Published online Mar 28, 2026. doi: 10.4329/wjr.v18.i3.118143
Revised: January 25, 2026
Accepted: March 5, 2026
Published online: March 28, 2026
Processing time: 91 Days and 23 Hours
Mullerian anomalies are frequently associated with endometriosis, particularly in the presence of genital tract obstruction; however, the anatomical distribution and determinants of disease in this population remain incompletely characterized. Most existing data are derived from surgical series, with limited use of standar
To characterize the prevalence and distribution of endometriosis in patients with Mullerian anomalies using pelvic magnetic resonance imaging (MRI) and the Enzian classification.
This retrospective observational study included pelvic MRI examinations per
Seventy-five patients with Mullerian anomalies were included. MRI findings consistent with endometriosis were identified in 22 patients (29.3%). Ovarian endometriomas were the most common manifestation, present in 16 patients (21.3%), while tubo-ovarian involvement corresponding to Enzian T2-T3 disease was seen in 11 patients (14.7%). Endometriosis was more frequently observed in complex uterine configurations, particularly bicorporeal septate uterus (U3c), aplastic uterus with a functional rudimentary cavity (U5a), and in patients classified as U0 with associated cervicovaginal anomalies. Vaginal aplasia (V4) was associated with a significantly lower pre
Endometriosis in patients with Mullerian anomalies demonstrates variable distribution on MRI, with higher prevalence in complex and obstructive uterine configurations. MRI combined with standardized Society of Human Reproduction and Embryology/European Society for Gynaecological Endoscopy and Enzian classifications provides a structured, anatomy-driven approach for evaluating disease patterns in this heterogeneous population.
Core Tip: This study uses magnetic resonance imaging with structured interpretation to characterize endometriosis in patients with Mullerian anomalies, an association traditionally explored through surgical series. By integrating the European Society of Human Reproduction and Embryology/European Society for Gynaecological Endoscopy classification of Mullerian anomalies with Enzian mapping of endometriosis, the study demonstrates that disease prevalence and distribution vary with uterine configuration and are strongly influenced by genital tract obstruction. The findings highlight the value of an anatomy-driven, magnetic resonance imaging-based approach for evaluating this complex and heterogeneous patient population.
- Citation: Kumar I, Kumari S, Ojha R, Kumari R, Kushwaha K, Singh PK, Verma A. Characterization of endometriosis in patients with Mullerian anomalies using the Enzian classification: A retrospective cohort study. World J Radiol 2026; 18(3): 118143
- URL: https://www.wjgnet.com/1949-8470/full/v18/i3/118143.htm
- DOI: https://dx.doi.org/10.4329/wjr.v18.i3.118143
Mullerian anomalies are congenital developmental disorders of the female genital tract resulting from abnormal formation, fusion, or resorption of the Mullerian ducts, producing a broad spectrum of uterine, cervical, and vaginal abnormalities. Accurate classification is fundamental for imaging interpretation, clinical communication, and management decisions. The American Society for Reproductive Medicine classification (formerly American Fertility Society classification) remains widely used and categorizes anomalies primarily on uterine morphology, offering simplicity and clinical familiarity but limited characterization of cervical and vaginal involvement. To address these limitations, the European Society of Human Reproduction and Embryology (ESHRE)-European Society for Gynaecological Endoscopy (ESGE) classification, proposed by the ESHRE and the ESGE, introduced a modular, anatomy-based system that independently classifies uterine, cervical, and vaginal anomalies, making it particularly suited to magnetic resonance imaging (MRI)-based anatomical assessment. Nevertheless, concerns have been raised regarding certain morphological criteria of the ESHRE-ESGE system, especially the reliance on uterine wall thickness for defining septate uterus, which may lead to overdiagnosis and influence surgical decision-making[1-4].
Endometriosis, defined by the presence of endometrium-like tissue outside the uterine cavity, has long been associated with Mullerian abnormalities[5]. Retrograde menstruation, particularly in obstructive anomalies, is a well-recognized mechanism for ectopic implantation However, its occurrence in patients without obstruction, including those with Mullerian agenesis, supports alternative mechanisms such as coelomic metaplasia and persistence of embryologic Mullerian remnants[6-11].
Accurate and standardized characterization of endometriosis in patients with Mullerian anomalies is essential for effective clinic radiologic correlation and management. MRI is central in this setting, as it provides comprehensive evaluation of complex congenital pelvic anatomy and allows accurate depiction of ovarian, peritoneal, and deep infiltrating endometriosis. Several classification systems exist for endometriosis. The revised American Society for Repro
The aim of this study was therefore to characterize the anatomical distribution of endometriosis in patients with a spectrum of Mullerian anomalies using pelvic MRI, and to assess its relationship with anomaly type and the presence or absence of obstruction, using standardized classification systems.
This retrospective observational study was conducted at a tertiary care university based hospital and included pelvic MRI examinations performed over a period of 15 months. Patient selection was carried out by reviewing stored MRI reports maintained in departmental archives during the study period. All patients whose MRI reports described or suggested the presence of a Mullerian anomaly were identified and included in the study. The corresponding MRI image datasets of these patients, stored in the departmental archives, were retrieved and re-evaluated. Images were reviewed for the presence and extent of endometriosis. Institutional ethics committee approval was obtained prior to initiation of the study. Owing to the retrospective design and use of existing anonymized imaging data, informed consent was waived in accordance with institutional ethical guidelines.
Pelvic MRI was performed on a 3.0-Tesla Philips Ingenia MRI system (Philips Healthcare, Best, The Netherlands) using a phased-array pelvic coil with the patient in the supine position. A standardized protocol included high-resolution T2-weighted images in sagittal, axial oblique (perpendicular to the uterine axis), and coronal planes for assessment of uterine morphology and pelvic compartments. Axial T1-weighted images, with and without fat suppression, were acquired for detection of hemorrhagic lesions and endometriomas. Diffusion-weighted imaging (b values 0, 400, and 800-1000 seconds/mm2) with corresponding apparent diffusion coefficient maps was obtained as an adjunct for lesion detection. Typical slice thickness was 3-4 mm with a field of view of 22-26 cm. Intravenous contrast was not routinely administered. All MRI examinations were reviewed in consensus by two radiologists with experience of 12 years and 24 years of experience respectively.
Uterine anomalies were classified using the ESHRE-ESGE classification system. Class U0 (normal uterus) included uteri with a straight or mildly curved interstitial line and an internal fundal indentation ≤ 50% of the uterine wall thickness. In this study, arcuate uteri, which present with a broad-based, smooth internal fundal indentation of < 10 mm, were considered U0. Class U1 (dysmorphic uterus) comprised uteri with a normal external outline but abnormal cavity shape, excluding septa. Subtypes included U1a (T-shaped uterus) with thickened lateral walls and a corpus-to-cervix ratio of approximately 2:1, U1b (infantile uterus) with a narrow cavity and inverse corpus-to-cervix ratio, and U1c (other minor dysmorphic forms) with inner fundal indentation < 50% of uterine wall thickness. Class U2 (septate uterus) included cases with normal external uterine contour and an internal fundal indentation > 50% of uterine wall thickness due to abnormal septal resorption. U2a represented partial septate uterus (above the internal os), and U2b complete septate uterus, with or without associated cervical or vaginal septa. Class U3 (bicorporeal uterus) represented fusion defects characterized by an external fundal indentation > 50% of uterine wall thickness. This included U3a (partial bicorporeal), U3b (complete bicorporeal), and U3c (bicorporeal septate uterus), the latter showing combined fusion and absorption defects. Class U4 (hemi-uterus) described unilateral uterine development with a functional hemi-cavity, subdivided into U4a (with a functional rudimentary horn) and U4b (without a functional contralateral cavity). Class U5 (aplastic uterus) included uterine aplasia or severe hypoplasia, subdivided into U5a (with a rudimentary functional cavity) and U5b (without any functional cavity), commonly associated with vaginal aplasia or MRKH syndrome. Class U6 was reserved for rare, complex, or unclassifiable anomalies. Accessory cavitated uterine masses, seen as isolated uterine mass with a functional endometrial cavity but hypoplastic or dysplastic surrounding myometrium, were classified as U6.
Co-existing cervical anomalies were recorded as C0 (normal), C1 (septate), C2 (double cervix), C3 (unilateral cervical aplasia), or C4 (cervical aplasia/severe dysgenesis). Co-existing vaginal anomalies were classified as V0 (normal), V1 (longitudinal non-obstructing septum), V2 (longitudinal obstructing septum), V3 (transverse septum/imperforate hymen), or V4 (vaginal aplasia).
The presence of genital tract outflow obstruction was specifically assessed on MRI. Obstruction was considered present when there was imaging evidence of hemorrhagic or fluid collections proximal to an anatomic narrowing or discontinuity within the genital tract. MRI was evaluated for hematocolpos, hematocervix, and hematometra, either in isolation or in combination. Hemorrhagic collections were identified based on characteristic signal features, including T1-weighted hyperintensity with variable T2-weighted signal intensity. The level of obstruction was determined by correlating the site of hemorrhagic collection with associated cervical or vaginal anomalies, such as obstructing vaginal septum, cervical aplasia, or a non-communicating rudimentary uterine horn.
Endometriosis was evaluated using the Enzian classification system for deep infiltrating endometriosis: A (anterior compartment): Rectovaginal septum, retrocervical region, vagina. B (parametrium compartment): Parametrium and uterosacral ligaments (assessed left and right separately). C (posterior compartment): Rectosigmoid colon. O (ovarian involvement): Sum of all endometriotic cysts, each side separately (O1: < 3 cm, O2: 3-7 cm, O3: > 7 cm). T (tubo-ovarian findings): Adhesions to the pelvic wall, uterus, or bowel; tubal patency (each side separately). P (peritoneal endometriosis): Sum of all visible peritoneal lesions (P1: < 3 cm, P2: 3-7 cm, P3: > 7 cm). Adenomyosis (F): Diffuse or focal and other defined locations.
Descriptive statistics were used to summarize categorical variables as n (%). Associations between uterine anomaly category and presence of endometriosis or obstruction were assessed with χ2 tests. P < 0.05 was considered statistically significant. Statistical analyses were performed using Jamovi (The jamovi project, 2022; Version 2.3, https://www.jamovi.org).
Seventy-five patients with Mullerian anomalies were identified and included in the analysis with mean age 21.3 ± 9.6 years. The most frequent category was U5b (aplasia/hypoplasia), comprising 25 patients (33.3%). Other common categories included U2a (partial septate) in 14 patients (18.7%) and U3c (bicorporeal subtype) in 13 patients (17.3%). Additional categories and frequencies are summarized in Table 1.
| Component | Category | n | % |
| Uterus (U) | Arcuate/U0 | 4 | 5.3 |
| U1b | 6 | 30.7 | |
| U2a | 14 | 18.67 | |
| U2b | 3 | 4 | |
| U3c | 13 | 17.33 | |
| U4a | 2 | 2.67 | |
| U4b | 1 | 1.33 | |
| U5a-U5b | 29 | 38.6 | |
| Accessory cavitated uterine masses/U6 | 3 | 4.0 | |
| Cervix (C) | Normal/C0 | 29 | 38.7 |
| Mild anomaly/C1-C2 | 17 | 22.7 | |
| Severe anomaly/C4 | 29 | 38.7 | |
| Vagina (V) | Normal/V0 | 46 | 61.3 |
| Anomaly/V4 | 29 | 38.7 | |
| Obstruction | No/N | 62 | 82.7 |
| Yes/Y | 13 | 17.3 |
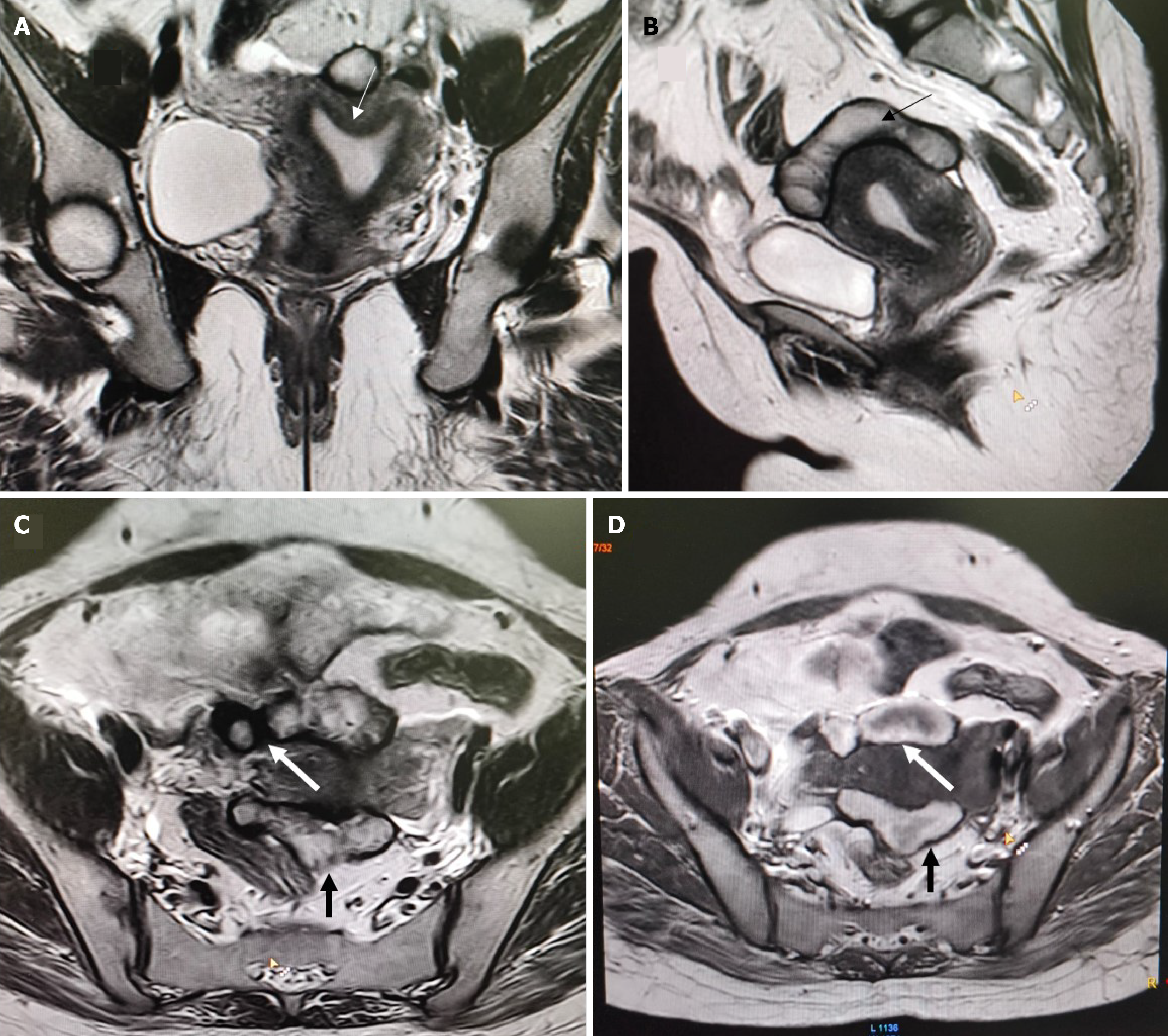
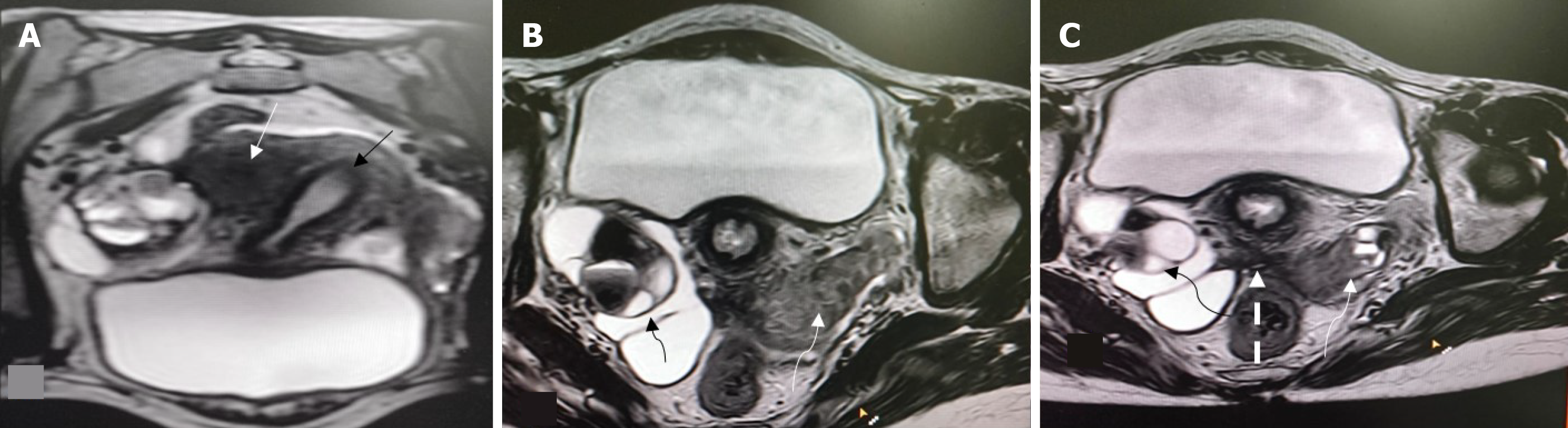
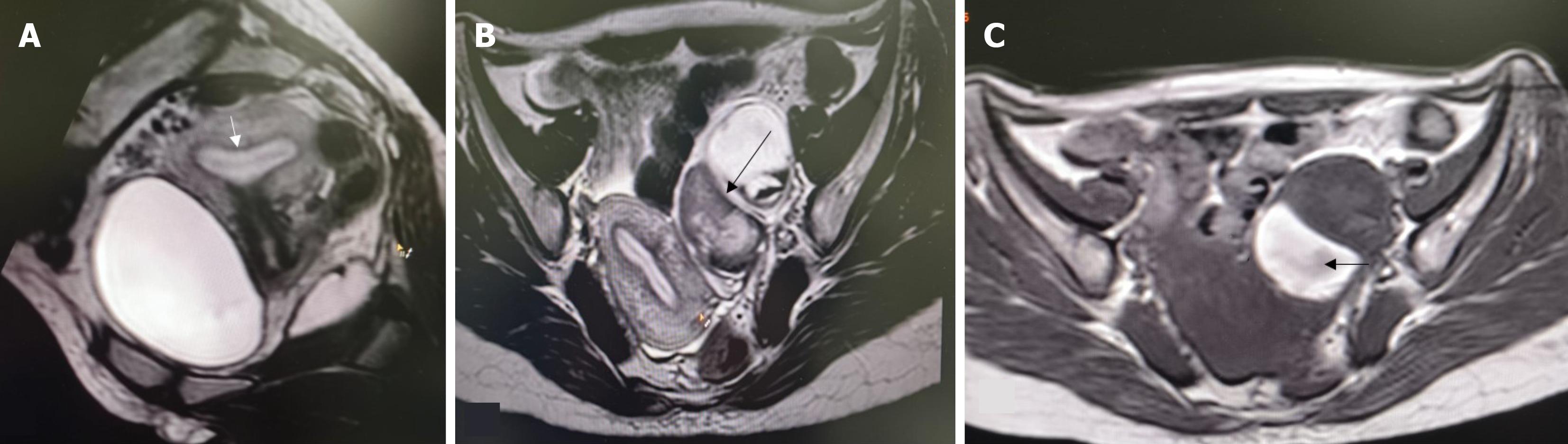
Overall, 22 patients had MRI findings consistent with endometriosis. Peritoneal disease (P1-P3) was not identified in this series. Ovarian endometriomas were present in 16 patients (21.3%), most commonly in the 3-7 cm range (O2, 12 patients; 16.0%). Tubo-ovarian adhesions or involvement (T2-T3) occurred in 11 patients (14.7%) (Figures 1, 2 and 3). Deep endometriosis nodules were observed primarily in the parametrial/uterosacral compartment (B) in 6 patients (8.0%). No rectosigmoid (C) or anterior compartment (A) endometriosis lesions were seen. Adenomyosis was identified in 4 patients (5.3%; diffuse in 2, focal in 2). A summary is presented in Table 2.
| Feature | Subtype/Location | Total | Percentage of total (%) | Percentage of patients with endometriosis, % (n = 22) |
| Peritoneum (P) | All | 0 | 0 | 0 |
| Ovary (O) | O1 (< 3 cm) | 1 | 1.3 | 4.5 |
| O2 (3-7 cm) | 12 | 16.0 | 54.5 | |
| O3 (> 7 cm) | 3 | 4.0 | 13.6 | |
| Tubo-ovarian/adhesions (T) | T2 | 8 | 10.7 | 36.4 |
| T3 | 3 | 4.0 | 13.6 | |
| Deep endometriosis | A: (Rectovaginal/retro-cervical/vagina) | 0 | 0 | 0 |
| B: (parametrium/uterosacral ligament) | 6 | 8.0 | 27.3 | |
| C (rectosigmoid) | 0 | 0 | 0 | |
| Adenomyosis (F) | Diffuse | 2 | 2.7 | 9.1 |
| Focal | 2 | 2.7 | 9.1 |
Table 3 highlights the association between uterine anomaly type, outflow obstruction, and the presence of endometriosis. Endometriosis was more frequently observed in patients with complex uterine anomalies, particularly U3c (bicorporeal septate uterus), U5a (aplastic uterus with a functional rudimentary cavity), and U0 (arcuate uterus), whereas a markedly low prevalence was noted in U5b (aplastic uterus without a functional cavity). A statistically significant association was seen between uterine category and endometriosis (P = 0.006).
| Feature | Category | Total (n) | Presence of endometriosis | Percentage with endometriosis (%) | P value |
| Uterus (U) | U0 | 4 | 3 | 75 | 0.006 |
| U1b | 6 | 1 | 16.7 | ||
| U2a | 14 | 3 | 21.4 | ||
| U2b | 3 | 1 | 33.3 | ||
| U3c | 13 | 7 | 53.8 | ||
| U4a | 2 | 1 | 50 | ||
| U4b | 1 | 1 | 100 | ||
| U5a | 4 | 3 | 75 | ||
| U5b | 25 | 1 | 4 | ||
| U6 | 3 | 1 | 33.3 | ||
| Cervix (C) | C0 | 29 | 10 | 34.5 | 0.084 |
| C1 | 3 | 1 | 33.3 | ||
| C2 | 14 | 7 | 50 | ||
| C4 | 29 | 4 | 13.8 | ||
| Vagina (V) | V0 | 46 | 18 | 39.1 | 0.019 |
| V4 | 29 | 4 | 13.8 | ||
| Obstruction | N | 62 | 14 | 22.6 | 0.005 |
| Y | 13 | 8 | 61.5 | ||
| Rudimentary horn | N | 69 | 18 | 26.1 | 0.036 |
| Y | 6 | 4 | 66.7 |
With respect to associated anomalies, vaginal aplasia (V4) showed a significantly lower prevalence of endometriosis compared to patients with a normal vagina (P = 0.019), while cervical anomaly subtype did not demonstrate a statistically significant association. Overall, these findings suggest that functional endometrial tissue combined with altered outflow or complex uterine morphology plays a key role in determining the distribution of endometriosis in patients with Mullerian anomalies. Patients with a rudimentary horn containing a functional cavity had a significantly higher prevalence of endometriosis compared to those without a rudimentary horn (66.7% vs 26.1%, P = 0.036).
Among the 75 patients evaluated, obstruction was identified in 13 patients (17.3%). Within uterine anomaly groups (U), obstruction was most frequent in U3c anomalies, where 6 of 13 patients (46%) demonstrated obstruction. The presence of genital tract obstruction showed a strong correlation with endometriosis, with affected patients demonstrating nearly threefold higher prevalence compared to those without obstruction (61.5% vs 22.6%, P = 0.005). In this subgroup, obstruction affected both endometriosis (3/7; 43%) and non-endometriosis (3/6; 50%) patients. In U0 uteri (arcuate), 2 of 4 patients affected (50%), split equally between endometriosis (1/3) and non-endometriosis (1/1). U4a had obstruction in 1 of 2 patients (50%), single patients showed signs of obstruction in each group of U4b, U5a, U6. It was rare in U2a (1/14; 7%) and U5b (1/25; 4%). Across cervical categories (C), obstruction occurred predominantly in patients with normal morphology (C0), where 10 of 29 patients (34.5%) had endometriosis. In C2, 7 of 14 patients had endometriosis, whereas severe anomalies (C4) were associated with endometriosis in 4 of 29 patients (13.8%). Despite these endometriosis distributions, obstruction overall remained infrequent and did not cluster within any specific cervical subtype. Vaginal morphology similarly showed limited correlation with obstruction. Most patients with normal vaginal anatomy (V0) did not exhibit obstruction, despite 18 of 46 having endometriosis (39.1%). Among those with V4 anomalies, endometriosis was identified in 4 of 29 patients (13.8%). Overall, obstruction occurred sporadically across uterine, cervical, and vaginal classifications. Obstruction was most frequent in U3c (46% of that subgroup) but was distributed among both endometriosis and nonendometriosis patients.
This MRI-based evaluation of Mullerian anomalies and endometriosis provides a structured, anatomy-driven appraisal of disease patterns that have traditionally been difficult to characterize. In this study, endometriosis was identified in a substantial proportion (30%) of patients with Mullerian anomalies, reinforcing the well-recognized association between congenital uterine abnormalities and ectopic endometrial disease[5-11,16-20]. Ovarian involvement was the most common manifestation, with endometriomas forming the predominant imaging finding, while tubo-ovarian involvement was also frequently observed, indicating that disease in this population is not limited to superficial implants.
Previous works have also showed that specific morphologies (unicornuate uteri) may have higher endometriosis prevalence even among nonobstructive anomalies, suggesting morphologyspecific risks[19]. Our findings reiterate that uterine morphology and the presence of functional endometrium influence the likelihood of endometriosis. The distribution of endometriosis varied across uterine anomaly types, with higher prevalence noted in complex configurations such as bicorporeal septate uterus and aplastic uterus with a functional rudimentary cavity. Vaginal aplasia (V4) was associated with a significantly lower prevalence of endometriosis compared with normal vaginal anatomy (P = 0.019). By integrating the ESHRE/ESGE classification of Mullerian anomalies with Enzian-based mapping of endometriosis, this study demonstrates how specific uterine anatomy and functional outflow characteristics influence both the likelihood and distribution of endometriotic disease.
Obstructive Mullerian anomalies remain the most consistently documented risk factor for endometriosis. Multiple prior series have demonstrated a substantially higher prevalence of endometriosis in patients with obstructive Mullerian anomalies compared with non-obstructive anomalies. A pooled estimate reported by Vercellini et al[6] found an overall mean prevalence of endometriosis of 47% (95% confidence interval: 36%-58%) in obstructive Mullerian anomalies vs 19% (95% confidence interval: 15%-24%) in non-obstructive anomalies. Takahashi et al[11] observed endometriosis in 6 of 12 (50%) young patients with obstructive Mullerian anomalies, with an especially high rate of ipsilateral ovarian endo
The Mayer-Rokitansky-Kuster-Hauser syndrome (MRKH) subgroup further illustrates this complexity. Marsh et al[20] reported that nearly half of MRKH patients had identifiable uterine remnants, and among those presenting with pelvic pain, over half had laparoscopically confirmed endometriosis, predominantly stage I-III disease. Uğur et al[18] docu
An important limitation of the ESHRE-ESGE classification relates to the handling of the arcuate uterus. In this system, arcuate morphology is not assigned a dedicated category and is generally subsumed under class U0 (normal uterus), based on the assumption that it represents a normal anatomical variant without clinical significance. At the same time, the reliance on proportional fundal indentation and myometrial thickness thresholds has been criticized for potentially overclassifying some arcuate uteri as septate, contributing to diagnostic ambiguity[3,4]. In the present study, uteri classified as U0, included those with arcuate morphology, demonstrated a relatively high prevalence of endometriosis (75%). While this observation suggests that arcuate uteri may not be entirely benign in all cases, it must be interpreted cautiously, as a clear selection bias exists: Asymptomatic arcuate uteri are unlikely to undergo MRI, whereas those included in this cohort were imaged in the context of pelvic symptoms or associated pathology. Nevertheless, the association observed in this selected population raises the possibility that arcuate morphology may represent a subtle developmental abnormality rather than a purely normal variant, and supports reconsideration of its placement within the “normal” category in future classification refinements.
An important factor influencing the observed prevalence of endometriosis in this study is the use of MRI-based structured interpretation, rather than surgical confirmation. Several earlier studies have relied on laparoscopic or operative findings to identify endometriosis and have consequently reported higher prevalence rates, reflecting referral bias toward more symptomatic patients undergoing surgery. In contrast, MRI primarily detects ovarian endometriomas and deep infiltrating endometriosis, while superficial peritoneal implants may remain occult, potentially resulting in lower prevalence estimates. Despite this limitation, MRI provides a comprehensive, non-invasive evaluation of pelvic anatomy and disease extent, which is particularly advantageous in patients with Mullerian anomalies[21,22]. Compared with transvaginal ultrasound, MRI offers superior soft-tissue contrast, multiplanar assessment, and reproducibility, allowing more reliable evaluation of deep pelvic compartments and associated congenital abnormalities. The use of standardized MRI-based classification systems in this study therefore represents a pragmatic approach that complements, rather than replaces, surgical staging and may better reflect disease patterns encountered in routine imaging practice.
This study has several limitations. Its retrospective design introduces inherent selection bias, as only patients who underwent MRI and were reported to have Mullerian anomalies were included; consequently, asymptomatic or incidentally detected anomalies are under-represented. The relatively small sample size within individual anomaly subgroups limits the strength of statistical comparisons, particularly for rare uterine, cervical, and vaginal classes. Some rare anomaly subtypes (e.g., U6 and U0) included in this study might be influenced by selection bias. Correlation with surgical or histopathological findings was not available in all cases, and MRI served as the reference standard for diagnosis of endometriosis, which may underestimate superficial peritoneal disease. During our analysis, potential confounding factors such as age and symptom severity were not adjusted for, which may have influenced the observed associations. In addition, the assessment of obstruction was based on imaging evidence of hemorrhagic collections at a single time point and may not reflect intermittent or partially relieved obstruction. Finally, As MRI interpretation was performed in consensus between two radiologists, formal assessment of inter-observer variability could not be done.
In conclusion, our findings highlight that an anatomy-based framework remains essential for understanding endometriosis risk across Mullerian anomaly subtypes. While obstruction is a major driver, remnant functionality, cavity architecture, and anomaly-specific flow patterns significantly modify risk. MRI enables a more nuanced and clinically relevant understanding of these relationships. Patients with obstructive anomalies or functional remnants warrant targeted surveillance for endometriosis and early gynecologic referral. At the same time, the presence of endometriosis in non-obstructive anomalies emphasizes the need for individualized assessment rather than reliance solely on broad anomaly categories.
| 1. | Pfeifer SM, Attaran M, Goldstein J, Lindheim SR, Petrozza JC, Rackow BW, Siegelman E, Troiano R, Winter T, Zuckerman A, Ramaiah SD. ASRM müllerian anomalies classification 2021. Fertil Steril. 2021;116:1238-1252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 320] [Cited by in RCA: 223] [Article Influence: 44.6] [Reference Citation Analysis (0)] |
| 2. | Grimbizis GF, Gordts S, Di Spiezio Sardo A, Brucker S, De Angelis C, Gergolet M, Li TC, Tanos V, Brölmann H, Gianaroli L, Campo R. The ESHRE/ESGE consensus on the classification of female genital tract congenital anomalies. Hum Reprod. 2013;28:2032-2044. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 683] [Cited by in RCA: 494] [Article Influence: 38.0] [Reference Citation Analysis (0)] |
| 3. | Ludwin A, Ludwin I. Comparison of the ESHRE-ESGE and ASRM classifications of Müllerian duct anomalies in everyday practice. Hum Reprod. 2015;30:569-580. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 137] [Cited by in RCA: 95] [Article Influence: 8.6] [Reference Citation Analysis (1)] |
| 4. | Dixit R, Duggireddy CS, Pradhan GS. Mullerian anomalies: revisiting imaging and classification. Insights Imaging. 2025;16:40. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 12.0] [Reference Citation Analysis (1)] |
| 5. | Konrad L, Dietze R, Kudipudi PK, Horné F, Meinhold-Heerlein I. Endometriosis in MRKH cases as a proof for the coelomic metaplasia hypothesis? Reproduction. 2019;158:R41-R47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 49] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 6. | Vercellini P, Salmeri N, Somigliana E, Piccini M, Caprara F, Viganò P, De Matteis S. Müllerian anomalies and endometriosis as potential explanatory models for the retrograde menstruation/implantation and the embryonic remnants/celomic metaplasia pathogenic theories: a systematic review and meta-analysis. Hum Reprod. 2024;39:1460-1470. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 22] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 7. | Signorile PG, Baldi F, Bussani R, D'Armiento M, De Falco M, Boccellino M, Quagliuolo L, Baldi A. New evidence of the presence of endometriosis in the human fetus. Reprod Biomed Online. 2010;21:142-147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 46] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 8. | Pitot MA, Bookwalter CA, Dudiak KM. Müllerian duct anomalies coincident with endometriosis: a review. Abdom Radiol (NY). 2020;45:1723-1740. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 33] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 9. | Tian W, Chen N, Liang Z, Song S, Wang Y, Ye Y, Duan J, Zhu L. Clinical Features and Management of Endometriosis among Patients with MRKH and Functional Uterine Remnants. Gynecol Obstet Invest. 2021;86:518-524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 10. | Bhamidipaty-Pelosi S, Kyei-Barffour I, Volpert M, O'Neill N, Grimshaw A, Eriksson L, Vash-Margita A, Pelosi E. Müllerian anomalies and endometriosis: associations and phenotypic variations. Reprod Biol Endocrinol. 2024;22:157. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 11. | Takahashi N, Harada M, Kanatani M, Wada-Hiraike O, Hirota Y, Osuga Y. The Association between Endometriosis and Obstructive Müllerian Anomalies. Biomedicines. 2024;12:651. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 12. | Revised American Society for Reproductive Medicine classification of endometriosis: 1996. Fertil Steril. 1997;67:817-821. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2443] [Cited by in RCA: 2125] [Article Influence: 73.3] [Reference Citation Analysis (1)] |
| 13. | Keckstein J, Saridogan E, Ulrich UA, Sillem M, Oppelt P, Schweppe KW, Krentel H, Janschek E, Exacoustos C, Malzoni M, Mueller M, Roman H, Condous G, Forman A, Jansen FW, Bokor A, Simedrea V, Hudelist G. The #Enzian classification: A comprehensive non-invasive and surgical description system for endometriosis. Acta Obstet Gynecol Scand. 2021;100:1165-1175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 262] [Cited by in RCA: 210] [Article Influence: 42.0] [Reference Citation Analysis (1)] |
| 14. | Burla L, Scheiner D, Samartzis EP, Seidel S, Eberhard M, Fink D, Boss A, Imesch P. The ENZIAN score as a preoperative MRI-based classification instrument for deep infiltrating endometriosis. Arch Gynecol Obstet. 2019;300:109-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 37] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 15. | Guerriero S, Condous G, van den Bosch T, Valentin L, Leone FP, Van Schoubroeck D, Exacoustos C, Installé AJ, Martins WP, Abrao MS, Hudelist G, Bazot M, Alcazar JL, Gonçalves MO, Pascual MA, Ajossa S, Savelli L, Dunham R, Reid S, Menakaya U, Bourne T, Ferrero S, Leon M, Bignardi T, Holland T, Jurkovic D, Benacerraf B, Osuga Y, Somigliana E, Timmerman D. Systematic approach to sonographic evaluation of the pelvis in women with suspected endometriosis, including terms, definitions and measurements: a consensus opinion from the International Deep Endometriosis Analysis (IDEA) group. Ultrasound Obstet Gynecol. 2016;48:318-332. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 729] [Cited by in RCA: 596] [Article Influence: 59.6] [Reference Citation Analysis (1)] |
| 16. | Koninckx PR, Ussia A, Adamyan L, Wattiez A, Gomel V, Martin DC. Pathogenesis of endometriosis: the genetic/epigenetic theory. Fertil Steril. 2019;111:327-340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 347] [Cited by in RCA: 278] [Article Influence: 39.7] [Reference Citation Analysis (2)] |
| 17. | Olive DL, Henderson DY. Endometriosis and mullerian anomalies. Obstet Gynecol. 1987;69:412-415. [PubMed] |
| 18. | Uğur M, Turan C, Mungan T, Kuşçu E, Senöz S, Ağiş HT, Gökmen O. Endometriosis in association with Müllerian anomalies. Gynecol Obstet Invest. 1995;40:261-264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 72] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 19. | Fedele L, Bianchi S, Di Nola G, Franchi D, Candiani GB. Endometriosis and nonobstructive Müllerian anomalies. Obstet Gynecol. 1992;79:515-517. [PubMed] |
| 20. | Marsh CA, Will MA, Smorgick N, Quint EH, Hussain H, Smith YR. Uterine remnants and pelvic pain in females with Mayer-Rokitansky-Küster-Hauser syndrome. J Pediatr Adolesc Gynecol. 2013;26:199-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 43] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 21. | Kido A, Himoto Y, Moribata Y, Kurata Y, Nakamoto Y. MRI in the Diagnosis of Endometriosis and Related Diseases. Korean J Radiol. 2022;23:426-445. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 35] [Article Influence: 8.8] [Reference Citation Analysis (1)] |
| 22. | Zuber M, Shoaib M, Kumari S. Magnetic resonance imaging of endometriosis: a common but often hidden, missed, and misdiagnosed entity. Pol J Radiol. 2022;87:e448-e461. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (1)] |