Published online Mar 26, 2026. doi: 10.4330/wjc.v18.i3.117999
Revised: January 12, 2026
Accepted: February 10, 2026
Published online: March 26, 2026
Processing time: 93 Days and 1.5 Hours
Arrhythmogenic right ventricular cardiomyopathy (ARVC) is an inherited car
A 57-year-old male was hospitalized with palpitations and was diagnosed with definite ARVC in 2021. He presented initially with sustained ventricular tach
A thorough assessment of atrial involvement is essential for ARVC patients.
Core Tip: This case highlights a rare arrhythmogenic right ventricular cardiomyopathy (ARVC) with sick sinus syndrome, para-Hisian focal atrial tachycardia, and extensive right atrial myocardial scarring confirmed by electroanatomic mapping. Beyond ventricular involvement, the patient demonstrated profound atrial electrical and structural remodeling, sinus node dysfunction and ischemic strokes despite successful ventricular tachycardia control. Therefore, atrial myocardial involve
- Citation: Li A, Zhu YJ, Ma XX, Jin Y, Yang J, Feng QT. Arrhythmogenic right ventricular cardiomyopathy with atrial tachycardia and sick sinus syndrome: A case report. World J Cardiol 2026; 18(3): 117999
- URL: https://www.wjgnet.com/1949-8462/full/v18/i3/117999.htm
- DOI: https://dx.doi.org/10.4330/wjc.v18.i3.117999
Arrhythmogenic right ventricular cardiomyopathy (ARVC) is a hereditary cardiomyopathy that mostly affects the right ventricle (RV), left ventricle (LV), or both. We report a rare case of ARVC involving both the right atrium (RA) and RV. The patient not only presented with ventricular tachycardia (VT) originating from the RV, but also with sick sinus syn
A 57-year-old male patient had recurrent palpitations for more than 30 years, with worsening for one day.
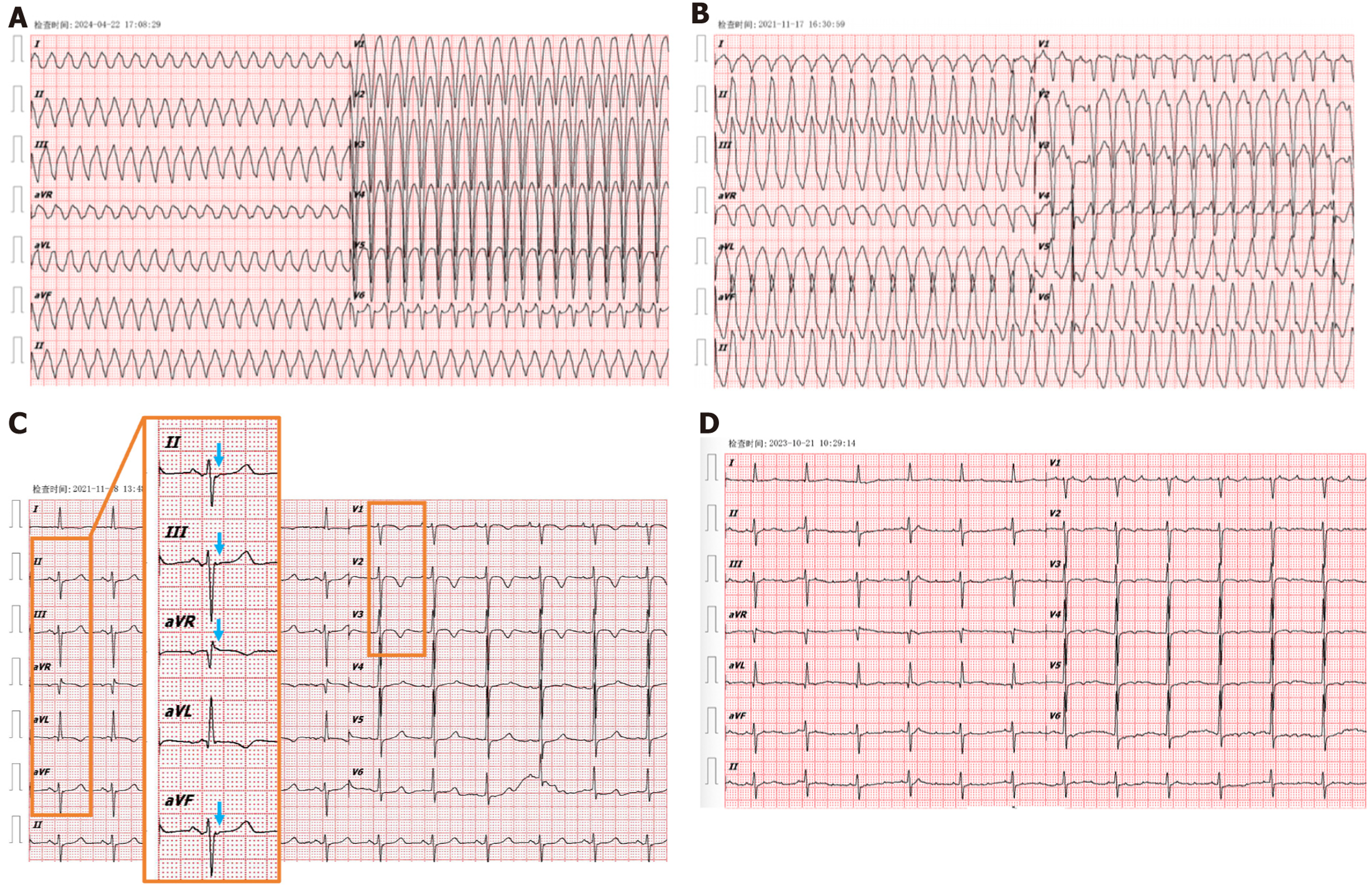
The patient presented to our hospital with palpitations. The emergency electrocardiography (ECG) showed sustained VT with left bundle branch block morphology and inferior axis, consistent with RV origin (Figure 1A). He began experiencing recurrent palpitations 30 years ago. He denied chest pain, chest tightness, dizziness, syncope, or radiating pain to the shoulder or left arm.
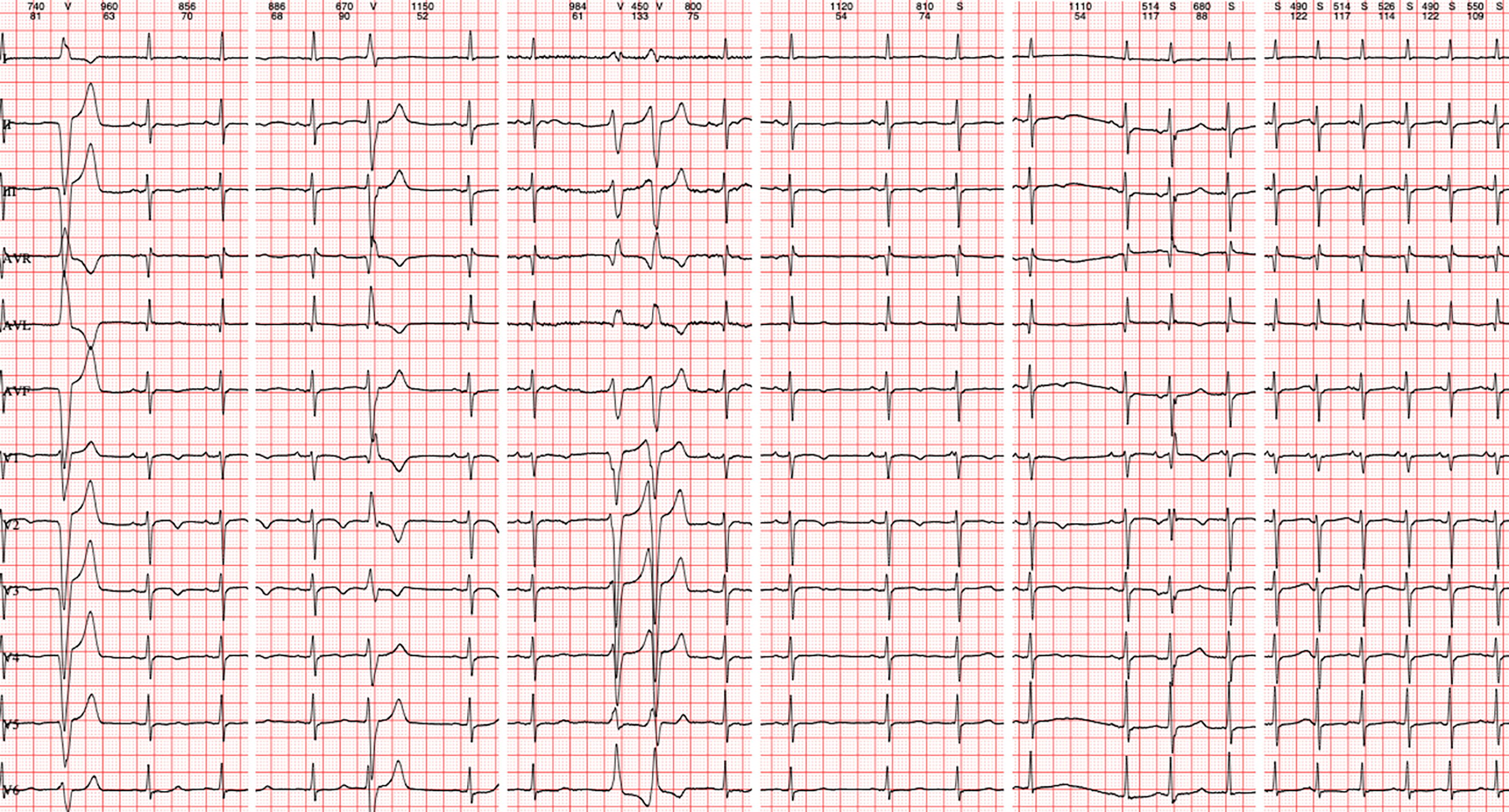
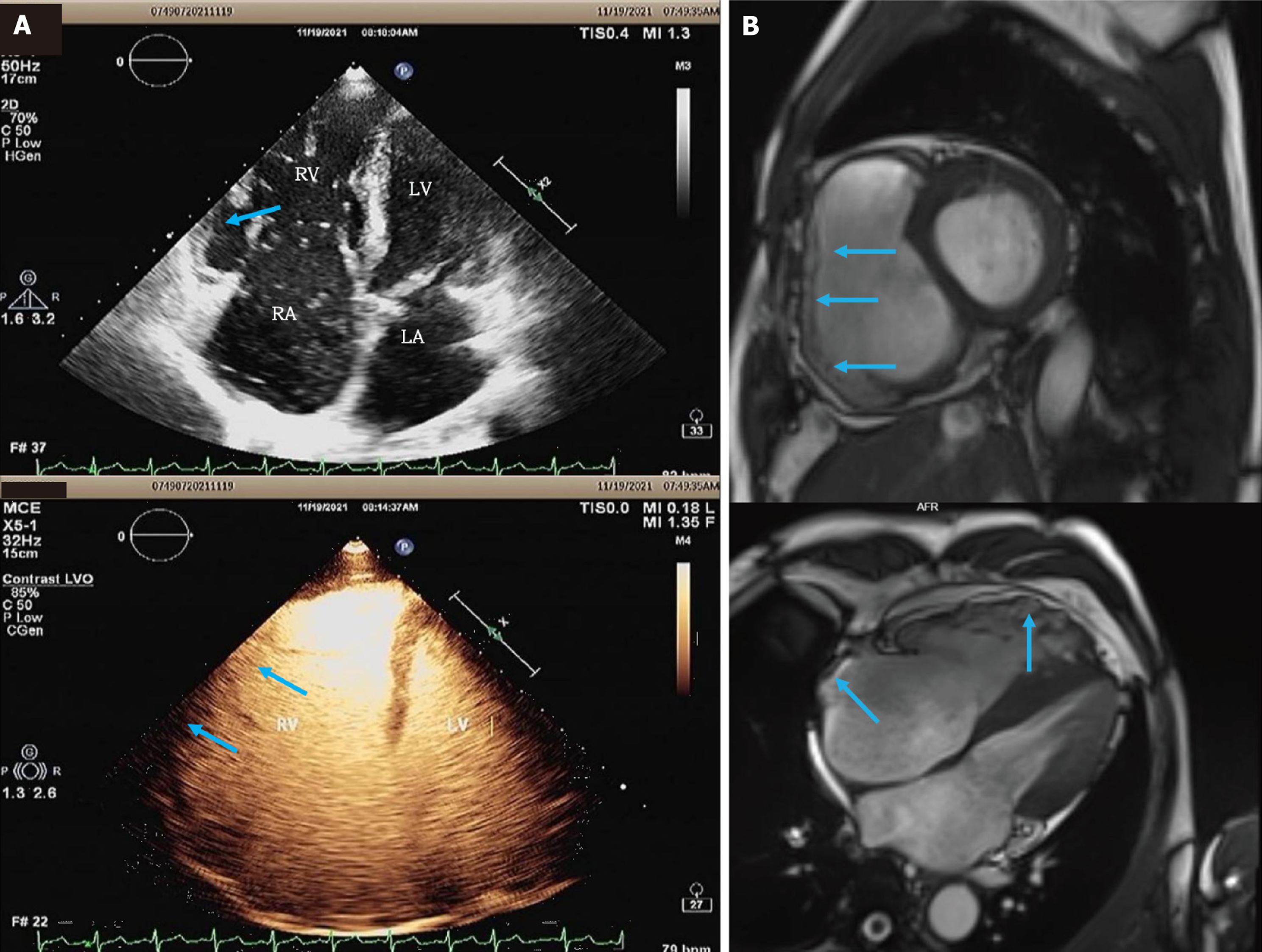
He once experienced sudden fatigue followed by loss of consciousness while working, but received no treatment at the time. In 2021, he was taken to the emergency department due to VT and sinus rhythm was restored after electrical cardioversion (Figure 1B and C). He was diagnosed with definite ARVC after undergoing Holter (Figure 2), echocardiography (Figure 3A), cardiac magnetic resonance (Figure 3B), and genetic testing. In 2023, he was hospitalized with ischemic stroke and ECG showed AT with 2:1 atrioventricular conduction (Figure 1D). He had no history of hyperte
The patient had no history of smoking or alcohol abuse. His father and elder brother had both died suddenly from un
Physical examination revealed no remarkable abnormalities. Following admission, the patient had reverted to sinus rhythm with a regular heart rate and rhythm. Cardiac auscultation showed no murmurs and no edema in the lower extre
Laboratory tests showed the following: Cardiac troponin T (1478.0 ng/L), cardiac troponin I (10.29 ng/mL), and C-reactive protein (13.1 mg/L). Coagulation parameters, brain natriuretic peptide levels, potassium and thyroid function test results showed no discernible abnormalities.
Echocardiography revealed cardiac chamber enlargement, mild mitral regurgitation, moderate-to-severe tricuspid regurgitation, reduced right ventricular systolic function, and a left ventricular ejection fraction of 58%. RV angiography revealed a ventricular aneurysm near the tricuspid annulus on the free wall of the RV (Video).
Arrhythmogenic right ventricular cardiomyopathy, SSS, and para-Hisian focal atrial tachycardia.
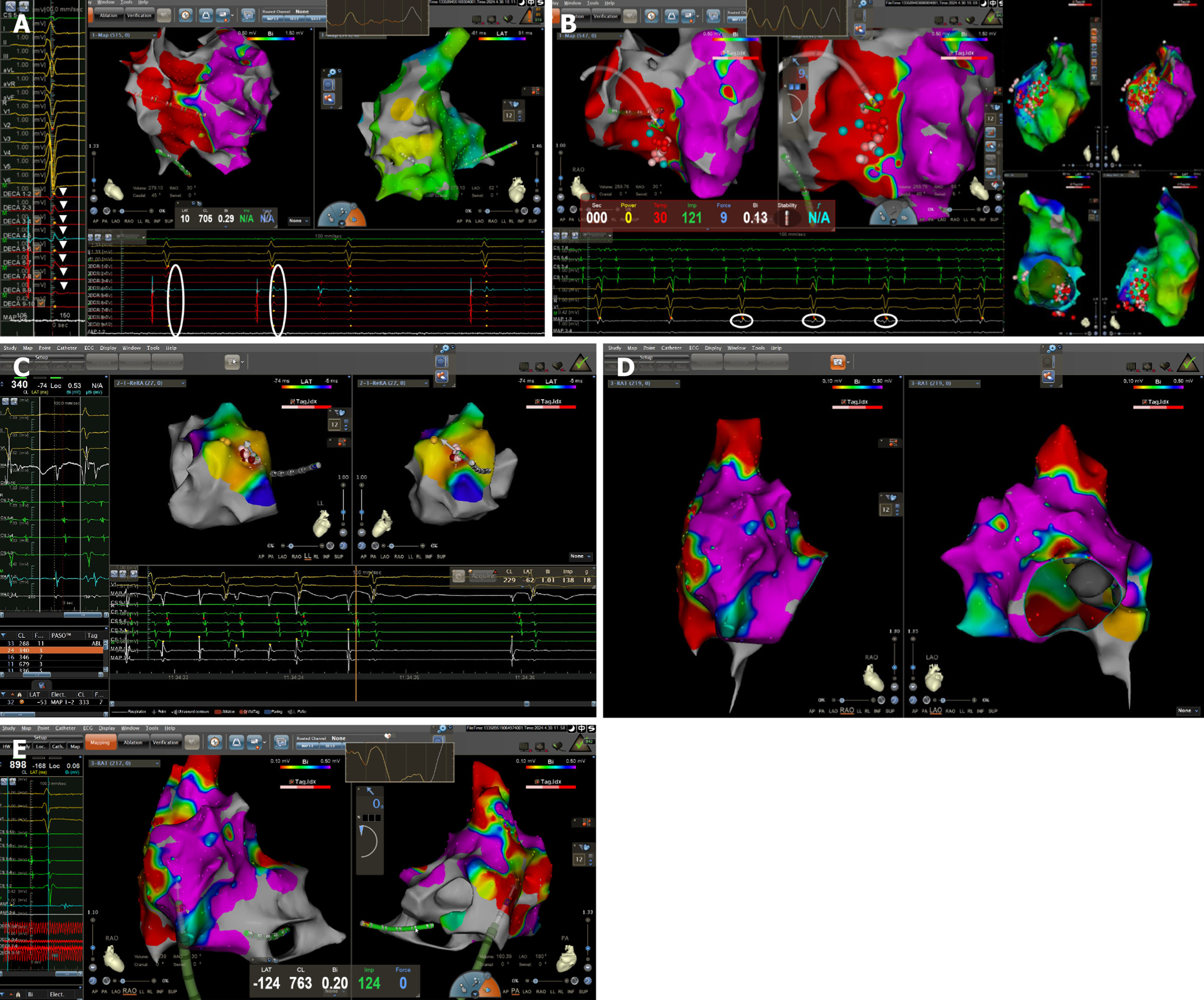
We adopted the method described by Wu et al[1] to induce VT through fast-rate (≥ 250 beats/minute) right ventricular burst stimulation. However, the patient could not tolerate the process, and induction was terminated. Endocardial substrate mapping during sinus rhythm showed extensive scarring and low-voltage areas on the RV free wall (ventricular scar was defined as bipolar electrogram amplitude between 0.5 mV and 1.5 mV during sinus rhythm and dense scar was defined as < 0.5 mV)[2]. Low-frequency, fractionated late potentials were observed along the right ventricular free wall, extending from the tricuspid annulus at the 10 o’clock position down to the low 6 o’clock position (Figure 4A). Concurrently, C-type isolation ring ablation was performed in the low-voltage areas, and homogenization modification was conducted targeting the areas with low-frequency, fractionated late potentials within the isolation rings (Figure 4B).
After confirming bidirectional block and adequate substrate modification, sustained AT was induced during the withdrawal of the mapping catheter. Activation mapping localized the earliest atrial activation site to the para-Hisian region focal AT below the interatrial septum. Mechanical friction around the target site could repeatedly terminate and re-induce AT, long RR pauses and marked sinus bradycardia were observed after AT termination. Electrophysiological study (EPS) showed a sinus node recovery time of 1660 milliseconds. Detailed mapping around the target site identified the earliest atrial activation occurring 37 milliseconds earlier than the surface ECG. Local ablation was performed in sinus rhythm with temperature control of 20 W and a flow rate of 2 mL/minute (500 U/500 mL heparinized saline), and res
Repeated atrial programmed stimulation and burst stimulation, as well as ventricular programmed stimulation, failed to induce the clinical AT or VT. Substrate mapping of the RA revealed extensive low-voltage and scarred areas on the right atrial free wall, posterior wall, and the sinus-node region (atrial dense scar was defined as bipolar electrogram voltage < 0.5 mV during sinus rhythm, and the low-voltage area was defined as abnormal myocardial tissue with voltages < 0.5 mV)[3] (Figure 4D and E). These findings were consistent with the abnormal signal regions seen on pre-procedural cardiac magnetic resonance (CMR) imaging. The post-ablation diagnosis was para-Hisian focal AT and SSS.
An implantable cardioverter defibrillator (ICD) was recommended per European Society of Cardiology (ESC) guidelines (class I indication)[4], but the patient declined and was discharged on oral aspirin therapy. At the time of writing, no recurrent atrial or ventricular arrhythmias have since been recorded. However, the patient experienced a second ischemic stroke. At the most recent follow-up, the patient remained at persistent risk due to refusal of ICD implantation and rec
| Time | Clinical events | Results |
| November 2021 | Admitted for palpitations. ARVC was diagnosed after complete examination | Requested conservative treatment, discharged |
| October 2023 | Admitted for ischemic stroke. ECG showed AT with 2:1 AV conduction | Symptoms improved, discharged |
| April 2024 | Admitted for palpitations. ECG showed VT and underwent radiofrequency ablation | Declined ICD implantation, discharged |
| August 2025 | Admitted for a second ischemic stroke with no VT | Symptoms improved, discharged |
ARVC is an inherited cardiomyopathy caused by mutations in desmosomal protein genes. It is a major cause of sudden cardiac death in young adults and athletes. The clinical presentation of ARVC varies widely, and its arrhythmic manifestations are often complex, which can pose considerable diagnostic challenges[5,6]. The diagnostic criteria for ARVC have progressed from the original 1994 task force criteria to the revised 2010 Task Force Criteria, and most recently, to the 2020 Padua Criteria. The condition involves morpho-functional ventricular abnormalities, structural myocardial abnormalities, repolarization and depolarization abnormalities, ventricular arrhythmias, and family genetics[7]. Electrocardiographic repolarization abnormalities, particularly T-wave inversion in the right precordial leads (V1-V3), are among the earliest and most common findings in ARVC. The extent of T-wave inversion usually correlates with the degree of fibrofatty rep
During cardiac development, major components of the RV and portions of the RA arise primarily from the second heart field (SHF). Pathogenic PKP2 variants have been shown to suppress Wnt signaling in epicardial SHF-derived pro
Traditionally, ARVC has been regarded as a condition affecting the RV, LV, or both. In its early stages, ARVC typically affects the “triangle of dysplasia” (the right ventricular inflow tract, outflow tract, and apex), with occasional involvement of the inferolateral left ventricular wall[13]. Recent evidence has demonstrated that atrial involvement is not uncommon in ARVC. Fibrofatty replacement can extend from the ventricles into the atria, leading to structural and electrical rem
Clinical management of ARVC should focus on not only ventricular pathology but also atrial involvement. Atrial structural and electrical abnormalities can affect the rhythmic contraction of the atria, and may facilitate thrombus for
Once ARVC is diagnosed, a thorough assessment of atrial involvement is essential. Echocardiography, CMR, and EPS are valuable methods for detecting atrial structural and electrical abnormalities. Early recognition of atrial involvement plays a critical role in comprehensive disease evaluation and in guiding optimal clinical management.
| 1. | Wu LM, Bao JR, Yao Y, Hou BB, Zheng LH, Zhang S. Fast rate (≥ 250 beats/min) right ventricular burst stimulation is useful for ventricular tachycardia induction in arrhythmogenic right ventricular cardiomyopathy. J Geriatr Cardiol. 2016;13:70-74. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 2. | Wu N, Chen H, Ju W, Li M, Gu K, Wang Z, Liu H, Shi J, Jiang X, Cui C, Cai C, Yang G, Chen M. Arrhythmogenic Right Ventricular Cardiomyopathy With Extensive Abnormal Substrate: Is Isolation Possible? JACC Clin Electrophysiol. 2023;9:1455-1463. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 3. | Butcher C, Roney C, Wharmby A, Ahluwalia N, Chow A, Lambiase PD, Hunter RJ, Honarbakhsh S. In Atrial Fibrillation, Omnipolar Voltage Maps More Accurately Delineate Scar Than Bipolar Voltage Maps. JACC Clin Electrophysiol. 2023;9:1500-1512. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 4. | Arbelo E, Protonotarios A, Gimeno JR, Arbustini E, Barriales-Villa R, Basso C, Bezzina CR, Biagini E, Blom NA, de Boer RA, De Winter T, Elliott PM, Flather M, Garcia-Pavia P, Haugaa KH, Ingles J, Jurcut RO, Klaassen S, Limongelli G, Loeys B, Mogensen J, Olivotto I, Pantazis A, Sharma S, Van Tintelen JP, Ware JS, Kaski JP; ESC Scientific Document Group. 2023 ESC Guidelines for the management of cardiomyopathies. Eur Heart J. 2023;44:3503-3626. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2099] [Cited by in RCA: 1740] [Article Influence: 580.0] [Reference Citation Analysis (2)] |
| 5. | Hu Y, Chen Z, Shi A, Li Z, Chen Z, Zheng Y, Zhao X, Dai S, Lin Y, Li Y, Yang B, Zhao X, Li G, Zhou X, Hu S, Wu L, Chen L. China Multi-Center Cohort Study on Risk Evaluation of Arrhythmogenic Cardiomyopathy: The ChinaCORE ACM Registry. JACC Asia. 2025;5:914-923. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 6. | Kreimer F, Saguner AM, Akin I, Milting H, Eckardt L, El-Battrawy I. Arrhythmogenic Right Ventricular Cardiomyopathy: Diagnosis, Risk Stratification, and Treatment. Dtsch Arztebl Int. 2025;122:229-234. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 7. | Corrado D, Perazzolo Marra M, Zorzi A, Beffagna G, Cipriani A, Lazzari M, Migliore F, Pilichou K, Rampazzo A, Rigato I, Rizzo S, Thiene G, Anastasakis A, Asimaki A, Bucciarelli-Ducci C, Haugaa KH, Marchlinski FE, Mazzanti A, McKenna WJ, Pantazis A, Pelliccia A, Schmied C, Sharma S, Wichter T, Bauce B, Basso C. Diagnosis of arrhythmogenic cardiomyopathy: The Padua criteria. Int J Cardiol. 2020;319:106-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 516] [Cited by in RCA: 388] [Article Influence: 64.7] [Reference Citation Analysis (0)] |
| 8. | Aimo A, Milandri A, Barison A, Pezzato A, Morfino P, Vergaro G, Merlo M, Argirò A, Olivotto I, Emdin M, Finocchiaro G, Sinagra G, Elliott P, Rapezzi C. Electrocardiographic abnormalities in patients with cardiomyopathies. Heart Fail Rev. 2024;29:151-164. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 17] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 9. | Cipriani A, Mattesi G, Bariani R, Cecere A, Martini N, De Michieli L, Da Pozzo S, Corradin S, De Conti G, Zorzi A, Motta R, De Lazzari M, Bauce B, Iliceto S, Basso C, Corrado D, Perazzolo Marra M. Cardiac magnetic resonance imaging of arrhythmogenic cardiomyopathy: evolving diagnostic perspectives. Eur Radiol. 2023;33:270-282. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 40] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 10. | Krahn AD, Wilde AAM, Calkins H, La Gerche A, Cadrin-Tourigny J, Roberts JD, Han HC. Arrhythmogenic Right Ventricular Cardiomyopathy. JACC Clin Electrophysiol. 2022;8:533-553. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 83] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 11. | Kohela A, van Rooij E. Fibro-fatty remodelling in arrhythmogenic cardiomyopathy. Basic Res Cardiol. 2022;117:22. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 12. | Shen L, Liu S, Zhang Z, Xiong Y, Lai Z, Hu F, Zheng L, Yao Y. Catheter ablation of ventricular tachycardia in patients with arrhythmogenic right ventricular cardiomyopathy and biventricular involvement. Europace. 2024;26:euae059. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 13. | Corrado D, van Tintelen PJ, McKenna WJ, Hauer RNW, Anastastakis A, Asimaki A, Basso C, Bauce B, Brunckhorst C, Bucciarelli-Ducci C, Duru F, Elliott P, Hamilton RM, Haugaa KH, James CA, Judge D, Link MS, Marchlinski FE, Mazzanti A, Mestroni L, Pantazis A, Pelliccia A, Marra MP, Pilichou K, Platonov PGA, Protonotarios A, Rampazzo A, Saffitz JE, Saguner AM, Schmied C, Sharma S, Tandri H, Te Riele ASJM, Thiene G, Tsatsopoulou A, Zareba W, Zorzi A, Wichter T, Marcus FI, Calkins H; International Experts. Arrhythmogenic right ventricular cardiomyopathy: evaluation of the current diagnostic criteria and differential diagnosis. Eur Heart J. 2020;41:1414-1429. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 310] [Cited by in RCA: 211] [Article Influence: 35.2] [Reference Citation Analysis (0)] |
| 14. | Sharma S, Giri G, Khadka S, Goswami R. Atrial Tachyarrhythmia in Arrhythmogenic Right Ventricular Cardiomyopathy: A Case Report. Clin Case Rep. 2025;13:e70154. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 15. | Mizia-Stec K, Caforio ALP, Charron P, Gimeno JR, Elliott P, Kaski JP, Maggioni AP, Tavazzi L, Rigopoulos AG, Laroche C, Frigy A, Zachara E, Pena-Pena ML, Olusegun-Joseph A, Pinto Y, Sala S, Drago F, Blagova O, Reznik E, Tendera M. Atrial fibrillation, anticoagulation management and risk of stroke in the Cardiomyopathy/Myocarditis registry of the EURObservational Research Programme of the European Society of Cardiology. ESC Heart Fail. 2020;7:3601-3609. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 16. | Li G, Fontaine GH, Fan S, Yan Y, Bode PK, Duru F, Frank R, Saguner AM. Right atrial pathology in arrhythmogenic right ventricular dysplasia. Cardiol J. 2019;26:736-743. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 17. | Liang E, Wu L, Fan S, Li X, Hu F, Zheng L, Fan X, Chen G, Ding L, Yao Y. Bradyarrhythmias in Arrhythmogenic Right Ventricular Cardiomyopathy. Am J Cardiol. 2019;123:1690-1695. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 18. | Van Gelder IC, Rienstra M, Bunting KV, Casado-Arroyo R, Caso V, Crijns HJGM, De Potter TJR, Dwight J, Guasti L, Hanke T, Jaarsma T, Lettino M, Løchen ML, Lumbers RT, Maesen B, Mølgaard I, Rosano GMC, Sanders P, Schnabel RB, Suwalski P, Svennberg E, Tamargo J, Tica O, Traykov V, Tzeis S, Kotecha D; ESC Scientific Document Group. 2024 ESC Guidelines for the management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J. 2024;45:3314-3414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2038] [Cited by in RCA: 1782] [Article Influence: 891.0] [Reference Citation Analysis (0)] |