Published online Mar 28, 2026. doi: 10.4329/wjr.v18.i3.119025
Revised: February 2, 2026
Accepted: March 9, 2026
Published online: March 28, 2026
Processing time: 68 Days and 22 Hours
Incidental findings are increasingly common in radiology yet reporting and follow-up remain inconsistent. We introduce Incidental-Reporting and Data System (I-RADS) as a classification system across imaging modalities and anato
Core Tip: Although several guidelines on managing specific incidental findings (IFs) already exist and largely enable clinical practice, standardized recommendations accommodated within written radiology reports are lacking. We propose the use of the Incidental Reporting and Data System (I-RADS) to promote clarifying the necessity of next steps and management of IFs within radiology reports, similar to other “Reporting and Data System” systems. I-RADS attempts to improve communication among phy
- Citation: Arkoudis NA, Moschovaki-Zeiger O, Koutserimpas C, Lama N, Velonakis G, Filippiadis D, Spiliopoulos S, Kelekis N. Proposing Incidental-Reporting and Data System: A classification system for incidental findings in radiology. World J Radiol 2026; 18(3): 119025
- URL: https://www.wjgnet.com/1949-8470/full/v18/i3/119025.htm
- DOI: https://dx.doi.org/10.4329/wjr.v18.i3.119025
In radiology, an incidental finding (IF) is typically defined as an imaging observation discovered unexpectedly while performing a study [i.e., computed tomography (CT), magnetic resonance imaging (MRI), etc.] for an unrelated clinical indication[1]. Given the continuous expansion of clinical indications for imaging and the ongoing de
Furthermore, substantial variation is noted in how IFs are being handled, from no reporting strategy to inconsistent recommendations[6]. This variability contributes to miscommunication between radiologists and referring physicians, which remains a pressing issue[7]. The lack of standardization for IFs results in subjective judgments and varying impressions in reports, as well as the suggested recommendations[8]. This situation also creates challenges in follow-up adherence and data collection for research, and contributes to overall variability in patient care.
The American College of Radiology (ACR) has released a number of significant white papers and guidelines via its IFs committee, addressing several topics including IFs on thoracic and abdominal CT[9,10], and incidental liver lesions, renal masses, adrenal masses and pancreatic cysts (to name a few)[11-14]. These documents offer valuable evidence-based information but are specialized to particular organs and scenarios. Radiologists may frequently have to consult various different guidelines, each with distinct language and thresholds, a task that can be demanding and time-consuming in routine practice. Beyond organ-specific guidelines, structured attempts to categorize IFs have also been proposed in defined clinical contexts. For example, a trauma-specific classification termed IF-Reporting and Data System (RADS) has been presented in abstract form to stratify IFs detected on whole-body CT in polytrauma patients[15]. While this work highlights the feasibility and value of structured incidental-finding categorization, it remains limited to a single modality and clinical scenario and does not address the need for a universal, modality-agnostic framework applicable across routine radiology practice.
We propose Incidental-RADS (I-RADS), a modality-agnostic, anatomy-agnostic structured reporting framework for classifying and managing IFs across imaging modalities and body regions. In this manuscript, I-RADS refers specifically to the Incidental-Reporting and Data System and is used exclusively in the context of structured reporting and management of IFs, distinct from other uses of the acronym “I-RADS”. Modeled after well-known successful systems like Liver Imaging RADS[16], Prostate Imaging RADS[17], and Breast Imaging RADS[18], I-RADS can be used by the reporting radiologist to assign each IF a category corresponding to the likelihood of clinical significance and the recommended course of action. In the context of I-RADS, the term clinical significance refers to the likelihood that an IF warrants further evaluation or intervention, or may have a potential impact on patient outcomes.
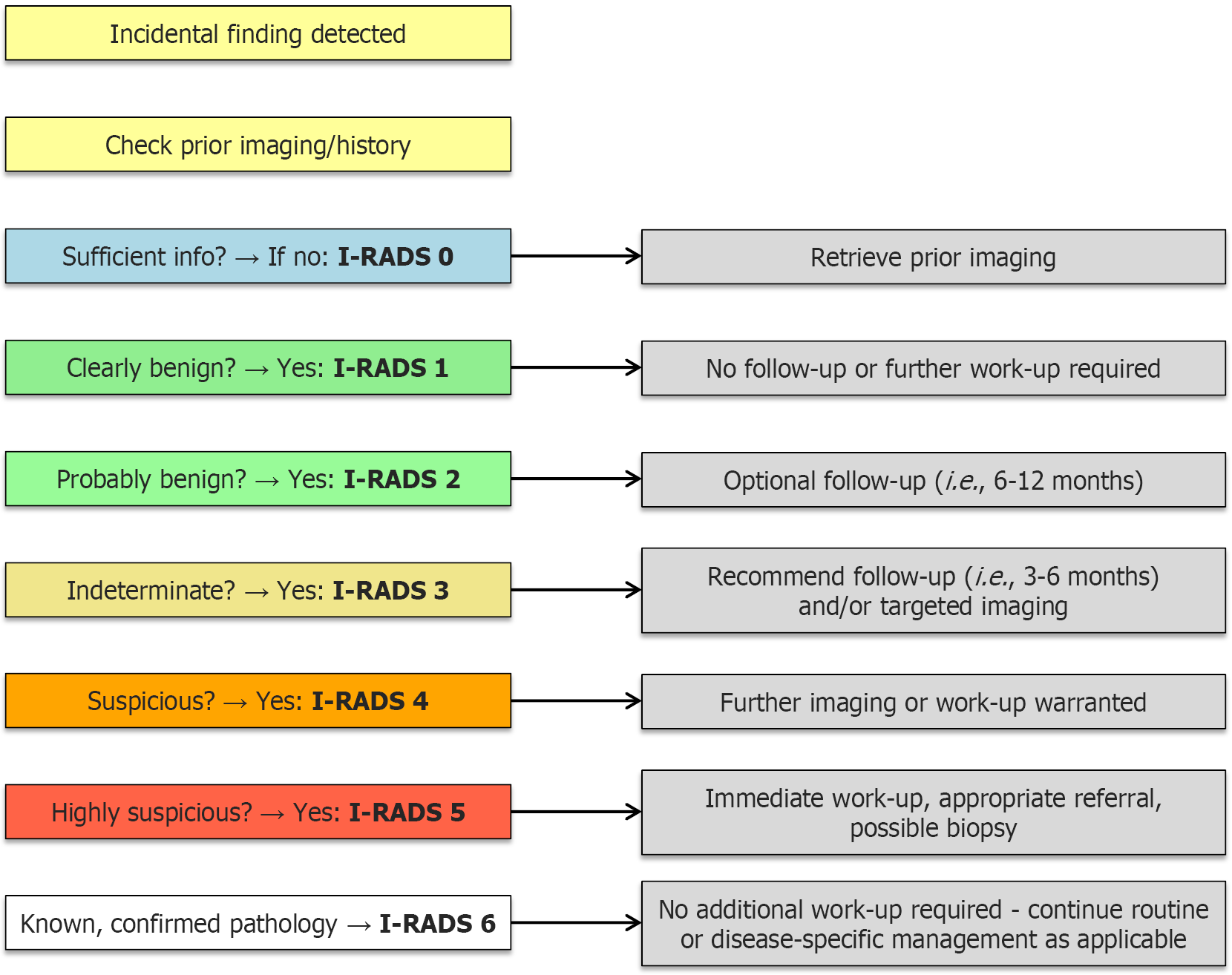
However, unlike organ-specific systems, I-RADS is deliberately designed as a universal framework applicable to any IF. Table 1 provides a description of each category of the proposed I-RADS scoring, as well as the currently proposed corresponding recommendations. A simplified, linear I-RADS flowchart showing a practical step-by-step IF classification decision process is also illustrated in Figure 1. In addition, to further illustrate how I-RADS categories could be applied in practice, we provide Table 2, listing a representative example for each category (0-6), thereby contextualizing the classification system with some real-world everyday radiological scenarios. For I-RADS 0, if prior imaging cannot be retrieved, escalation to targeted imaging or reassignment to a higher category may be appropriate at the discretion of the radiologist. For I-RADS 2, the term “low-risk patient” is explicitly defined as a context-dependent assessment incorporating clinical history and known risk factors rather than fixed thresholds. Findings categorized as I-RADS 6 should be supported by reliable prior documentation, such as histopathologic confirmation, prior imaging demonstrating stability or progression consistent with a known diagnosis, or a clearly documented diagnosis in the medical record. Standardized reporting language (e.g., “known per prior imaging”, “known per pathology”, or “known per chart history”) is recommended to clearly convey the basis for classification. Findings that are suspected but not yet confirmed should not be classified as I-RADS 6 and should instead be categorized according to their level of concern (e.g., I-RADS 4 or 5), with appropriate recommendations for further evaluation.
| I-RADS category | Description | Recommended action |
| I-RADS 0 | Incomplete/insufficient information | Retrieve prior imaging (if not feasible, consider escalation to targeted imaging or reassignment to a higher category) |
| I-RADS 1 | Incidental benign finding - no clinical significance | No follow-up or further work-up required |
| I-RADS 2 | Probably benign - minimal concern | Optional follow-up (i.e., 6-12 months) |
| I-RADS 3 | Indeterminate - moderate concern | Recommend follow-up (i.e., 3-6 months) and/or targeted imaging |
| I-RADS 4 | Suspicious - likely pathology | Further imaging or work-up warranted |
| I-RADS 5 | Highly suspicious - urgent concern | Immediate work-up, appropriate referral, possible biopsy |
| I-RADS 6 | Known/confirmed pathology | No additional work-up required - continue routine or disease-specific management as applicable |
| I-RADS category | Indicative example |
| I-RADS 0 | A 1.5 cm hypodense liver lesion detected on a non-contrast abdominal CT performed for suspected renal colic; a multiphase abdominal CT has been performed previously on the patient but is not available to the reporting radiologist |
| I-RADS 1 | A simple renal cyst measuring 1.5 cm detected on abdominal CT performed due to suspected acute appendicitis |
| I-RADS 2 | A small (3 mm) incidental pulmonary nodule detected on a cardiac CT, in a low-risk, nonsmoking patient |
| I-RADS 3 | A 2 cm indeterminate adrenal lesion discovered incidentally on trauma CT |
| I-RADS 4 | A 2.5 cm spiculated pulmonary nodule incidentally identified on a CT angiogram of the chest performed for suspected pulmonary embolism; morphology and margins highly concerning for malignancy |
| I-RADS 5 | A 3.5 cm irregular mass in the pancreatic head with upstream biliary ductal dilatation, found incidentally on an abdominal MRI performed to evaluate hepatic steatosis; findings concerning for pancreatic carcinoma |
| I-RADS 6 | Patient with biopsy-proven metastatic colon cancer and multiple known hepatic metastases, incidentally detected in the lower slices (upper abdominal slices) of a chest CT angiogram performed to exclude pulmonary embolism |
In general, reassignment may occur if additional clinical or imaging data become available. When multiple IFs are identified on a single examination, each finding may be assigned its own I-RADS category. For reporting purposes, higher-grade findings, or those deemed most clinically relevant, may be emphasized in the Impression section at the discretion of the reporting radiologist.
The I-RADS approach seeks to promote structured reporting of recommendations for IFs, as such reporting has demonstrated effectiveness in improving communication between radiologists and referring physicians[19]. Although guidelines on several findings may already exist, standardized recommendations and wording in radiology reports is lacking and could potentially be addressed by the implementation of the presented system. After all several other now refined-RADS classification systems, aim to standardize recommendations occurring from radiology reports instead of allowing wording that could otherwise be misinterpreted.
Therefore, the objective is to provide consistency in terminology and decision-making for IFs while ensuring a transparent and reproducible connection between IFs and recommendations. Additionally, I-RADS seeks to enable data collection and ultimately establish an infrastructure for AI algorithms to learn from clearly labeled standardized datasets enabling machine learning, registries and big-data research in ways current fragmented guidelines cannot. In addition, I-RADS can be incorporated into structured reporting platforms, streamlining diagnostic workflow.
I-RADS implementation may have additional significant clinical impacts. By minimizing uncertainties and use of vague language in radiological reports, it could assist referring physicians in prioritizing follow-up and enhance patient care. Through consistent, understandable language, it can help prevent unnecessary testing in IFs that are obviously benign while improving compliance with follow-up advice or further recommended actions.
I-RADS provides a systematic data framework for research purposes as well. By categorizing each IF into a consistent classification, multicenter retrospective and prospective studies can be conducted without the need for extensive data harmonization. This could simplify large-scale studies and support cost-effectiveness evaluations of follow-up recom
Besides the above, I-RADS can serve an educational purpose, giving trainees a straightforward framework for IFs, guiding them to make consistent, structured decisions and unifying how trainees, radiologists, and referring clinicians discuss IFs. It also strengthens quality assurance, allowing institutions to measure performance more reliably and to use structured data for auditing and improving reporting practices.
From an implementation standpoint, I-RADS is intended to be compatible with structured reporting workflows and existing reporting systems. Practical adoption will depend on local institutional resources, infrastructure, and clinical practice patterns. Clear and patient-centered communication of IFs and recommended follow-up is also important to support understanding and compliance. Detailed technical integration strategies and patient education frameworks are beyond the scope of this initial conceptual proposal and represent areas for future work.
Implementation of I-RADS may be associated with potential downsides, including increased follow-up imaging, patient anxiety, radiation exposure, and resource utilization. I-RADS is not intended to promote uniform escalation of IFs, and over-surveillance may be mitigated through risk stratification, appropriate threshold setting, alignment with organ-specific guidelines, and shared decision-making. Existing RADS-based systems for specific incidental lesions (e.g., bone-RADS)[20] illustrate how structured approaches can support targeted management rather than a one-size-fits-all strategy.
I-RADS is not intended to replace existing IF guidance (such as ACR recommendations) but to complement and combine it into a single, simplified cross-sectional system. For example, an I-RADS 3 adrenal lesion could route to the ACR adrenal white paper for more detailed management[14]. By offering a consistent all-encompassing framework, I-RADS har
| Feature | ACR incidental findings guidelines | I-RADS proposal |
| Scope | Organ- and condition-specific | Universal, modality-agnostic, and pan-anatomical |
| Format | White papers and detailed flowcharts, often requiring consultation of multiple documents | Single classification system with 6 categories applicable to any incidental finding |
| Goal | Provide evidence-based management recommendations for specific incidental findings | Provide standardized language and structured framework to classify all incidental findings |
| Strengths | Evidence-based, specialty consensus; addresses details of particular lesions | Simple, unified terminology, allowing easier communication, reporting consistency, and research data harmonization |
| Limitations | Spread across multiple documents, not integrated into a universal framework | Broad by design, requires integration with organ-specific guidelines for detailed management |
| Complementarity | Offers depth for specific lesions | Provides an all-embracing framework into which ACR guidelines can be fused |
The proposed I-RADS system is a conceptual framework intended to provide a unified and simplified approach to the classification and communication of IFs across imaging modalities and anatomical regions. At this stage, I-RADS has not yet been clinically validated, and no formal interobserver agreement or reproducibility testing has been performed. The current categories and recommended follow-up intervals are therefore intentionally broad and illustrative, rather than evidence-defining thresholds. For example, the suggested time windows for optional follow-up in I-RADS 2 (6-12 months) and recommended follow-up in I-RADS 3 (3-6 months) are designed to reflect relative levels of clinical concern and to promote consistency in reporting language, rather than to replace or supersede organ-specific, evidence-based guidelines.
The clinical impact of I-RADS implementation (including effects on patient outcomes, follow-up adherence, rates of missed or delayed diagnoses, downstream imaging utilization, and resource allocation) has not yet been evaluated. These aspects represent essential areas for future investigation. Prospective validation studies, interobserver reliability assessments, and multicenter research initiatives will be required to assess the reproducibility, clinical utility, and real-world consequences of structured IF classification using I-RADS.
Future evaluation of I-RADS will require staged research efforts, including multicenter case libraries or vignette-based studies across imaging modalities and anatomical regions to assess grading reliability, followed by prospective studies evaluating the impact of structured IF reporting on follow-up completion, clinical outcomes, referral patterns, and resource utilization. Detailed study design elements, such as specific endpoints, sample size calculations, and ad
Future development of I-RADS should follow an iterative process similar to other established RADS systems, incorporating expert consensus, empirical evidence, and feedback from routine clinical use. Subsequent refinements may include more detailed category definitions, incorporation of additional common and less common IFs, and closer integration with organ-specific guidelines as supporting data emerge. In this context, I-RADS is intended as a reproducible starting point that facilitates standardization and research, while remaining adaptable to ongoing advances in imaging technology, clinical practice, and evidence generation.
To conclude, although guidelines on several IFs may already exist, consistent recommendations integrated within radiology reports are lacking. I-RADS aims to improve communication among physicians, standardize recommendations, and promote research. Along with preexisting and ongoing guidelines for organ-specific IFs, adoption of I-RADS could harmonize management, improve patient outcomes, and support multicenter studies. We welcome colleagues to engage in this initiative and invite feedback and recommendations from the radiology community to refine I-RADS into a functional and broadly embraced system.
| 1. | Lumbreras B, Donat L, Hernández-Aguado I. Incidental findings in imaging diagnostic tests: a systematic review. Br J Radiol. 2010;83:276-289. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 284] [Cited by in RCA: 279] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 2. | O'Sullivan JW, Muntinga T, Grigg S, Ioannidis JPA. Prevalence and outcomes of incidental imaging findings: umbrella review. BMJ. 2018;361:k2387. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 234] [Cited by in RCA: 221] [Article Influence: 27.6] [Reference Citation Analysis (1)] |
| 3. | Evans CS, Arthur R, Kane M, Omofoye F, Chung AE, Moreton E, Moore C. Incidental Radiology Findings on Computed Tomography Studies in Emergency Department Patients: A Systematic Review and Meta-Analysis. Ann Emerg Med. 2022;80:243-256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 25] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 4. | Safari S, Dizaji SR, Yousefifard M, Taheri MS, Sharifi A. Prevalence and clinical significance of incidental findings in chest and abdominopelvic CT scans of trauma patients; A cross-sectional study. Am J Emerg Med. 2024;82:117-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 5. | Rowley PA, Paukner MJ, Eisenmenger LB, Field AS, Davidson RJ, Johnson SC, Asthana S, Chin NA, Prabhakaran V, Bendlin BB, Postle BR, Goldsmith HH, Carlsson CM, Brooks MA, Kalin NH, Williams LE, Rowley HA. Incidental Findings from 16,400 Brain MRI Examinations of Research Volunteers. AJNR Am J Neuroradiol. 2023;44:417-423. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 16] [Reference Citation Analysis (0)] |
| 6. | Booth TC, Waldman AD, Wardlaw JM, Taylor SA, Jackson A. Management of incidental findings during imaging research in "healthy" volunteers: current UK practice. Br J Radiol. 2012;85:11-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 34] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 7. | Arkoudis NA, Karofylakis E, Moschovaki-Zeiger O, Prountzos S, Efthymiou E, Samonis G, Koutserimpas C. Interdepartmental miscommunication regarding radiology: Addressing chronic challenges and exploring solutions. World J Radiol. 2024;16:109-114. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 8. | Cochon LR, Kapoor N, Carrodeguas E, Ip IK, Lacson R, Boland G, Khorasani R. Variation in Follow-up Imaging Recommendations in Radiology Reports: Patient, Modality, and Radiologist Predictors. Radiology. 2019;291:700-707. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 46] [Article Influence: 6.6] [Reference Citation Analysis (1)] |
| 9. | Berland LL, Silverman SG, Gore RM, Mayo-Smith WW, Megibow AJ, Yee J, Brink JA, Baker ME, Federle MP, Foley WD, Francis IR, Herts BR, Israel GM, Krinsky G, Platt JF, Shuman WP, Taylor AJ. Managing incidental findings on abdominal CT: white paper of the ACR incidental findings committee. J Am Coll Radiol. 2010;7:754-773. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 726] [Cited by in RCA: 583] [Article Influence: 36.4] [Reference Citation Analysis (1)] |
| 10. | Munden RF, Carter BW, Chiles C, MacMahon H, Black WC, Ko JP, McAdams HP, Rossi SE, Leung AN, Boiselle PM, Kent MS, Brown K, Dyer DS, Hartman TE, Goodman EM, Naidich DP, Kazerooni EA, Berland LL, Pandharipande PV. Managing Incidental Findings on Thoracic CT: Mediastinal and Cardiovascular Findings. A White Paper of the ACR Incidental Findings Committee. J Am Coll Radiol. 2018;15:1087-1096. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 118] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 11. | Gore RM, Pickhardt PJ, Mortele KJ, Fishman EK, Horowitz JM, Fimmel CJ, Talamonti MS, Berland LL, Pandharipande PV. Management of Incidental Liver Lesions on CT: A White Paper of the ACR Incidental Findings Committee. J Am Coll Radiol. 2017;14:1429-1437. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 99] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 12. | Herts BR, Silverman SG, Hindman NM, Uzzo RG, Hartman RP, Israel GM, Baumgarten DA, Berland LL, Pandharipande PV. Management of the Incidental Renal Mass on CT: A White Paper of the ACR Incidental Findings Committee. J Am Coll Radiol. 2018;15:264-273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 213] [Cited by in RCA: 189] [Article Influence: 23.6] [Reference Citation Analysis (1)] |
| 13. | Megibow AJ, Baker ME, Morgan DE, Kamel IR, Sahani DV, Newman E, Brugge WR, Berland LL, Pandharipande PV. Management of Incidental Pancreatic Cysts: A White Paper of the ACR Incidental Findings Committee. J Am Coll Radiol. 2017;14:911-923. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 278] [Cited by in RCA: 245] [Article Influence: 27.2] [Reference Citation Analysis (0)] |
| 14. | Mayo-Smith WW, Song JH, Boland GL, Francis IR, Israel GM, Mazzaglia PJ, Berland LL, Pandharipande PV. Management of Incidental Adrenal Masses: A White Paper of the ACR Incidental Findings Committee. J Am Coll Radiol. 2017;14:1038-1044. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 322] [Cited by in RCA: 254] [Article Influence: 28.2] [Reference Citation Analysis (1)] |
| 15. | Vogele D, Hensinger T, Efinger K, Beer M, Kildal D. IF-RADS classification of incidental findings on whole-body computed tomography. In: European Congress of Radiology (ECR); 2023 Mar; Vienna. Vienna: European Society of Radiology, 2023. |
| 16. | Chernyak V, Fowler KJ, Kamaya A, Kielar AZ, Elsayes KM, Bashir MR, Kono Y, Do RK, Mitchell DG, Singal AG, Tang A, Sirlin CB. Liver Imaging Reporting and Data System (LI-RADS) Version 2018: Imaging of Hepatocellular Carcinoma in At-Risk Patients. Radiology. 2018;289:816-830. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 990] [Cited by in RCA: 903] [Article Influence: 112.9] [Reference Citation Analysis (4)] |
| 17. | Weinreb JC, Barentsz JO, Choyke PL, Cornud F, Haider MA, Macura KJ, Margolis D, Schnall MD, Shtern F, Tempany CM, Thoeny HC, Verma S. PI-RADS Prostate Imaging - Reporting and Data System: 2015, Version 2. Eur Urol. 2016;69:16-40. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2544] [Cited by in RCA: 2343] [Article Influence: 234.3] [Reference Citation Analysis (4)] |
| 18. | Magny SJ, Shikhman R, Keppke AL. Breast Imaging Reporting and Data System. 2023 Aug 28. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2026 Jan. [PubMed] |
| 19. | Schwartz LH, Panicek DM, Berk AR, Li Y, Hricak H. Improving communication of diagnostic radiology findings through structured reporting. Radiology. 2011;260:174-181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 305] [Cited by in RCA: 347] [Article Influence: 23.1] [Reference Citation Analysis (0)] |
| 20. | Caracciolo JT, Ali S, Chang CY, Degnan AJ, Flemming DJ, Henderson ER, Kransdorf MJ, Letson GD, Madewell JE, Murphey MD. Bone Tumor Risk Stratification and Management System: A Consensus Guideline from the ACR Bone Reporting and Data System Committee. J Am Coll Radiol. 2023;20:1044-1058. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 8.3] [Reference Citation Analysis (0)] |