Published online Mar 22, 2026. doi: 10.4291/wjgp.v17.i1.113373
Revised: October 2, 2025
Accepted: January 9, 2026
Published online: March 22, 2026
Processing time: 209 Days and 10.4 Hours
Acute cholangitis is a potentially life-threatening infection of the biliary tract and its mortality rate is between 10%-30%. Early risk stratification is essential for the best possible outcome. Serum albumin, an index of the inflammatory and nu
To assess the value of hypoalbuminemia as a predictor of mortality in acute cholangitis.
A systematic search of PubMed, Web of Science, Semantic Scholar, Cochrane Library, and Google Scholar was performed up to May 2025. Eligible studies included adults diagnosed with acute cholangitis and reported mor
Eight retrospectively assembled cohort studies enrolling a total of 2215 of patients with acute cholangitis were incorporated in this meta-analysis. Mean age of the patient was greater than 65 years. Among them, 52.2% were males. Out 2215 patients, 242 (10.9%) died. Hypoalbuminemia was highly associated with increased mortality. Dichotomous outcome analysis demonstrated strong evidence of association between hypoalbuminemia and mortality (OR = 8.02, 95%CI: 3.41-18.83; P ≤ 0.001; I² = 50%). Continuous outcome analysis demonstrated a 23% increased risk of mortality associated with every 1 g/dL fall in serum albumin (OR = 0.77, 95%CI: 0.64-0.93; P = 0.005; I² = 66%). All the incorporated studies were of high methodological quality with minimal evidence of publication bias.
Hypoalbuminemia on hospital admission is a strong and independent predictor of death in patients with acute cholangitis. Admission serum albumin measurement provides a simple, cost-effective, and universally available method of early risk stratification. Prospective studies in the future need to examine whether correction of hypoalbuminemia improves clinical outcomes.
Core Tip: Early recognition of high-risk patients is essential in acute cholangitis. This meta-analysis shows that hypoalbuminemia is strongly linked to mortality, making serum albumin a practical bedside marker. Being inexpensive and widely available, albumin measurement at admission can guide clinicians toward intensified monitoring, early biliary drainage, and tailored therapy, ultimately improving survival outcomes in this life-threatening condition.
- Citation: Khan RTY, Ahsam S, Kumar SK, Khan K, Kakar MT, Hyder A, Malik W, Mubarak M, Luck NH. Hypoalbuminemia as a predictor of mortality in patients with acute cholangitis: A systematic review and meta-analysis. World J Gastrointest Pathophysiol 2026; 17(1): 113373
- URL: https://www.wjgnet.com/2150-5330/full/v17/i1/113373.htm
- DOI: https://dx.doi.org/10.4291/wjgp.v17.i1.113373
Acute cholangitis is a potentially life-threatening infection of the biliary tree that occurs due to the obstruction of biliary tree and ascending infection, most commonly due to gallstones, malignancies, or strictures[1]. This condition is consi
In patients with cholangitis, early risk stratification is imperative for obtaining desired outcomes. Early identification of the patients who are at high-risk for adverse outcome allows timely application of directed intensive therapy, such as intensive care monitoring, directed antimicrobial therapy, and timely biliary drainage[6]. Current guidelines, such as the Tokyo Guidelines, incorporate clinical, laboratory, and imaging parameters for the diagnosis and grading of the severity of cholangitis, but no clear-cut, reproducible, and openly accessible prognostic markers have been determined[7]. Appro
Several models have been proposed to predict mortality in patients with acute cholangitis. Schneider et al[9] proposed a model in predicting mortality in cholangitis in German population with a sensitivity and specificity of 82.9% and 85.1% respectively. Similarly, Yıldız et al[10] proposed a model for prediction of mortality in acute cholangitis in Turkish population. Similarly, Ismail et al[11] proposed in South Asian population. However, all these models were complex and relied upon multiple laboratory and clinical parameters.
One of the most commonly measured laboratory parameters, serum albumin, is not only a well-recognized marker of nutritional and inflammation status; but also a consistent predictor of adverse outcome in various disease states, e.g., sepsis, pneumonia, heart failure, and malignancy[12,13]. Decreased serum albumin levels are not only associated with increased susceptibility to infections but can also result in delayed wound healing, prolonged hospitalization, and increased mortality[14]. As a result of its prognostic implication in various disease states, hypoalbuminemia is developing as a promising marker for the purpose of risk stratification in the context of acute diseases[15].
The rationale for the utility of decreased serum albumin levels as a prognostic indicator in acute cholangitis relies on pathophysiological and clinical grounds. Inflammatory illnesses, like cholangitis, cause enhanced capillary permeability, redistribution and loss of albumin from the vascular space, usually complicated by underlying malnutrition or liver disease[16]. Consequently, hypoalbuminemia can be an integrated marker of the intensity of systemic inflammation and the patient's pre-existing nutritional and hepatic reserve[17,18]. Characterization of hypoalbuminemia as an independent mortality predictor in acute cholangitis could offer the clinician a readily available, inexpensive tool toward the improve
Comprehensive literature search was performed by two authors independently using PubMed, Google Scholar, Semantic Scholar, Web of Science, and the Cochrane Library. A literature search was performed based on the following search string “[("Cholangitis"[Mesh] OR acute cholangitis) AND ("Hypoalbuminemia"[Mesh] OR hypoalbuminemia OR low serum albumin OR albumin) AND ("Mortality" OR death OR outcome OR prognosis)]” (Supplementary Table 1). The search included all relevant articles published up to May 31, 2025. Additional references were identified through manual screening of bibliographies of selected articles.
All those studies were selected, which fulfill the selection criteria described as PICOS: (1) P (Population): Adults ( ≥ 18 years) diagnosed with acute cholangitis, confirmed by clinical, laboratory, and/or imaging criteria (e.g., Tokyo Guide
Inclusion criteria: Retrospective cohort studies that reported data on serum albumin levels and mortality outcomes in patients with acute cholangitis were enrolled in the study. The study population was restricted to adults aged 18 years or older with a diagnosis of acute cholangitis (as defined by the Tokyo guidelines 2018)[7]. The exposure of interest was hypoalbuminemia, typically defined as a serum albumin level below 3.5 g/dL, or according to the threshold specified in each study. The comparator group comprised of the patients with normal serum albumin levels. The primary outcome was all-cause in-hospital mortality or 30-day mortality.
Exclusion criteria: Case reports, reviews, pediatric studies, and articles lacking relevant data were excluded from the study.
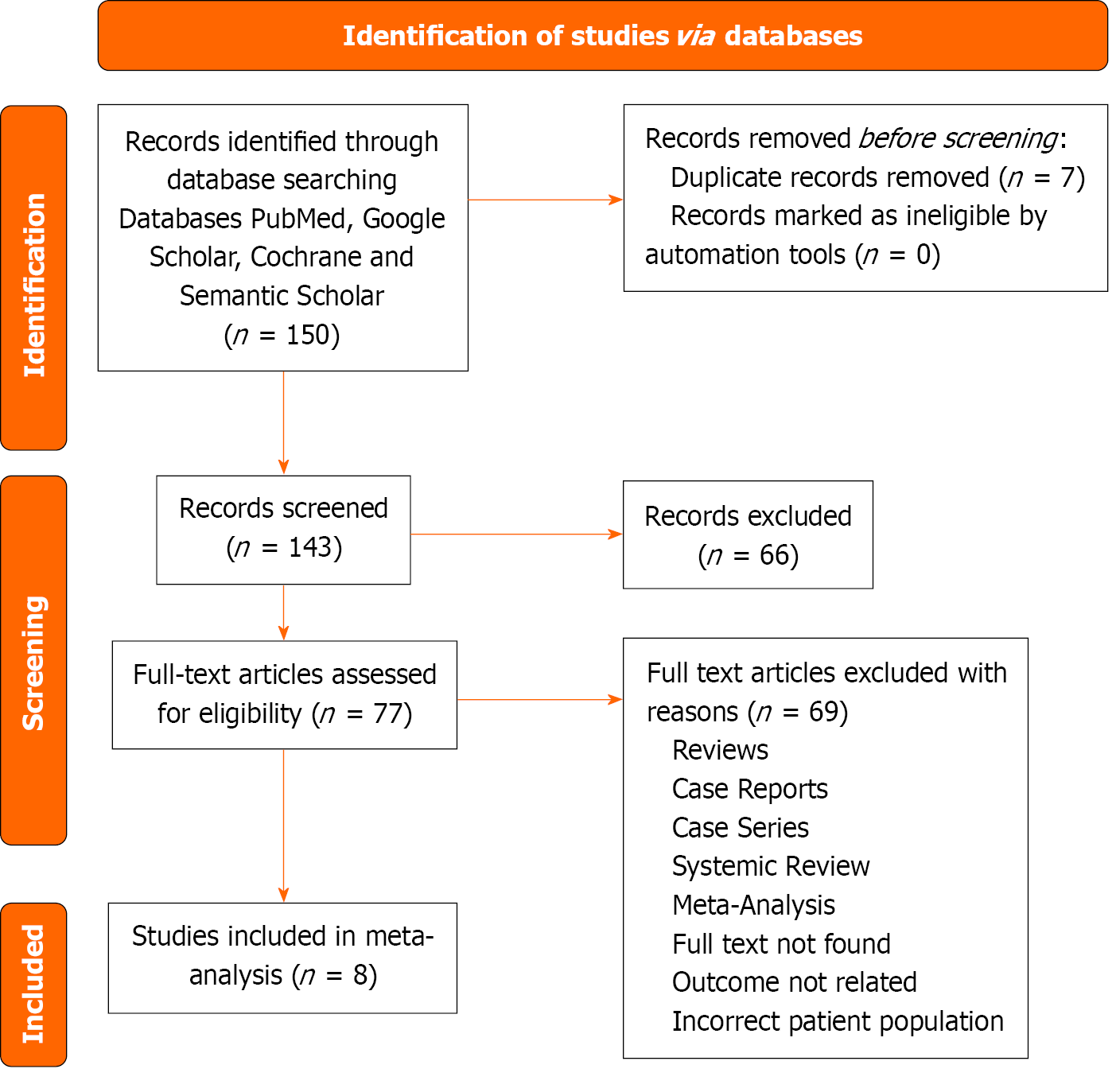
Two independent reviewers (Khan RTY and Kumar SK) screened titles and abstracts for relevance. Full texts were retrieved for potentially eligible studies. Discrepancies were resolved through mutual discussion and also by consulting the third reviewer (Hyder A). Of the 150 initially identified records, 143 were retained after removing 7 duplicates. Full-text screening was conducted for 77 articles, and finally, 8 studies were included in the meta-analysis. A structured data extraction form was used to collect relevant variables from each study. Extracted data included: Author name, pub
Each included study was assessed for methodological quality using the Newcastle–Ottawa Scale (NOS), which evaluates participant selection, comparability, and outcome assessment. Studies were classified as high (7–9 points), moderate (5–6 points), or low (< 5 points) quality.
Statistical analysis was carried out by using the Review Manager (version 5.4; Copenhagen: The Nordic Cochrane Center, The Cochrane Collaboration, 2020). Pooled odds ratios (OR) or risk ratios (RR) with corresponding 95%CI were calculated to assess the association between hypoalbuminemia and mortality in acute cholangitis. A random-effects model was used to account for between-study heterogeneity. Statistical heterogeneity was assessed using the I² statistic, with values above 50% considered substantial. Publication bias was evaluated by visual inspection of funnel plots.
A total of 150 studies were initially identified through the search strategy. After removing 7 duplicates, 143 articles were screened by the title and abstract. Full texts were assessed for 77 studies, and ultimately 8 studies met the eligibility criteria for inclusion[5,6,19-24]. These studies were published between 2012 and 2024; and encompassed a cumulative population of 2914 patients admitted with cholangitis from countries including Turkey, China, Singapore, Japan and Germany. All these studies were retrospective cohort studies comprising of adult population (age 18 years or older) admitted with acute cholangitis. The process of study selection is summarized in the PRISMA flow diagram (Figure 1).
Out of 2215 patients, 1157 (52.2%) patients were males; while 1058 (47.8%) were females. The mean or median age of participants of this meta-analysis was generally above 65 years. The definition of hypoalbuminemia varied, with 4 out of eight studies using mean albumin levels; while the other studies using an albumin as a dichotomous variable. Two out of remaining 4 studies used serum albumin cutoff of 2.8 g/dL; while the remaining 2 studies used the serum albumin cutoffs of 2.5 g/dL and 3.5 g/dL respectively. Out 2215 patients, mortality occurred in 242 (10.9%) patients. Table 1 summarizes the baseline characteristics of the studies included in this meta-analysis[5,6,19-24].
| Ref. | Country | Study type | Sample size (n) | Age | Males/females | Definition of hypoalbuminemia | Incidence rates of hypoalbuminemia | Mortality rates | Type of mortality reported |
| Acehan et al[19], 2023 | Türkiye | Retrospective study | 309 | 77.2 ± 8.0 | 149/160 (48.2/51.8) | Albumin level | 155 (50.2) | 27 (8.7) | In-hospital mortality |
| Comoglu et al[5], 2024 | Türkiye | Retrospective study | 250 | 76 (69-82) | 125/125 (50/50) | < 3.5 g/dL | 63 (25.2) | 23 (9.2) | In-hospital mortality |
| Sun et al[20], 2013 | China | Retrospective study | 120 | 66 ± 15.1 | 83/37 (69.2/30.8) | < 2.5 g/dL | 21 (17.5) | 6 (5) | 30-day mortality |
| Hunyady et al[21], 2023 | Germany | Retrospective study | 127 | 76 (19-87) | 93/34 (73.2/26.8) | Albumin level | Not mentioned | 45 (35.4) | Not mentioned |
| Inan et al[6], 2023 | Türkiye | Retrospective study | 300 | 77.2 ± 8.0 | 144/156 (48/52) | Albumin level | Not mentioned | 23 (7.7) | In-hospital mortality |
| Mohan et al[22], 2021 | Singapore | Retrospective study | 388 | 83.77 (81.86-85.61) | 162/226 (41.8/58.2) | < 2.8 g/dL | Not mentioned | 38 (9.8) | In-hospital mortality |
| Pan et al[23], 2023 | China | Retrospective study | 506 | 72.92 (60.86-83.17) | 263/243 (52/48) | Albumin level | Not mentioned | 71 (14) | In-hospital mortality |
| Tsuyuguchi et al[24], 2012 | Japan | Retrospective study | 215 | 66.7 ± 10.3 | 138/77 (64.2/35.8) | < 2.8 g/dL | 40 (18.6) | 9 (4.2) | Not mentioned |
All included studies were subjected to quality assessment using the NOS. All the eight studies were labelled as high quality on the basis of NOS with an overall low risk of bias (6 studies scored 9 points on NOS and 2 studies scored 8 points). All the eight studies achieved ≥ 8 points, reflecting strong selection procedures, adequate adjustment for confounders, and reliable outcome ascertainment (Supplementary Table 2).
A total of 8 studies evaluated the role of hypoalbuminemia as a predictor of mortality in acute cholangitis. There was a significant relationship between hypoalbuminemia and survival in patients with acute cholangitis; with low levels of serum albumin predicting increased mortality.
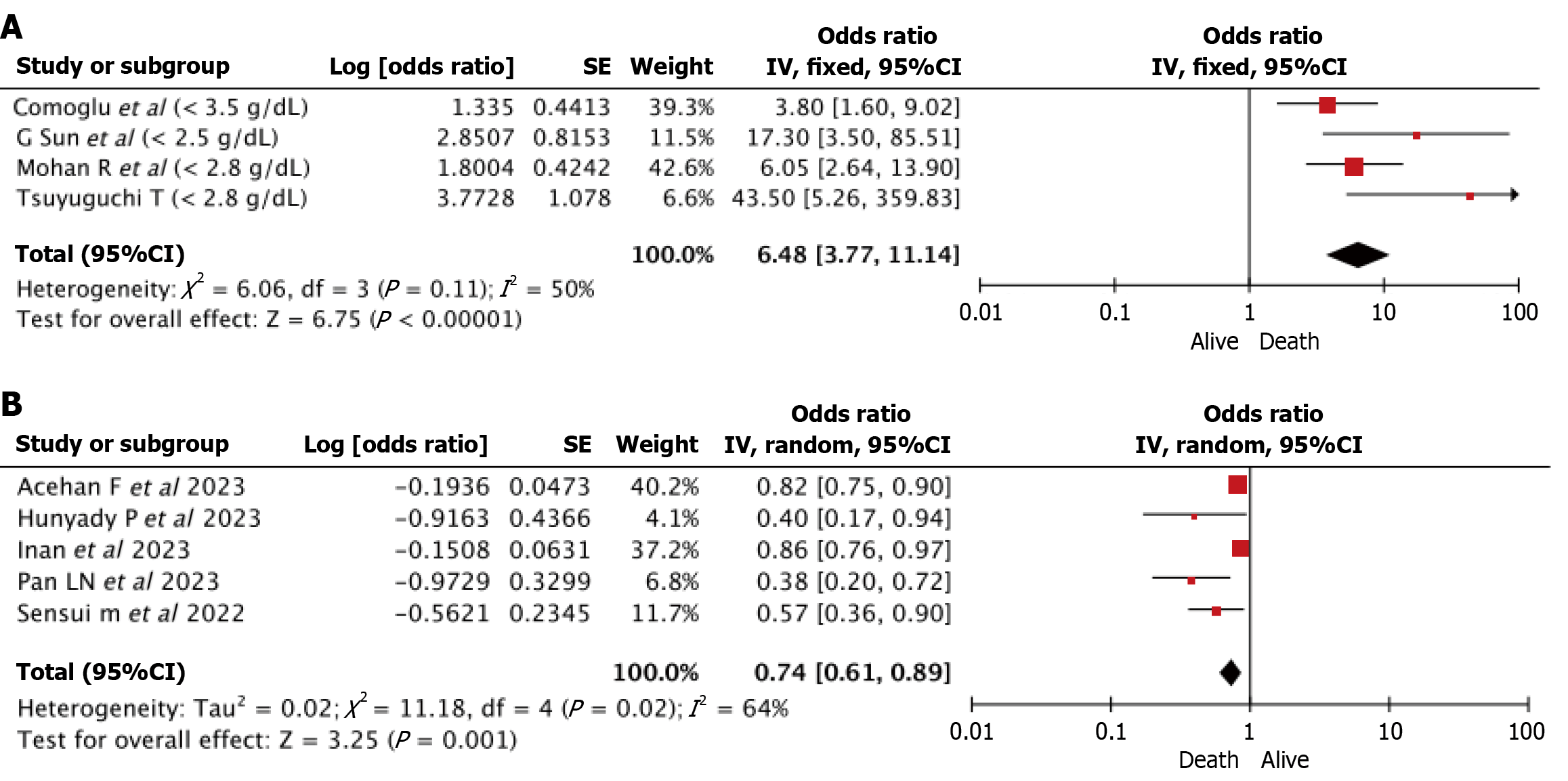
Four out of 8 studies reported mortality using study-defined albumin cutoff values. Two studies used albumin cutoff of < 2.8 g/dL; one study used 2.5 g/dL and one used 3.5 g/dL respectively. The results of random-effect meta-analysis revealed that patients with hypoalbuminemia on admission were at increased risk of in-hospital or 30-days mortality in acute cholangitis [OR = 8.02 (3.41-18.83); P ≤ 0.001]. Moderate heterogeneity was observed among studies with dichotomous albumin values (I2 = 50%; P = 0.11) (Figure 2A).
In four studies, mean serum albumin levels was used for the evaluation of its role as a predictor of mortality in acute cholangitis. The results of random-effect meta-analysis showed the inverse relation between mean serum albumin levels and risk of mortality. Here, we observed that lower serum albumin levels were significantly associated with increased mortality in acute cholangitis [OR = 0.77 (0.64-0.93); P = 0.005] (for each 1 g/dL decrease in serum albumin levels, the mortality rate increased by 23%). Substantial heterogeneity was observed among the studies using mean serum albumin levels to predict mortality (I2 = 66%; P = 0.03) (Figure 2B).
The funnel plot showed symmetrical distribution with minimal evidence of publication bias for types of analysis done for serum albumin in this study. These findings were further supported by Egger’s regression test which were statistically non-significant (P > 0.05 for both analyses); further confirming that publication bias was unlikely to have a substantial impact on the outcome of meta-analysis (Supplementary Figures 1 and 2).
The results of this meta-analysis provided the convincing role of hypoalbuminemia as a significant predictor of mortality in patients with acute cholangitis. Using two types of analysis performed in this study i.e. dichotomous (using specific albumin cutoffs) and continuous (mean albumin levels) analyses, we observed a strong association between low serum albumin levels at admission and increased risk of in-hospital and 30-day mortality. This evidence compels the need of clinical assessment for hypoalbuminemia in acute cholangitis at presentation, as it is not only a marker of nutritional and hepatic reserve but can also reflect the ongoing systemic inflammatory response in acute cholangitis.
Our findings are consistent with prior studies regarding the prognostic role of hypoalbuminemia in a variety of acute medical conditions, including sepsis, pneumonia, and other infectious diseases, where low serum albumin has been independently linked to worse clinical outcomes and higher mortality[12]. Jellinge et al[25] proposed the role of hypoalbuminemia in predicting 30-day mortality in hospitalized patients. Similarly, Perel et al[26] predicted the role of hypoalbuminemia as a significant prognostic factor in predicting in-hospital mortality among patients admitted to intensive care unit. Kumar et al[27] proposed the role of low albumin levels as a predictor of mortality in Indian population with sepsis.
The association between serum albumin levels and mortality in acute cholangitis is quite understandable by the fact that serum albumin not only indicates the nutritional state of the patient but also predicts the intensity of systemic inflammation, hepatic synthetic capacity and increased capillary permeability[15,28]. Severe acute cholangitis presents with an overactive inflammatory response and hepatic impairment, both conditions ultimately resulting in decline in serum albumin levels. In this scenario, hypoalbuminemia not only function to serve as an indirect marker of inflammation, but also predicts the patient's overall physiologic reserve[19,29,30]. Additionally, albumin possesses immunomodulatory as well as antioxidant capabilities, and therefore its deficiency could directly impair the body's defense mechanisms, hence potentially rendering patients susceptible to sepsis, organ failure and decreased survival[31,32]. The magnitude of association observed in this meta-analysis-an OR of over 8 for mortality in patients with hypoalbuminemia-highlights the clinical importance of routinely monitoring serum albumin in patients admitted with acute cholangitis. The finding that every 1 g/dL decrease in serum albumin was associated with a 23% increase in mortality risk further reinforces the prog
There are several mechanisms that can explain the association between hypoalbuminemia and mortality from acute cholangitis. Reduced serum albumin is frequently an indicator of poor nutritional status, which impairs immune protection, aggravates delayed recovery, and impairs physiological reserve in acute illness[33]. Malnutrition is typical among elderly or chronically sick patients and predisposes to severe infection[34]. Acute cholangitis induces a vigorous inflammatory reaction resulting in capillary hyperpermeability, fluid shifts, and decreased hepatic albumin production. Cytokines like IL-6 repress albumin synthesis, whereas albumin itself exerts antioxidant and immunomodulatory activities that limit inflammation[35]. When serum albumin levels fall below a certain threshold, its protective activities are lost and can predispose the patients to sepsis and organ failure[36]. Last but not least, hepatic synthetic failure can precipitate hypoalbuminemia when it becomes severe in patients or when severe infection is present. Reduced hepatic reserve indicates the presence of either chronic illness or an acute hepatic insult and amplifies the prognostic significance of low albumin[37]. Collectively, hypoalbuminemia is a composite indicator of nutritional depletion, inflammatory burden, and hepatic failure that explains its strong and persistent association with mortality especially in a condition like acute cholangitis.
An important clinical consideration is determining which serum albumin cutoff should be adopted for risk stratification in patients with acute cholangitis. Across the included studies, the cutoff values for hypoalbuminemia varied between 2.5 g/dL and 3.5 g/dL, reflecting differences in population characteristics and study methodology. From a practical standpoint, a threshold of 3.5 g/dL is most widely recognized in clinical practice as the lower limit of normal for serum albumin. Using this cutoff has two advantages. First, it aligns with routine laboratory reporting standards, ensuring easy applicability without the need for additional calculations. Second, the pooled OR in our analysis indicate that patients with albumin levels below this threshold face a substantially higher risk of mortality. Therefore, adopting 3.5 g/dL as a standardized clinical cutoff may offer a simple and effective way to identify high-risk patients early during hospital admission.
The decision regarding optimal cutoff selection, however, must balance sensitivity and specificity. A lower cutoff (e.g., 2.5–2.8 g/dL) may increase specificity for adverse outcomes but risks missing patients at earlier stages of deterioration[20,24]. Conversely, a higher cutoff of 3.5 g/dL improves sensitivity and allows for broader identification of at-risk patients who might benefit from closer monitoring or earlier intervention[5].
Although our study did not perform a formal Decision Curve Analysis (DCA), such analysis could provide useful insights into the net clinical benefit of applying different albumin thresholds for guiding clinical decision-making. DCA integrates both the sensitivity/specificity of a test and the clinical consequences of true and false positives.
There are several prognostic scoring systems, including Acute Physiology and Chronic Health Evaluation II and Sequential Organ Failure Assessment, that are widely applied to predict outcomes among critically ill patients, including those with acute cholangitis. They involve multiple clinical and laboratory parameters and have been found to have excellent predictive role in predicting mortality[38]. However, these parameters require a large number of parameters, specialized tools or gadgets for calculation, and sometimes there is a requirement of repeated measurements; and the
There are several strengths that can be attributed to this study. First, included studies had high methodologic quality with low risk of bias as assessed according to the NOS. Consistency in findings from various study designs, geographical locations, and thresholds for albumin further increases the strength and generalizability of findings. In addition, meta-analysis showed limited evidence of publication bias, which further confirms the validity of these findings.
Despite these strengths, this study had certain limitations. First, the studies included in this meta-analysis were all retrospective in nature, which may introduce residual confounding and selection bias. While most studies adjusted for key covariates, unmeasured confounders may still have influenced the observed associations. Second, there was some heterogeneity in the definition of hypoalbuminemia and albumin cutoff values used across studies, although this did not significantly alter the overall direction or significance of the results. Third, the included studies were conducted primarily in Asian and European populations, which may limit the applicability of the findings to other regions or healthcare set
The results of this meta-analysis suggest that serum albumin levels at presentation have significant impact on predicting mortality in acute cholangitis. Therefore, the serum albumin measurement at admission should be incorporated into the initial risk assessment of patients with acute cholangitis. Early identification of patients with hypoalbuminemia may prompt clinicians to consider more intensive monitoring, timely biliary drainage, and multidisciplinary management. Therefore, future studies need to emphasize on exploring not only the association between correction of hypoalbuminemia with the improvement in the clinical outcomes in patients with cholangitis; but should also compare the pro
| 1. | Lan Cheong Wah D, Christophi C, Muralidharan V. Acute cholangitis: current concepts. ANZ J Surg. 2017;87:554-559. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 44] [Article Influence: 4.9] [Reference Citation Analysis (1)] |
| 2. | Lee JG. Diagnosis and management of acute cholangitis. Nat Rev Gastroenterol Hepatol. 2009;6:533-541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 101] [Article Influence: 5.9] [Reference Citation Analysis (1)] |
| 3. | Florescu V, Pârvuleţu R, Ardelean M, Angelescu M, Angelescu GA, Enciu O, Iordache N. The Emergency Endoscopic Treatment in Acute Cholangitis. Chirurgia (Bucur). 2021;116:42-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 4. | Mohammad Alizadeh AH. Cholangitis: Diagnosis, Treatment and Prognosis. J Clin Transl Hepatol. 2017;5:404-413. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 22] [Article Influence: 2.4] [Reference Citation Analysis (2)] |
| 5. | Comoglu M, Acehan F, Sahiner ES, Kalkan C, Comoglu E, Yılmaz Y, Canlı T, Ates I. Clinical Features and Prognosis of Acute Cholangitis in Octogenarians: A Prospective Comparative Study. Medicina (Kaunas). 2024;60:1759. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 6. | Inan O, Sahiner ES, Ates I. Factors associated with clinical outcome in geriatric acute cholangitis patients. Eur Rev Med Pharmacol Sci. 2023;27:3313-3321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (2)] |
| 7. | Gravito-Soares E, Gravito-Soares M, Gomes D, Almeida N, Tomé L. Clinical applicability of Tokyo guidelines 2018/2013 in diagnosis and severity evaluation of acute cholangitis and determination of a new severity model. Scand J Gastroenterol. 2018;53:329-334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 18] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 8. | Murayama KM. Acute Cholangitis Management: Prevention of Organ Failure and Death. JAMA Surg. 2016;151:1045. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (1)] |
| 9. | Schneider J, Hapfelmeier A, Thöres S, Obermeier A, Schulz C, Pförringer D, Nennstiel S, Spinner C, Schmid RM, Algül H, Huber W, Weber A. Mortality Risk for Acute Cholangitis (MAC): a risk prediction model for in-hospital mortality in patients with acute cholangitis. BMC Gastroenterol. 2016;16:15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 42] [Article Influence: 4.2] [Reference Citation Analysis (1)] |
| 10. | Yıldız BD, Özden S, Saylam B, Martlı F, Tez M. Simplified scoring system for prediction of mortality in acute suppurative cholangitis. Kaohsiung J Med Sci. 2018;34:415-419. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 9] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 11. | Ismail H, Yaseen RT, Danish M, Tasneem AA, Hanif F, Hanif F, Jariko A, Laeeq SM, Majid Z, Luck NH. Role of "HinCh Score" as a Non-invasive Predictor of Post-endoscopic Retrograde Cholangiopancreatography Cholangitis. Euroasian J Hepatogastroenterol. 2022;12:19-23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (1)] |
| 12. | Jin X, Li J, Sun L, Zhang J, Gao Y, Li R, Ren J, Hou Y, Su D, Liu J, Wang X, Chen D, Wang G, Wiedermann CJ. Prognostic Value of Serum Albumin Level in Critically Ill Patients: Observational Data From Large Intensive Care Unit Databases. Front Nutr. 2022;9:770674. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 34] [Article Influence: 8.5] [Reference Citation Analysis (1)] |
| 13. | Yoshioka G, Tanaka A, Goriki Y, Node K. The role of albumin level in cardiovascular disease: a review of recent research advances. J Lab Precis Med. 2023;8:7. [DOI] [Full Text] |
| 14. | Minatoguchi S, Nomura A, Imaizumi T, Sasaki S, Ozeki T, Uchida D, Kawarazaki H, Sasai F, Tomita K, Shimizu H, Fujita Y. Low serum albumin as a risk factor for infection-related in-hospital death among hemodialysis patients hospitalized on suspicion of infectious disease: a Japanese multicenter retrospective cohort study. Ren Replace Ther. 2018;4:30. [RCA] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 15. | Eckart A, Struja T, Kutz A, Baumgartner A, Baumgartner T, Zurfluh S, Neeser O, Huber A, Stanga Z, Mueller B, Schuetz P. Relationship of Nutritional Status, Inflammation, and Serum Albumin Levels During Acute Illness: A Prospective Study. Am J Med. 2020;133:713-722.e7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 573] [Cited by in RCA: 476] [Article Influence: 79.3] [Reference Citation Analysis (1)] |
| 16. | Artigas A, Wernerman J, Arroyo V, Vincent JL, Levy M. Role of albumin in diseases associated with severe systemic inflammation: Pathophysiologic and clinical evidence in sepsis and in decompensated cirrhosis. J Crit Care. 2016;33:62-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 200] [Cited by in RCA: 169] [Article Influence: 16.9] [Reference Citation Analysis (2)] |
| 17. | Oster HS, Dolev Y, Kehat O, Weis-Meilik A, Mittelman M. Serum Hypoalbuminemia Is a Long-Term Prognostic Marker in Medical Hospitalized Patients, Irrespective of the Underlying Disease. J Clin Med. 2022;11:1207. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 32] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 18. | Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JP, Clarke M, Devereaux PJ, Kleijnen J, Moher D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ. 2009;339:b2700. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 19. | Acehan F, Çamlı H, Kalkan C, Tez M, Altiparmak E, Ates I. Characteristics and clinical outcomes of acute cholangitis in older patients. Eur Geriatr Med. 2023;14:263-273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 20. | Sun G, Han L, Yang YS, Linghu EQ, Li W, Cai FC, Kong JY, Wang XD, Meng JY, Du H, Wang HB, Huang QY, Hyder Q, Zhang XL. Verification of the Tokyo guidelines for acute cholangitis secondary to benign and malignant biliary obstruction: experience from a Chinese tertiary hospital. Hepatobiliary Pancreat Dis Int. 2013;12:400-407. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 21. | Hunyady P, Streller L, Rüther DF, Groba SR, Bettinger D, Fitting D, Hamesch K, Marquardt JU, Mücke VT, Finkelmeier F, Sekandarzad A, Wengenmayer T, Bounidane A, Weiss F, Peiffer KH, Schlevogt B, Zeuzem S, Waidmann O, Hollenbach M, Kirstein MM, Kluwe J, Kütting F, Mücke MM. Secondary Sclerosing Cholangitis Following Coronavirus Disease 2019 (COVID-19): A Multicenter Retrospective Study. Clin Infect Dis. 2023;76:e179-e187. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 28] [Article Influence: 9.3] [Reference Citation Analysis (2)] |
| 22. | Mohan R, Goh SWL, Tan G, Junnarkar SP, Huey C, Shelat VG. Validation of TG07 and TG13/TG18 criteria for acute cholangitis and predictors of in-hospital mortality in patients over 80 years old. Clin Exp Hepatol. 2021;7:396-405. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 23. | Pan LN, Pan SA, Hong GL, Chen KW. A New Nomogram for Predicting 30-Day In-Hospital Mortality Rate of Acute Cholangitis Patients in the Intensive Care Unit. Emerg Med Int. 2023;2023:9961438. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 24. | Tsuyuguchi T, Sugiyama H, Sakai Y, Nishikawa T, Yokosuka O, Mayumi T, Kiriyama S, Yokoe M, Takada T. Prognostic factors of acute cholangitis in cases managed using the Tokyo Guidelines. J Hepatobiliary Pancreat Sci. 2012;19:557-565. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 31] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 25. | Jellinge ME, Henriksen DP, Hallas P, Brabrand M. Hypoalbuminemia is a strong predictor of 30-day all-cause mortality in acutely admitted medical patients: a prospective, observational, cohort study. PLoS One. 2014;9:e105983. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 134] [Cited by in RCA: 121] [Article Influence: 10.1] [Reference Citation Analysis (1)] |
| 26. | Perel N, Taha L, Farkash R, Steinmetz Y, Shaheen F, Levi N, Dadon Z, Karameh H, Karmi M, Maller T, Hamyil K, Turyan A, Manatzra M, Bayya F, Glikson M, Asher E. Level of Hypoalbuminemia as a Prognostic Factor in Patients admitted to a Tertiary Care Intensive Coronary Care Unit. Cardiol Cardiovasc Med. 2022;6:536-541. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 27. | Kumar HG, Kanakaraju K, Manikandan VAC, Patel V, Pranay C. The Relationship Between Serum Albumin Levels and Sepsis in Patients Admitted to a Tertiary Care Center in India. Cureus. 2024;16:e59424. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 28. | Sheinenzon A, Shehadeh M, Michelis R, Shaoul E, Ronen O. Serum albumin levels and inflammation. Int J Biol Macromol. 2021;184:857-862. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 337] [Cited by in RCA: 259] [Article Influence: 51.8] [Reference Citation Analysis (1)] |
| 29. | Anderson CF, Wochos DN. The utility of serum albumin values in the nutritional assessment of hospitalized patients. Mayo Clin Proc. 1982;57:181-184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 30. | Nazha B, Moussaly E, Zaarour M, Weerasinghe C, Azab B. Hypoalbuminemia in colorectal cancer prognosis: Nutritional marker or inflammatory surrogate? World J Gastrointest Surg. 2015;7:370-377. [PubMed] [DOI] [Full Text] |
| 31. | Han T, Cheng T, Liao Y, Tang S, Liu B, He Y, Gu Z, Lei C, Cao Y, Cao Y. Analysis of the Value of the Blood Urea Nitrogen to Albumin Ratio as a Predictor of Mortality in Patients with Sepsis. J Inflamm Res. 2022;15:1227-1235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 34] [Article Influence: 8.5] [Reference Citation Analysis (1)] |
| 32. | Cao Y, Su Y, Guo C, He L, Ding N. Albumin Level is Associated with Short-Term and Long-Term Outcomes in Sepsis Patients Admitted in the ICU: A Large Public Database Retrospective Research. Clin Epidemiol. 2023;15:263-273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 41] [Article Influence: 13.7] [Reference Citation Analysis (2)] |
| 33. | Soeters PB, Wolfe RR, Shenkin A. Hypoalbuminemia: Pathogenesis and Clinical Significance. JPEN J Parenter Enteral Nutr. 2019;43:181-193. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1096] [Cited by in RCA: 913] [Article Influence: 130.4] [Reference Citation Analysis (1)] |
| 34. | Daud M, Ullah F, Uzair M, Siddiq A, Siddiq U, Riaz FB, Ibrar M, Khan AH. Malnutrition and Its Influence on Sepsis Outcomes in Elderly Patients. Cureus. 2024;16:e63433. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 35. | Li D, Sun J, Qi C, Fu X, Gao F. Predicting severity of inpatient acute cholangitis: combined neutrophil-to-lymphocyte ratio and prognostic nutritional index. BMC Gastroenterol. 2024;24:468. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 36. | Nicholson JP, Wolmarans MR, Park GR. The role of albumin in critical illness. Br J Anaesth. 2000;85:599-610. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 727] [Cited by in RCA: 586] [Article Influence: 22.5] [Reference Citation Analysis (1)] |
| 37. | Sun L, Yin H, Liu M, Xu G, Zhou X, Ge P, Yang H, Mao Y. Impaired albumin function: a novel potential indicator for liver function damage? Ann Med. 2019;51:333-344. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 134] [Cited by in RCA: 117] [Article Influence: 16.7] [Reference Citation Analysis (1)] |
| 38. | Shahi S, Paneru H, Ojha R, Karn R, Rajbhandari R, Gajurel BP. SOFA and APACHE II scoring systems for predicting outcome of neurological patients admitted in a tertiary hospital intensive care unit. Ann Med Surg (Lond). 2024;86:1895-1900. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (1)] |