Published online Mar 26, 2026. doi: 10.4330/wjc.v18.i3.118227
Revised: January 16, 2026
Accepted: February 14, 2026
Published online: March 26, 2026
Processing time: 86 Days and 18.3 Hours
Heart failure (HF) is a leading cause of cardiovascular morbidity and mortality, affecting over 64 million individuals worldwide. Despite advances in guideline-directed medical therapy, patients with HF continue to experience poor outcomes, highlighting the need for novel therapeutic approaches. Sodium glucose tran
Core Tip: Heart failure (HF) affects over 64 million people worldwide, with persistently poor outcomes despite advances in treatment. Sodium glucose transporter 2 inhibitors, originally developed for diabetes, have emerged as breakthrough HF therapies with cardiovascular benefits beyond glucose control, including reduced risk of hospitalization and mortality. Landmark trials (Dapagliflozin and Prevention of Adverse Outcomes in Heart Failure, Empagliflozin Outcome Trial in Patients with Chronic Heart Failure with Reduced Ejection Fraction, Empagliflozin Outcome Trial in Patients with Chronic Heart Failure with Preserved Ejection Fraction, Dapagliflozin Evaluation to Improve the Lives of Patients with Preserved Ejection Fraction Heart Failure) have demonstrated efficacy across all HF types, regardless of diabetes status. Their benefits stem from improved cardiac energetics, enhanced mitochondrial function, reduced oxidative stress and inflammation, and favorable cardiac remodeling. This review examines current evidence, mechanistic pathways, and future prospects for sodium glucose transporter 2 inhibitor use in cardiovascular medicine.
- Citation: Usuda D, Furukawa D, Imaizumi R, Ono R, Kaneoka Y, Nakajima E, Kato M, Sugawara Y, Shimizu R, Inami T, Sakurai R, Kawai K, Matsubara S, Tanaka R, Suzuki M, Shimozawa S, Hotchi Y, Osugi I, Katou R, Ito S, Mishima K, Kondo A, Mizuno K, Takami H, Komatsu T, Nomura T, Sugita M. Sodium glucose transporter 2 inhibitors for heart failure. World J Cardiol 2026; 18(3): 118227
- URL: https://www.wjgnet.com/1949-8462/full/v18/i3/118227.htm
- DOI: https://dx.doi.org/10.4330/wjc.v18.i3.118227
Heart failure (HF) is a multifaceted clinical syndrome in which the heart cannot pump blood sufficiently to meet the metabolic needs of peripheral tissues[1]. In developed countries, HF affects approximately 1%-2% of the adult population, with a prevalence of over 10% in individuals aged ≥ 70 years[2]. The worldwide burden of HF continues to increase, dri
Classification of HF is traditionally based on left ventricular ejection fraction (LVEF), with HF with reduced ejection fraction (HFrEF) defined by an LVEF ≤ 40%, HF with mildly reduced ejection fraction defined by an LVEF (41%-49%), and HF with preserved ejection fraction (HFpEF) defined by an LVEF ≥ 50%[4]. Each phenotype exhibits unique path
HF pathophysiology involves complex neurohormonal activation, including the renin-angiotensin-aldosterone and sympathetic nervous systems, resulting in vasoconstriction, sodium retention, and progressive cardiac remodeling[6]. Current guidelines for medical treatment of HFrEF recommend angiotensin-converting enzyme inhibitors or angiotensin receptor blockers, beta-blockers, mineralocorticoid receptor antagonists, and more recently, angiotensin receptor-nep
The diverse and complex nature of HF, especially HFpEF, poses major therapeutic challenges[9]. Characterized by diastolic dysfunction, increased left ventricular stiffness, and impaired relaxation, HFpEF is also frequently associated with diabetes, obesity, chronic kidney disease (CKD), and atrial fibrillation[9]. The limited treatment options for HFpEF highlights the urgent need for novel therapeutic approaches targeting the pathophysiological mechanisms common to both HFrEF and HFpEF[10].
Sodium glucose transporter 2 (SGLT2) inhibitors are a groundbreaking class of antidiabetic drugs initially designed to treat type 2 diabetes mellitus (T2DM) by selective inhibition of renal glucose reabsorption; however, unexpectedly, they have demonstrated significant cardiovascular benefits that extend beyond glycemic control[11].
The development of SGLT2 inhibitors originated from 19th-century discovery of phlorizin, a natural compound from apple tree bark that induces glycosuria[12]. However, phlorizin’s non-selective inhibition of both SGLT1 and SGLT2 and poor pharmacokinetic properties have limited its clinical use[12]. Modern SGLT2 inhibitors have been developed through structure-activity relationship studies to selectively target SGLT2 and inhibit it, while maintaining favorable pharmacological profiles[13].
Canagliflozin was the first SGLT2 inhibitor approved in 2013, followed by dapagliflozin and empagliflozin[14]. These drugs block approximately 90% of filtered glucose reabsorption in the proximal tubule of the kidney, causing glucosuria and modest insulin-independent improvements in glycemic control[15]. This mechanism also promotes caloric loss, wei
The paradigm shift was driven by the results of cardiovascular outcome trials assessing the cardiovascular safety of SGLT2 inhibitors in patients with T2DM and established cardiovascular disease. The 2015 EMPA-REG OUTCOME trial showed that empagliflozin significantly reduced the risk of cardiovascular death (CD), myocardial infarction, and stroke in high-risk patients with diabetes[17], while reducing HF hospitalizations by 35%, an effect that was evident early and independent of baseline HF status[18].
Subsequent cardiovascular outcome trials on canagliflozin (CANVAS Program) and dapagliflozin (DECLARE-TIMI 58) confirmed consistent cardiovascular benefits across different SGLT2 inhibitors, particularly reduced HF hospitalizations[14,19]. These results suggested that SGLT2 inhibitors may exert direct cardiovascular benefits independent of glycemic control, prompting further studies on patients with established HF regardless of diabetes status[20].
Since SGLT2 inhibitors have emerged as a feasible pharmacological strategy, no paper has systematically summarized the latest evidence and knowledge, and a review and update on this topic is urgently needed. Therefore, this narrative review aims to provide a focused overview of SGLT2 inhibitors, including their structure, expression, regulatory roles, effect on the heart and HF, evidence from clinical trials, adverse effects, and prospects.
We searched the PubMed database to identify key human studies reporting the most recent advances in SGLT2 inhibitor research. We identified relevant literature, and the findings are summarized below by category.
SGLT2 inhibitors are small-molecule compounds exhibiting common structural similarities while maintaining distinct pharmacokinetic and pharmacodynamic profiles[21]. In terms of molecular structure, clinically available SGLT2 inhi
SGLT2 is a high-capacity, low-affinity glucose transporter expressed primarily in the S1 segment of the renal proximal tubule[23]. SGLT2 reabsorbs approximately 90% of filtered glucose, with SGLT1 reabsorbing the remaining 10% in the distal proximal tubule segments[23]. Glucose uptake is driven against its concentration gradient using the sodium gra
Recent studies show that SGLT2 is expressed beyond the kidney, extending to the heart, skeletal muscle, liver, and intestine, though at much lower levels[25]. In the heart, SGLT2 is present in cardiomyocytes, with expression elevated in HF and diabetic cardiomyopathy[26]. This extrarenal expression may contribute to the direct cardiovascular effects of SGLT2 inhibitors, independent of their renal effects.
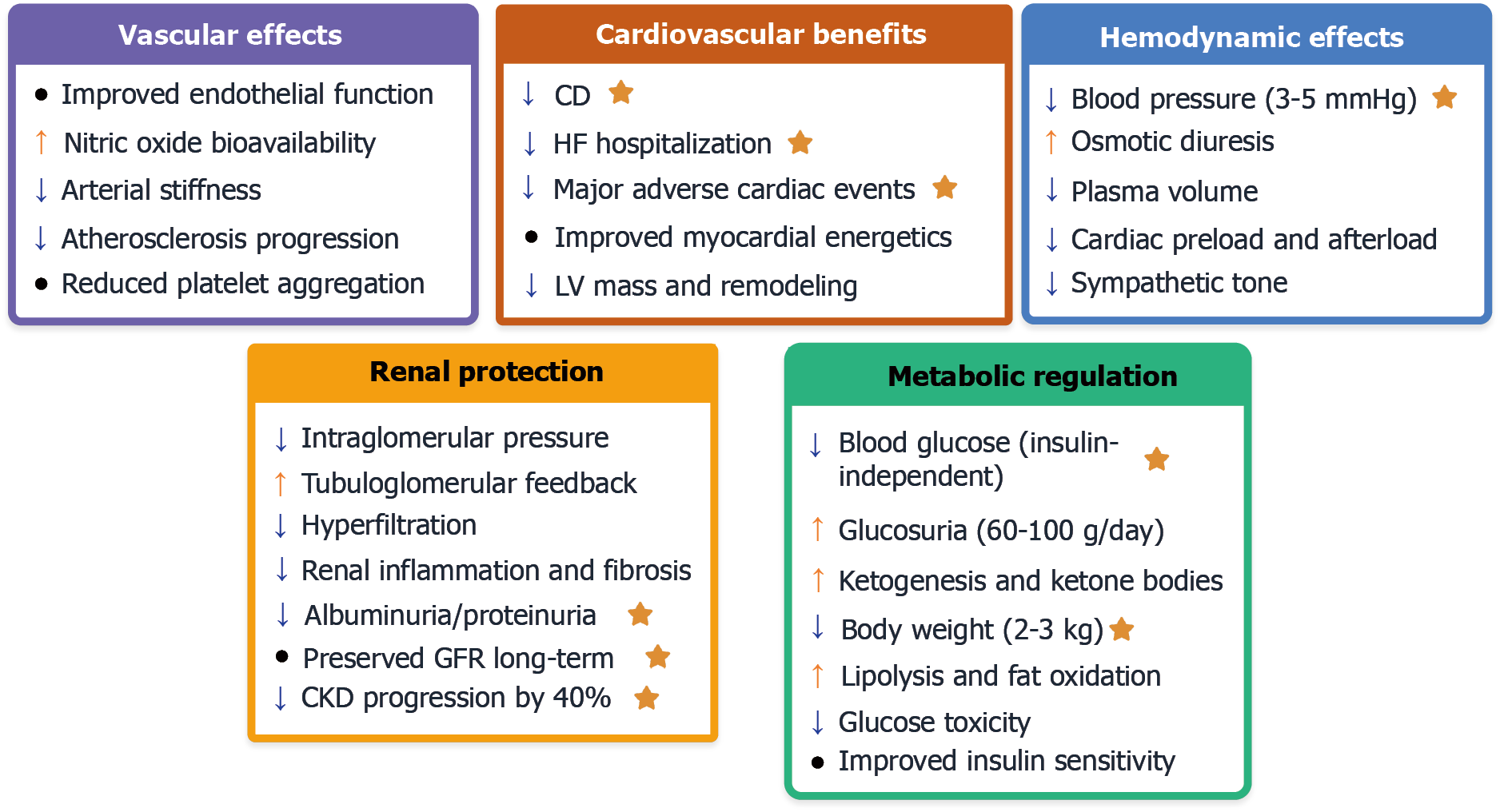
Multiple transcriptional and post-transcriptional factors are associated with the regulatory mechanisms governing SGLT2 expression[27]. High glucose levels upregulate SGLT2 expression via signaling pathways such as the protein kinase C activation and transforming growth factor-β signaling pathways[27]. Additionally, inflammatory cytokines, oxidative stress, and neurohormonal activation typical of HF can modulate SGLT2 expression and activity[28]. Figure 1 summarizes the systemic effects of SGLT2 inhibitors and their pleiotropic effects across multiple organ systems.
SGLT2 inhibitors are highly select for SGLT2 over SGLT1, with selectivity ratios of approximately 2500-fold for empagliflozin and 1200-fold for canagliflozin[29]. This selectivity is important because SGLT1 mediates intestinal glucose absorption and plays key roles in the heart and kidneys[29]. Pharmacokinetically, SGLT2 inhibitors exhibit good oral bioavailability, extensive protein binding, and are primarily metabolized in the liver with minimal renal elimination of the parent drug[30]. The characteristics of each SGLT2 inhibitor – particularly their pharmacological properties and stru
The cardiovascular benefits of SGLT2 inhibitors extend beyond renal glucose inhibition[31] encompassing hemodynamic, metabolic, and direct cellular effects that synergistically promote cardiovascular health and slow HF progression[32].
SGLT2 inhibitors induce favorable hemodynamic effects that contribute to benefits in HF[33]. Glucosuria-driven osmotic diuresis produces a mild natriuretic effect, reducing plasma volume and decreasing preload without significantly acti
Additionally, the benefits of SGLT2 inhibitors on arterial stiffness and blood pressure contribute to decreased afterload and improved ventricular-arterial coupling[35]. Clinical studies have shown reductions in both systolic and diastolic blood pressure by 3-5 mmHg with SGLT2 inhibitors, and greater effects are observed in patients with baseline hyp
Cardiac metabolism is profoundly altered by SGLT2 inhibitors via promotion of a shift from glucose to more efficient fuel sources, notably ketone bodies and fatty acids[38]. This metabolic reprogramming is especially beneficial in HF, where myocardial energy efficiency is impaired[38]. Elevated ketone availability, particularly β-hydroxybutyrate, provides a high-efficiency energy source, generating more adenosine triphosphate per oxygen molecule than glucose[39].
The “thrifty fuel hypothesis” suggests that ketone bodies act as a “super fuel” for the failing heart, enhancing cardiac efficiency and function[40]. Clinical studies show that SGLT2 inhibitors increase circulating ketone levels, even in patients without diabetes, underscoring the contribution of ketone metabolism to cardiovascular health[16]. Moreover, SGLT2 inhibitors improve myocardial glucose uptake by enhancing insulin sensitivity and promoting glucose transporter type 4 translocation[41].
Mitochondrial dysfunction is a hallmark of HF, characterized by impaired oxidative phosphorylation, excessive pro
Preclinical studies have shown that empagliflozin improves mitochondrial respiratory capacity and reduces produc
The ketone body β-hydroxybutyrate is produced during SGLT2 inhibitor therapy and also functions as a signaling molecule, activating longevity pathways such as sirtuin-1 and 5’-adenosine monophosphate-activated protein kinase. Activation of these pathways promotes mitochondrial biogenesis and resistance to cellular stress[47]. In addition, SGLT2 inhibitors have been shown to reduce mitochondrial fission while promoting fusion, thereby improving mitochondrial network dynamics[48].
Chronic inflammation is a key contributor to HF pathogenesis, driving myocardial dysfunction, adverse remodeling, and disease progression[49]. SGLT2 inhibitors exhibit potent anti-inflammatory effects through multiple mechanisms, inc
Clinical studies have shown that treatment with SGLT2 inhibitors lowers circulating levels of inflammatory markers such as C-reactive protein, interleukin-6, and tumor necrosis factor-α[51]. These anti-inflammatory actions are mediated in part by inhibition of the nucleotide-binding and oligomerization domain-like receptors protein 3 inflammasome, a key regulator of sterile inflammation in cardiovascular disease[52]. Furthermore, SGLT2 inhibitors promote immune home
The molecular basis of these anti-inflammatory effects involves modulation of nuclear factor kappa B signaling, acti
Adverse cardiac remodeling – characterized by myocyte hypertrophy, interstitial fibrosis, and chamber dilatation – is a fundamental mechanism driving HF progression[56]. SGLT2 inhibitors demonstrate beneficial effects on cardiac rem
Preclinical studies show that SGLT2 inhibitor treatment reduces cardiac fibrosis by suppressing transforming growth factor-β signaling and limiting collagen deposition[58]. These antifibrotic effects are mediated by the downregulation of profibrotic genes and proteins, such as α-smooth muscle actin and collagen types I and III[59]. In addition, SGLT2 inhi
Cardiac magnetic resonance imaging studies have confirmed that SGLT2 inhibitor therapy is associated with regression of myocardial fibrosis and improvements in left ventricular function in patients with HF[61]. Notably, these remodeling benefits have been observed across different HF phenotypes and appear to be independent of diabetes status[62].
Robust clinical evidence supports the use of SGLT2 inhibitors in HF management, including multiple landmark ran
The Dapagliflozin and Prevention of Adverse Outcomes in Heart Failure (DAPA-HF) trial was a pivotal randomized, double-blind, placebo-controlled study that enrolled 4744 patients with HFrEF, irrespective of diabetes status[64]. Dapag
Importantly, the benefits of dapagliflozin were consistent across subgroups stratified by diabetes status, with com
Secondary analyses of the DAPA-HF trial revealed additional clinical benefits including improved quality of life (assessed using the Kansas City Cardiomyopathy Questionnaire), reduced HF hospitalizations, and preserved renal function[68]. The safety profile was favorable, with low rates of diabetic ketoacidosis, amputations, and fractures, and no increased risk of hypoglycemia among patients without diabetes[69].
The Empagliflozin Outcome Trial in Patients with Chronic Heart Failure with Reduced Ejection Fraction (EMPEROR-Reduced) trial enrolled 3730 patients with HFrEF, demonstrating that empagliflozin (10 mg daily) reduced the rates of CD and HF hospitalization by 25% (HR = 0.75, 95%CI: 0.65-0.86, P < 0.001) compared with a placebo[33].
This trial confirmed a class effect of SGLT2 inhibitors in HF, with consistent benefits observed irrespective of diabetes status. Empagliflozin reduced HF hospitalizations by 30% and showed a favorable trend toward reduced CD. Notably, the EMPEROR-Reduced population included patients with more severe HF, including lower mean LVEF and higher baseline N-terminal pro-brain natriuretic peptide levels, compared with that of the DAPA-HF[70]. A rapid onset of benefit was observed, with early separation of event curves within the first month of therapy[71]. In addition, empa
The Empagliflozin Outcome Trial in Patients with Chronic Heart Failure with Preserved Ejection Fraction (EMPEROR-Preserved) trial marked a breakthrough in HFpEF therapy, being the first large, randomized trial to demonstrate sig
The observed benefit was primarily driven by a 29% reduction in HF hospitalizations, with no significant effect on CD[74]. Importantly, the benefits were consistent across the full spectrum of ejection fraction, with comparable effects observed in patients with an LVEF of 41%-49% and those with an LVEF of ≥ 50%[75]. Moreover, benefits were observed in patients with and without diabetes[76].
Subgroup analyses indicated the greatest benefits in patients with an LVEF < 60%, suggesting that SGLT2 inhibitors may be particularly effective for mildly reduced or borderline preserved ejection fraction[77]. Treatment with empagli
The Dapagliflozin Evaluation to Improve the Lives of Patients with Preserved Ejection Fraction Heart Failure (DELIVER) trial complemented EMPEROR-Preserved by evaluating dapagliflozin in 6263 patients with HFpEF[79]. This trial dem
DELIVER confirmed the class effect of SGLT2 inhibitors in HFpEF, with benefits primarily driven by reductions in HF events. The trial enrolled a broader population than EMPEROR-Preserved, including patients with LVEF up to 65% and application of less stringent natriuretic peptide criteria[72]. The safety profile was consistent with prior SGLT2 inhibitor trials[80].
Consistent benefits of SGLT2 inhibitors across HF populations have been confirmed in several meta-analyses. A comprehensive meta-analysis including five major HF trials (DAPA-HF, EMPEROR-Reduced, EMPEROR-Preserved, DELIVER, and SOLOIST-WHF) and encompassing 21947 patients demonstrated a reduced risk of CD or HF hospitalization of 23% (HR = 0.77, 95%CI: 0.72-0.82)[55].
The benefits were consistent across the full spectrum of ejection fractions, with similar relative risk reductions in HFrEF and HFpEF populations[81]. Another meta-analysis confirmed the safety of SGLT2 inhibitors, with no increased risk of major adverse events such as diabetic ketoacidosis, amputations, or fractures[82]. The number required to treat for the primary endpoint was approximately 19 patients over 2 years.
SGLT2 inhibitors – including canagliflozin, dapagliflozin, empagliflozin, and ertugliflozin – are commonly used to treat T2DM, HF, and CKD because of their cardiovascular and renal protective effects[83,84]. Clinicians, however, should be mindful of potential adverse effects.
The most frequent class-related side effects include genital mycotic infections, occurring three to five times more often than with a placebo, presenting as vulvovaginal candidiasis in women and balanitis in men[83,84]. Euglycemic diabetic ketoacidosis is a rare but life-threatening complication, marked by metabolic acidosis with ketosis despite blood glucose levels of less than 250 mg/dL[85], with risk factors including surgery, fasting, low carbohydrate intake, and acute illness[85]. Osmotic diuresis may cause volume depletion, leading to hypotension and dizziness, though severe cases are uncommon with provided patients are properly monitored and stay hydrated[84]. Dapagliflozin is particularly associated with increased risk of urinary tract infection, while canagliflozin has been linked to an elevated risk of lower-limb amputations, particularly in patients with pre-existing peripheral vascular disease[83,84,86]. In 2020, the Food and Drug Administration removed its boxed warning for canagliflozin based on updated safety data[84]. Contrary to initial concerns, current evidence does not indicate an increased risk of acute kidney injury or bone fracture with SGLT2 inhi
Prevention strategies include educating patients on genital hygiene, maintaining adequate hydration, and recognizing early warning signs[84]. SGLT2 inhibitors should be temporarily discontinued 3-4 days before scheduled surgeries to minimize the risk of euglycemic DKA[85,86]. Most adverse effects can be managed without permanent discontinuation of therapy, enabling patients to continue receiving the substantial cardiovascular and renal protective effects of these medications[83,84].
The success of SGLT2 inhibitors in HF has created multiple avenues for future research. Ongoing clinical trials are exploring their effects in specific HF populations and clinical scenarios, while mechanistic studies continue to identify novel therapeutic targets[87].
The role of SGLT2 inhibitors in acute HF is under intense investigation. The DAPA-ACT HF-TIMI 68 trial is assessing the safety and efficacy of initiating dapagliflozin during HF hospitalization[88]. Early findings suggest that these agents can be initiated safely in the acute setting, potentially accelerating decongestion and shortening hospital stay[89].
Mechanistic benefits include rapid onset of natriuretic effects, improved renal function, and reduced neurohormonal activation[90]. Future studies should focus on determining the optimal timing of treatment initiation and identifying patients most likely to benefit from early SGLT2 inhibitor therapy[91].
Research evaluating the benefits of combining SGLT2 inhibitors with other novel HF therapies is ongoing. Combining SGLT2 inhibitors and glucagon-like peptide-1 receptor agonists is of particular interest because of the complementary mechanisms of action and potential synergistic cardiovascular benefits[92]. Preclinical studies suggest additive effects on metabolism, inflammation, and cardiac remodeling[93].
Dual SGLT1/SGLT2 inhibitors, such as sotagliflozin, have shown promising results in HF trials by targeting SGLT1 inhibition in the heart and other tissues[94]. The ideal balance between SGLT1 and SGLT2 inhibition for optimal car
Future research will also need to focus on identification of biomarkers and patient characteristics that can predict the response to SGLT2 inhibitor therapy. Genomic studies are exploring genetic variants that influence SGLT2 inhibitor efficacy and safety[96], while metabolomics and proteomics may uncover novel biomarkers for personalized treatment.
Incorporating clinical, laboratory, imaging, and genetic data into predictive models may enable precision medicine approaches to optimize SGLT2 inhibitor treatment. Machine learning algorithms are also being developed to predict optimal dosing strategies and identify patients most likely to benefit from SGLT2 inhibitor therapy[97].
The pleiotropic effects of SGLT2 inhibitors have prompted evaluation of their benefits in other cardiovascular conditions. Ongoing trials are investigating the use of SGLT2 inhibitors in patients with myocardial infarction, atrial fibrillation, and peripheral artery disease[98], given that the anti-inflammatory and metabolic effects of SGLT2 inhibitors may provide benefits across the spectrum of cardiovascular diseases.
Researchers are also exploring the potential role of SGLT2 inhibitors in preventing HF in high-risk populations, lev
Ongoing research into the mechanisms underlying SGLT2 inhibitor cardiovascular benefits will guide future drug development and optimize therapy. Key areas of investigation include cardiac SGLT2 expression, the contribution of various metabolic pathways, and the interaction between renal and cardiac effects[99].
Advanced imaging techniques, such as positron emission tomography and cardiac magnetic resonance spectroscopy, are being used to assess the metabolic effects of SGLT2 inhibitors in real-time, offering insights into the temporal rela
In this study, we identified key human studies reporting the most recent advances in SGLT2 inhibitor research. However, despite the established cardiovascular and renal benefits, several questions surrounding SGLT2 inhibitors remain unanswered[100]. The precise mechanisms driving cardioprotection extend beyond glycosuria and natriuresis[100], with uncertainties surrounding the relative contributions of metabolic shifts toward ketone utilization, anti-inflammatory pathways, improved myocardial sodium homeostasis via sodium-hydrogen exchanger 1 inhibition, and autophagy activation[100]. Notably, empagliflozin retains cardioprotective effects in SGLT2 knockout mice, suggesting potential off-target effects[101]. Additional unresolved issues include the optimal timing for perioperative management to prevent euglycemic diabetic ketoacidosis, the lack of long-term safety data in specific populations such as kidney transplant recipients, and the durability of benefits across different CKD stages[100]. The precise mechanisms mediating arrhythmia reduction, and whether benefits are truly class effects or agent-specific, also remain areas of active research[100,101]. Clarifying these mechanistic gaps and population-specific safety concerns are critical priorities for optimizing SGLT2 inhibitor therapy in clinical practice.
The use of SGLT2 inhibitors represents a paradigm shift in HF management, delivering significant clinical benefits across all HF phenotypes, independent of diabetes status. Landmark clinical trials – including DAPA-HF, EMPEROR-Reduced, EMPEROR-Preserved, and DELIVER – have firmly established SGLT2 inhibitors as integral to guideline-directed HF therapy.
Moving forward, the integration of SGLT2 inhibitors into clinical practice guidelines and routine HF care will be essential for reducing the global burden of HF. The transformation of these glucose-lowering agents into cornerstone HF therapies exemplifies the importance of continued cardiovascular outcome research and highlights the potential for unex
| 1. | McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, Böhm M, Burri H, Butler J, Čelutkienė J, Chioncel O, Cleland JGF, Coats AJS, Crespo-Leiro MG, Farmakis D, Gilard M, Heymans S, Hoes AW, Jaarsma T, Jankowska EA, Lainscak M, Lam CSP, Lyon AR, McMurray JJV, Mebazaa A, Mindham R, Muneretto C, Francesco Piepoli M, Price S, Rosano GMC, Ruschitzka F, Kathrine Skibelund A; ESC Scientific Document Group. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J. 2021;42:3599-3726. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10992] [Cited by in RCA: 9188] [Article Influence: 1837.6] [Reference Citation Analysis (4)] |
| 2. | Savarese G, Becher PM, Lund LH, Seferovic P, Rosano GMC, Coats AJS. Global burden of heart failure: a comprehensive and updated review of epidemiology. Cardiovasc Res. 2023;118:3272-3287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2589] [Cited by in RCA: 2155] [Article Influence: 718.3] [Reference Citation Analysis (1)] |
| 3. | Virani SS, Alonso A, Aparicio HJ, Benjamin EJ, Bittencourt MS, Callaway CW, Carson AP, Chamberlain AM, Cheng S, Delling FN, Elkind MSV, Evenson KR, Ferguson JF, Gupta DK, Khan SS, Kissela BM, Knutson KL, Lee CD, Lewis TT, Liu J, Loop MS, Lutsey PL, Ma J, Mackey J, Martin SS, Matchar DB, Mussolino ME, Navaneethan SD, Perak AM, Roth GA, Samad Z, Satou GM, Schroeder EB, Shah SH, Shay CM, Stokes A, VanWagner LB, Wang NY, Tsao CW; American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. Heart Disease and Stroke Statistics-2021 Update: A Report From the American Heart Association. Circulation. 2021;143:e254-e743. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4769] [Cited by in RCA: 4012] [Article Influence: 802.4] [Reference Citation Analysis (4)] |
| 4. | Heidenreich PA, Bozkurt B, Aguilar D, Allen LA, Byun JJ, Colvin MM, Deswal A, Drazner MH, Dunlay SM, Evers LR, Fang JC, Fedson SE, Fonarow GC, Hayek SS, Hernandez AF, Khazanie P, Kittleson MM, Lee CS, Link MS, Milano CA, Nnacheta LC, Sandhu AT, Stevenson LW, Vardeny O, Vest AR, Yancy CW; ACC/AHA Joint Committee Members. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2022;145:e895-e1032. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1736] [Cited by in RCA: 1858] [Article Influence: 464.5] [Reference Citation Analysis (2)] |
| 5. | Borlaug BA. Evaluation and management of heart failure with preserved ejection fraction. Nat Rev Cardiol. 2020;17:559-573. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 537] [Cited by in RCA: 446] [Article Influence: 74.3] [Reference Citation Analysis (3)] |
| 6. | Hartupee J, Mann DL. Neurohormonal activation in heart failure with reduced ejection fraction. Nat Rev Cardiol. 2017;14:30-38. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 588] [Cited by in RCA: 509] [Article Influence: 56.6] [Reference Citation Analysis (0)] |
| 7. | McMurray JJV, Solomon SD, Inzucchi SE, Køber L, Kosiborod MN, Martinez FA, Ponikowski P, Sabatine MS, Anand IS, Bělohlávek J, Böhm M, Chiang CE, Chopra VK, de Boer RA, Desai AS, Diez M, Drozdz J, Dukát A, Ge J, Howlett JG, Katova T, Kitakaze M, Ljungman CEA, Merkely B, Nicolau JC, O'Meara E, Petrie MC, Vinh PN, Schou M, Tereshchenko S, Verma S, Held C, DeMets DL, Docherty KF, Jhund PS, Bengtsson O, Sjöstrand M, Langkilde AM; DAPA-HF Trial Committees and Investigators. Dapagliflozin in Patients with Heart Failure and Reduced Ejection Fraction. N Engl J Med. 2019;381:1995-2008. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5807] [Cited by in RCA: 5074] [Article Influence: 724.9] [Reference Citation Analysis (6)] |
| 8. | Jones NR, Roalfe AK, Adoki I, Hobbs FDR, Taylor CJ. Survival of patients with chronic heart failure in the community: a systematic review and meta-analysis. Eur J Heart Fail. 2019;21:1306-1325. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 518] [Cited by in RCA: 428] [Article Influence: 61.1] [Reference Citation Analysis (0)] |
| 9. | Redfield MM, Borlaug BA. Heart Failure With Preserved Ejection Fraction: A Review. JAMA. 2023;329:827-838. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 499] [Cited by in RCA: 415] [Article Influence: 138.3] [Reference Citation Analysis (0)] |
| 10. | Shah SJ, Borlaug BA, Chung ES, Cutlip DE, Debonnaire P, Fail PS, Gao Q, Hasenfuß G, Kahwash R, Kaye DM, Litwin SE, Lurz P, Massaro JM, Mohan RC, Ricciardi MJ, Solomon SD, Sverdlov AL, Swarup V, van Veldhuisen DJ, Winkler S, Leon MB; REDUCE LAP-HF II investigators. Atrial shunt device for heart failure with preserved and mildly reduced ejection fraction (REDUCE LAP-HF II): a randomised, multicentre, blinded, sham-controlled trial. Lancet. 2022;399:1130-1140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 241] [Cited by in RCA: 192] [Article Influence: 48.0] [Reference Citation Analysis (0)] |
| 11. | Fonseca-Correa JI, Correa-Rotter R. Sodium-Glucose Cotransporter 2 Inhibitors Mechanisms of Action: A Review. Front Med (Lausanne). 2021;8:777861. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 215] [Cited by in RCA: 164] [Article Influence: 32.8] [Reference Citation Analysis (1)] |
| 12. | Vallon V, Verma S. Effects of SGLT2 Inhibitors on Kidney and Cardiovascular Function. Annu Rev Physiol. 2021;83:503-528. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 416] [Cited by in RCA: 377] [Article Influence: 75.4] [Reference Citation Analysis (10)] |
| 13. | Ghezzi C, Loo DDF, Wright EM. Physiology of renal glucose handling via SGLT1, SGLT2 and GLUT2. Diabetologia. 2018;61:2087-2097. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 304] [Cited by in RCA: 257] [Article Influence: 32.1] [Reference Citation Analysis (0)] |
| 14. | Neal B, Perkovic V, Mahaffey KW, de Zeeuw D, Fulcher G, Erondu N, Shaw W, Law G, Desai M, Matthews DR; CANVAS Program Collaborative Group. Canagliflozin and Cardiovascular and Renal Events in Type 2 Diabetes. N Engl J Med. 2017;377:644-657. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6523] [Cited by in RCA: 5717] [Article Influence: 635.2] [Reference Citation Analysis (7)] |
| 15. | Wright EM. Renal Na(+)-glucose cotransporters. Am J Physiol Renal Physiol. 2001;280:F10-F18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 442] [Cited by in RCA: 380] [Article Influence: 15.2] [Reference Citation Analysis (3)] |
| 16. | Ferrannini E, Mark M, Mayoux E. CV Protection in the EMPA-REG OUTCOME Trial: A "Thrifty Substrate" Hypothesis. Diabetes Care. 2016;39:1108-1114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 845] [Cited by in RCA: 781] [Article Influence: 78.1] [Reference Citation Analysis (3)] |
| 17. | Zinman B, Wanner C, Lachin JM, Fitchett D, Bluhmki E, Hantel S, Mattheus M, Devins T, Johansen OE, Woerle HJ, Broedl UC, Inzucchi SE; EMPA-REG OUTCOME Investigators. Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes. N Engl J Med. 2015;373:2117-2128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10169] [Cited by in RCA: 8821] [Article Influence: 801.9] [Reference Citation Analysis (8)] |
| 18. | Fitchett D, Zinman B, Wanner C, Lachin JM, Hantel S, Salsali A, Johansen OE, Woerle HJ, Broedl UC, Inzucchi SE; EMPA-REG OUTCOME® trial investigators. Heart failure outcomes with empagliflozin in patients with type 2 diabetes at high cardiovascular risk: results of the EMPA-REG OUTCOME® trial. Eur Heart J. 2016;37:1526-1534. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 817] [Cited by in RCA: 727] [Article Influence: 72.7] [Reference Citation Analysis (4)] |
| 19. | Wiviott SD, Raz I, Bonaca MP, Mosenzon O, Kato ET, Cahn A, Silverman MG, Zelniker TA, Kuder JF, Murphy SA, Bhatt DL, Leiter LA, McGuire DK, Wilding JPH, Ruff CT, Gause-Nilsson IAM, Fredriksson M, Johansson PA, Langkilde AM, Sabatine MS; DECLARE–TIMI 58 Investigators. Dapagliflozin and Cardiovascular Outcomes in Type 2 Diabetes. N Engl J Med. 2019;380:347-357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5288] [Cited by in RCA: 4640] [Article Influence: 662.9] [Reference Citation Analysis (4)] |
| 20. | Joshi SS, Singh T, Newby DE, Singh J. Sodium-glucose co-transporter 2 inhibitor therapy: mechanisms of action in heart failure. Heart. 2021;107:1032-1038. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 151] [Cited by in RCA: 136] [Article Influence: 27.2] [Reference Citation Analysis (0)] |
| 21. | Chen S, Coronel R, Hollmann MW, Weber NC, Zuurbier CJ. Direct cardiac effects of SGLT2 inhibitors. Cardiovasc Diabetol. 2022;21:45. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 149] [Cited by in RCA: 135] [Article Influence: 33.8] [Reference Citation Analysis (0)] |
| 22. | Kaplan A, Abidi E, El-Yazbi A, Eid A, Booz GW, Zouein FA. Direct cardiovascular impact of SGLT2 inhibitors: mechanisms and effects. Heart Fail Rev. 2018;23:419-437. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 82] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 23. | DeFronzo RA, Norton L, Abdul-Ghani M. Renal, metabolic and cardiovascular considerations of SGLT2 inhibition. Nat Rev Nephrol. 2017;13:11-26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 474] [Cited by in RCA: 419] [Article Influence: 46.6] [Reference Citation Analysis (3)] |
| 24. | Wright EM, Loo DD, Hirayama BA. Biology of human sodium glucose transporters. Physiol Rev. 2011;91:733-794. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1230] [Cited by in RCA: 1063] [Article Influence: 70.9] [Reference Citation Analysis (0)] |
| 25. | Vrhovac I, Balen Eror D, Klessen D, Burger C, Breljak D, Kraus O, Radović N, Jadrijević S, Aleksic I, Walles T, Sauvant C, Sabolić I, Koepsell H. Localizations of Na(+)-D-glucose cotransporters SGLT1 and SGLT2 in human kidney and of SGLT1 in human small intestine, liver, lung, and heart. Pflugers Arch. 2015;467:1881-1898. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 259] [Cited by in RCA: 240] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 26. | Banerjee SK, McGaffin KR, Pastor-Soler NM, Ahmad F. SGLT1 is a novel cardiac glucose transporter that is perturbed in disease states. Cardiovasc Res. 2009;84:111-118. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 200] [Cited by in RCA: 177] [Article Influence: 10.4] [Reference Citation Analysis (4)] |
| 27. | Rahmoune H, Thompson PW, Ward JM, Smith CD, Hong G, Brown J. Glucose transporters in human renal proximal tubular cells isolated from the urine of patients with non-insulin-dependent diabetes. Diabetes. 2005;54:3427-3434. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 656] [Cited by in RCA: 596] [Article Influence: 28.4] [Reference Citation Analysis (3)] |
| 28. | Meng W, Ellsworth BA, Nirschl AA, McCann PJ, Patel M, Girotra RN, Wu G, Sher PM, Morrison EP, Biller SA, Zahler R, Deshpande PP, Pullockaran A, Hagan DL, Morgan N, Taylor JR, Obermeier MT, Humphreys WG, Khanna A, Discenza L, Robertson JG, Wang A, Han S, Wetterau JR, Janovitz EB, Flint OP, Whaley JM, Washburn WN. Discovery of dapagliflozin: a potent, selective renal sodium-dependent glucose cotransporter 2 (SGLT2) inhibitor for the treatment of type 2 diabetes. J Med Chem. 2008;51:1145-1149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 552] [Cited by in RCA: 471] [Article Influence: 26.2] [Reference Citation Analysis (3)] |
| 29. | Grempler R, Thomas L, Eckhardt M, Himmelsbach F, Sauer A, Sharp DE, Bakker RA, Mark M, Klein T, Eickelmann P. Empagliflozin, a novel selective sodium glucose cotransporter-2 (SGLT-2) inhibitor: characterisation and comparison with other SGLT-2 inhibitors. Diabetes Obes Metab. 2012;14:83-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 507] [Cited by in RCA: 475] [Article Influence: 33.9] [Reference Citation Analysis (3)] |
| 30. | Scheen AJ. Pharmacokinetics, Pharmacodynamics and Clinical Use of SGLT2 Inhibitors in Patients with Type 2 Diabetes Mellitus and Chronic Kidney Disease. Clin Pharmacokinet. 2015;54:691-708. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 139] [Article Influence: 12.6] [Reference Citation Analysis (3)] |
| 31. | Verma S, McMurray JJV. SGLT2 inhibitors and mechanisms of cardiovascular benefit: a state-of-the-art review. Diabetologia. 2018;61:2108-2117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 826] [Cited by in RCA: 733] [Article Influence: 91.6] [Reference Citation Analysis (3)] |
| 32. | Maejima Y. SGLT2 Inhibitors Play a Salutary Role in Heart Failure via Modulation of the Mitochondrial Function. Front Cardiovasc Med. 2019;6:186. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 108] [Cited by in RCA: 102] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 33. | Packer M, Anker SD, Butler J, Filippatos G, Pocock SJ, Carson P, Januzzi J, Verma S, Tsutsui H, Brueckmann M, Jamal W, Kimura K, Schnee J, Zeller C, Cotton D, Bocchi E, Böhm M, Choi DJ, Chopra V, Chuquiure E, Giannetti N, Janssens S, Zhang J, Gonzalez Juanatey JR, Kaul S, Brunner-La Rocca HP, Merkely B, Nicholls SJ, Perrone S, Pina I, Ponikowski P, Sattar N, Senni M, Seronde MF, Spinar J, Squire I, Taddei S, Wanner C, Zannad F; EMPEROR-Reduced Trial Investigators. Cardiovascular and Renal Outcomes with Empagliflozin in Heart Failure. N Engl J Med. 2020;383:1413-1424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4235] [Cited by in RCA: 3674] [Article Influence: 612.3] [Reference Citation Analysis (6)] |
| 34. | Jhund PS, Solomon SD, Docherty KF, Heerspink HJL, Anand IS, Böhm M, Chopra V, de Boer RA, Desai AS, Ge J, Kitakaze M, Merkley B, O'Meara E, Shou M, Tereshchenko S, Verma S, Vinh PN, Inzucchi SE, Køber L, Kosiborod MN, Martinez FA, Ponikowski P, Sabatine MS, Bengtsson O, Langkilde AM, Sjöstrand M, McMurray JJV. Efficacy of Dapagliflozin on Renal Function and Outcomes in Patients With Heart Failure With Reduced Ejection Fraction: Results of DAPA-HF. Circulation. 2021;143:298-309. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 267] [Cited by in RCA: 237] [Article Influence: 47.4] [Reference Citation Analysis (3)] |
| 35. | Cherney DZI, Zinman B, Inzucchi SE, Koitka-Weber A, Mattheus M, von Eynatten M, Wanner C. Effects of empagliflozin on the urinary albumin-to-creatinine ratio in patients with type 2 diabetes and established cardiovascular disease: an exploratory analysis from the EMPA-REG OUTCOME randomised, placebo-controlled trial. Lancet Diabetes Endocrinol. 2017;5:610-621. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 328] [Cited by in RCA: 294] [Article Influence: 32.7] [Reference Citation Analysis (3)] |
| 36. | Weber MA, Mansfield TA, Alessi F, Iqbal N, Parikh S, Ptaszynska A. Effects of dapagliflozin on blood pressure in hypertensive diabetic patients on renin-angiotensin system blockade. Blood Press. 2016;25:93-103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 93] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 37. | Mone P, Varzideh F, Jankauskas SS, Pansini A, Lombardi A, Frullone S, Santulli G. SGLT2 Inhibition via Empagliflozin Improves Endothelial Function and Reduces Mitochondrial Oxidative Stress: Insights From Frail Hypertensive and Diabetic Patients. Hypertension. 2022;79:1633-1643. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 188] [Cited by in RCA: 175] [Article Influence: 43.8] [Reference Citation Analysis (0)] |
| 38. | Mudaliar S, Alloju S, Henry RR. Can a Shift in Fuel Energetics Explain the Beneficial Cardiorenal Outcomes in the EMPA-REG OUTCOME Study? A Unifying Hypothesis. Diabetes Care. 2016;39:1115-1122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 515] [Cited by in RCA: 474] [Article Influence: 47.4] [Reference Citation Analysis (0)] |
| 39. | Nielsen R, Møller N, Gormsen LC, Tolbod LP, Hansson NH, Sorensen J, Harms HJ, Frøkiær J, Eiskjaer H, Jespersen NR, Mellemkjaer S, Lassen TR, Pryds K, Bøtker HE, Wiggers H. Cardiovascular Effects of Treatment With the Ketone Body 3-Hydroxybutyrate in Chronic Heart Failure Patients. Circulation. 2019;139:2129-2141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 461] [Cited by in RCA: 420] [Article Influence: 60.0] [Reference Citation Analysis (0)] |
| 40. | Lopaschuk GD, Verma S. Mechanisms of Cardiovascular Benefits of Sodium Glucose Co-Transporter 2 (SGLT2) Inhibitors: A State-of-the-Art Review. JACC Basic Transl Sci. 2020;5:632-644. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 737] [Cited by in RCA: 686] [Article Influence: 114.3] [Reference Citation Analysis (5)] |
| 41. | Baartscheer A, Schumacher CA, Wüst RC, Fiolet JW, Stienen GJ, Coronel R, Zuurbier CJ. Empagliflozin decreases myocardial cytoplasmic Na(+) through inhibition of the cardiac Na(+)/H(+) exchanger in rats and rabbits. Diabetologia. 2017;60:568-573. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 567] [Cited by in RCA: 523] [Article Influence: 58.1] [Reference Citation Analysis (0)] |
| 42. | Rosano GM, Vitale C, Seferovic P. Heart Failure in Patients with Diabetes Mellitus. Card Fail Rev. 2017;3:52-55. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 159] [Cited by in RCA: 131] [Article Influence: 14.6] [Reference Citation Analysis (4)] |
| 43. | Li X, Flynn ER, do Carmo JM, Wang Z, da Silva AA, Mouton AJ, Omoto ACM, Hall ME, Hall JE. Direct Cardiac Actions of Sodium-Glucose Cotransporter 2 Inhibition Improve Mitochondrial Function and Attenuate Oxidative Stress in Pressure Overload-Induced Heart Failure. Front Cardiovasc Med. 2022;9:859253. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 40] [Article Influence: 10.0] [Reference Citation Analysis (1)] |
| 44. | Sawicki KT, Ben-Sahra I, McNally EM. SGLT2 Inhibition on Cardiac Mitochondrial Function: Searching for a Sweet Spot. J Am Heart Assoc. 2021;10:e021949. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 45. | Xing YJ, Liu BH, Wan SJ, Cheng Y, Zhou SM, Sun Y, Yao XM, Hua Q, Meng XJ, Cheng JH, Zhong M, Zhang Y, Lv K, Kong X. A SGLT2 Inhibitor Dapagliflozin Alleviates Diabetic Cardiomyopathy by Suppressing High Glucose-Induced Oxidative Stress in vivo and in vitro. Front Pharmacol. 2021;12:708177. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 59] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 46. | Durak A, Olgar Y, Degirmenci S, Akkus E, Tuncay E, Turan B. A SGLT2 inhibitor dapagliflozin suppresses prolonged ventricular-repolarization through augmentation of mitochondrial function in insulin-resistant metabolic syndrome rats. Cardiovasc Diabetol. 2018;17:144. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 150] [Cited by in RCA: 146] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 47. | Zhou H, Wang S, Zhu P, Hu S, Chen Y, Ren J. Empagliflozin rescues diabetic myocardial microvascular injury via AMPK-mediated inhibition of mitochondrial fission. Redox Biol. 2018;15:335-346. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 474] [Cited by in RCA: 473] [Article Influence: 59.1] [Reference Citation Analysis (0)] |
| 48. | Bode D, Semmler L, Wakula P, Hegemann N, Primessnig U, Beindorff N, Powell D, Dahmen R, Ruetten H, Oeing C, Alogna A, Messroghli D, Pieske BM, Heinzel FR, Hohendanner F. Dual SGLT-1 and SGLT-2 inhibition improves left atrial dysfunction in HFpEF. Cardiovasc Diabetol. 2021;20:7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 105] [Cited by in RCA: 96] [Article Influence: 19.2] [Reference Citation Analysis (0)] |
| 49. | Murphy SP, Kakkar R, McCarthy CP, Januzzi JL Jr. Inflammation in Heart Failure: JACC State-of-the-Art Review. J Am Coll Cardiol. 2020;75:1324-1340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 591] [Cited by in RCA: 523] [Article Influence: 87.2] [Reference Citation Analysis (0)] |
| 50. | Quagliariello V, De Laurentiis M, Rea D, Barbieri A, Monti MG, Carbone A, Paccone A, Altucci L, Conte M, Canale ML, Botti G, Maurea N. The SGLT-2 inhibitor empagliflozin improves myocardial strain, reduces cardiac fibrosis and pro-inflammatory cytokines in non-diabetic mice treated with doxorubicin. Cardiovasc Diabetol. 2021;20:150. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 315] [Cited by in RCA: 299] [Article Influence: 59.8] [Reference Citation Analysis (0)] |
| 51. | Wang C, Qin Y, Zhang X, Yang Y, Wu X, Liu J, Qin S, Chen K, Xiao W. Effect of Dapagliflozin on Indicators of Myocardial Fibrosis and Levels of Inflammatory Factors in Heart Failure Patients. Dis Markers. 2022;2022:5834218. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 52. | Yang Z, Li T, Xian J, Chen J, Huang Y, Zhang Q, Lin X, Lu H, Lin Y. SGLT2 inhibitor dapagliflozin attenuates cardiac fibrosis and inflammation by reverting the HIF-2α signaling pathway in arrhythmogenic cardiomyopathy. FASEB J. 2022;36:e22410. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 63] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 53. | Chen B, Guo J, Ye H, Wang X, Feng Y. Role and molecular mechanisms of SGLT2 inhibitors in pathological cardiac remodeling (Review). Mol Med Rep. 2024;29:73. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 25] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 54. | Albulushi A, Askari KM, Al-Abedi AM, Al-Kulaibi MA, Hasan MS, Hosseini Z, Al-Rahman MT, Tanoh DB, Hasan AS, Al-Helli Y, Basouni A. Impact of SGLT2 inhibitors on myocardial fibrosis in diabetic HFpEF: a longitudinal study. Eur J Med Res. 2025;30:592. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 55. | Zannad F, Ferreira JP, Pocock SJ, Zeller C, Anker SD, Butler J, Filippatos G, Hauske SJ, Brueckmann M, Pfarr E, Schnee J, Wanner C, Packer M. Cardiac and Kidney Benefits of Empagliflozin in Heart Failure Across the Spectrum of Kidney Function: Insights From EMPEROR-Reduced. Circulation. 2021;143:310-321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 253] [Cited by in RCA: 227] [Article Influence: 45.4] [Reference Citation Analysis (3)] |
| 56. | Nassif ME, Windsor SL, Borlaug BA, Kitzman DW, Shah SJ, Tang F, Khariton Y, Malik AO, Khumri T, Umpierrez G, Lamba S, Sharma K, Khan SS, Chandra L, Gordon RA, Ryan JJ, Chaudhry SP, Joseph SM, Chow CH, Kanwar MK, Pursley M, Siraj ES, Lewis GD, Clemson BS, Fong M, Kosiborod MN. The SGLT2 inhibitor dapagliflozin in heart failure with preserved ejection fraction: a multicenter randomized trial. Nat Med. 2021;27:1954-1960. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 528] [Cited by in RCA: 448] [Article Influence: 89.6] [Reference Citation Analysis (0)] |
| 57. | Pabel S, Hamdani N, Singh J, Sossalla S. Potential Mechanisms of SGLT2 Inhibitors for the Treatment of Heart Failure With Preserved Ejection Fraction. Front Physiol. 2021;12:752370. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 58. | Heath R, Johnsen H, Strain WD, Evans M. Emerging Horizons in Heart Failure with Preserved Ejection Fraction: The Role of SGLT2 Inhibitors. Diabetes Ther. 2022;13:241-250. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 59. | Jhund PS. SGLT2 Inhibitors and Heart Failure with Preserved Ejection Fraction. Heart Fail Clin. 2022;18:579-586. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 60. | De Marzo V, Savarese G, Porto I, Metra M, Ameri P. Efficacy of SGLT2-inhibitors across different definitions of heart failure with preserved ejection fraction. J Cardiovasc Med (Hagerstown). 2023;24:537-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 9] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 61. | Jaiswal A, Jaiswal V, Ang SP, Hanif M, Vadhera A, Agrawal V, Kumar T, Nair AM, Borra V, Garimella V, Ishak A, Wajid Z, Song D, Attia AM, Huang H, Aguilera Alvarez VH, Shrestha AB, Biswas M. SGLT2 inhibitors among patients with heart failure with preserved ejection fraction: A meta-analysis of randomised controlled trials. Medicine (Baltimore). 2023;102:e34693. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 20] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 62. | Wagdy K, Nagy S. EMPEROR-Preserved: SGLT2 inhibitors breakthrough in the management of heart failure with preserved ejection fraction. Glob Cardiol Sci Pract. 2021;2021:e202117. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 63. | De Lorenzi AB, Kaplinsky E, Zambrano MR, Chaume LT, Rosas JM. Emerging concepts in heart failure management and treatment: focus on SGLT2 inhibitors in heart failure with preserved ejection fraction. Drugs Context. 2023;12:2022-7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 64. | Leo I, Salerno N, Figliozzi S, Cersosimo A, Ielapi J, Stankowski K, Bisaccia G, Dellegrottaglie S, Canino G, De Rosa S, Sorrentino S, Bucciarelli-Ducci C, Torella D. Effect of SGLT2 inhibitors on cardiac structure and function assessed by cardiac magnetic resonance: a systematic review and meta-analysis. Cardiovasc Diabetol. 2025;24:345. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 65. | Petrie MC, Verma S, Docherty KF, Inzucchi SE, Anand I, Belohlávek J, Böhm M, Chiang CE, Chopra VK, de Boer RA, Desai AS, Diez M, Drozdz J, Dukát A, Ge J, Howlett J, Katova T, Kitakaze M, Ljungman CEA, Merkely B, Nicolau JC, O'Meara E, Vinh PN, Schou M, Tereshchenko S, Køber L, Kosiborod MN, Langkilde AM, Martinez FA, Ponikowski P, Sabatine MS, Sjöstrand M, Solomon SD, Johanson P, Greasley PJ, Boulton D, Bengtsson O, Jhund PS, McMurray JJV. Effect of Dapagliflozin on Worsening Heart Failure and Cardiovascular Death in Patients With Heart Failure With and Without Diabetes. JAMA. 2020;323:1353-1368. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 422] [Cited by in RCA: 389] [Article Influence: 64.8] [Reference Citation Analysis (4)] |
| 66. | Docherty KF, Jhund PS, Inzucchi SE, Køber L, Kosiborod MN, Martinez FA, Ponikowski P, DeMets DL, Sabatine MS, Bengtsson O, Sjöstrand M, Langkilde AM, Desai AS, Diez M, Howlett JG, Katova T, Ljungman CEA, O'Meara E, Petrie MC, Schou M, Verma S, Vinh PN, Solomon SD, McMurray JJV. Effects of dapagliflozin in DAPA-HF according to background heart failure therapy. Eur Heart J. 2020;41:2379-2392. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 159] [Cited by in RCA: 145] [Article Influence: 24.2] [Reference Citation Analysis (4)] |
| 67. | Martinez FA, Serenelli M, Nicolau JC, Petrie MC, Chiang CE, Tereshchenko S, Solomon SD, Inzucchi SE, Køber L, Kosiborod MN, Ponikowski P, Sabatine MS, DeMets DL, Dutkiewicz-Piasecka M, Bengtsson O, Sjöstrand M, Langkilde AM, Jhund PS, McMurray JJV. Efficacy and Safety of Dapagliflozin in Heart Failure With Reduced Ejection Fraction According to Age: Insights From DAPA-HF. Circulation. 2020;141:100-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 187] [Cited by in RCA: 163] [Article Influence: 27.2] [Reference Citation Analysis (3)] |
| 68. | Kosiborod MN, Jhund PS, Docherty KF, Diez M, Petrie MC, Verma S, Nicolau JC, Merkely B, Kitakaze M, DeMets DL, Inzucchi SE, Køber L, Martinez FA, Ponikowski P, Sabatine MS, Solomon SD, Bengtsson O, Lindholm D, Niklasson A, Sjöstrand M, Langkilde AM, McMurray JJV. Effects of Dapagliflozin on Symptoms, Function, and Quality of Life in Patients With Heart Failure and Reduced Ejection Fraction: Results From the DAPA-HF Trial. Circulation. 2020;141:90-99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 313] [Cited by in RCA: 273] [Article Influence: 45.5] [Reference Citation Analysis (3)] |
| 69. | Jackson AM, Dewan P, Anand IS, Bělohlávek J, Bengtsson O, de Boer RA, Böhm M, Boulton DW, Chopra VK, DeMets DL, Docherty KF, Dukát A, Greasley PJ, Howlett JG, Inzucchi SE, Katova T, Køber L, Kosiborod MN, Langkilde AM, Lindholm D, Ljungman CEA, Martinez FA, O'Meara E, Sabatine MS, Sjöstrand M, Solomon SD, Tereshchenko S, Verma S, Jhund PS, McMurray JJV. Dapagliflozin and Diuretic Use in Patients With Heart Failure and Reduced Ejection Fraction in DAPA-HF. Circulation. 2020;142:1040-1054. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 142] [Article Influence: 23.7] [Reference Citation Analysis (0)] |
| 70. | Anker SD, Butler J, Filippatos G, Khan MS, Marx N, Lam CSP, Schnaidt S, Ofstad AP, Brueckmann M, Jamal W, Bocchi EA, Ponikowski P, Perrone SV, Januzzi JL, Verma S, Böhm M, Ferreira JP, Pocock SJ, Zannad F, Packer M. Effect of Empagliflozin on Cardiovascular and Renal Outcomes in Patients With Heart Failure by Baseline Diabetes Status: Results From the EMPEROR-Reduced Trial. Circulation. 2021;143:337-349. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 304] [Cited by in RCA: 266] [Article Influence: 53.2] [Reference Citation Analysis (3)] |
| 71. | Ferreira JP, Zannad F, Pocock SJ, Anker SD, Butler J, Filippatos G, Brueckmann M, Jamal W, Steubl D, Schueler E, Packer M. Interplay of Mineralocorticoid Receptor Antagonists and Empagliflozin in Heart Failure: EMPEROR-Reduced. J Am Coll Cardiol. 2021;77:1397-1407. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 135] [Article Influence: 27.0] [Reference Citation Analysis (3)] |
| 72. | Zannad F, Ferreira JP, Pocock SJ, Anker SD, Butler J, Filippatos G, Brueckmann M, Ofstad AP, Pfarr E, Jamal W, Packer M. SGLT2 inhibitors in patients with heart failure with reduced ejection fraction: a meta-analysis of the EMPEROR-Reduced and DAPA-HF trials. Lancet. 2020;396:819-829. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1182] [Cited by in RCA: 983] [Article Influence: 163.8] [Reference Citation Analysis (3)] |
| 73. | Anker SD, Butler J, Filippatos G, Ferreira JP, Bocchi E, Böhm M, Brunner-La Rocca HP, Choi DJ, Chopra V, Chuquiure-Valenzuela E, Giannetti N, Gomez-Mesa JE, Janssens S, Januzzi JL, Gonzalez-Juanatey JR, Merkely B, Nicholls SJ, Perrone SV, Piña IL, Ponikowski P, Senni M, Sim D, Spinar J, Squire I, Taddei S, Tsutsui H, Verma S, Vinereanu D, Zhang J, Carson P, Lam CSP, Marx N, Zeller C, Sattar N, Jamal W, Schnaidt S, Schnee JM, Brueckmann M, Pocock SJ, Zannad F, Packer M; EMPEROR-Preserved Trial Investigators. Empagliflozin in Heart Failure with a Preserved Ejection Fraction. N Engl J Med. 2021;385:1451-1461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3998] [Cited by in RCA: 3426] [Article Influence: 685.2] [Reference Citation Analysis (6)] |
| 74. | Butler J, Filippatos G, Jamal Siddiqi T, Brueckmann M, Böhm M, Chopra VK, Pedro Ferreira J, Januzzi JL, Kaul S, Piña IL, Ponikowski P, Shah SJ, Senni M, Vedin O, Verma S, Peil B, Pocock SJ, Zannad F, Packer M, Anker SD. Empagliflozin, Health Status, and Quality of Life in Patients With Heart Failure and Preserved Ejection Fraction: The EMPEROR-Preserved Trial. Circulation. 2022;145:184-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 150] [Article Influence: 37.5] [Reference Citation Analysis (0)] |
| 75. | Verma S, Mazer CD, Fitchett D, Inzucchi SE, Pfarr E, George JT, Zinman B. Empagliflozin reduces cardiovascular events, mortality and renal events in participants with type 2 diabetes after coronary artery bypass graft surgery: subanalysis of the EMPA-REG OUTCOME® randomised trial. Diabetologia. 2018;61:1712-1723. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 106] [Cited by in RCA: 93] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 76. | Bhatt DL, Szarek M, Steg PG, Cannon CP, Leiter LA, McGuire DK, Lewis JB, Riddle MC, Voors AA, Metra M, Lund LH, Komajda M, Testani JM, Wilcox CS, Ponikowski P, Lopes RD, Verma S, Lapuerta P, Pitt B; SOLOIST-WHF Trial Investigators. Sotagliflozin in Patients with Diabetes and Recent Worsening Heart Failure. N Engl J Med. 2021;384:117-128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1568] [Cited by in RCA: 1389] [Article Influence: 277.8] [Reference Citation Analysis (3)] |
| 77. | Butler J, Usman MS, Filippatos G, Ferreira JP, Böhm M, Brueckmann M, Januzzi JL, Kaul S, Piña IL, Ponikowski P, Senni M, Sumin M, Verma S, Zaremba-Pechmann L, Pocock SJ, Packer M, Anker S. Safety and Efficacy of Empagliflozin and Diuretic Use in Patients with Heart Failure and Preserved Ejection Fraction: A Post Hoc Analysis of the EMPEROR-Preserved Trial. JAMA Cardiol. 2023;8:640-649. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 49] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 78. | Jhund PS, Kondo T, Butt JH, Docherty KF, Claggett BL, Desai AS, Vaduganathan M, Gasparyan SB, Bengtsson O, Lindholm D, Petersson M, Langkilde AM, de Boer RA, DeMets D, Hernandez AF, Inzucchi SE, Kosiborod MN, Køber L, Lam CSP, Martinez FA, Sabatine MS, Shah SJ, Solomon SD, McMurray JJV. Dapagliflozin across the range of ejection fraction in patients with heart failure: a patient-level, pooled meta-analysis of DAPA-HF and DELIVER. Nat Med. 2022;28:1956-1964. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 253] [Cited by in RCA: 198] [Article Influence: 49.5] [Reference Citation Analysis (0)] |
| 79. | Solomon SD, McMurray JJV, Claggett B, de Boer RA, DeMets D, Hernandez AF, Inzucchi SE, Kosiborod MN, Lam CSP, Martinez F, Shah SJ, Desai AS, Jhund PS, Belohlavek J, Chiang CE, Borleffs CJW, Comin-Colet J, Dobreanu D, Drozdz J, Fang JC, Alcocer-Gamba MA, Al Habeeb W, Han Y, Cabrera Honorio JW, Janssens SP, Katova T, Kitakaze M, Merkely B, O'Meara E, Saraiva JFK, Tereshchenko SN, Thierer J, Vaduganathan M, Vardeny O, Verma S, Pham VN, Wilderäng U, Zaozerska N, Bachus E, Lindholm D, Petersson M, Langkilde AM; DELIVER Trial Committees and Investigators. Dapagliflozin in Heart Failure with Mildly Reduced or Preserved Ejection Fraction. N Engl J Med. 2022;387:1089-1098. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2275] [Cited by in RCA: 1958] [Article Influence: 489.5] [Reference Citation Analysis (6)] |
| 80. | Vaduganathan M, Docherty KF, Claggett BL, Jhund PS, de Boer RA, Hernandez AF, Inzucchi SE, Kosiborod MN, Lam CSP, Martinez F, Shah SJ, Desai AS, McMurray JJV, Solomon SD. SGLT-2 inhibitors in patients with heart failure: a comprehensive meta-analysis of five randomised controlled trials. Lancet. 2022;400:757-767. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 797] [Cited by in RCA: 642] [Article Influence: 160.5] [Reference Citation Analysis (3)] |
| 81. | Park S, Jeong HE, Lee H, You SC, Shin JY. Association of sodium-glucose cotransporter 2 inhibitors with post-discharge outcomes in patients with acute heart failure with type 2 diabetes: a cohort study. Cardiovasc Diabetol. 2023;22:191. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 82. | Modzelewski KL, Pipilas A, Bosch NA. Comparative Outcomes of Empagliflozin to Dapagliflozin in Patients With Heart Failure. JAMA Netw Open. 2024;7:e249305. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 42] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 83. | Pelletier R, Ng K, Alkabbani W, Labib Y, Mourad N, Gamble JM. Adverse events associated with sodium glucose co-transporter 2 inhibitors: an overview of quantitative systematic reviews. Ther Adv Drug Saf. 2021;12:2042098621989134. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 56] [Article Influence: 11.2] [Reference Citation Analysis (1)] |
| 84. | Kodur N, Sharew B, Lansang MC, Tang WHW. Managing the side effects of sodium-glucose cotransporter-2 inhibitors. Cleve Clin J Med. 2025;92:503-511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 85. | Bazan DZ, Esqueda L, Ibrahim A, Gonzalez L, Skubic JJ, Reilly J, Cavazos R, Verduzco R Jr. Sodium-Glucose Cotransporter-2 Inhibitors and Euglycemic Diabetic Ketoacidosis: A Case Series of Three Post-surgical Patients. Cureus. 2025;17:e84665. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 86. | Ueda P, Svanström H, Melbye M, Eliasson B, Svensson AM, Franzén S, Gudbjörnsdottir S, Hveem K, Jonasson C, Pasternak B. Sodium glucose cotransporter 2 inhibitors and risk of serious adverse events: nationwide register based cohort study. BMJ. 2018;363:k4365. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 306] [Cited by in RCA: 271] [Article Influence: 33.9] [Reference Citation Analysis (4)] |
| 87. | Voors AA, Angermann CE, Teerlink JR, Collins SP, Kosiborod M, Biegus J, Ferreira JP, Nassif ME, Psotka MA, Tromp J, Borleffs CJW, Ma C, Comin-Colet J, Fu M, Janssens SP, Kiss RG, Mentz RJ, Sakata Y, Schirmer H, Schou M, Schulze PC, Spinarova L, Volterrani M, Wranicz JK, Zeymer U, Zieroth S, Brueckmann M, Blatchford JP, Salsali A, Ponikowski P. The SGLT2 inhibitor empagliflozin in patients hospitalized for acute heart failure: a multinational randomized trial. Nat Med. 2022;28:568-574. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 760] [Cited by in RCA: 651] [Article Influence: 162.8] [Reference Citation Analysis (7)] |
| 88. | Bhatt DL, Szarek M, Pitt B, Cannon CP, Leiter LA, McGuire DK, Lewis JB, Riddle MC, Inzucchi SE, Kosiborod MN, Cherney DZI, Dwyer JP, Scirica BM, Bailey CJ, Díaz R, Ray KK, Udell JA, Lopes RD, Lapuerta P, Steg PG; SCORED Investigators. Sotagliflozin in Patients with Diabetes and Chronic Kidney Disease. N Engl J Med. 2021;384:129-139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1002] [Cited by in RCA: 877] [Article Influence: 175.4] [Reference Citation Analysis (6)] |
| 89. | Cannon CP, Pratley R, Dagogo-Jack S, Mancuso J, Huyck S, Masiukiewicz U, Charbonnel B, Frederich R, Gallo S, Cosentino F, Shih WJ, Gantz I, Terra SG, Cherney DZI, McGuire DK; VERTIS CV Investigators. Cardiovascular Outcomes with Ertugliflozin in Type 2 Diabetes. N Engl J Med. 2020;383:1425-1435. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1284] [Cited by in RCA: 1112] [Article Influence: 185.3] [Reference Citation Analysis (3)] |
| 90. | Natali A, Nesti L, Tricò D, Ferrannini E. Effects of GLP-1 receptor agonists and SGLT-2 inhibitors on cardiac structure and function: a narrative review of clinical evidence. Cardiovasc Diabetol. 2021;20:196. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 49] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 91. | Wang A, Li Z, Zhuo S, Gao F, Zhang H, Zhang Z, Ren G, Ma X. Mechanisms of Cardiorenal Protection With SGLT2 Inhibitors in Patients With T2DM Based on Network Pharmacology. Front Cardiovasc Med. 2022;9:857952. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 92. | Reifsnider OS, Kansal AR, Gandhi PK, Cragin L, Brand SB, Pfarr E, Fahrbach K, Ustyugova A. Cost-effectiveness of empagliflozin versus canagliflozin, dapagliflozin, or standard of care in patients with type 2 diabetes and established cardiovascular disease. BMJ Open Diabetes Res Care. 2021;9:e001313. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 93. | Nakagaito M, Joho S, Ushijima R, Nakamura M, Kinugawa K. Comparison of Canagliflozin, Dapagliflozin and Empagliflozin Added to Heart Failure Treatment in Decompensated Heart Failure Patients With Type 2 Diabetes Mellitus. Circ Rep. 2019;1:405-413. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 23] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 94. | Figtree GA, Rådholm K, Barrett TD, Perkovic V, Mahaffey KW, de Zeeuw D, Fulcher G, Matthews DR, Shaw W, Neal B. Effects of Canagliflozin on Heart Failure Outcomes Associated With Preserved and Reduced Ejection Fraction in Type 2 Diabetes Mellitus. Circulation. 2019;139:2591-2593. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 118] [Article Influence: 16.9] [Reference Citation Analysis (3)] |
| 95. | McMurray JJV, Wheeler DC, Stefánsson BV, Jongs N, Postmus D, Correa-Rotter R, Chertow GM, Greene T, Held C, Hou FF, Mann JFE, Rossing P, Sjöström CD, Toto RD, Langkilde AM, Heerspink HJL; DAPA-CKD Trial Committees and Investigators. Effect of Dapagliflozin on Clinical Outcomes in Patients With Chronic Kidney Disease, With and Without Cardiovascular Disease. Circulation. 2021;143:438-448. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 103] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 96. | McEwan P, Morgan AR, Boyce R, Green N, Song B, Huang J, Bergenheim K. Cardiorenal disease in the United States: Future health care burden and potential impact of novel therapies. J Manag Care Spec Pharm. 2022;28:415-424. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 97. | Luconi M, Cantini G, Crescioli C. Repurposing glucose-lowering drugs for cancer therapy. Trends Cancer. 2025;11:691-710. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 98. | Bea S, Ko HY, Bae JH, Cho YM, Chang Y, Ryu S, Byrne CD, Shin JY. Risk of hepatic events associated with use of sodium-glucose cotransporter-2 inhibitors versus glucagon-like peptide-1 receptor agonists, and thiazolidinediones among patients with metabolic dysfunction-associated steatotic liver disease. Gut. 2025;74:284-294. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 42] [Article Influence: 42.0] [Reference Citation Analysis (0)] |
| 99. | Butler J, Anker SD, Lund LH, Coats AJS, Filippatos G, Siddiqi TJ, Friede T, Fabien V, Kosiborod M, Metra M, Piña IL, Pinto F, Rossignol P, van der Meer P, Bahit C, Belohlavek J, Böhm M, Brugts JJ, Cleland JGF, Ezekowitz J, Bayes-Genis A, Gotsman I, Goudev A, Khintibidze I, Lindenfeld J, Mentz RJ, Merkely B, Montes EC, Mullens W, Nicolau JC, Parkhomenko A, Ponikowski P, Seferovic PM, Senni M, Shlyakhto E, Cohen-Solal A, Szecsödy P, Jensen K, Dorigotti F, Weir MR, Pitt B. Patiromer for the management of hyperkalemia in heart failure with reduced ejection fraction: the DIAMOND trial. Eur Heart J. 2022;43:4362-4373. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 179] [Cited by in RCA: 166] [Article Influence: 41.5] [Reference Citation Analysis (0)] |
| 100. | Savage P, Dixon L, Grieve D, Watson C. SGLT2 Inhibition in Heart Failure: Clues to Cardiac Effects? Cardiol Rev. 2025;33:509-516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 101. | Forzano I, Wilson S, Lombardi A, Jankauskas SS, Kansakar U, Mone P, Varzideh F, Santulli G. SGLT2 inhibitors: an evidence-based update on cardiovascular implications. Expert Opin Investig Drugs. 2023;32:839-847. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 6.7] [Reference Citation Analysis (0)] |