Published online Apr 26, 2026. doi: 10.4330/wjc.v18.i4.118879
Revised: February 14, 2026
Accepted: March 19, 2026
Published online: April 26, 2026
Processing time: 91 Days and 12.9 Hours
Chronic kidney disease (CKD) and heart failure (HF) frequently coexist, creating a complex clinical challenge. Mineralocorticoid receptor antagonists (MRAs) have shown promise in managing both conditions, primarily by mitigating the adverse effects of aldosterone. Finerenone, a novel nonsteroidal MRA, has demonstrated significant benefits in reducing cardiovascular and renal events in patients with CKD and type 2 diabetes. However, while the efficacy and safety of finerenone have been extensively evaluated in CKD popu
To investigate the efficacy and safety of finerenone in patients with HF.
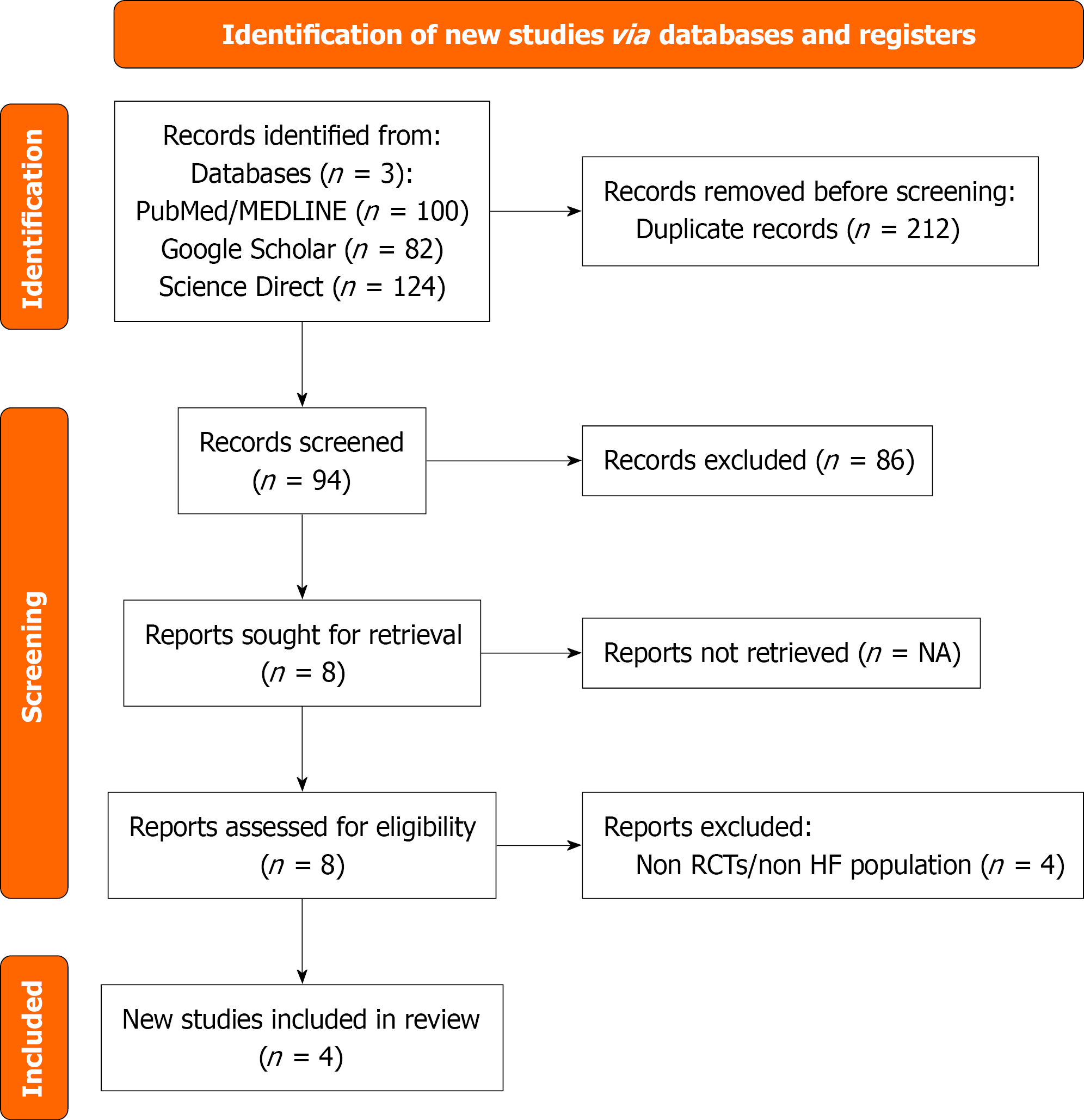
Per the PRISMA guidelines, a thorough search was conducted in PubMed/MEDLINE, Science Direct, and Google Scholar, and relevant randomized control trials (RCTs) comparing finerenone to placebo or other agents on clinical outcomes in patients with HF were included.
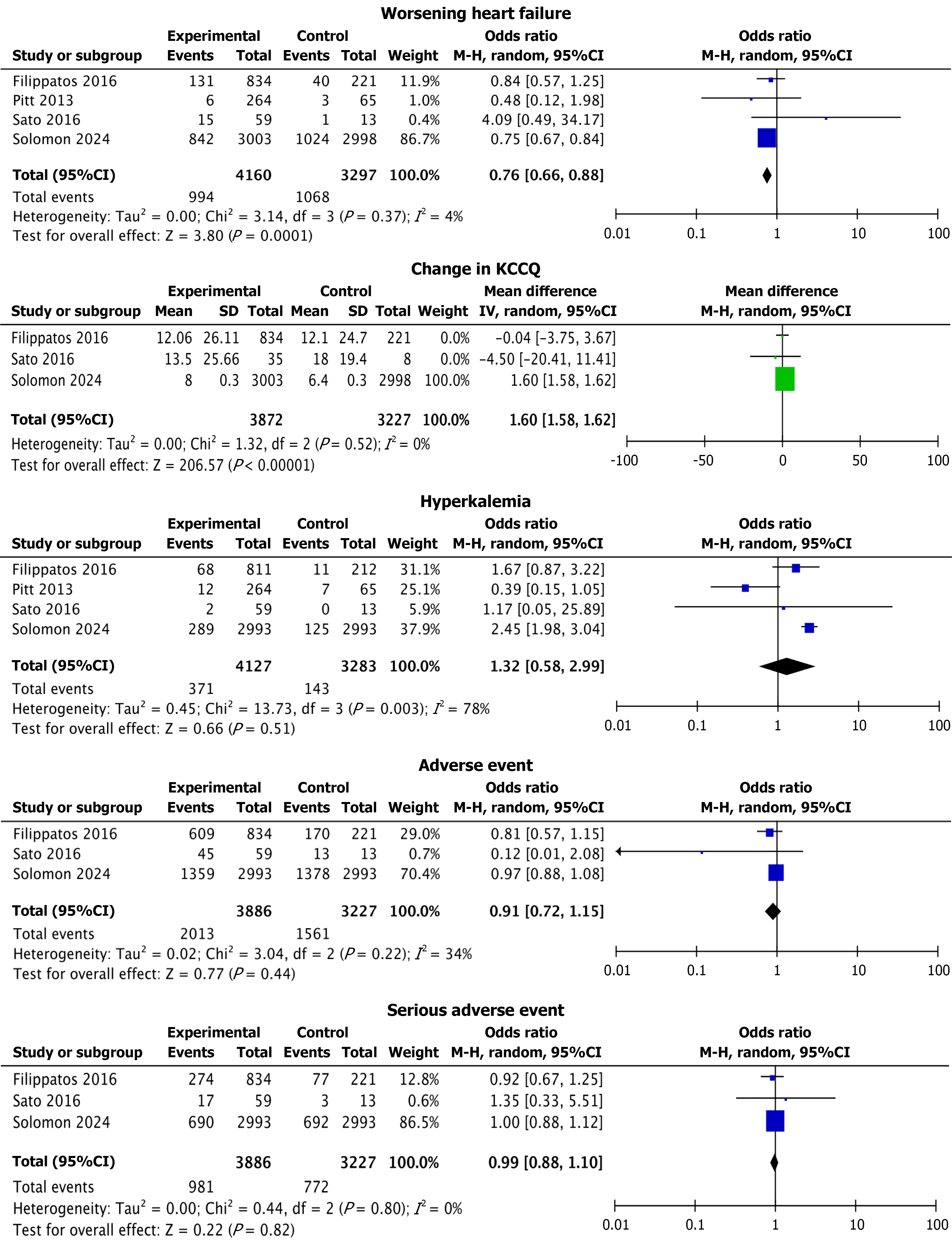
Four RCTs involving 7457 patients with HF (mean age, 71.85 years; females, 41.0%) were included in the study. Of them, 4160 received finerenone (mean age, 71.7 years) and 3297 received a placebo (mean age, 72.04 years). The finerenone group had significantly lower odds of worsening HF [odds ratio (OR) = 0.76; 95%CI: 0.66-0.88; P = 0.0001] and improvement in the Kansas City Cardiomyopathy Questionnaire (mean differences: 1.60; 95%CI: 1.58-1.62; P < 0.0001). Furthermore, higher odds of hyperkalemia (OR = 1.32; 95%CI: 0.58-2.99; P = 0.51) and lower odds of adverse event (AE) (OR = 0.91; 95%CI: 0.72-1.15; P = 0.44) and severe AE (OR = 0.99; 95%CI: 0.88-1.10; P = 0.82).
Our meta-analysis demonstrates that finerenone significantly reduces the risk of worsening of HF and improves quality of life in HF. However, finerenone was associated with increased risks of hyperkalemia, requiring clinical vigilance. Future research should focus on long-term outcomes and the optimal dosing of finerenone in specific HF subpopulations, including those receiving other guideline-directed medical therapies.
Core Tip: Finerenone, a novel nonsteroidal mineralocorticoid receptor antagonist, has shown clear benefits in chronic kidney disease, but its specific impact on heart failure (HF) has required further synthesis. Our meta-analysis of 7457 patients reveals that finerenone significantly decreases the odds of worsening HF and enhances quality of life scores. However, potential safety signals, including hyperkalemia, suggest that while finerenone is a potent tool for managing HF, its use requires vigilant monitoring to balance clinical benefits against adverse events.
- Citation: Mylavarapu M, Kiyani M, Ndakotsu A, Vats V, Karnan N, Palaparthi EC, Anjum M, Kodali LSM, Jamshed A, Cabrera FEP. Clinical outcomes of finerenone in heart failure: A systematic review and meta-analysis. World J Cardiol 2026; 18(4): 118879
- URL: https://www.wjgnet.com/1949-8462/full/v18/i4/118879.htm
- DOI: https://dx.doi.org/10.4330/wjc.v18.i4.118879
Heart failure (HF) remains one of the most common and challenging cardiovascular conditions worldwide. It affects over 64 million people and continues to drive hospitalizations, morbidity, and mortality despite major strides in both medications and devices[1,2]. While significant progress has been made in managing HF, outcomes remain suboptimal across all ejection fraction phenotypes[3].
Guideline-directed medical therapy (GDMT) has improved prognosis by targeting neurohormonal pathways, particularly the renin-angiotensin-aldosterone system[4]. Mineralocorticoid receptor antagonists (MRAs), such as spironolactone and eplerenone, are important components of GDMT and have demonstrated significant benefit in reducing morbidity and mortality in patients with HF with reduced ejection fraction (HFrEF)[5,6]. However, in real-world practice, their use is often limited due to hyperkalemia, worsening renal function, and endocrine-related side effects including gynecomastia and menstrual irregularities[7,8].
Finerenone, a newer nonsteroidal selective MRA, has gained attention as a potential alternative. It has greater mineralocorticoid receptor selectivity and exhibits both anti-fibrotic and anti-inflammatory properties, with a more favorable safety profile compared to traditional steroidal MRAs[9,10]. Importantly, finerenone has reduced affinity for androgen and progesterone receptors, which may help mitigate hormonal side effects[10]. The FIDELIO-DKD and FIGARO-DKD trials demonstrated that finerenone significantly reduced cardiovascular and renal events in patients with chronic kidney disease (CKD) and type 2 diabetes, including HF hospitalizations[11,12].
However, its role in broader HF populations, including those with different ejection fraction phenotypes and co
Per the PRISMA guidelines[15,16], a comprehensive literature search was conducted in prominent databases, including PubMed/MEDLINE, Google Scholar, and Science Direct. Subject headings and keywords for “Heart Failure”, “HFrEF”, “HFpEF”, “Finerenone”, and “Nonsteroidal MRA” were used along with appropriate Boolean operators. The references of the selected studies were also examined to verify the comprehensiveness of the search, using the snowballing method. The search strategy utilized for the study is outlined in Supplementary Table 1.
Screening of the title and abstract was done independently by two reviewers, Ndakotsu A and Palaparthi EC, and conflicts were resolved by a third reviewer, Kiyani M. Full-text screening was done independently by two reviewers, Anjum M and Kodali LSM, and conflicts were resolved by a third reviewer, Mylavarapu M. RCTs that reported clinical outcomes in patients with HF treated with finerenone were included in our analysis. A detailed list of inclusion and exclusion criteria was outlined in Supplementary Table 2.
The risk of bias assessment was conducted using the adaptations of the risk of bias in randomized trials by Cochrane Library[17]. Data extraction was carried out by Kiyani M and the extraction was validated by Mylavarapu M. Outcomes were divided into efficacy, consisting worsening HF (WHF; hospitalization for HF or urgent HF-related clinical deterioration) and change in Change in Kansas City Cardiomyopathy Questionnaire (KCCQ) and safety outcomes, including hyperkalemia, adverse event (any registered adverse event in the trial), and serious adverse event (SAE; grade 3-5 adverse event). Binary random effects were used to estimate the odds ratio (OR), and continuous random effects were used to estimate the mean differences (MD). Heterogeneity was assessed using the (χ2) test and I2 statistics. The I2 statistic was interpreted as follows: < 25% (low heterogeneity), 25%-50% (moderate heterogeneity), and > 50% (high heterogeneity). A leave-one-out (LOO) sensitivity analysis was performed to address the heterogeneity and test for the robustness of the results. Visual inspection of funnel plots was utilized to estimate for publication bias. All the statistical analyses were performed using the Review Manager version 5.4.1. A P value < 0.05 was considered to be statistically significant.
In total, four RCTs[18-21] with 7457 patients (41% females; 71.85 mean age) were included in our analysis. The two groups were similar to the control group in terms of age (mean age 71.7 vs 72.04). However, the finerenone group had lesser number of females compared to the control group (1613, 38.77% vs 1444, 43.8%). Figure 1 depicts the study selection process[22]. Table 1 summarizes the baseline characteristics of the included studies.
| Ref. | Year | Study type | Patient population | Age | Sex (female) | |||
| Finerenone | Control | Finerenone | Control | Finerenone | Control | |||
| Solomon et al[18] | 2024 | RCT | 3003 | 2998 | 71.9 ± 9.6 | 72.0 ± 9.7 | 1355 | 1377 |
| Filippatos et al[19] | 2016 | RCT | 834 | 221 | 70.83 ± 10.17 | 72.4 ± 9.9 | 188 | 51 |
| Sato et al[20] | 2016 | RCT | 59 | 13 | 76.11 ± 9.06 | 76.5 ± 10.89 | 18 | 1 |
| Pitt et al[21] | 2013 | RCT | 264 | 65 | 71.27 ± 8.24 | 71.74 ± 7.54 | 52 | 15 |
Finerenone group had significantly lower odds of WHF (OR = 0.76; 95%CI: 0.66-0.88; P = 0.0001; I2 = 4%) and impro
Regarding safety outcomes, although the finerenone group had higher odds of hyperkalemia (OR = 1.32; 95%CI: 0.58-2.99; P = 0.51), the odds of AEs (OR = 0.91; 95%CI: 0.72-1.15; P = 0.44) and SAEs (OR = 0.99; 95%CI: 0.88-1.10; P = 0.82) compared to the control group were lower. However, these outcomes did not reach statistical significance (Figure 2).
Our analyses revealed low heterogeneity in most of outcomes assessed, including WHF (I2 = 4%), improvement in KCCQ (I2 = 0%), and SAE (I2 = 0%), except hyperkalemia (I2 = 78%). Supplementary Figure 1 and Supplementary Table 3 outlines the heterogeneity assessment and LOO sensitivity analyses, respectively. Regarding risk of bias assessment, all of the included studies demonstrated a low risk across most domains except for D3: Missing Outcomes Data (1/4 studies) and D4: Measurements of Outcomes (1/4 studies) for the majority of the literature. Supplementary Table 4 reports the risk of bias assessment of the included studies. Supplementary Figures 2-6 outlines the funnel plots for publication bias.
To our knowledge, this is the most comprehensive meta-analysis to date evaluating finerenone in HF, comprising four RCTs with a total population of 7457 patients. Our pooled analysis demonstrates that finerenone, when added to standard-of-care, yields clear improvements in WHF events and KCCQ scores. Statistical heterogeneity remained low for most outcomes despite variations in trial design, with the substantial FINE-ARTS HF trial contributing the majority of the analytical weight[18]. While early-phase studies were limited by sample size, they demonstrated consistent trends toward improved biomarkers, including natriuretic peptide reduction, even at lower dosages[19-21].
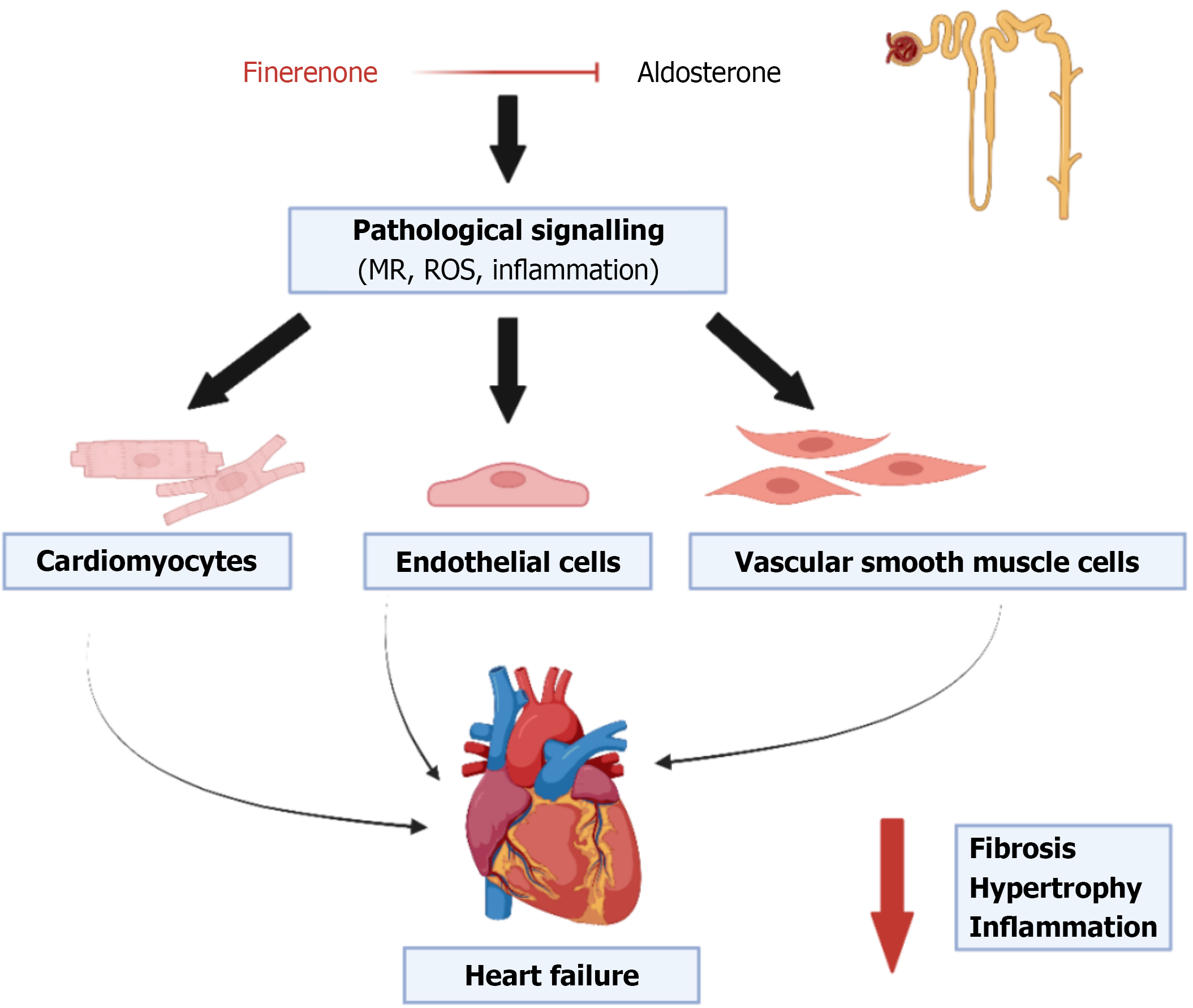
Finerenone has emerged as a pivotal therapeutic option addressing the intersection of cardiovascular, kidney, and metabolic pathologies. While steroidal MRAs are established class I recommendations for HFrEF, their utility in HF with midrange ejection fraction (HFmrEF) and HF with preserved ejection fraction (HFpEF) remained equivocal following the mixed results of the TOPCAT trial[23]. Finerenone possesses a distinct pharmacodynamic profile compared to steroidal MRAs; its nonsteroidal structure confers high affinity and selectivity for the mineralocorticoid receptor with negligible activity at androgen receptors. This distinct receptor interplay promotes a balanced cardiorenal distribution, effectively attenuating myocardial and renal fibrosis, inflammation, and oxidative stress[24]. By inhibiting mineralocorticoid receptor overactivation, finerenone prevents the recruitment of transcriptional cofactors responsible for the expression of proinflammatory and profibrotic genes[25]. A detailed mechanism of action is illustrated in Figure 3.
The reduction in WHF events, comprising urgent visits and hospitalizations, is clinically paramount, as recurrent decompensation drives progressive myocardial injury and mortality[26]. In the FINE-ARTS HF trial, finerenone reduced the composite of WHF and cardiovascular mortality by 16%, a benefit primarily driven by the prevention of WHF[18]. Notably, these benefits were consistent across the ejection fraction spectrum, underscoring finerenone’s efficacy in phenotypes with historically limited therapeutic options. Earlier dose-finding studies, such as ARTS-HF and ARTS-HF Japan, corroborated these findings, showing that lower doses (5-10 mg) reduced natriuretic peptides and cardiovascular events comparably to eplerenone, yet with a more favorable safety profile regarding hyperkalemia[27]. Similarly, Pei et al[28] reported that 10 mg of finerenone elicited anti-remodeling effects equivalent to 25-50 mg of steroidal MRAs, while maintaining higher epidermal growth factor receptor (eGFR) levels and lower serum potassium, suggesting superior cardiorenal protection.
Our findings align with decades of evidence supporting mineralocorticoid receptor antagonism in HF. The landmark RALES trial established that spironolactone reduces all-cause mortality in HFrEF[29]. Subsequently, EPHESUS demonstrated that eplerenone reduced mortality and hospitalization in post-myocardial infarction patients with left ventricular dysfunction, while EMPHASIS-HF extended these benefits to mildly symptomatic (NYHA II) patients[6,30]. In contrast, the TOPCAT trial, focusing on HFpEF, showed only a modest reduction in hospitalizations at the cost of significant hyperkalemia and renal adverse events[5]. The results of our meta-analysis suggest that finerenone bridges this gap. While the reduction in WHF is robust, head-to-head comparisons with steroidal MRAs across HFrEF and HFpEF phenotypes are required to definitively establish its comparative efficacy.
Our analysis revealed statistically significant improvements in KCCQ scores with finerenone. Although the mean improvement remains numerically below the 5-point threshold often cited as the minimal clinically important difference for individuals, the population-level significance is substantial. Health-status improvements are incremental; indeed, many foundational HF therapies produce modest KCCQ gains of 1-3 points[31]. Observational data from HF-ACTION indicate that KCCQ shifts are prognostically potent: A 5-point decline correlates with a 7% increase in mortality/hospitalization risk and quantifiable reductions in functional capacity[31,32]. In FINE-ARTS HF, finerenone not only improved total KCCQ scores but also preserved NYHA class better than placebo, indicating a tangible attenuation of symptom progression[18].
Clinically, while a 1-2 points KCCQ gain may be imperceptible daily to an individual, it reflects a meaningful shift in symptom burden across a population that may accumulate over time. Importantly, these quality-of-life benefits were achieved without a concomitant increase in severe adverse events. As the KCCQ captures physical limitations, symptom frequency, and social participation, even modest improvements facilitate better adherence to GDMT and maintenance of functional independence. Clinicians should view finerenone not as a standalone solution for symptom management, but as a component of comprehensive therapy that improves overall health status while reducing clinical events.
The safety profile of finerenone requires nuanced interpretation, particularly regarding hyperkalemia. In our meta-analysis, the OR for hyperkalemia was elevated but not statistically significant. Granular data from FINE-ARTS HF showed that while finerenone increased potassium levels > 5.5 mmol/L (14.3% vs 6.9%) and > 6.0 mmol/L (3.0% vs 1.4%) compared to placebo, the clinical consequences were minimal. Hyperkalemia-related hospitalization occurred in only 0.5% of the treatment arm, with no attributed deaths[18]. This dissociation between laboratory hyperkalemia and adverse clinical outcomes suggests that with routine monitoring and dose modification, the risk is manageable and does not negate the cardiovascular benefit[33]. As expected, patients with lower baseline eGFR (< 45 mL/minute/1.73 m2) exhibited higher rates of hyperkalemia compared to those with preserved renal function[25].
Comparatively, finerenone demonstrates a superior safety profile to steroidal MRAs. In the ARTS trial, finerenone resulted in significantly lower rates of hyperkalemia (5.3% vs 12.7%) and fewer treatment discontinuations than spironolactone[34]. This safety advantage is likely attributable to finerenone’s nonsteroidal structure, optimizing tissue distribution and minimizing off-target hormonal effects. In FINE-ARTS HF, discontinuation rates were virtually identical between finerenone (20.4%) and placebo (20.6%), reinforcing its tolerability. Minor signals for increased creatinine and hypotension were observed but did not translate into SAE[35].
The integration of finerenone with SGLT2 inhibitors represents a significant advancement in quadruple therapy. Prespecified analyses from FINE-ARTS HF confirm that finerenone’s efficacy is independent of background SGLT2 inhibitor use (P = 0.76), with recent data suggesting additive cardiovascular protection when combined[36]. The CONFIDENCE trial further supports this combination, demonstrating a 52% reduction in urinary albumin-to-creatinine ratio, implying a synergistic effect on renal preservation[37]. Modeling studies project that the combination of SGLT2 inhibitors, GLP-1 receptor agonists, and finerenone could extend major cardiovascular and kidney event-free survival by several years[38].
A primary strength of this meta-analysis is the inclusion of the recent, large-scale FINE-ARTS HF trial, which provides high-fidelity evidence for finerenone in HFmrEF/HFpEF[18]. Large trials minimize bias and improve the precision of treatment effect estimates[39]. Furthermore, the consistency of benefit across prespecified subgroups-including ejection fraction and concurrent SGLT2 inhibitor use enhances the external validity of our results[18,40-42].
However, limitations exist. The relatively small number of eligible RCTs constrains the power of subgroup analyses and the detection of rare safety signals[43,44]. While statistical heterogeneity was low, this must be interpreted with caution given the low power of Cochran’s Q in small meta-analyses[42]. Additionally, our results are heavily influenced by FINE-ARTS HF; while robust, reliance on a single dominant trial limits the assessment of consistency across diverse clinical settings[39]. Finally, the median follow-up of 32 months may not fully capture the long-term cumulative benefits or late-emerging risks inherent to lifelong HF therapy[43-45].
This meta-analysis establishes that finerenone significantly improves clinical outcomes in patients with HF, particularly those with mildly reduced or preserved ejection fraction, a phenotype with historically few evidence-based therapies. The consistency of benefit across the ejection fraction spectrum and regardless of background SGLT2 inhibitor use supports finerenone as a high-value addition to GDMT. The safety profile is favorable, with hyperkalemia proving manageable and rarely leading to treatment discontinuation. Future research should prioritize long-term outcome data and real-world effectiveness. Ongoing trials, including REDEFINE-HF, CONFIRMATION-HF, and FINALITY-HF, will further elucidate the role of finerenone across the continuum of HF.
| 1. | Shahim B, Kapelios CJ, Savarese G, Lund LH. Global Public Health Burden of Heart Failure: An Updated Review. Card Fail Rev. 2023;9:e11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 358] [Reference Citation Analysis (1)] |
| 2. | Lippi G, Sanchis-gomar F. Global epidemiology and future trends of heart failure. AME Med J. 2020;5:15-15. [DOI] [Full Text] |
| 3. | Roger VL. Epidemiology of heart failure. Circ Res. 2013;113:646-659. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1039] [Cited by in RCA: 1228] [Article Influence: 94.5] [Reference Citation Analysis (1)] |
| 4. | Heidenreich PA, Bozkurt B, Aguilar D, Allen LA, Byun JJ, Colvin MM, Deswal A, Drazner MH, Dunlay SM, Evers LR, Fang JC, Fedson SE, Fonarow GC, Hayek SS, Hernandez AF, Khazanie P, Kittleson MM, Lee CS, Link MS, Milano CA, Nnacheta LC, Sandhu AT, Stevenson LW, Vardeny O, Vest AR, Yancy CW; ACC/AHA Joint Committee Members. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2022;145:e895-e1032. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1736] [Cited by in RCA: 1858] [Article Influence: 464.5] [Reference Citation Analysis (2)] |
| 5. | Pitt B, Zannad F, Remme WJ, Cody R, Castaigne A, Perez A, Palensky J, Wittes J. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized Aldactone Evaluation Study Investigators. N Engl J Med. 1999;341:709-717. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7483] [Cited by in RCA: 6058] [Article Influence: 224.4] [Reference Citation Analysis (5)] |
| 6. | Zannad F, McMurray JJ, Krum H, van Veldhuisen DJ, Swedberg K, Shi H, Vincent J, Pocock SJ, Pitt B; EMPHASIS-HF Study Group. Eplerenone in patients with systolic heart failure and mild symptoms. N Engl J Med. 2011;364:11-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2600] [Cited by in RCA: 2166] [Article Influence: 144.4] [Reference Citation Analysis (6)] |
| 7. | Juurlink DN, Mamdani MM, Lee DS, Kopp A, Austin PC, Laupacis A, Redelmeier DA. Rates of hyperkalemia after publication of the Randomized Aldactone Evaluation Study. N Engl J Med. 2004;351:543-551. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1425] [Cited by in RCA: 1163] [Article Influence: 52.9] [Reference Citation Analysis (4)] |
| 8. | Vardeny O, Wu DH, Desai A, Rossignol P, Zannad F, Pitt B, Solomon SD; RALES Investigators. Influence of baseline and worsening renal function on efficacy of spironolactone in patients With severe heart failure: insights from RALES (Randomized Aldactone Evaluation Study). J Am Coll Cardiol. 2012;60:2082-2089. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 170] [Cited by in RCA: 214] [Article Influence: 15.3] [Reference Citation Analysis (4)] |
| 9. | Fuchs A, Kühl JT, Sigvardsen PE, Knudsen AD, Nilsson EJP, Stisen ZR, Jeppesen JL, Nordestgaard BG, Køber LV, Kofoed KF. Arterial hypertension and morphologic abnormalities of cardiac chambers: results from the Copenhagen General Population Study. J Hypertens. 2021;39:703-710. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 10. | Nicholls M. Australia and colchicine for coronary heart disease. Eur Heart J. 2021;42:367-368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 11. | Bakris GL, Agarwal R, Anker SD, Pitt B, Ruilope LM, Rossing P, Kolkhof P, Nowack C, Schloemer P, Joseph A, Filippatos G; FIDELIO-DKD Investigators. Effect of Finerenone on Chronic Kidney Disease Outcomes in Type 2 Diabetes. N Engl J Med. 2020;383:2219-2229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2319] [Cited by in RCA: 1953] [Article Influence: 325.5] [Reference Citation Analysis (10)] |
| 12. | Pitt B, Filippatos G, Agarwal R, Anker SD, Bakris GL, Rossing P, Joseph A, Kolkhof P, Nowack C, Schloemer P, Ruilope LM; FIGARO-DKD Investigators. Cardiovascular Events with Finerenone in Kidney Disease and Type 2 Diabetes. N Engl J Med. 2021;385:2252-2263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1436] [Cited by in RCA: 1167] [Article Influence: 233.4] [Reference Citation Analysis (8)] |
| 13. | Ruilope LM, Agarwal R, Anker SD, Filippatos G, Pitt B, Rossing P, Sarafidis P, Schmieder RE, Joseph A, Rethemeier N, Nowack C, Bakris GL; FIDELIO-DKD Investigators. Blood Pressure and Cardiorenal Outcomes With Finerenone in Chronic Kidney Disease in Type 2 Diabetes. Hypertension. 2022;79:2685-2695. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 62] [Reference Citation Analysis (1)] |
| 14. | Hageman S, Pennells L, Ojeda F, Kaptoge S, Dorresteijn J, Di Angelantonio E; SCORE2 working group and ESC Cardiovascular Risk Collaboration. SCORE2 models allow consideration of sex-specific cardiovascular disease risks by region. Eur Heart J. 2022;43:241-242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 15. | Moher D, Liberati A, Tetzlaff J, Altman DG; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6:e1000097. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57226] [Cited by in RCA: 48219] [Article Influence: 2836.4] [Reference Citation Analysis (11)] |
| 16. | Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, Moher D. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9803] [Reference Citation Analysis (0)] |
| 17. | Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, Cates CJ, Cheng HY, Corbett MS, Eldridge SM, Emberson JR, Hernán MA, Hopewell S, Hróbjartsson A, Junqueira DR, Jüni P, Kirkham JJ, Lasserson T, Li T, McAleenan A, Reeves BC, Shepperd S, Shrier I, Stewart LA, Tilling K, White IR, Whiting PF, Higgins JPT. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22257] [Cited by in RCA: 20409] [Article Influence: 2915.6] [Reference Citation Analysis (7)] |
| 18. | Solomon SD, McMurray JJV, Vaduganathan M, Claggett B, Jhund PS, Desai AS, Henderson AD, Lam CSP, Pitt B, Senni M, Shah SJ, Voors AA, Zannad F, Abidin IZ, Alcocer-Gamba MA, Atherton JJ, Bauersachs J, Chang-Sheng M, Chiang CE, Chioncel O, Chopra V, Comin-Colet J, Filippatos G, Fonseca C, Gajos G, Goland S, Goncalvesova E, Kang S, Katova T, Kosiborod MN, Latkovskis G, Lee AP, Linssen GCM, Llamas-Esperón G, Mareev V, Martinez FA, Melenovský V, Merkely B, Nodari S, Petrie MC, Saldarriaga CI, Saraiva JFK, Sato N, Schou M, Sharma K, Troughton R, Udell JA, Ukkonen H, Vardeny O, Verma S, von Lewinski D, Voronkov L, Yilmaz MB, Zieroth S, Lay-Flurrie J, van Gameren I, Amarante F, Kolkhof P, Viswanathan P; FINEARTS-HF Committees and Investigators. Finerenone in Heart Failure with Mildly Reduced or Preserved Ejection Fraction. N Engl J Med. 2024;391:1475-1485. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 550] [Article Influence: 275.0] [Reference Citation Analysis (0)] |
| 19. | Filippatos G, Anker SD, Böhm M, Gheorghiade M, Køber L, Krum H, Maggioni AP, Ponikowski P, Voors AA, Zannad F, Kim SY, Nowack C, Palombo G, Kolkhof P, Kimmeskamp-Kirschbaum N, Pieper A, Pitt B. A randomized controlled study of finerenone vs. eplerenone in patients with worsening chronic heart failure and diabetes mellitus and/or chronic kidney disease. Eur Heart J. 2016;37:2105-2114. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 290] [Cited by in RCA: 338] [Article Influence: 33.8] [Reference Citation Analysis (8)] |
| 20. | Sato N, Ajioka M, Yamada T, Kato M, Myoishi M, Yamada T, Kim SY, Nowack C, Kolkhof P, Shiga T; ARTS-HF Japan study group. A Randomized Controlled Study of Finerenone vs. Eplerenone in Japanese Patients With Worsening Chronic Heart Failure and Diabetes and/or Chronic Kidney Disease. Circ J. 2016;80:1113-1122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 58] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 21. | Pitt B, Kober L, Ponikowski P, Gheorghiade M, Filippatos G, Krum H, Nowack C, Kolkhof P, Kim SY, Zannad F. Safety and tolerability of the novel non-steroidal mineralocorticoid receptor antagonist BAY 94-8862 in patients with chronic heart failure and mild or moderate chronic kidney disease: a randomized, double-blind trial. Eur Heart J. 2013;34:2453-2463. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 309] [Cited by in RCA: 483] [Article Influence: 37.2] [Reference Citation Analysis (8)] |
| 22. | Haddaway NR, Page MJ, Pritchard CC, McGuinness LA. PRISMA2020: An R package and Shiny app for producing PRISMA 2020-compliant flow diagrams, with interactivity for optimised digital transparency and Open Synthesis. Campbell Syst Rev. 2022;18:e1230. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 649] [Cited by in RCA: 1480] [Article Influence: 370.0] [Reference Citation Analysis (5)] |
| 23. | Jhund PS, Talebi A, Henderson AD, Claggett BL, Vaduganathan M, Desai AS, Lam CSP, Pitt B, Senni M, Shah SJ, Voors AA, Zannad F, Solomon SD, McMurray JJV. Mineralocorticoid receptor antagonists in heart failure: an individual patient level meta-analysis. Lancet. 2024;404:1119-1131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 107] [Article Influence: 53.5] [Reference Citation Analysis (1)] |
| 24. | Lima Posada I, Soulié M, Stephan Y, Palacios Ramirez R, Bonnard B, Nicol L, Pitt B, Kolkhof P, Mulder P, Jaisser F. Nonsteroidal Mineralocorticoid Receptor Antagonist Finerenone Improves Diastolic Dysfunction in Preclinical Nondiabetic Chronic Kidney Disease. J Am Heart Assoc. 2024;13:e032971. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (1)] |
| 25. | Mc Causland FR, Vaduganathan M, Claggett BL, Kulac IJ, Desai AS, Jhund PS, Henderson AD, Brinker M, Perkins R, Scheerer MF, Schloemer P, Lam CSP, Senni M, Shah SJ, Voors AA, Zannad F, Pitt B, McMurray JJV, Solomon SD. Finerenone and Kidney Outcomes in Patients With Heart Failure: The FINEARTS-HF Trial. J Am Coll Cardiol. 2025;85:159-168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 51] [Article Influence: 51.0] [Reference Citation Analysis (0)] |
| 26. | Huusko J, Tuominen S, Studer R, Corda S, Proudfoot C, Lassenius M, Ukkonen H. Recurrent hospitalizations are associated with increased mortality across the ejection fraction range in heart failure. ESC Heart Fail. 2020;7:2406-2417. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 38] [Article Influence: 6.3] [Reference Citation Analysis (1)] |
| 27. | Khullar D, Gupta AK, Singh K. Finerenone: Will It Be a Game-changer? Card Fail Rev. 2024;10:e19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 28. | Pei H, Wang W, Zhao D, Wang L, Su GH, Zhao Z. The use of a novel non-steroidal mineralocorticoid receptor antagonist finerenone for the treatment of chronic heart failure: A systematic review and meta-analysis. Medicine (Baltimore). 2018;97:e0254. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 46] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 29. | Pitt B, Pfeffer MA, Assmann SF, Boineau R, Anand IS, Claggett B, Clausell N, Desai AS, Diaz R, Fleg JL, Gordeev I, Harty B, Heitner JF, Kenwood CT, Lewis EF, O'Meara E, Probstfield JL, Shaburishvili T, Shah SJ, Solomon SD, Sweitzer NK, Yang S, McKinlay SM; TOPCAT Investigators. Spironolactone for heart failure with preserved ejection fraction. N Engl J Med. 2014;370:1383-1392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2427] [Cited by in RCA: 2092] [Article Influence: 174.3] [Reference Citation Analysis (4)] |
| 30. | Pitt B, Remme W, Zannad F, Neaton J, Martinez F, Roniker B, Bittman R, Hurley S, Kleiman J, Gatlin M; Eplerenone Post-Acute Myocardial Infarction Heart Failure Efficacy and Survival Study Investigators. Eplerenone, a selective aldosterone blocker, in patients with left ventricular dysfunction after myocardial infarction. N Engl J Med. 2003;348:1309-1321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4141] [Cited by in RCA: 3362] [Article Influence: 146.2] [Reference Citation Analysis (4)] |
| 31. | Stogios N, Fezza G, Wong JV, Ross HJ, Farkouh ME, Nolan RP. Current challenges for using the Kansas City Cardiomyopathy Questionnaire to obtain a standardized patient-reported health status outcome. Eur J Heart Fail. 2021;23:205-207. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 32. | O'Connor CM, Whellan DJ, Lee KL, Keteyian SJ, Cooper LS, Ellis SJ, Leifer ES, Kraus WE, Kitzman DW, Blumenthal JA, Rendall DS, Miller NH, Fleg JL, Schulman KA, McKelvie RS, Zannad F, Piña IL; HF-ACTION Investigators. Efficacy and safety of exercise training in patients with chronic heart failure: HF-ACTION randomized controlled trial. JAMA. 2009;301:1439-1450. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1686] [Cited by in RCA: 1535] [Article Influence: 90.3] [Reference Citation Analysis (1)] |
| 33. | Vardeny O, Vaduganathan M, Claggett BL, Desai AS, Jhund PS, Lam CSP, Senni M, Shah SJ, Voors AA, Zannad F, Pitt B, Matsumoto S, Merkely B, Zieroth S, Yilmaz MB, Lay-Flurrie J, Viswanathan P, Horvat-Broecker A, Scalise A, McMurray JJV, Solomon SD. Finerenone, Serum Potassium, and Clinical Outcomes in Heart Failure With Mildly Reduced or Preserved Ejection Fraction. JAMA Cardiol. 2025;10:42-48. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 20] [Article Influence: 20.0] [Reference Citation Analysis (1)] |
| 34. | Kaul S. Evidentiary Landscape of Heart Failure Therapies, Regulatory Decisions, and Translation Into Guidelines. J Am Coll Cardiol. 2025;86:610-624. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 35. | Savarese G, Lund LH. Global Public Health Burden of Heart Failure. Card Fail Rev. 2017;3:7-11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2169] [Cited by in RCA: 1866] [Article Influence: 207.3] [Reference Citation Analysis (1)] |
| 36. | Vaduganathan M, Claggett BL, Kulac IJ, Miao ZM, Desai AS, Jhund PS, Henderson AD, Brinker M, Lay-Flurrie J, Viswanathan P, Scheerer MF, Lage A, Lam CSP, Senni M, Shah SJ, Voors AA, Zannad F, Pitt B, McMurray JJV, Solomon SD. Effects of the Nonsteroidal MRA Finerenone With and Without Concomitant SGLT2 Inhibitor Use in Heart Failure. Circulation. 2025;151:149-158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 40] [Article Influence: 40.0] [Reference Citation Analysis (1)] |
| 37. | de Boer IH, Khunti K, Sadusky T, Tuttle KR, Neumiller JJ, Rhee CM, Rosas SE, Rossing P, Bakris G. Diabetes Management in Chronic Kidney Disease: A Consensus Report by the American Diabetes Association (ADA) and Kidney Disease: Improving Global Outcomes (KDIGO). Diabetes Care. 2022;45:3075-3090. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 670] [Cited by in RCA: 558] [Article Influence: 139.5] [Reference Citation Analysis (6)] |
| 38. | Ostrominski JW, Cheng AYY, Nelson AJ, Neuen BL, Sattar N, Tuttle KR, Vaduganathan M. Cardiovascular, kidney, and metabolic health: an actionable vision for heart failure prevention. Lancet. 2025;406:1171-1192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 9] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 39. | Dechartres A, Altman DG, Trinquart L, Boutron I, Ravaud P. Association between analytic strategy and estimates of treatment outcomes in meta-analyses. JAMA. 2014;312:623-630. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 167] [Article Influence: 13.9] [Reference Citation Analysis (1)] |
| 40. | Chang J, Ambrosy AP, Vardeny O, Van Spall HGC, Mentz RJ, Sauer AJ. Mineralocorticoid Antagonism in Heart Failure: Established and Emerging Therapeutic Role. JACC Heart Fail. 2024;12:1979-1993. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 16] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 41. | Cordero CP, Dans AL. Key concepts in clinical epidemiology: detecting and dealing with heterogeneity in meta-analyses. J Clin Epidemiol. 2021;130:149-151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 54] [Article Influence: 10.8] [Reference Citation Analysis (1)] |
| 42. | Rao G, Lopez-Jimenez F, Boyd J, D'Amico F, Durant NH, Hlatky MA, Howard G, Kirley K, Masi C, Powell-Wiley TM, Solomonides AE, West CP, Wessel J; American Heart Association Council on Lifestyle and Cardiometabolic Health; Council on Cardiovascular and Stroke Nursing; Council on Cardiovascular Surgery and Anesthesia; Council on Clinical Cardiology; Council on Functional Genomics and Translational Biology; and Stroke Council. Methodological Standards for Meta-Analyses and Qualitative Systematic Reviews of Cardiac Prevention and Treatment Studies: A Scientific Statement From the American Heart Association. Circulation. 2017;136:e172-e194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 192] [Article Influence: 21.3] [Reference Citation Analysis (0)] |
| 43. | Seide SE, Röver C, Friede T. Likelihood-based random-effects meta-analysis with few studies: empirical and simulation studies. BMC Med Res Methodol. 2019;19:16. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 78] [Cited by in RCA: 108] [Article Influence: 15.4] [Reference Citation Analysis (0)] |
| 44. | Lin L. Bias caused by sampling error in meta-analysis with small sample sizes. PLoS One. 2018;13:e0204056. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 132] [Cited by in RCA: 213] [Article Influence: 26.6] [Reference Citation Analysis (1)] |
| 45. | Perego C, Sbolli M, Specchia C, Fiuzat M, McCaw ZR, Metra M, Oriecuia C, Peveri G, Wei LJ, O'Connor CM, Psotka MA. Utility of Restricted Mean Survival Time Analysis for Heart Failure Clinical Trial Evaluation and Interpretation. JACC Heart Fail. 2020;8:973-983. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 43] [Article Influence: 7.2] [Reference Citation Analysis (1)] |