©Author(s) (or their employer(s)) 2026.
World J Radiol. Feb 28, 2026; 18(2): 116486
Published online Feb 28, 2026. doi: 10.4329/wjr.v18.i2.116486
Published online Feb 28, 2026. doi: 10.4329/wjr.v18.i2.116486
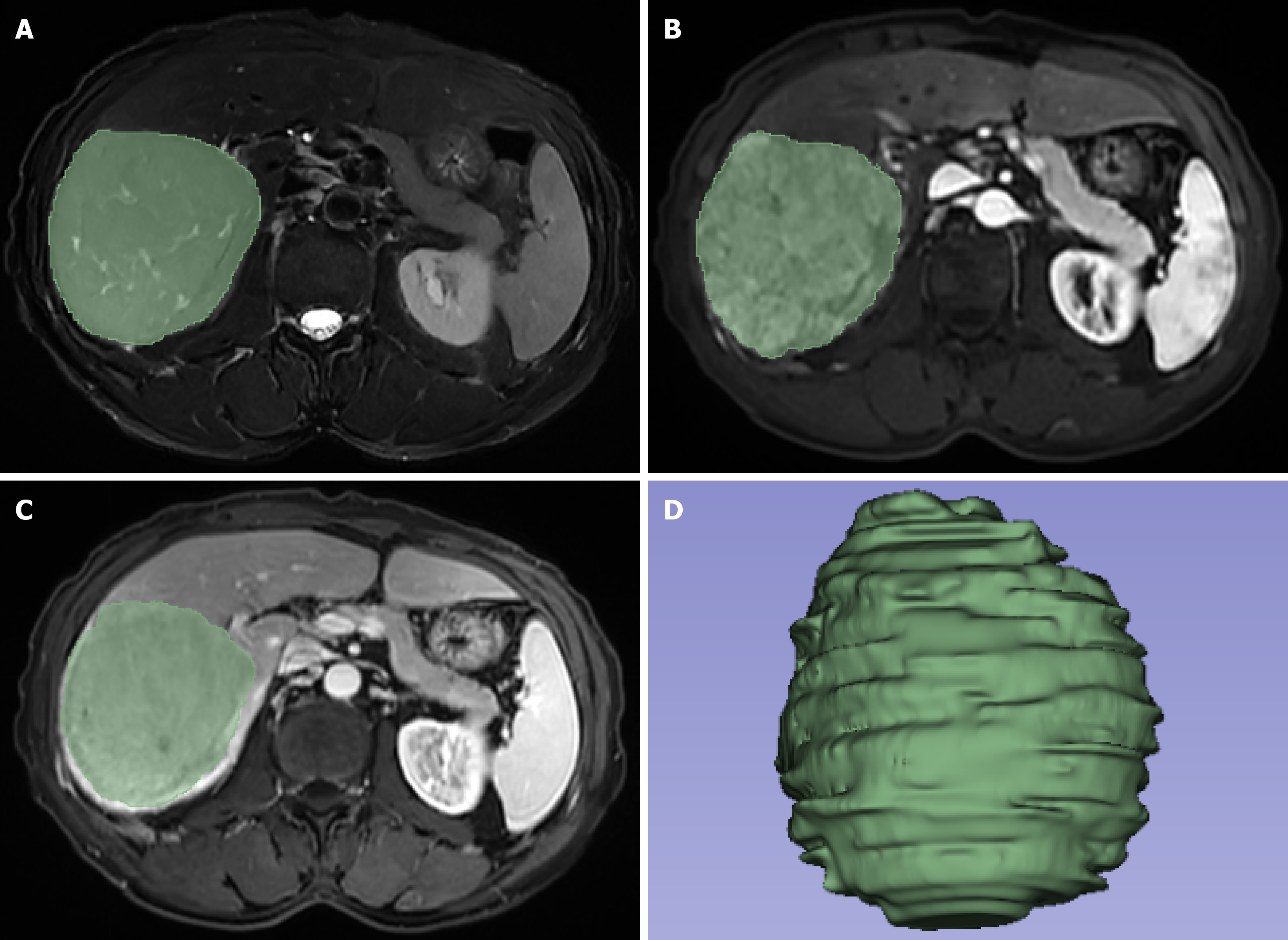
Figure 1 Region of interests along the edge of the lesion.
A: Fat-saturation T2-weighted imaging; B: Arterial phase image; C: Portal venous phase image; D: Three-dimensional image of the lesion.
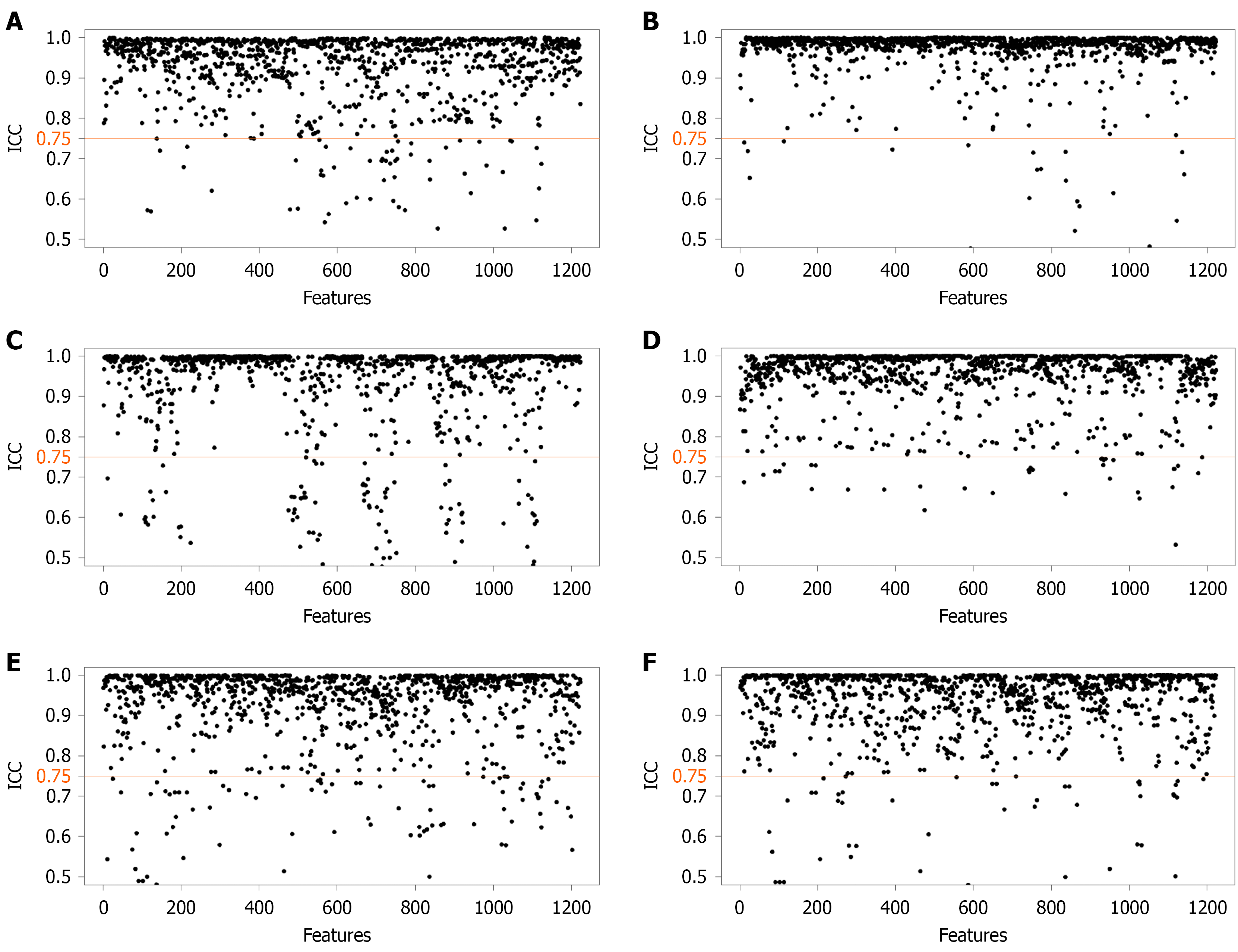
Figure 2 Assessment of the consistency of magnetic resonance imaging radiomic feature extraction via the intraclass correlation coefficient.
A: Inter-observer agreement assessment of fat-saturation T2-weighted imaging; B: Intra-observer reliability assessment of fat-saturation T2-weighted imaging; C: Inter-observer agreement assessment of arterial phase; D: Intra-observer reliability assessment of arterial phase; E: Inter-observer agreement assessment of portal venous phase; F: Intra-observer reliability assessment of portal venous phase. ICC: Intraclass correlation coefficient.
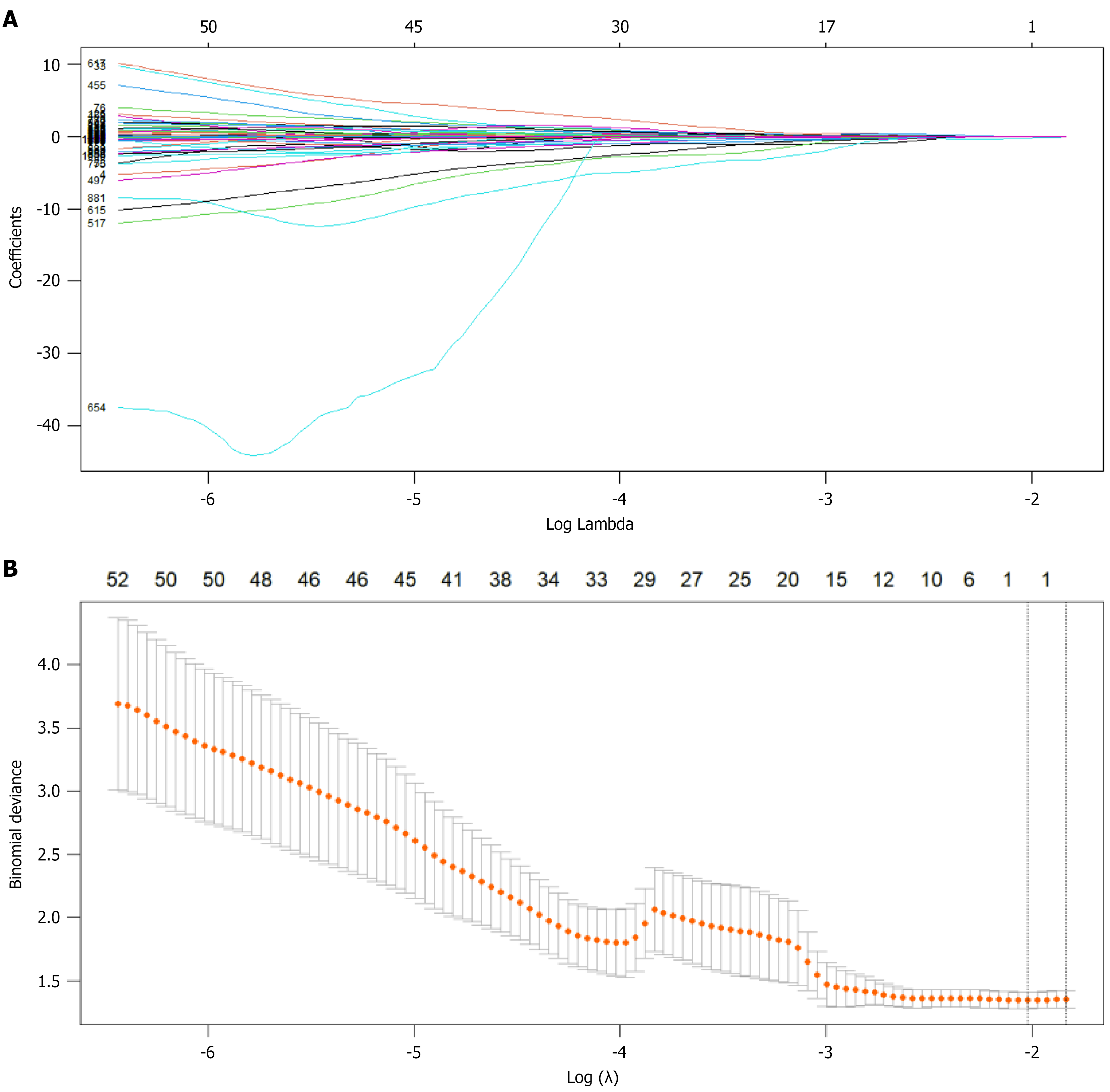
Figure 3 Feature selection via least absolute shrinkage and selection operator regression.
A: Least absolute shrinkage and selection operator regression coefficient path diagram. As the penalty parameter increases, the feature coefficients gradually decrease toward zero. The features with nonzero coefficients at the λ (1-standard error) line were ultimately selected, resulting in 3, 2, and 1 optimal feature from the features of the fat-saturation T2-weighted imaging, arterial phase, and portal venous phase datasets for subsequent model construction; B: Least absolute shrinkage and selection operator regression parameter diagram. The two vertical dashed lines indicate the selected values using cross-validation: The optimal value was obtained by applying the minimum criteria and 1 of the minimum criteria (1-standard error criteria).
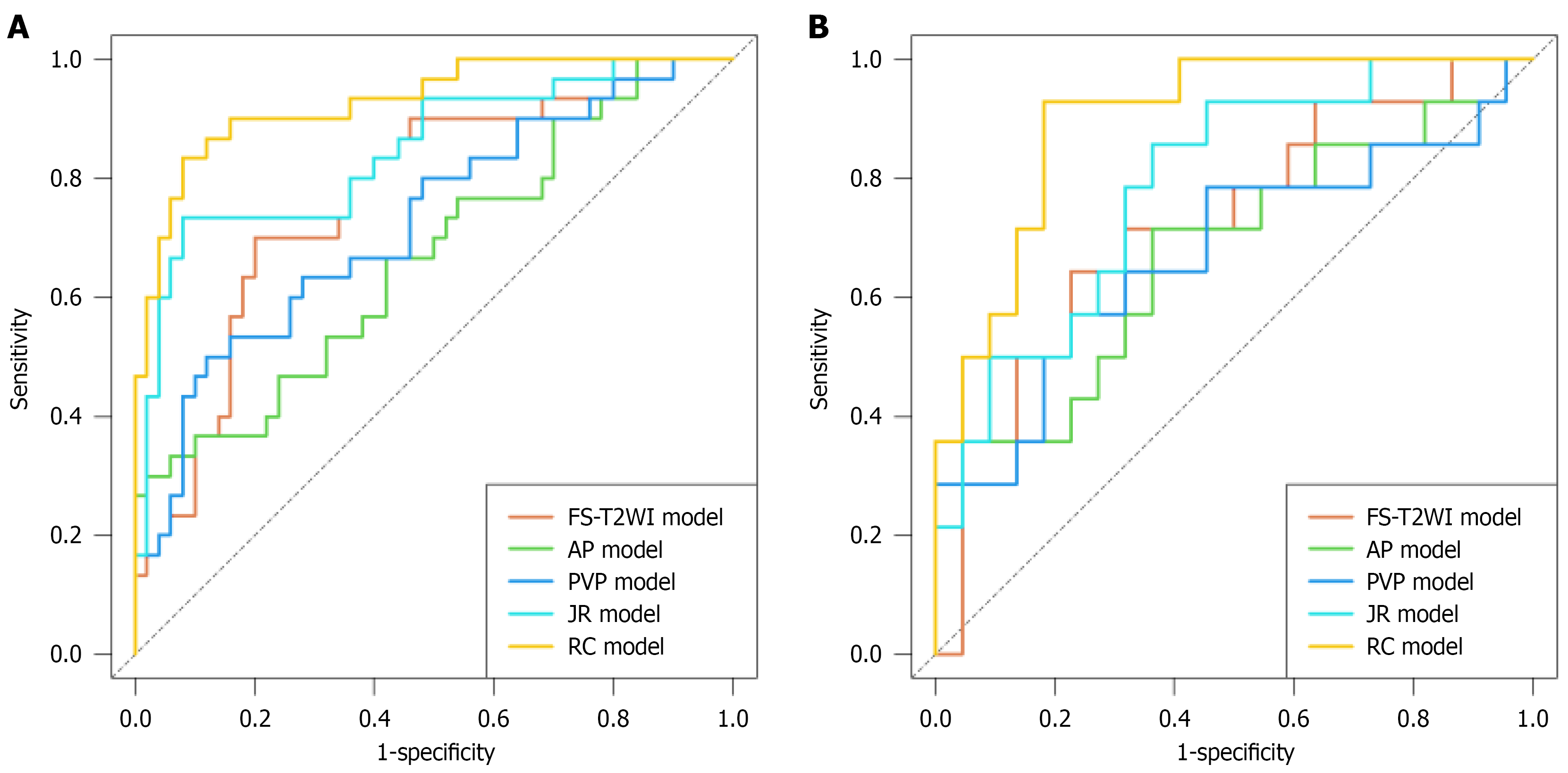
Figure 4 Receiver operating characteristic curves.
The showing the performance of the fat-saturation T2-weighted imaging, arterial phase, portal venous phase, joint-radiomic model, and radiomic-clinical model in predicting responses to transarterial chemoembolization. A: Training group; B: Validation group. FS-T2WI: Fat-saturation T2-weighted imaging; AP: Arterial phase; PVP: Portal venous phase; JR: Joint-radiomic; RC: Radiomic-clinical.
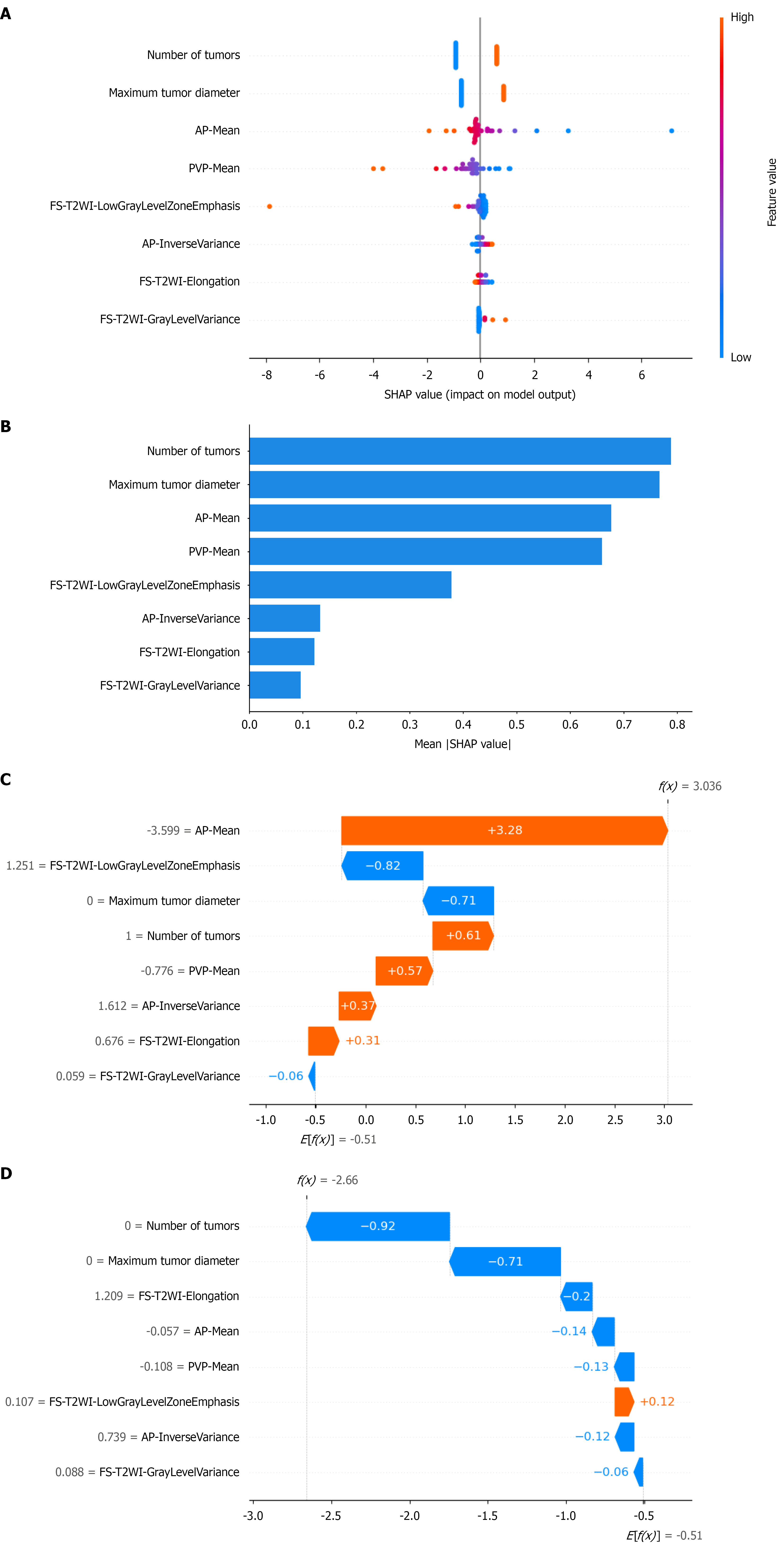
Figure 5 SHapley Additive exPlanations analysis plot of each predictor in the radiomic-clinical model.
A: SHapley Additive exPlanations (SHAP) summary plot shows the distribution of the SHAP values of each predictor in all the samples; B: The SHAP bar plot shows the importance of each predictor in predicting early responses to transarterial chemoembolization (TACE); C: SHAP waterfall plot showing the predicted values for individual samples with the predicted outcome of non-response to TACE; D: SHAP waterfall plot showing the predicted values for individual samples with the predicted outcome of early response to TACE. AP: Arterial phase; PVP: Portal venous phase; FS-T2WI: Fat-saturation T2-weighted imaging; SHAP: SHapley Additive exPlanations.
- Citation: Mao Q, Zhang P, Zhou MT, Shi Y, Min XL, Xu H, Yang L, Zhang XM. Interpretable radiomics model based on magnetic resonance imaging to predict responses to transarterial chemoembolization for hepatocellular carcinoma. World J Radiol 2026; 18(2): 116486
- URL: https://www.wjgnet.com/1949-8470/full/v18/i2/116486.htm
- DOI: https://dx.doi.org/10.4329/wjr.v18.i2.116486