INTRODUCTION
Small intestinal hemangioma is a rare benign tumor of the digestive tract, accounting for approximately 0.05% of all intestinal tumors[1]. To date, no clear clinical practice guidelines or expert consensus statements have been established to guide the diagnosis and management of such lesions. Intestinal serosal hemangiomas are even rarer, with neither epidemiological research data available nor their pathogenesis clearly understood. This article reports a case of giant cavernous hemangioma of the ileal serosa, summarizes its diagnostic and therapeutic strategies, and reviews its pathophysiological characteristics based on the literature, aiming to enhance clinical understanding and provide references for diagnostic and therapeutic approaches.
CASE PRESENTATION
Chief complaints
The patient was found to have a space-occupying lesion in the terminal ileum one month after a cesarean section.
History of present illness
On September 5, 2021, a 29-year-old female patient was found to have an occupying lesion in the terminal ileum during a cesarean section procedure (which had not been detected preoperatively due to limited medical conditions at the time). Since the nature of the mass remained unclear, no specific treatment was administered. One month later, the patient presented to our hospital for further evaluation. Upon admission, the patient exhibited no symptoms of gastrointestinal bleeding or chronic anemia.
History of past illness
The patient denies any past medical history.
Personal and family history
The patient denies any personal or family medical history.
Physical examination
The abdomen is soft and flat, with no tenderness or rebound pain, and no obvious abnormalities are palpable. Percussion reveals tympany, and bowel sounds are normal.
Laboratory examinations
No obvious abnormalities detected.
Imaging examinations
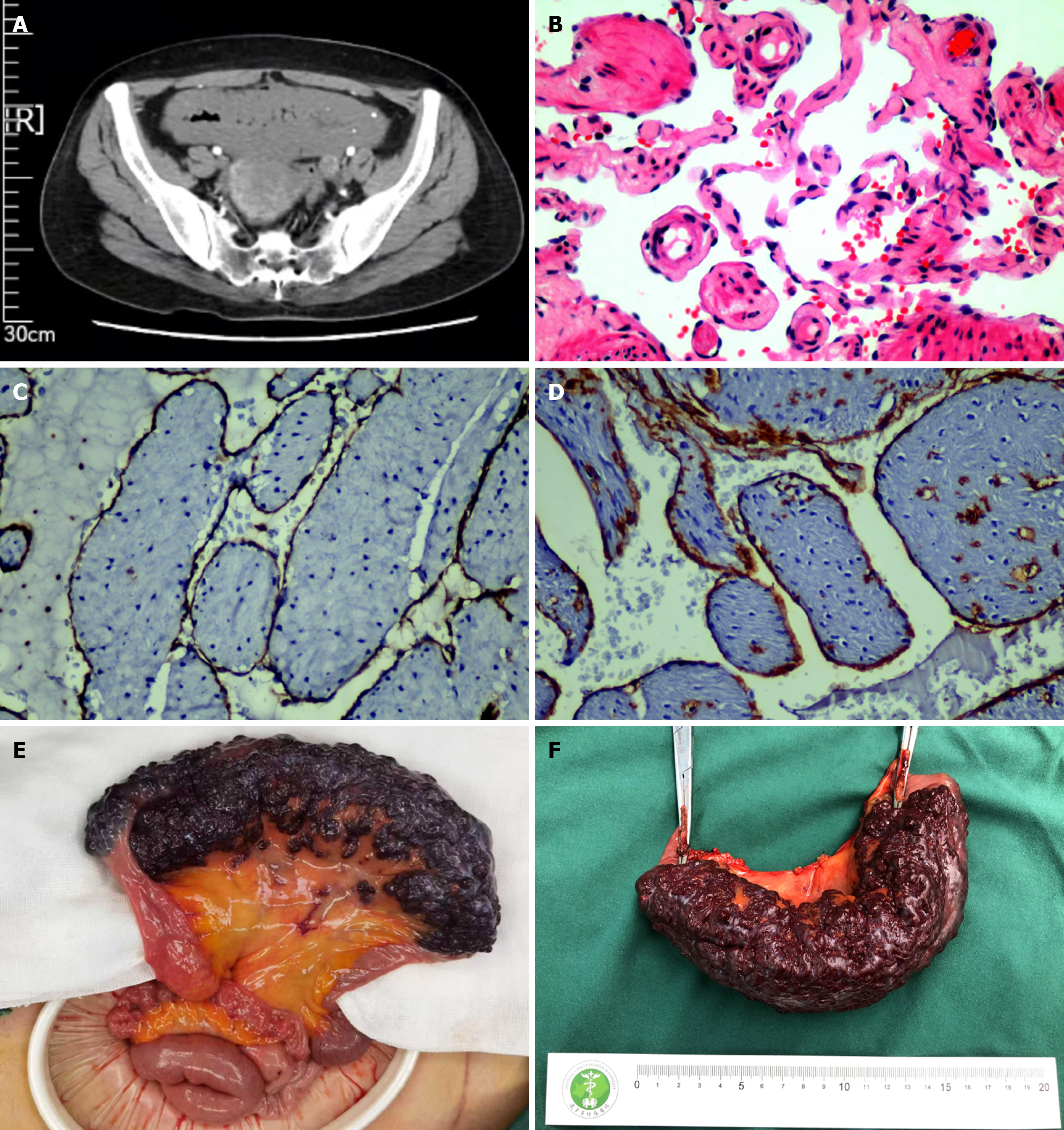
Ultrasonography revealed a heterogeneous echo adjacent to the right ovary, measuring approximately 12.3 cm × 10.8 cm. Contrast-enhanced computed tomography showed focal thickening of the small intestinal wall (Figure 1A). The initial diagnosis was small intestinal lymphoma. Electronic colonoscopy yielded normal results. Double-balloon enteroscopy was not performed due to technical limitations.
Figure 1 Examinations and treatments related to the case.
A: Pelvic computed tomography: Local thickening of the small bowel wall in the lower abdomen with punctate high-density foci, suggestive of a vascular lesion or tumor with calcifications; B: Hematoxylin and eosin staining (× 200): Diffuse proliferation of vascular structures; C and D: Immunohistochemistry (× 200), CD31 and CD34: Positive expression. CD31 is primarily expressed in mature vascular endothelial cells; CD34 is mainly expressed in immature vascular endothelial cells. Positivity for both markers indicates active angiogenesis in the lesion area; E and F: Gross specimen - a 15 cm lesion along the ileal serosa shows dark purple, rough, and irregular mucosa with a spongy appearance, consistent with hemangiomatous hyperplasia.
TREATMENT
Laparoscopic exploration revealed a dense, highly vascular, sinus-like, well-defined lesion on the serosal layer of the ileum, measuring approximately 15 cm in length and located about 1.5 m from the ileocecal junction (Figure 1E and F). Based on gross morphological features, the lesion was suspected to be a hemangioma. Due to its large size (approximately 15 cm) and dense vascular sinuses, the decision was made to convert to open surgery for segmental enterectomy. A 20-cm segment of intestine containing the entire lesion was resected, and intestinal anastomosis was performed.
DISCUSSION
This case report presents a rare instance of cavernous hemangioma of the ileal serosa. Although commonly regarded as a “benign tumor” in clinical practice, the International Society for the Study of Vascular Anomalies classifies it as a vascular malformation caused by structural anomalies, rather than a true neoplasm originating from abnormal endothelial cell proliferation[2]. This clarification of clinical and academic understanding is crucial for comprehending its clinical manifestations, growth patterns, and treatment strategies.
Etiology and molecular mechanisms
Cavernous hemangioma originates from aberrant angiogenesis and vasculogenesis in the embryonic stage and is characterized by thin-walled, dilated venous channels[3]. Although germline mutations may contribute to the pathogenesis, sporadic cases are more frequently associated with somatic mutations[4]. In over 50% of sporadic venous malformations, there are acquired gain-of-function mutations in the endothelial tyrosine kinase receptor TIE2 (encoded by the TEK gene), while approximately 25% of cases harbor activating mutations in phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit alpha (PIK3CA); these two mutations are typically mutually exclusive[5,6]. TEK mutations lead to ligand-independent constitutive activation of the receptor, while PIK3CA mutations cause persistent aberrant activation of the PI3K enzyme; both ultimately drive hyperactivation of the phosphatidylinositol 3-kinase/protein kinase B/mammalian target of rapamycin signaling pathway[7]. This aberrant signaling does not primarily drive excessive cell proliferation, but rather leads to vascular endothelial cell dysfunction, impaired migration, and disruption of structural integrity, ultimately resulting in the formation of malformed vascular networks[8-10]. Based on the molecular mechanisms described above, targeted therapeutic strategies focusing on TIE2/PIK3CA have emerged as a key direction in precision medicine, demonstrating significant efficacy and promising potential, particularly in refractory cases[11].
Structural malformations and non-neoplastic growth
Cavernous hemangiomas are fundamentally structural anomalies arising from aberrant vasculogenesis and angiogenesis during embryonic development. The core pathological characteristic is the failure to form a well-structured and fully functional capillary-arteriole-venous network, which is replaced by abundant abnormal vascular channels featuring thin-walled, tortuous pathways filled with slow-flowing blood[12]. Unlike true neoplasms, these lesions exhibit no significant endothelial cell proliferative activity and lack autonomous, uncontrolled clonal proliferation. The expansion of the lesion is a passive and progressive process that occurs alongside bodily development. Due to the absence of a normal smooth muscle layer in the malformed vessels, the vascular walls gradually stretch and extend along natural spaces such as tissue interstices under long-term hemodynamic forces[13]. Although their growth does not rely on cellular proliferation, vascular malformations are often sensitive to hormonal environments - during periods of significant hormonal fluctuations such as puberty, pregnancy, or exogenous hormone use, the lesions may undergo noticeable and irreversible expansion due to increased blood flow and altered vascular wall compliance[14]. This phenomenon highlights the interaction between inherent structural defects and the body's physiological processes, with the underlying mechanism being rooted in hemodynamic alterations rather than neoplastic growth.
Pseudo-invasive growth pattern
Cavernous hemangiomas lack the biological invasiveness characteristic of malignant tumors but may exhibit a pseudo-invasive growth pattern resembling “infiltrative growth”[15]. This phenomenon essentially represents the gradual extension of malformed vessels along natural anatomical planes - such as tissue interstices or fascial surfaces - with encasement of adjacent structures, rather than stemming from destructive cellular invasion. Their vascular endothelial cells demonstrate low proliferative activity, absence of cytological atypia, and do not metastasize[16,17]. Although this case presented with a root-like infiltrative morphology, the relatively spacious abdominal cavity and confinement by outer muscular tissues resulted in predominantly intra-abdominal growth, characterized more by encasement than transmural invasion. However, this very pattern of encasement tends to lead to entanglement or compression of critical structures such as nerves and blood vessels, significantly increasing the difficulty of surgical dissection and often necessitating combined partial organ resection[18]. Additionally, the mass can directly encase and compress the intestinal tract and mesenteric vessels, compromising the blood supply to the intestinal wall and leading to secondary intestinal obstruction[19]. Therefore, although the lesion is categorized as benign, its pseudo-invasive growth pattern still results in significant local tissue destruction and poses considerable challenges for clinical management.
Delayed rupture risk of the mass
Unlike common submucosal hemangiomas, hemangiomas located on the serosal side have a distinct, potentially more dangerous clinical course, with their core risk lying in “insidious growth and delayed rupture”. The fundamental reason for this difference lies in the direction of growth: In its early stages, the tumor expands toward the peritoneal side due to the intestinal wall barrier, without invading the mucosa. This allows it to avoid early bleeding caused by erosion or friction from digestive fluids, resulting in concealed clinical symptoms and enabling prolonged silent growth[20]. However, it is precisely this “protective effect” that leads to the accumulation and qualitative change of risk. As the tumor enlarges, the vessel wall undergoes structural degeneration and increased fragility[21,22]. Meanwhile, according to Laplace’s law, the increasing radius leads to continuously rising wall tension[21,22]. The slow blood flow within the tumor is prone to triggering chronic inflammation, further compromising the integrity of the vascular wall[23]. Coupled with the long-term mechanical stress from gravity, intestinal peristalsis, and hemodynamic forces, these factors collectively render the tumor extremely fragile in its advanced stages[24]. When this fragility reaches a critical point, a common trigger - such as coughing or a sudden increase in abdominal pressure - can lead to rupture[25]. The consequence is not the typical gastrointestinal bleeding but rather a sudden, rapid, massive intra-abdominal hemorrhage, often directly manifesting as life-threatening hemorrhagic shock[26,27]. In addition, larger tumors may also trigger acute abdominal conditions such as intussusception or volvulus. Therefore, clinical management strategies must undergo a fundamental shift - from passively awaiting symptoms to proactive early warning: Emphasizing early recognition of their essential differences from submucosal hemangiomas, conducting regular monitoring through dynamic imaging, and timely adopting minimally invasive interventional therapy before high rupture risk emerges, so as to prevent catastrophic outcomes[28,29].
Clinical treatment
The management of serosal cavernous hemangioma of the small intestine should be individualized, with the core principle being risk stratification based on lesion characteristics and patient status to guide the decision between observation and surgical intervention. Specifically, very low-risk lesions (incidental, < 3 cm, asymptomatic) may be managed with active surveillance and regular imaging follow-up. Low-risk lesions (3-5 cm, mild symptoms) warrant close monitoring or consideration of elective minimally invasive surgery. Moderate-risk lesions (> 5 cm, or features suggesting pseudo-invasiveness, proximity to critical structures, etc.) are advised to undergo prophylactic elective surgery, particularly in patients planning pregnancy. High-risk lesions (large, complex, causing compression or complications) or growing symptomatic lesions require radical resection. In cases of acute emergencies, such as hemorrhage or obstruction, emergency surgery is indicated.
For serosal lesions requiring surgical intervention, if intraoperative examination confirms a solitary and well-localized lesion, laparoscopic intestinal segment resection can be performed[30,31]. If a large-volume lesion, highly vascularized tissue, complex morphology (such as root-like infiltration), or difficult exposure is encountered, immediate conversion to open laparotomy is necessary to ensure safe and complete resection[31]. Preoperative contrast-enhanced three-dimensional computed tomography reconstruction is essential for accurately assessing the lesion extent, identifying multifocal lesions, and formulating the surgical plan[1].