Published online Mar 28, 2026. doi: 10.4329/wjr.v18.i3.116736
Revised: January 26, 2026
Accepted: February 27, 2026
Published online: March 28, 2026
Processing time: 127 Days and 12.8 Hours
Objective assessment of pain relief and adequacy of decompression following spine surgery remains challenging. Infrared thermography (IRT) offers a non-invasive, radiation-free modality to visualize physiological changes in regional blood flow and inflammation.
To evaluate diagnostic concurrence and postoperative normalization of IRT in patients undergoing spine surgery, and to identify clinical predictors of recovery in thermal patterns.
This retrospective study included 35 adult patients who underwent lumbar decompression or fusion procedures for spinal pathologies such as degenerative conditions, deformity, tumors and adjacent segment disease. The pre-operative and post-operative IRT scans were analyzed to quantify the focal temperature asymmetry corresponding to the symptomatic spinal levels. Two blinded reviewers independently evaluated the concurrence and post
The mean age of the cohort was 66.3 ± 10.7 years (males: 63.6%). Multilevel disease was present in 63.6% of pa
IRT demonstrated correlation with symptomatic spinal levels preoperatively and showed moderate association with postoperative thermal normalization following spine surgery. These preliminary findings suggest that IRT may provide additional, non-invasive information to complement standard imaging in the assessment of neural decompression and recovery. Larger, prospective studies are warranted to confirm its clinical utility and define its role in routine postoperative monitoring.
Core Tip: Infrared thermography (IRT) provides a novel, non-invasive biomarker for assessing symptomatic spinal levels and recovery after surgery. In this retrospective study, IRT demonstrated strong preoperative concurrence with clinical loca
- Citation: Muthu S, Natarajan KP, Viswanathan VK, Kolarpatti Ponnusamy DV, Satish Kumar RC, Sharun K, Jang HJ. Infrared thermography as adjunctive imaging in spine surgery: Evaluating thermal asymmetry for predicting symptomatic level and recovery. World J Radiol 2026; 18(3): 116736
- URL: https://www.wjgnet.com/1949-8470/full/v18/i3/116736.htm
- DOI: https://dx.doi.org/10.4329/wjr.v18.i3.116736
Accurate identification of the symptomatic spinal levels and objective evaluation of postoperative recovery are still critical challenges in patients undergoing spinal surgery[1]. Conventional imaging modalities like magnetic resonance imaging (MRI) and CT provide practical morphological and anatomical details; however, more often than not, they may fail to reflect any physiological or functional changes after spinal decompression[2]. Specifically, infrared thermography (IRT) has emerged as a promising non-invasive tool capable of detecting subtle changes in the skin surface temperature[2-6], which may correspond to neural or vascular dysfunction[7-11].
IRT has been successfully employed in the diagnosis of diverse musculoskeletal and neurological conditions[1-5], such as complex regional pain syndrome[6-10], peripheral neuropathy and radiculopathy[11-15]. Previous studies have pur
Although prior studies have demonstrated the diagnostic utility of IRT in spinal disorders, evidence regarding its prognostic role in the postoperative setting remains limited[13-15]. Most published work has focused on preoperative localization or machine-learning-based classification, with little emphasis on postoperative normalization as a surrogate of recovery. Our study addresses this gap by evaluating both preoperative concurrence and postoperative normalization of thermal asymmetry in a surgical cohort. The retrospective design was chosen to leverage complete imaging datasets, and the 7-14 day follow-up window was selected to minimize transient inflammatory effects while capturing early re
This retrospective observational study was conducted at a tertiary care academic center and included 35 adult patients who underwent spine surgery for degenerative, deformity, trauma, tumor, or adjacent segment disease. This re
The inclusion criteria were as follows: (1) Availability of both preoperative and postoperative IRT scans; (2) Docu
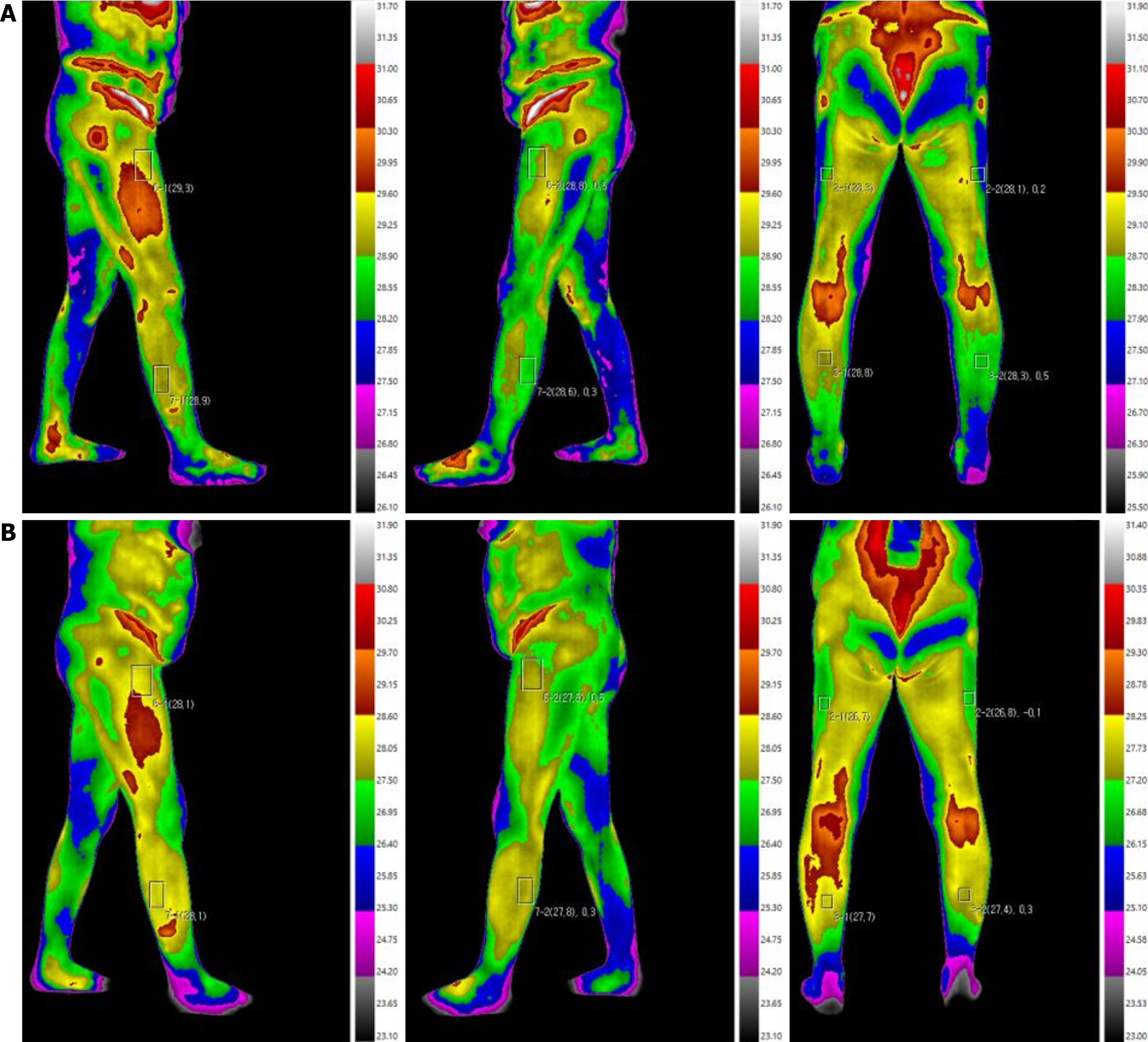
Infrared thermographic scans were acquired using the IRIS ST 9000 system (IRIS Systems, Seoul, South Korea) under standardized ambient conditions. Each patient underwent full-body thermography in upright posture, with anterior, posterior, lateral, and plantar views captured. All scans were performed in a temperature-controlled room (22-24 °C) with standardized ambient lighting. Patients were positioned upright with arms abducted to 30° and feet shoulder-width apart. Regions of interest were delineated manually at the decompression level and adjacent dermatomes. Temperature gradients were quantified using pixel-intensity histograms and temperature difference (ΔT) mapping. ΔT thresholds were defined as ≥ 0.5 °C difference between dermatomes. Image processing was performed using ImageJ software version 1.53 (National Institutes of Health, Bethesda, MD, United States). To address heterogeneity, subgroup analyses were conducted by pathology type (degenerative, tumor, trauma) to mitigate confounding. Preoperative scans were obtained within 48 hours prior to surgery, and postoperative scans were acquired between 7 days and 14 days following the procedure in order to minimize the transient inflammatory effects. The image-based interpretation was focused on identifying focal thermal asymmetry corresponding to the symptomatic dermatomes and its resolution following surgery.
Each pair of thermographic images (preoperative and postoperative) was independently reviewed by two experts: A radiologist and a spine surgeon, who evaluated three different parameters: Preoperative concurrence of thermal asym
The clinical parameters included age, sex, diagnosis, decompression level, procedure type - posterior lumbar interbody fusion (PLIF), transforaminal lumbar interbody fusion (TLIF), oblique lumbar interbody fusion (OLIF), anterior cervical discectomy and fusion (ACDF), unilateral biportal endoscopy (UBE), or decompression alone (laminectomy or laminoplasty), and presence of multilevel disease (defined as involvement of two or more contiguous spinal levels).
Descriptive statistics were computed for all demographic and clinical parameters (such as diagnosis, procedure type, thermography concurrence, normalization, and presence of multilevel disease). Associations between multilevel disease and thermography outcomes were assessed using the χ2 tests or Fisher’s exact test, as appropriate. Subgroup analyses were performed to compare normalization rates across the diagnosis and procedure types.
A binary logistic regression model was constructed to identify predictors of thermography normalization, including age, sex, diagnosis, procedure type, and multilevel disease. Model performance was evaluated using receiver operating characteristic curve analysis, and statistical significance was defined as P < 0.05. All the studies were conducted using Python version 3.10 and R version 4.2.2.
A total of 35 patients (22 males, mean age: 66.3 ± 10.7 years) were included in the analysis. Multilevel disease was present in 22 patients (63.6%). The most common indications for surgery were spinal stenosis (51.5%, n = 18), followed by spondylolisthesis (18.2%, n = 6), ossification of the posterior longitudinal ligament (OPLL; 12.1%, n = 4); and other con
| Category | Subcategory | |
| Age (years) | 66.3 ± 10.7 | |
| Sex | Male | 22 (63.6) |
| Female | 13 (36.4) | |
| Multilevel disease | Present | 22 (63.6) |
| Absent | 13 (36.4) | |
| Diagnosis | Stenosis | 18 (51.5) |
| Listhesis | 6 (18.2) | |
| OPLL | 4 (12.1) | |
| Other (CSM, PJF, trauma) | 7 (18.2) | |
| Procedure type | PLIF | 17 (48.5) |
| Decompression only | 10 (27.3) | |
| TLIF | 5 (15.2) | |
| OLIF/ACDF/UBE | 3 (9) | |
| Thermography concurrence | Yes | 26 (78.8) |
| No | 7 (21.2) | |
| Thermography normalization | Yes | 22 (63.6) |
| No | 13 (36.4) |
The most frequently performed procedure was PLIF (48.5%, n = 17), followed by decompression only (27.3%, n = 10), TLIF (15.2%, n = 5), and other interventional techniques [such as OLIF, ACDF, and UBE (9.0%, n = 3)].
Based on consensus image analysis, preoperative thermography concurrence - defined as focal thermal asymmetry corresponding to the level of decompression - was observed in 78.8% of patients (n = 26). Postoperative normalization of thermal asymmetry was achieved in 63.6% of cases (n = 22), reflecting partial restoration of bilateral thermal balance as illustrated in Figure 1. Normalization was defined quantitatively as a reduction in inter-dermatomal ΔT to ≤ 0.5 °C.
Thermography concurrence was observed in 77.3% (n = 17/22) of multilevel cases and 81.8% (n = 9/11) of single-level instances, with no statistically significant association (P = 1.000). Postoperative normalization was achieved in 59.1% (n = 13/22) of multilevel cases, and 72.7% (n = 8/11) of single-level cases (P = 0.516).
Subgroup analysis showed variation in normalization rates across procedure types. TLIF demonstrated the highest normalization rate at 80% (4/5), followed by decompression-alone procedures at 66.7% (6/9), and PLIF at 62.5% (10/16). OLIF, ACDF, and UBE each demonstrated 100% normalization, though the subgroup size was small (n = 3). When stratified by diagnosis, spinal stenosis showed the highest normalization rate at 76.5% (13/17), followed by OPLL at 75% (3/4), and spondylolisthesis at 50% (3/6). No normalization was observed in patients with CSM, PJF, or IDEM.
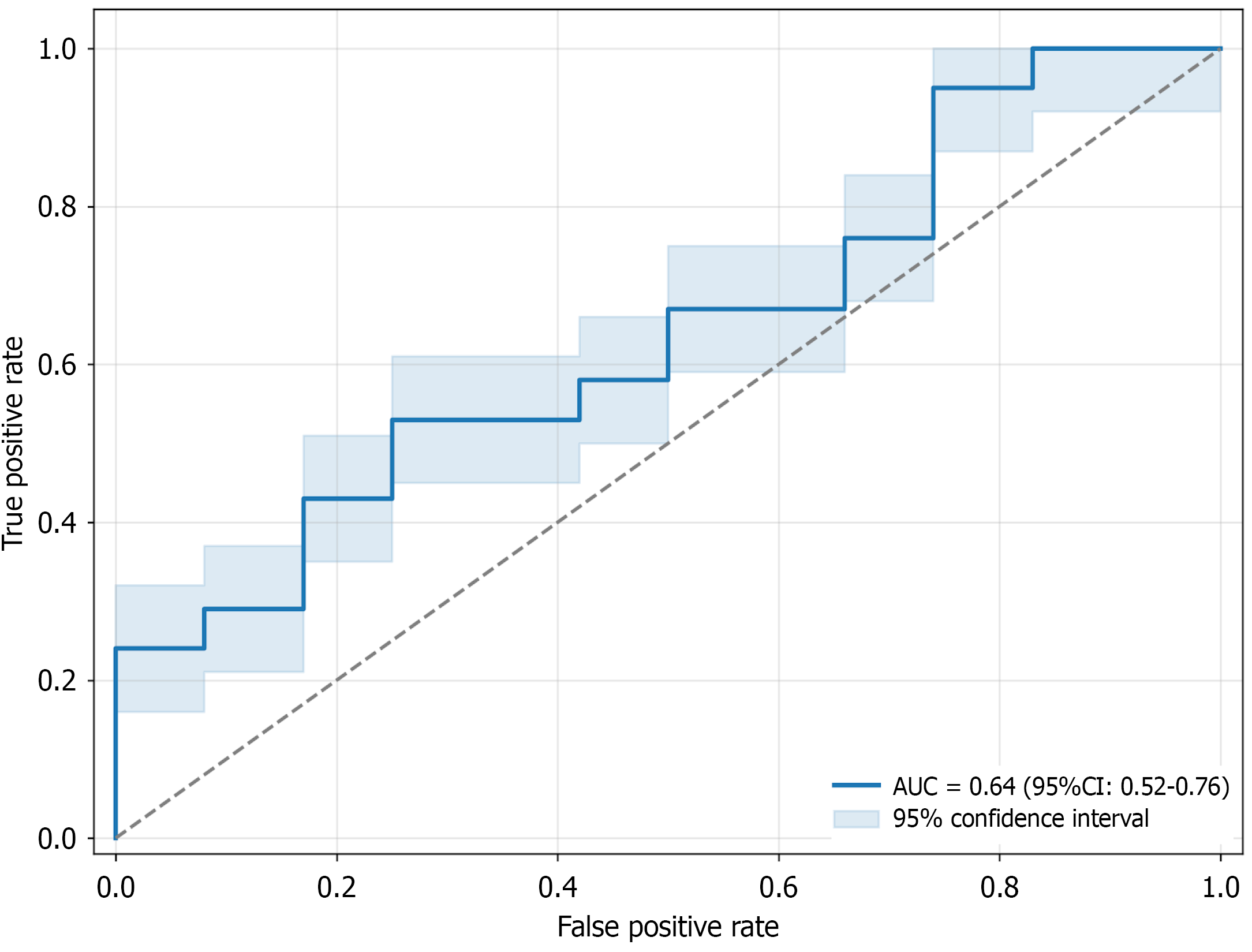
Multivariate logistic regression analysis identified female sex, diagnosis of stenosis, and TLIF procedure as positive predictors of thermography normalization; whereas multilevel disease was a negative predictor, as shown in Table 2. The regression model should be interpreted as exploratory given the modest sample size (n = 35), which limits statistical power and increases risk of overfitting. Model performance demonstrated moderate predictive accuracy [area under the curve = 0.64, 95% confidence interval (CI): 0.52-0.76]. CIs for odds ratios are reported in Table 2, and receiver operating characteristic CIs are displayed in Figure 2. These findings suggest moderate predictive performance rather than depen
| Predictor | Coefficient (β) | Odds ratio | 95%CI | P value |
| Age (per year) | -0.02 | 0.98 | 0.94-1.02 | 0.28 |
| Sex (female) | +0.85 | 2.34 | 1.01-5.42 | 0.048 |
| Diagnosis: Stenosis | +1.12 | 3.06 | 1.18-7.92 | 0.021 |
| Diagnosis: Listhesis | -0.44 | 0.64 | 0.21-1.96 | 0.42 |
| Procedure: TLIF | +1.39 | 4.01 | 1.02-15.72 | 0.046 |
| Procedure: PLIF | Reference | |||
| Multilevel disease | -0.77 | 0.46 | 0.19-1.12 | 0.09 |
This study demonstrates that IRT can provide meaningful physiological insights regarding the symptom definition and recovery patterns in patients undergoing spine surgery. The preoperative focal thermal asymmetry corresponded with the decompression level in approximately 80% of individuals, and postoperative normalization was observed in approximately two-thirds, indicating that IRT may serve as a functional correlate of symptomatic neural compression and the success of surgical decompression.
The high rate of preoperative concurrence between thermal asymmetry and levels of symptomatology in our study supports the prior evidence linking temperature gradients with lumbar radiculopathy and nerve root irritation[14]. Segmental sympathetic dysfunction and altered microvascular flow have been implicated as the mechanism underlying these observations, consistent with the thermographic patterns described in lumbar disc herniation and cervical myelopathy[4,13].
In another retrospective review by Kim et al[2,12], they applied the IRT to the machine learning algorithm (which is the “bag of visual words” method) in the differential screening of L5 and S1 lumbosacral radiculopathy. The IRT dataset included data from healthy individuals and radiculopathy patients with lumbar disc herniation at L4-5 and L5-S1 levels. A total of 842 patients were identified, and the dataset was divided into a 7:3 ratio as the training algorithm and test da
In a retrospective study by Park et al[15] involving 224 patients (180 patients and 44 patients in hypothermia and hyperthermia groups, respectively) undergoing IRT at a single center, diverse demographic and symptom-related factors that can potentially influence the results of IRT were compared. In general, the thermal pattern with IRT in patients with unilateral spinal radicular pain demonstrates relative hypothermia in comparison with the contralateral side. Based on their observation, the presence of trauma history (13.9% vs 31.8%, odds ratio: 2.893, P = 0.008), shorter duration of sym
Postoperative normalization in 63.6% of patients in our series suggests that the sympathetic tone and neurological status are at least partially restored within 2 weeks of surgery. The absence of normalization in a particular proportion of individuals may be attributed to delayed physiological improvement, incomplete decompression and persistence of inflammation[14]. The higher normalization rate after TLIF (in our patients), in comparison with PLIF, can be correlated with more localized nerve root decompression as well as minimized posterior muscle disruption inherent to the transforaminal approach. Postoperative normalization observed at 7-14 days may reflect early modulation of local inflammation and sympathetic tone rather than complete neural recovery. Therefore, IRT should be considered a potential adjunctive biomarker rather than a definitive measure of neural restoration.
Although the role of IRT in evaluating the post-interventional normalization or physiological recovery in patients with lumbar spinal pathologies is not clearly established hitherto, in the study by Cañada-Soriano et al[4], the evaluation of thermal variations (measurement of skin ΔT) on IRT was identified as an effective tool in monitoring the patients’ res
Multivariate analysis revealed that female sex and a diagnosis of lumbar canal stenosis predicted favorable thermographic recovery, while multilevel disease was associated with lower normalization rates. These observations align with prior reports suggesting that multilevel pathologies demonstrate slower recovery patterns owing to widespread ischemia and prolonged neurological recovery time[16,17]. The stratified findings highlight that normalization rates varied across both procedure type and diagnosis. TLIF and decompression-alone procedures demonstrated relatively higher normalization, while PLIF showed moderate recovery. OLIF, ACDF, and UBE achieved complete normalization, though numbers were small. By diagnosis, stenosis and OPLL showed favorable recovery patterns, whereas spondylolisthesis demonstrated lower normalization and no recovery was observed in CSM, PJF, or IDEM. These results reinforce that IRT normalization is most reliable in degenerative conditions, particularly stenosis, and less consistent in trauma-related or complex pathologies.
IRT offers several practical advantages[2-6]: It is radiation-free, repeatable, inexpensive and provides immediate visual feedback[13,17]. It may complement MRI and computed tomography in localizing the actual pain-generating spinal levels, especially in ambiguous clinical situations or in patients with multilevel degenerative pathologies. Postoperatively, IRT could serve as an objective adjuvant in tracking the physiological recovery of patients, wound perfusion and detecting early complications such as infection and nerve irritation[13,14,16].
The findings of our study are consistent with the prior thermographic framework proposed by Muthu et al[14], who emphasized the diagnostic potential of digital thermography in the context of degenerative spinal pathologies. Their review underscored that temperature asymmetry, which may manifest as hypothermic or hyperthermic dermatomal zones, could reflect distinct physiological processes such as ischemia or inflammation, paralleling the pre-operative and post-operative trends observed in our patient cohort. Specifically, our results further validate the concept that focal thermal normalization following decompression can be considered as a surrogate marker for restored microvascular and sympathetic equilibrium. In addition, the integration of standardized acquisition protocols and quantitative ΔT mapping in our study directly addresses the methodological limitations highlighted in previous reviews, advancing thermography from a qualitative adjunct to a reproducible, image-based biomarker for surgical recovery. Future incorporation of arti
In another large-scale, prospective trial by Liu et al[13], IRT was systematically validated against intraoperative gold standards employed in patients with lumbosacral radicular pain. Their study identified an optimal ΔT range of 0.8 °C to 2.2 °C, yielding a sensitivity of 89% and an overall accuracy of 80%. Notably, integrating ΔT with a structured clinical certainty score improved the diagnostic area under the curve to 0.88, further underscoring the additive value of com
Our study is limited by its retrospective design, modest sample size, and heterogeneous pathology spectrum. In par
IRT may serve as a useful adjunctive tool for the functional assessment of patients undergoing spinal surgery. In this study, preoperative thermal asymmetry showed correlation with symptomatic levels, and postoperative changes sug
| 1. | Mensah EO, Chalif JI, Baker JG, Chalif E, Biundo J, Groff MW. Challenges in Contemporary Spine Surgery: A Comprehensive Review of Surgical, Technological, and Patient-Specific Issues. J Clin Med. 2024;13:5460. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 21] [Article Influence: 10.5] [Reference Citation Analysis (1)] |
| 2. | Kim GU, Chang MC, Kim TU, Lee GW. Diagnostic Modality in Spine Disease: A Review. Asian Spine J. 2020;14:910-920. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 55] [Article Influence: 9.2] [Reference Citation Analysis (1)] |
| 3. | Bagavathiappan S, Saravanan T, Philip J, Jayakumar T, Raj B, Karunanithi R, Panicker TM, Korath MP, Jagadeesan K. Infrared thermal imaging for detection of peripheral vascular disorders. J Med Phys. 2009;34:43-47. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 144] [Cited by in RCA: 96] [Article Influence: 5.6] [Reference Citation Analysis (5)] |
| 4. | Cañada-Soriano M, Priego-Quesada JI, Bovaira M, García-Vitoria C, Salvador Palmer R, Cibrián Ortiz de Anda R, Moratal D. Quantitative Analysis of Real-Time Infrared Thermography for the Assessment of Lumbar Sympathetic Blocks: A Preliminary Study. Sensors (Basel). 2021;21:3573. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 5. | Haddad DS, Brioschi ML, Arita ES. Thermographic and clinical correlation of myofascial trigger points in the masticatory muscles. Dentomaxillofac Radiol. 2012;41:621-629. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 71] [Cited by in RCA: 50] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 6. | Hildebrandt C, Raschner C, Ammer K. An overview of recent application of medical infrared thermography in sports medicine in Austria. Sensors (Basel). 2010;10:4700-4715. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 292] [Cited by in RCA: 164] [Article Influence: 10.3] [Reference Citation Analysis (1)] |
| 7. | Lahiri BB, Bagavathiappan S, Jayakumar T, Philip J. Medical applications of infrared thermography: A review. Infrared Phys Technol. 2012;55:221-235. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1031] [Cited by in RCA: 488] [Article Influence: 34.9] [Reference Citation Analysis (1)] |
| 8. | Zaproudina N, Varmavuo V, Airaksinen O, Närhi M. Reproducibility of infrared thermography measurements in healthy individuals. Physiol Meas. 2008;29:515-524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 127] [Article Influence: 7.1] [Reference Citation Analysis (1)] |
| 9. | Uematsu S, Jankel WR, Edwin DH, Kim W, Kozikowski J, Rosenbaum A, Long DM. Quantification of thermal asymmetry. Part 2: Application in low-back pain and sciatica. J Neurosurg. 1988;69:556-561. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 55] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 10. | Uematsu S, Edwin DH, Jankel WR, Kozikowski J, Trattner M. Quantification of thermal asymmetry. Part 1: Normal values and reproducibility. J Neurosurg. 1988;69:552-555. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 185] [Cited by in RCA: 135] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 11. | Ring EF, Ammer K. Infrared thermal imaging in medicine. Physiol Meas. 2012;33:R33-R46. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 778] [Cited by in RCA: 431] [Article Influence: 30.8] [Reference Citation Analysis (1)] |
| 12. | Kim GN, Zhang HY, Cho YE, Ryu SJ. Differential Screening of Herniated Lumbar Discs Based on Bag of Visual Words Image Classification Using Digital Infrared Thermographic Images. Healthcare (Basel). 2022;10:1094. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 13. | Liu H, Zhu Z, Jin X, Huang P. The diagnostic accuracy of infrared thermography in lumbosacral radicular pain: a prospective study. J Orthop Surg Res. 2024;19:409. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 14. | Muthu S, Kalanchiam GP, Shyam A. Thermographic Insights into Degenerative Spinal Disorders: Evaluating the Diagnostic Potential of Digital Infrared Thermography Imaging. J Orthop Case Rep. 2025;15:6-10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 15. | Park TY, Son S, Lim TG, Jeong T. Hyperthermia associated with spinal radiculopathy as determined by digital infrared thermographic imaging. Medicine (Baltimore). 2020;99:e19483. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 7] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 16. | Park JB, Lee GW. Choosing the right treatment for degenerative cervical myelopathy. J Clin Orthop Trauma. 2025;66:103014. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 17. | Ko JY, Choi H, Suh JH, Park KS, Lee JW, Ryu JS. The comparison of recovery patterns between ischemic spinal cord injury and traumatic spinal cord injury from acute to chronic phase. J Spinal Cord Med. 2021;44:288-298. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (1)] |