Syndrome After Excluding Small Intestinal Bacterial Overgrowth. J Neurogastroenterol Motil. 2018;24:307-316. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 21] [Article Influence: 2.6] [Reference Citation Analysis (1)] |
| 18. | Mishkin D, Sablauskas L, Yalovsky M, Mishkin S. Fructose and sorbitol malabsorption in ambulatory patients with functional dyspepsia: comparison with lactose maldigestion/malabsorption. Dig Dis Sci. 1997;42:2591-2598. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 44] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 19. | Sia T, Tanaka RO, Mousad A, Narayan AP, Si K, Bacchus L, Ouerghi H, Patel A, Patel A, Cunningham E, Epstein T, Fu J, Liu S, Khuda R, McDonald P, Mallik S, McNulty J, Pan M, Leung J. Fructose malabsorption and fructan malabsorption are associated in patients with irritable bowel syndrome. BMC Gastroenterol. 2024;24:143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 20. | Goebel-Stengel M, Stengel A, Schmidtmann M, Voort Iv, Kobelt P, Mönnikes H. Unclear abdominal discomfort: pivotal role of carbohydrate malabsorption. J Neurogastroenterol Motil. 2014;20:228-235. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 31] [Article Influence: 2.6] [Reference Citation Analysis (1)] |
| 21. | Nucera G, Gabrielli M, Lupascu A, Lauritano EC, Santoliquido A, Cremonini F, Cammarota G, Tondi P, Pola P, Gasbarrini G, Gasbarrini A. Abnormal breath tests to lactose, fructose and sorbitol in irritable bowel syndrome may be explained by small intestinal bacterial overgrowth. Aliment Pharmacol Ther. 2005;21:1391-1395. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 134] [Article Influence: 6.4] [Reference Citation Analysis (4)] |
| 22. | Johlin FC Jr, Panther M, Kraft N. Dietary fructose intolerance: diet modification can impact self-rated health and symptom control. Nutr Clin Care. 2004;7:92-97. [PubMed] |
| 23. | Melchior C, Gourcerol G, Déchelotte P, Leroi AM, Ducrotté P. Symptomatic fructose malabsorption in irritable bowel syndrome: A prospective study. United European Gastroenterol J. 2014;2:131-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 32] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 24. | Rasquin A, Di Lorenzo C, Forbes D, Guiraldes E, Hyams JS, Staiano A, Walker LS. Childhood functional gastrointestinal disorders: child/adolescent. Gastroenterology. 2006;130:1527-1537. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1292] [Cited by in RCA: 1087] [Article Influence: 54.4] [Reference Citation Analysis (5)] |
| 25. | Ghoshal UC, Srivastava D. Irritable bowel syndrome and small intestinal bacterial overgrowth: meaningful association or unnecessary hype. World J Gastroenterol. 2014;20:2482-2491. [PubMed] [DOI] [Full Text] |
| 26. | Rezaie A, Buresi M, Lembo A, Lin H, McCallum R, Rao S, Schmulson M, Valdovinos M, Zakko S, Pimentel M. Hydrogen and Methane-Based Breath Testing in Gastrointestinal Disorders: The North American Consensus. Am J Gastroenterol. 2017;112:775-784. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 721] [Cited by in RCA: 586] [Article Influence: 65.1] [Reference Citation Analysis (5)] |
| 27. | Mathur R, Amichai M, Chua KS, Mirocha J, Barlow GM, Pimentel M. Methane and hydrogen positivity on breath test is associated with greater body mass index and body fat. J Clin Endocrinol Metab. 2013;98:E698-E702. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 92] [Article Influence: 7.1] [Reference Citation Analysis (1)] |
| 28. | Escobar MA Jr, Lustig D, Pflugeisen BM, Amoroso PJ, Sherif D, Saeed R, Shamdeen S, Tuider J, Abdullah B. Fructose intolerance/malabsorption and recurrent abdominal pain in children. J Pediatr Gastroenterol Nutr. 2014;58:498-501. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 29] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 29. | Posovszky C, Roesler V, Becker S, Iven E, Hudert C, Ebinger F, Calvano C, Warschburger P. Roles of Lactose and Fructose Malabsorption and Dietary Outcomes in Children Presenting with Chronic Abdominal Pain. Nutrients. 2019;11:3063. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (1)] |
| 30. | Yuce O, Kalayci AG, Comba A, Eren E, Caltepe G. Lactose and Fructose Intolerance in Turkish Children with Chronic Abdominal Pain. Indian Pediatr. 2016;53:394-397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 31. | Wilder-Smith C, Lee SH, Olesen SS, Low JY, Kioh DYQ, Ferraris R, Materna A, Chan ECY. Fructose intolerance is not associated with malabsorption in patients with functional gastrointestinal disorders. Neurogastroenterol Motil. 2021;33:e14150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 32. | Wintermeyer P, Baur M, Pilic D, Schmidt-Choudhury A, Zilbauer M, Wirth S. Fructose malabsorption in children with recurrent abdominal pain: positive effects of dietary treatment. Klin Padiatr. 2012;224:17-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 33. | Beyerlein L, Pohl D, Delco F, Stutz B, Fried M, Tutuian R. Correlation between symptoms developed after the oral ingestion of 50 g lactose and results of hydrogen breath testing for lactose intolerance. Aliment Pharmacol Ther. 2008;27:659-665. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 36] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 34. | Sachdeva S, Rawat AK, Reddy RS, Puri AS. Small intestinal bacterial overgrowth (SIBO) in irritable bowel syndrome: frequency and predictors. J Gastroenterol Hepatol. 2011;26 Suppl 3:135-138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 56] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 35. | Chu H, Fox M, Zheng X, Deng Y, Long Y, Huang Z, Du L, Xu F, Dai N. Small Intestinal Bacterial Overgrowth in Patients with Irritable Bowel Syndrome: Clinical Characteristics, Psychological Factors, and Peripheral Cytokines. Gastroenterol Res Pract. 2016;2016:3230859. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 18] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 36. | Jiang Y, Greenwood-Van Meerveld B, Johnson AC, Travagli RA. Role of estrogen and stress on the brain-gut axis. Am J Physiol Gastrointest Liver Physiol. 2019;317:G203-G209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 53] [Article Influence: 7.6] [Reference Citation Analysis (1)] |
| 37. | Iovino P, Bucci C, Tremolaterra F, Santonicola A, Chiarioni G. Bloating and functional gastro-intestinal disorders: where are we and where are we going? World J Gastroenterol. 2014;20:14407-14419. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 39] [Cited by in RCA: 45] [Article Influence: 3.8] [Reference Citation Analysis (5)] |
| 38. | Choi YK, Johlin FC Jr, Summers RW, Jackson M, Rao SS. Fructose intolerance: an under-recognized problem. Am J Gastroenterol. 2003;98:1348-1353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 84] [Article Influence: 3.7] [Reference Citation Analysis (1)] |