Published online Apr 26, 2026. doi: 10.4330/wjc.v18.i4.118863
Revised: February 24, 2026
Accepted: March 18, 2026
Published online: April 26, 2026
Processing time: 91 Days and 3.3 Hours
Atrial electrode lead dislodgment leading to phrenic nerve capture is rare.
An elderly man with third-degree atrioventricular block underwent dual-chamber pacemaker implantation. During the procedure, both leads were introduced th
Atrial electrode lead displacement may capture the phrenic nerve, leading to diaphragmatic flutter.
Core Tip: During the dual-chamber pacemaker implantation through the same venous puncture site, a passive lead was positioned in the atrium and an active lead was positioned in the ventricle. The patient developed diaphragmatic flutter postoperatively. Electrophysiological evaluation confirmed poor atrial sensing. The frequency of diaphragmatic flutter varied according to the programmed atrial pacing rate, whereas ventricular sensing and pacing thresholds remained normal. After reprogramming the device to VVI mode, the diaphragmatic flutter resolved completely. X-ray examination revealed atrial lead displacement. The displaced atrial lead triggered diaphragmatic flutter by stimulating the phrenic nerve.
- Citation: Zhang XS, Wang F, Qi XW, Shafii P, Cui XN, Wang YH. Phrenic nerve capture caused by atrial electrode dislodgement: A case report. World J Cardiol 2026; 18(4): 118863
- URL: https://www.wjgnet.com/1949-8462/full/v18/i4/118863.htm
- DOI: https://dx.doi.org/10.4330/wjc.v18.i4.118863
In the early era of pacemaker implantation, most ventricular leads were passive fixation leads placed at the right ventricular apex, which is anatomically close to the diaphragm. High-output ventricular pacing could therefore stimulate both the ventricular myocardium and the diaphragm, resulting in diaphragmatic twitching during or after implantation[1]. Intraoperatively, this complication can usually be resolved by repositioning the ventricular lead, and postoperatively by reducing pacing output.
There have also been reports of ventricular active-fixation leads causing myocardial perforation or dislodgement toward the apex or base of the heart, leading to diaphragmatic twitching[2], as well as cases of micro-dislodgement of a right ventricular screw-in lead resulting in similar symptoms.
Passive atrial leads are typically positioned in the right atrial appendage, which is relatively distant from the diaphragm. Even if dislodgement occurs, diaphragmatic twitching is uncommon[3]. However, diaphragmatic twitching may also result from direct stimulation of the phrenic nerve rather than the diaphragm itself.
A 77-year-old man was admitted with a 4-year history of intermittent dizziness that had worsened, accompanied by presyncope for 1 day.
A 4-year history of intermittent dizziness that had worsened, accompanied by presyncope for 1 day.
No special notes.
No special notes.
No rales in the lungs or heart murmurs were heard. The heart rate was 37 beats per minute with a regular rhythm, and there was no edema in both lower extremities.
The routine blood test, blood lipid test, blood glucose test, electrolyte test, cardiac enzyme test, liver function test, and kidney function test all showed no abnormalities.
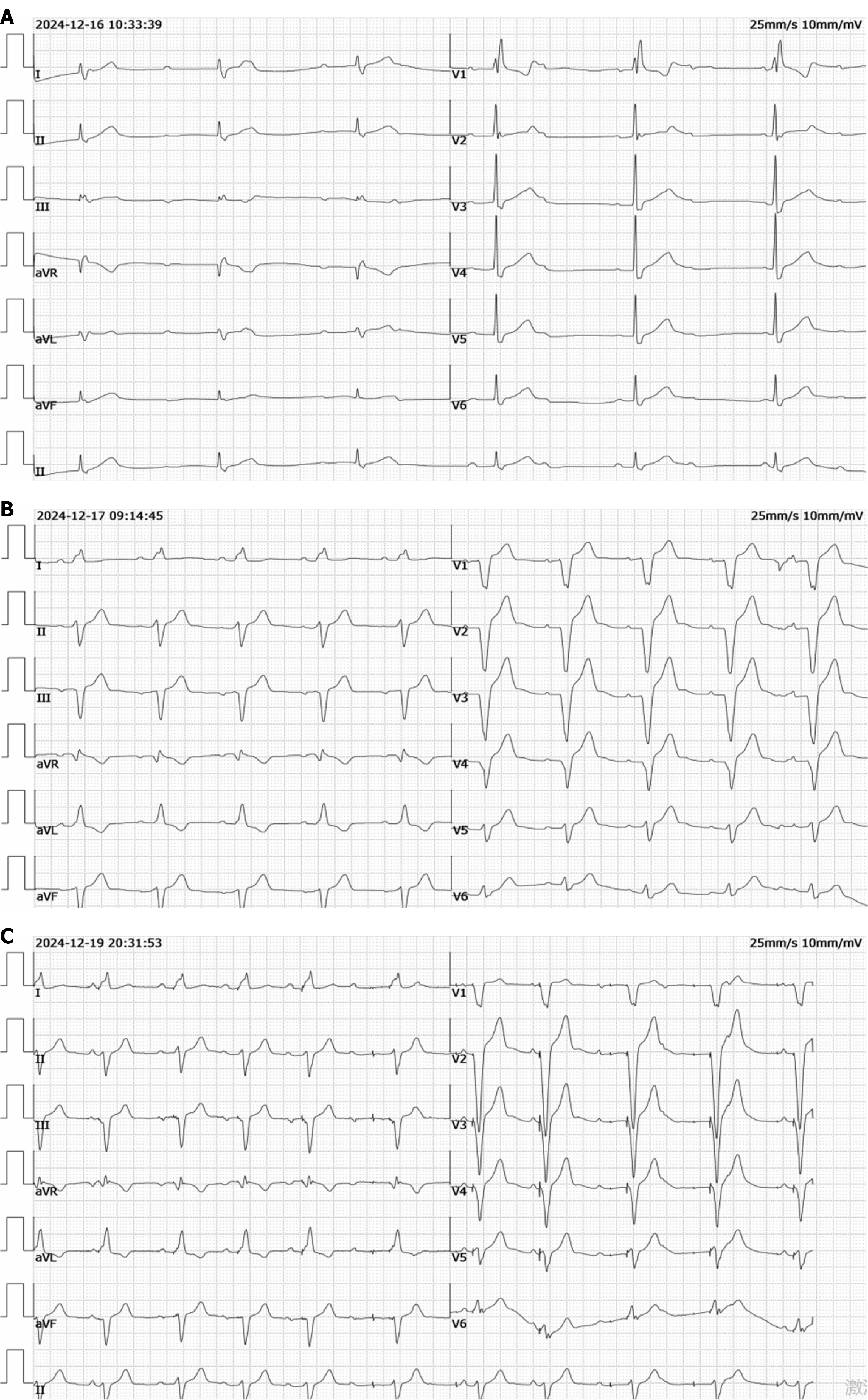
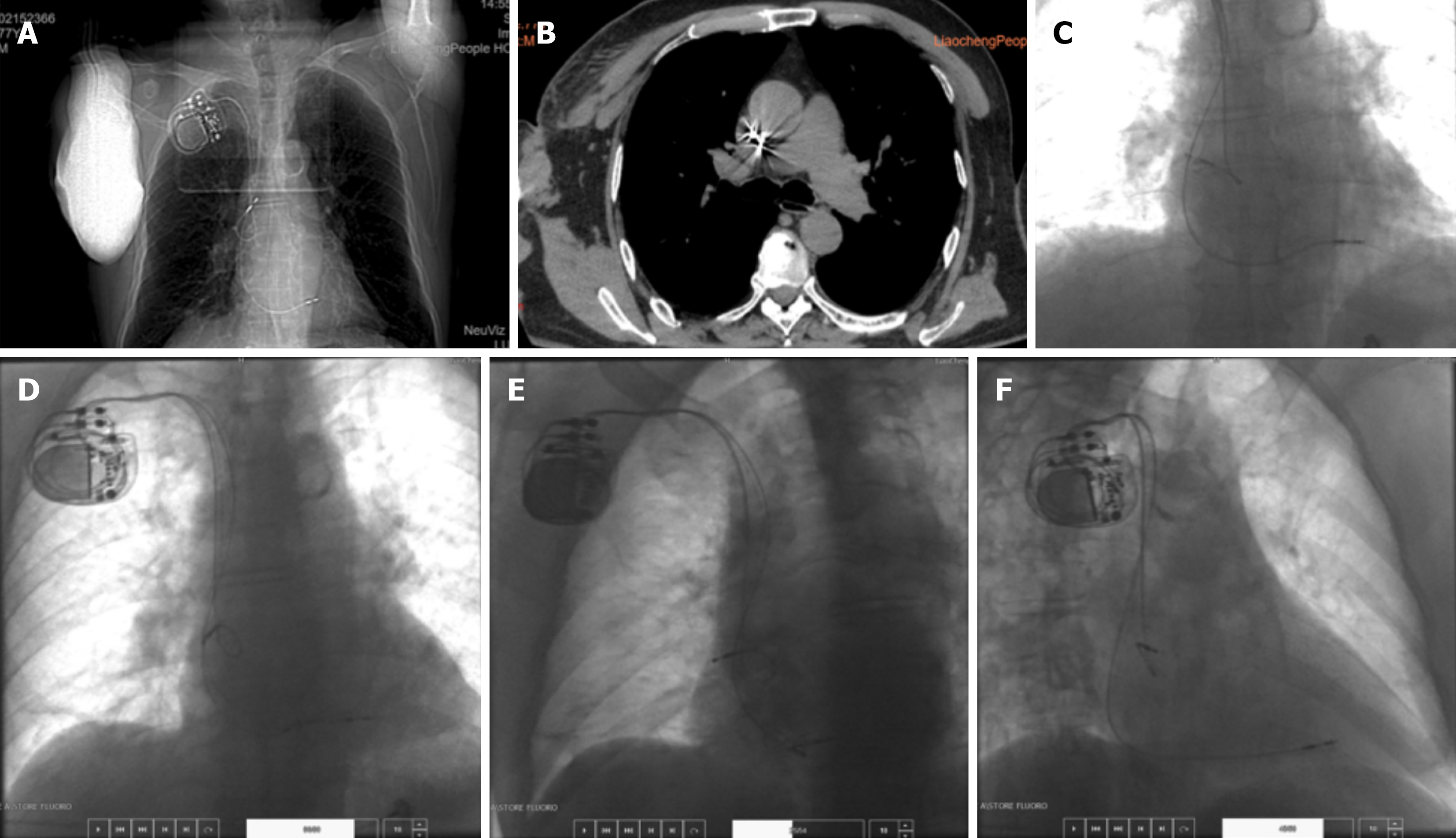
The admission electrocardiogram (ECG) showed third-degree atrioventricular block (III° AVB) (Figure 1), which was considered the cause of his symptoms. A dual-chamber pacemaker implantation was planned under local anesthesia. However, ECG demonstrated poor atrial sensing with normal ventricular sensing and pacing (Figure 1C). Repeat chest radiography and computed tomography (Figure 2A and B) revealed no change in ventricular lead position, but confirmed displacement of the atrial lead compared with its postoperative position. Device interrogation showed loss of atrial sensing. High-output atrial pacing failed to capture the atrium but induced high-frequency diaphragmatic twitching. The frequency of twitching corresponded precisely to the programmed atrial pacing rate. Ventricular impedance, sensing, and threshold remained unchanged, and no diaphragmatic twitching occurred during ventricular threshold testing.
Third-degree atrioventricular block; dual-chamber pacemaker implantation; atrial electrode dislodgement; phrenic nerve capture; and diaphragmatic flutter.
Initial attempts to access the left axillary and left subclavian veins were unsuccessful. The right subclavian vein was then punctured successfully, and the guidewire was advanced into the inferior vena cava. A second attempt at right subclavian vein puncture failed; therefore, we elected to use the same puncture site for both leads.
After skin incision and pocket creation, a sheath was introduced. The inner dilator was removed while the guidewire and outer sheath were maintained in place, and a second guidewire was inserted to facilitate lead placement. Resistance was encountered during sheath advancement, but positioning was ultimately successful. An active-fixation ventricular lead (model 2088TC) was implanted in the mid-interventricular septum, and a passive atrial lead (model 1944) was positioned in the right atrial appendage (Figure 2C). Intraoperative parameters were as follows: The atrial sensing voltage was 2.5 mV, the impedance was 680 Ω, and the threshold was 0.6 V; while the ventricular sensing voltage was 6.7 mV, the impedance was 640 Ω, and the threshold was 0.6 V, with a pulse width of 0.4 millisecond for both leads.
An Abbott PM2224 pulse generator was connected and placed in the pocket. The incision was closed. Routine postoperative anteroposterior, left anterior oblique, and right anterior oblique chest radiographs were obtained (Figure 2D-F). Compared with intraoperative images, the atrial lead showed slight rotation after wound closure; however, pacing parameters remained stable, and the procedure was completed. Lidocaine (100 mg) was used for subcutaneous local anesthesia during the procedure, and cephalosporin antibiotics were used for perioperative infection prevention. Immediate postoperative ECG showed atrial sensing with ventricular pacing (Figure 1B).
On postoperative day 3, the patient reported intermittent pulsations in the upper abdomen. Rhythmic abdominal wall movements were observed below the xiphoid process. Ventricular lead dislodgement with diaphragmatic stimulation was initially suspected.
The pacemaker was reprogrammed to ventricular demand pacing mode, after which the diaphragmatic twitching resolved completely. Ventricular lead dislodgement was excluded, and atrial lead dislodgement with phrenic nerve stimulation was diagnosed. Although lead repositioning was recommended, the patient declined reoperation.
During 6 months of follow-up, no recurrence of diaphragmatic twitching was observed.
Diaphragmatic flutter after cardiac pacemaker implantation is usually triggered by the pacing electrodes. In addition, factors such as perioperative medication exposure, anesthesia strategy, and autonomic or metabolic stressors may plausibly modulate phrenic nerve excitability and pacing thresholds. During local anesthesia, the patient received 100 mg of lidocaine. Lidocaine blocks sodium ion channels and inhibits abnormal excitation of the phrenic or vagus nerve, thereby reducing diaphragmatic excitability and contraction without inducing diaphragmatic flutter. This patient was also administered statins during the perioperative period. Statins can cause diaphragmatic inflammation and weakness but are not known to induce diaphragmatic flutter. This patient had no abnormal serum potassium or sodium titers, which does not support electrolyte imbalance as a cause of abnormal diaphragmatic movement. The patient received cephalosporin antibiotics for infection prophylaxis. Although cephalosporins may stimulate the vagus or phrenic nerve via the bloodstream and cause irregular diaphragmatic contractions, this patient’s diaphragmatic flutter was regular and consistent with the atrial pacing rate.
Pacing output variability, transient post-implantation inflammation, or short-term lead maturation effects should also be taken seriously. As noted above, diaphragmatic flutter can be triggered when the output voltage is too high. In the initial stage of pacemaker electrode implantation, the pacing threshold undergoes dynamic changes. Within 24 hours after implantation, the pacing threshold stabilizes or slightly decreases. From 7 days to 10 days after implantation, the myocardial tissue in contact with the electrode undergoes injury, inflammation, and fibrosis, causing the pacing threshold to rise significantly, generally not exceeding 1.5 V. About 2 weeks after implantation, the pacing threshold usually reaches its peak. Around 3 months after implantation, the pacing threshold gradually decreases and tends to stabilize. In recent years, with the application of technologies such as hormone-coated electrodes, the amplitude of the increase in the acute-phase threshold has decreased. Therefore, although pacing output variability and transient post-implantation inflammation were unlikely to be responsible for the diaphragmatic flutter in this case, they remain factors that warrant attention. In addition, variations in operator technique and patient-specific characteristics may also have contributed to the occurrence of diaphragmatic flutter.
In this case, the ventricular lead was positioned in the mid-septal region. Imaging and device interrogation excluded ventricular lead-related diaphragmatic stimulation.
After originating from the cervical plexus, the phrenic nerve enters the thoracic cavity between the right subclavian artery and vein and descends posterior to the superior vena cava (SVC). At this level, the phrenic nerve lies in close proximity to the atrial aspect of the SVC and is susceptible to electrical stimulation. For example, during radiofrequency isolation of SVC potentials or cryoablation of the right superior pulmonary vein, pacing within the SVC is often performed to localize and protect the phrenic nerve.
In this patient, computed tomography imaging after symptom onset demonstrated that the atrial lead tip had migrated to the left posterior aspect of the SVC (Figure 2B). Device interrogation showed absent atrial sensing and diaphragmatic twitching synchronized with atrial pacing stimuli. These findings strongly suggest that electrical stimulation from the displaced atrial lead directly activated the phrenic nerve, resulting in diaphragmatic flutter. Previous studies have also reported phrenic nerve stimulation when active atrial leads were positioned near the interatrial septum.
In this case, the two electrode leads passed through a single puncture point, resulting in the two electrode leads being in close contact and prone to mutual resistance, thus restricting the natural extension of the atrial electrode lead. At the end of the procedure for this patient, the atrial electrode had shifted clockwise to varying degrees (Figure 2D-F). At this time, the resistance between the leads may have prevented the complete release of the clockwise torsional force of the atrial electrode.
In this case, both leads were introduced through a single venous puncture site. The close proximity of the leads likely caused mechanical interaction and resistance, limiting the natural extension of the atrial lead. At the end of the procedure, slight clockwise rotation of the atrial lead was observed (Figure 2D-F). Residual torsional tension may not have been fully released due to friction between the two leads. Postoperative body movements, deep breathing, or coughing may have further released this torsional force, leading to complete atrial lead dislodgement. The displaced lead subsequently stimulated the adjacent phrenic nerve, producing diaphragmatic twitching.
To prevent the recurrence of such cases, the following key points should be strictly followed: First, a puncture site is selected as lateral as possible, preferably at the distal subclavian or axillary vein, to minimize lead compression at the entry site. Venography may be helpful if necessary. Second, it is important to avoid using a single puncture site for multiple leads to prevent mechanical interaction. Third, active-fixation leads are preferred in the atrium. Fourth, if passive leads are used, leaving excessive slack in the atrium should be avoided, as their preformed curvature increases the risk of dislodgement. Fifth, before final fixation, repeated physiological maneuvers (e.g., coughing, deep breathing) are performed to detect potential instability. Additionally, suturing the lead may cause micro-displacement due to traction. After fixation, confirmatory imaging and reassess pacing parameters should be obtained. Significant dis
Atrial electrode lead dislodgement can capture the phrenic nerve, resulting in diaphragmatic flutter. Some measures should be taken to avoid this complication.
We thank the medical and nursing staff of the catheterization laboratory and the Department of Cardiology for their support.
| 1. | Palmer TE, Finestone AJ, Leary J. Endocardiac pacemaker-induced diaphragmatic contractions. JAMA. 1967;200:1179-1180. [PubMed] |
| 2. | Nguyen D, Nguyen T, Almorad A, De Keyzer E. Right ventricular lead perforation revealed by diaphragmatic stimulation. Intern Emerg Med. 2021;16:783-784. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 3. | Arnsbo P, Møller M. Updated appraisal of pacing lead performance from the Danish Pacemaker Register: the reliability of bipolar pacing leads has improved. Pacing Clin Electrophysiol. 2000;23:1401-1406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 4. | Nicholson WJ, Tuohy KA, Tilkemeier P. Twiddler's Syndrome. N Engl J Med. 2003;348:1726-1727. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 59] [Article Influence: 2.6] [Reference Citation Analysis (2)] |
| 5. | Aniyathodiyil G, Nanjappa V. Reel Syndrome--A Variety of Twiddler's Syndrome. J Assoc Physicians India. 2015;63:65-67. [PubMed] |