Published online Apr 27, 2026. doi: 10.4240/wjgs.v18.i4.115864
Revised: December 26, 2025
Accepted: February 4, 2026
Published online: April 27, 2026
Processing time: 150 Days and 1.7 Hours
Anal fistula is a common acquired anorectal disorder characterized by an ab
To comparing the diagnostic value of multislice spiral computed tomography (MSCT), 1.5T magnetic resonance imaging (MRI), and 3.0T MRI for anal fistula.
A retrospective analysis was conducted on 119 patients with anal fistula admitted to Nanjing Hospital of Chinese Medicine between May 2017 and December 2023 and categorized into three groups based on diagnostic modality: MSCT group (n = 25), 1.5T MRI group (n = 45), and 3.0T MRI group (n = 49). Kappa tests were applied to assess diagnostic agreement between each group and surgical findings (gold standard). Diagnostic accuracy, sensitivity, specificity, positive and negative predictive values, and Youden indices for detecting internal openings, main tracts, and abscesses were calculated and compared.
Using surgical findings as the reference standard, 3.0T MRI demonstrated sig
Compared with MSCT and 1.5T MRI, 3.0T MRI demonstrated higher diagnostic accuracy, sensitivity, and spe
Core Tip: This study compared diagnostic efficacy of multislice spiral computed tomography, 1.5T magnetic resonance imaging (MRI), and 3.0T MRI for anal fistula in 119 patients. With surgery as gold standard, 3.0T MRI showed significantly higher detection rates, accuracy, sensitivity, specificity, and consistency with surgery for internal openings/main tracts than multislice spiral computed tomography and 1.5T MRI, proving its superior diagnostic value.
- Citation: Tang JJ, Wang TH, Jia XF. Detection rates, sensitivity, and specificity of multislice spiral computed tomography, 1.5T and 3.0T magnetic resonance imaging in anal fistula diagnosis. World J Gastrointest Surg 2026; 18(4): 115864
- URL: https://www.wjgnet.com/1948-9366/full/v18/i4/115864.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i4.115864
Anal fistula is a chronic inflammatory anorectal disease characterized by an abnormal tract connecting the anal canal or rectum to the perianal skin. It most commonly arises from cryptoglandular infection and may also be associated with Crohn’s disease, trauma, or postoperative complications[1]. The disease is prone to recurrence and progression to complex fistula patterns, posing significant challenges to clinical management[2].
Surgical intervention remains the primary treatment for anal fistula. However, incomplete identification of the internal opening, primary tract, secondary extensions, or associated abscesses are major causes of postoperative recurrence and anal sphincter injury[3]. Therefore, accurate preoperative imaging assessment is essential for guiding surgical planning, minimizing complications, and improving patient outcomes.
Imaging plays a critical role in the preoperative evaluation of anal fistula by accurately delineating fistula anatomy and its relationship with surrounding sphincter structures. Currently, multislice spiral computed tomography (MSCT), which provides rapid three-dimensional anatomical reconstruction, and magnetic resonance imaging (MRI), which offers soft-tissue contrast and multiplanar visualization, are the most used imaging modalities[4]. With advances in magnetic field strength, 3.0T MRI has been increasingly applied in anal fistula evaluation; however, comparative evidence regarding its diagnostic advantage over MSCT and 1.5T MRI remains limited[5].
Therefore, this study aimed to systematically compare the diagnostic performance of MSCT, 1.5T MRI, and 3.0T MRI in patients with anal fistula, using surgical findings as the gold standard, to determine the optimal imaging modality for clinical practice.
A retrospective analysis was conducted on the clinical data of patients with anal fistula admitted to Nanjing Hospital of Chinese Medicine between May 2017 and December 2023. The inclusion criteria were: (1) Diagnosis met the criteria outlined in the clinical diagnosis and treatment guidelines for anal fistula[6], presenting with perianal pain, skin ul
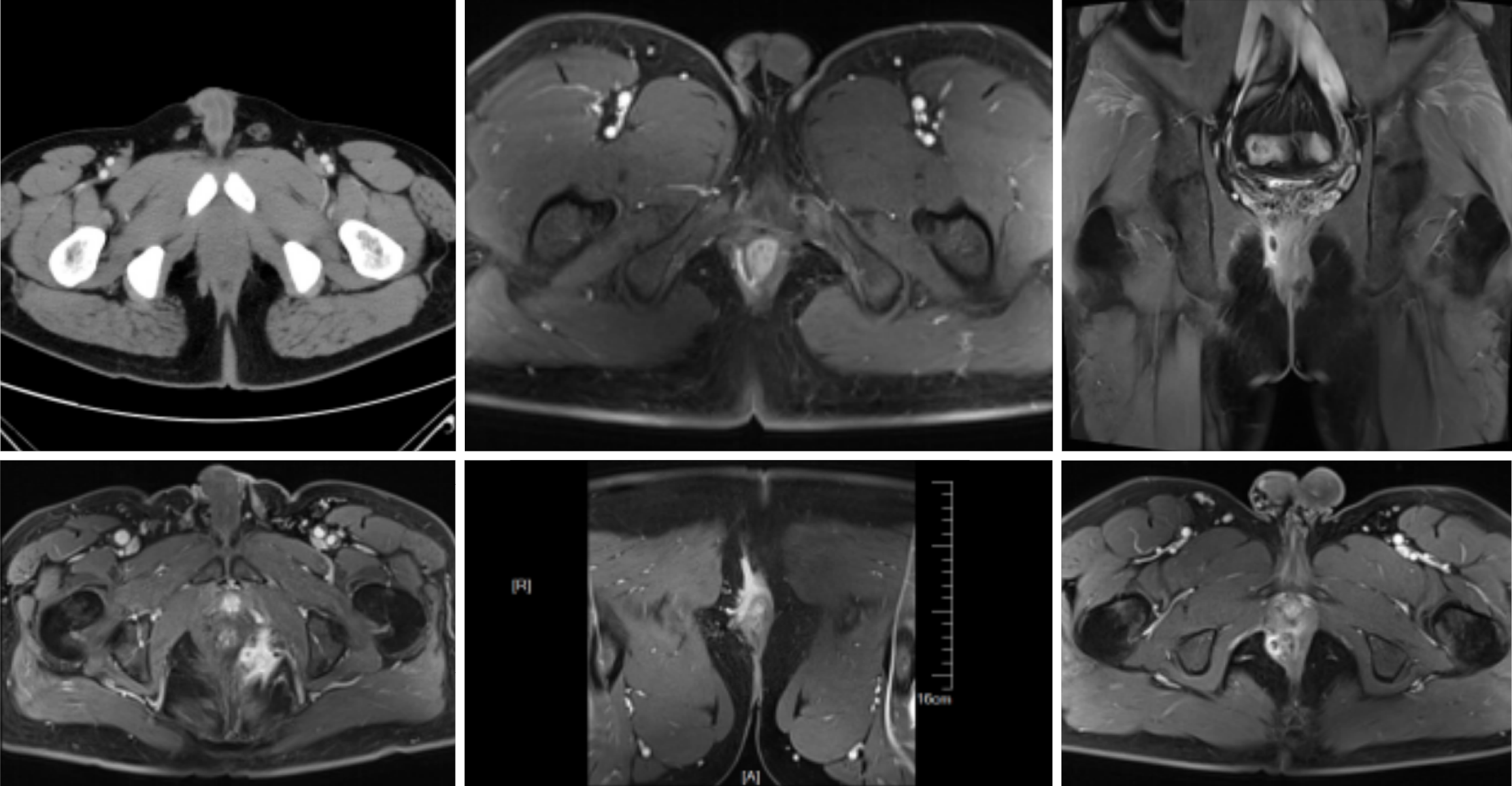
MSCT examination: Performed using Siemens force dual-source computed tomography (Germany). Parameters: Tube voltage 110-120 kV, tube current 180-200 mA, slice thickness 5 mm, slice spacing 5 mm, window setting 300 HU. Patients underwent bowel preparation and metallic object removal prior to scanning. Scanning was performed in the supine position. Non-contrast scanning was conducted first. Iopamidol (40 mL) was injected via the antecubital vein at 3.0-3.5 mL/second. Contrast-enhanced scanning was performed after 10 second. The acquired scans and images underwent back-end processing, and raw data were reconstructed.
MRI examination: Performed using a GE Signa HDe 1.5 magnetic resonance scanner (4-channel phased array body coil) and a magnetic resonance Siemens Prisma 3.0 superconducting scanner (18-channel phased array body coil). No special preparation was required prior to scanning. Scans were performed in standard sagittal, coronal, and axial planes perpendicular and parallel to the anal canal long axis. Diffusion-weighted imaging used diffusion sensitivity coefficients (b-values) of 0 second/mm2 or 800 second/mm2. 1.5T MRI utilized T1-weighted fat-suppressed sequences in the sagittal, coronal, and axial planes. 3.0T MRI employed gradient echo volume-weighted dynamic multi-phase contrast-enhanced sequences (T1 volume interpolated breath-hold examination fat suppression) with breath-hold. Contrast agent: Eu
| Parameter | Sequence | 1.5T MRI | 3.0T MRI |
| TR (milliseconds) | Sagittal T2WI | 4000 | 6000 |
| Coronal T2WI | 4050 | 6500 | |
| Axial T2WI | 4400 | 6700 | |
| Axial T1WI | 680 | 600 | |
| Axial T2WI FS | 5500 | 8500 | |
| TE (milliseconds) | Sagittal T2WI | 120 | 100 |
| Coronal T2WI | 80 | 102 | |
| Axial T2WI | 100 | 102 | |
| Axial T1WI | 16 | 20 | |
| Axial T2WI FS | 90 | 110 | |
| FOV (mm) | Sagittal T2WI | 280 × 280 | 300 × 300 |
| Coronal T2WI | 280 × 320 | 240 × 240 | |
| Axial T2WI | 240 × 300 | 240 × 240 | |
| Axial T1WI | 280 × 280 | 240 × 240 | |
| Axial T2WI FS | 280 × 300 | 240 × 240 | |
| Slice thickness (mm) | Sagittal T2WI | 4.0 | 4.0 |
| Coronal T2WI | 4.0 | 3.5 | |
| Axial T2WI | 4.0 | 3.5 | |
| Axial T1WI | 4.0 | 4.0 | |
| Axial T2WI FS | 4.0 | 3.5 | |
| Inter-slice gap (mm) | Sagittal T2WI | 1.0 | 0.4 |
| Coronal T2WI | 1.0 | 0.4 | |
| Axial T2WI | 1.0 | 0.4 | |
| Axial T1WI | 1.0 | 0.35 | |
| Axial T2WI FS | 1.0 | 0.35 | |
| Number of slices | All sequences | 24 | 25 |
Two radiologists with over 10 years of clinical experience in MSCT and MRI independently reviewed the images to assess the internal opening, main fistula tract, and abscess status. In cases of disagreement, a consensus was reached through discussion and analysis. Internal openings were localized using the lithotomy clock position method. Fistula tracts and abscesses were defined as < 10 mm-diameter and ≥ 10 mm-diameter fluid-filled structures, respectively[7,8].
Using surgical outcomes as the gold standard, the diagnostic accuracy, sensitivity, specificity, positive and negative predictive values, and Yorden index for MSCT, 1.5T MRI, and 3.0T MRI were calculated and compared in identifying internal openings, main fistula tracts, and abscesses in patients with anal fistula.
SPSS version 25.0 statistical software was used. Quantitative data were expressed as mean ± SD. Comparisons were performed using t-tests or F-tests. Count data were expressed as n (%). Comparisons were performed using χ2 tests. The consistency between MSCT/MRI diagnostic results and surgical findings for internal openings, main fistula tracts, and abscesses was assessed using Kappa tests. Kappa values (K) ≥ 0.78, 0.6-0.8, 0.4-0.6, and ≤ 0.4 indicated excellent, good, moderate, and poor agreement, respectively. The test level factor α = 0.05.
A total of 119 patients were included in this study. Based on the different diagnostic methods used, the patients were divided into three groups: The MSCT group (undergoing MSCT, n = 25), the 1.5T MRI group (undergoing 1.5T MRI, n = 45), and the 3.0T MRI group (undergoing 3.0T MRI, n = 49). Sex, age, disease duration, and body mass index (BMI) showed no statistically significant differences among the three groups (P > 0.05 L; Figure 1 and Table 2).
| Parameter | MSCT group | 1.5T MRI group | 3.0T MRI group | F/χ2 | P value |
| Sex (male/female) | 15/10 | 27/18 | 38/11 | χ2 = 1.321 | 0.517 |
| Age (years) | 39.63 ± 6.87 | 40.15 ± 7.59 | 37.87 ± 8.25 | F = 0.952 | 0.389 |
| Disease duration (months) | 9.86 ± 2.57 | 10.13 ± 2.65 | 9.39 ± 2.34 | F = 0.838 | 0.435 |
| BMI (kg/m2) | 24.52 ± 3.04 | 24.11 ± 2.78 | 23.96 ± 3.17 | F = 0.369 | 0.693 |
The detection rates of internal openings and main fistula tracts by 3.0T MRI (94.54% and 96.43%, respectively) were higher than those by 1.5T MRI (78.00% and 81.63%, respectively) and MSCT (76.67% and 77.42%, respectively), with statistically significant differences (P < 0.05). The consistency of 3.0T MRI results with surgical findings (K-values: 0.911, 0.926) was also higher than that of 1.5T MRI (K-values: 0.694, 0.726) and MSCT (K-values: 0.681, 0.711), with statistically significant differences (P < 0.05). The detection rates of internal openings and main fistula tracts were not statistically significantly different between 1.5T MRI and MSCT (P > 0.05). Additionally, the detection rates of abscesses were not statistically significantly different among the diagnostic methods (P > 0.05; Table 3).
| Methods | Items | Preoperative diagnosis | Detection rate | Surgical outcome | K | |||
| True positive | False positive | False negative | True negative | |||||
| MSCT | Internal opening | 23 | 5 | 2 | 13 | 76.67 (23/30) | 27 | 0.681 |
| Primary fistula | 24 | 5 | 2 | 14 | 77.42 (24/31) | 27 | 0.711 | |
| Abscess | 8 | 1 | 1 | 3 | 80.00 (8/10) | 8 | 0.845 | |
| 1.5T MRI | Internal opening | 39 | 7 | 4 | 22 | 78.00 (39/50) | 46 | 0.694 |
| Primary fistula | 40 | 7 | 2 | 21 | 81.63 (40/49) | 47 | 0.726 | |
| Abscess | 14 | 2 | 0 | 8 | 87.50 (14/16) | 14 | 0.851 | |
| 3.0T MRI | Internal opening | 52 | 2 | 1 | 25 | 94.54 (52/55)a | 52 | 0.911a |
| Primary fistula | 54 | 1 | 1 | 24 | 96.43 (54/56)a | 55 | 0.926a | |
| Abscess | 17 | 1 | 0 | 24 | 94.45 (17/18) | 17 | 0.906 | |
The diagnostic accuracy of 3.0T MRI for internal openings and main fistula tracts (96.25%, 97.50%) was higher than that of 1.5T MRI (83.72%, 84.44%) and MSCT (83.72%, 84.44%), with statistically significant differences (P < 0.05). Except for the sensitivity in abscess diagnosis, 3.0T MRI showed statistically significantly higher sensitivity, specificity, positive and negative predictive value, and Youden index in the diagnosis of internal openings, main fistula tracts, and abscesses than MSCT and 1.5T MRI (P < 0.05; Table 4).
| Modality | Finding | Sensitivity | Specificity | PPV | NPV | Diagnostic accuracy | Youden index |
| MSCT | Internal opening | 92.00% (23/25) | 72.22% (13/18) | 82.14% (23/28) | 86.67% (13/15) | 83.72% (36/43) | 0.64 |
| Primary tract | 92.30% (24/26) | 73.68% (14/19) | 82.76% (24/29) | 87.50% (14/16) | 84.44% (38/45) | 0.66 | |
| Abscess | 88.89% (8/9) | 75.00% (3/4) | 88.89% (8/9) | 75.00% (3/4) | 84.61% (11/13) | 0.69 | |
| 1.5T MRI | Internal opening | 90.69% (39/43) | 75.86% (22/29) | 84.78% (39/46) | 84.62% (22/26) | 84.72% (61/72) | 0.67 |
| Primary tract | 95.24% (40/42) | 75.00% (21/28) | 85.11% (40/47) | 91.30% (21/23) | 87.14% (61/70) | 0.7 | |
| Abscess | 100.00% (14/14) | 80.00% (8/10) | 87.50% (14/16) | 100.00% (8/8) | 91.67% (22/24) | 0.8 | |
| 3.0T MRI | Internal opening | 98.11% (52/53)a | 92.59% (25/27)a | 96.30% (52/54)a | 96.15% (25/26)a | 96.25% (77/80)a | 0.91a |
| Primary tract | 98.18% (54/55)a | 96.00% (24/25)a | 98.18% (54/55)a | 96.00% (24/25)a | 97.50% (78/80)a | 0.94a | |
| Abscess | 100.00% (17/17) | 96.00% (24/25)a | 94.44% (17/18)a | 100.00% (24/24) | 97.62% (41/42) | 0.96a |
Anal fistula is a common anorectal disease characterized by recurrent episodes and prolonged non-healing. Symptoms such as perianal exudate and pain after onset can significantly affect patients’ daily life and work[9]. Currently, surgery is the main treatment for anal fistula. During the clinical treatment process, factors such as the location of fistula tracts and internal openings, inflammation, and perianal tissue structure may easily lead to false-positive and false-negative results, resulting in misdiagnosis and missed diagnosis of anal fistula. Misdiagnosis can mislead surgical procedures and cause unnecessary damage to patients’ anorectal tissues and perianal structures, while missed diagnosis can lead to incomplete surgical resection, which is a major cause of recurrent anal fistula after surgery[10,11]. Therefore, improving the diagnostic accuracy of anal fistula is of great significance for enhancing patient prognosis and preventing anal fistula recurrence.
MSCT is a non-invasive imaging diagnostic method that can perform three-dimensional reconstruction of fistula tracts through multiple data channels. It can clearly display the location of internal openings, the shape and course of fistula tracts, and their relationship with surrounding tissues, thereby enabling rapid and accurate diagnosis[12]. In this study, among the 25 patients with anal fistula diagnosed by MSCT, the detection rates of internal openings, main fistula tracts, and abscesses were 76.67%, 77.42%, and 80.00%, respectively, with 83.72%, 84.44%, and 84.61% diagnostic accuracies, respectively. These results are basically consistent with those of previous relevant reports[13].
The diagnostic principle of MRI is to generate a strong magnetic field through the action of specific radiofrequency pulses. It utilizes the magnetic resonance phenomenon of hydrogen nuclei in the human body with different relaxation times to provide high-resolution soft tissue images, thereby enabling the accurate identification and localization of anal fistula internal openings, fistula tracts, and abscesses. 1.5T MRI and 3.0T MRI share the same diagnostic principle, but 3.0T MRI has a higher magnetic field strength and thinner image slices. Particularly, 3.0T MRI has a higher abscess detection rate. Mac Curtain et al[14] used thin-slice T2-weighted imaging with fat suppression sequence for anal fistula diagnosis and found that 3.0T MRI had good diagnostic efficacy for anal fistula internal openings and main fistula tracts. The results of this study showed that the detection rates of internal openings and main fistula tracts by 3.0T MRI were 94.54% and 96.43%, respectively, with diagnostic accuracies of 96.25% and 97.50%, respectively, which were higher than those of 1.5T MRI and MSCT. Meanwhile, the consistency between the detection of internal openings and main fistula tracts by 3.0T MRI and surgical findings was highly consistent, which was higher than that of 1.5T MRI and MSCT. These results are consistent with the report by Saboo et al[15]. In addition, in this study, 3.0T MRI showed higher sensitivity, specificity, positive and negative predictive values, and Youden index in the diagnosis of internal openings, main fistula tracts, and abscesses compared with MSCT and 1.5T MRI. Moreover, compared with 1.5T MRI and MSCT, 3.0T MRI has higher diagnostic efficacy for anal fistula internal openings and main fistula tracts, and the risk of false-positive and false-negative results in patients is lower. This is because 3.0T MRI has a stronger magnetic field strength. Compared with 1.5T MRI, 3.0T MRI has significantly improved temporal resolution, spatial resolution, and signal-to-noise ratio, with less interference from local inflammation and clearer images[15]. Although MSCT can perform three-dimensional recon
In addition to accurately displaying the shape and course of fistula tracts and the location of internal openings, MRI can also assess the severity of anal fistula, whether it is complicated with abscesses, and other perianal diseases such as anal sphincter injury[17]. In this study, the abscess detection rate by 3.0T MRI was 94.45%, which was higher than that of 1.5T MRI and MSCT, but the difference was not statistically significant. This may be related to the small sample size of this study. Clinical studies have shown that the causes of false-positive and false-negative results in MRI are mostly related to asymmetrically distributed small perianal blood vessels, uneven fat suppression, and signal interference from inflammatory skin tags[18,19]. In this study, the 3.0T MRI group had fewer false-positive and false-negative diagnosis of anal fistula internal openings, main fistula tracts, and abscesses: There were 2 false-positive cases of internal openings, and 1 false-positive case each for main fistula tracts and abscesses, with an incidence far lower than that in the 1.5T MRI and MSCT groups. Among the causes of false-positive internal openings in 3.0T MRI and 1.5T MRI, four cases were misdiagnosed owing to the passage of small perianal blood vessels through the mucosa, and the rest were caused by high signals from mucosal inflammation. The causes of false-negative internal openings were all due to interference from indirect internal openings. For MSCT, the causes of false-positive and false-negative results were all related to extrasphincteric anal fistula, and MSCT has certain limitations in the diagnosis of low anal fistula. Regarding the causes of false-positive main fistula tracts in MRI, three cases were misdiagnosed as main fistula tracts due to small perianal blood vessel branches, one case was misdiagnosed due to uneven fat suppression, and the rest were caused by signal interference from inflammatory mucosal skin tags. For MSCT, misdiagnosis and missed diagnosis were all caused by interference and confusion from mucosal inflammatory signals.
In conclusion, MSCT, 1.5T MRI, and 3.0T MRI are effective for diagnosing anal fistula. Compared with the first two methods, 3.0T MRI has better diagnostic accuracy, sensitivity, and specificity, and higher diagnostic efficacy for anal fistula. However, in practical clinical application, the diagnostic method should be selected based on the comprehensive consideration of examination costs and specific condition of patients to improve diagnostic accuracy.
| 1. | Limura E, Giordano P. Modern management of anal fistula. World J Gastroenterol. 2015;21:12-20. [PubMed] [DOI] [Full Text] |
| 2. | Mei Z, Wang Q, Zhang Y, Liu P, Ge M, Du P, Yang W, He Y. Risk Factors for Recurrence after anal fistula surgery: A meta-analysis. Int J Surg. 2019;69:153-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 115] [Article Influence: 16.4] [Reference Citation Analysis (1)] |
| 3. | Han L, Chen Y, Cheng W, Bai H, Wang J, Yu M. Deep Learning-Based CT Image Characteristics and Postoperative Anal Function Restoration for Patients with Complex Anal Fistula. J Healthc Eng. 2021;2021:1730158. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 4. | Wang X, Lv D, Song H, Deng L, Gao Q, Wu J, Shi Y, Li L. Multimodal preoperative evaluation system in surgical decision making for rectal cancer: a randomized controlled trial. Int J Colorectal Dis. 2010;25:351-358. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 5. | Tüney D, Altun E, Barlas A, Yegen C. Pancreatico-colonic fistula after acute necrotizing pancreatitis. Diagnosis with spiral CT using rectal water soluble contrast media. JOP. 2008;9:26-29. [PubMed] |
| 6. | Schneider J, Heinze T, Laubert T, Kahlke V, Wedel T, Heimke M. [Anal fistula disease : Anatomical foundations and surgical procedures]. Chirurgie (Heidelb). 2025;96:728-736. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 7. | Thipphavong S, Costa AF, Ali HA, Wang DC, Brar MS, Jhaveri KS. Structured reporting of MRI for perianal fistula. Abdom Radiol (NY). 2019;44:1295-1305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 21] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 8. | Sudoł-Szopińska I, Santoro GA, Kołodziejczak M, Wiaczek A, Grossi U. MRI template to standardize reporting of anal fistula. Tech Coloproctol. 2022;26:325-328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 9. | Seow-Choen F, Nicholls RJ. Anal fistula. Br J Surg. 1992;79:197-205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 84] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 10. | Cheng JW, Ahn JJ, Cain MP, Anderson JE, Smith CA, Rice-Townsend SE. Misdiagnosis of Congenital Posterior Urethroperineal Fistula and Comparison With Urethral Duplications and Rectourethral Fistula. Urology. 2021;158:193-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 11. | Sa YL, Xu YM, Feng C, Ye XX, Song LJ. Three-dimensional spiral computed tomographic cysto-urethrography for post-traumatic complex posterior urethral strictures associated with urethral-rectal fistula. J Xray Sci Technol. 2013;21:133-139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 12. | Yamamoto S, Yonezawa K, Fukata N, Takeshita K, Kodama M, Yamana T, Kiryu S, Okada Y. Value of apparent diffusion coefficient on MRI for prediction of histopathological type in anal fistula cancer. Medicine (Baltimore). 2023;102:e33281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 13. | Feng Y, Zhang W, Luo C. Evaluation of clinical application of multi-slice computerized tomography in primary retroperitoneal tumors. J Clin Lab Anal. 2020;34:e23169. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 14. | Mac Curtain BM, Qian W, Bell J, O'Mahony A, Temperly HC, Ng ZQ. Pre- and post-treatment FDG PET-CT as a predictor of patient outcomes in anal squamous cell carcinoma: A systematic review and meta-analysis. J Med Imaging Radiat Oncol. 2023;67:634-646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 15. | Saboo SS, Zukotynski K, Shinagare AB, Krajewski KM, Ramaiya N. Anal carcinoma: FDG PET/CT in staging, response evaluation, and follow-up. Abdom Imaging. 2013;38:728-735. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 29] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 16. | Klingenberg S, Jochumsen MR, Nielsen TF, Bouchelouche K. 68Ga-PSMA Uptake in Anal Fistula on PET/CT Scan. Clin Nucl Med. 2019;44:e54-e56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 17. | Erden A, Peker E, Gençtürk ZB. Chronic anal fissure: morphometric analysis of the anal canal at 3.0 Tesla MR imaging. Abdom Radiol (NY). 2017;42:423-434. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 18. | Zijta FM, Lakeman MM, Froeling M, van der Paardt MP, Borstlap CS, Bipat S, Montauban van Swijndregt AD, Strijkers GJ, Roovers JP, Nederveen AJ, Stoker J. Evaluation of the female pelvic floor in pelvic organ prolapse using 3.0-Tesla diffusion tensor imaging and fibre tractography. Eur Radiol. 2012;22:2806-2813. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 26] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 19. | Wang Y, Gu C, Huo Y, Han W, Yu J, Ding C, Zhao X, Meng Y, Li C. Diffusion tensor imaging for evaluating perianal fistula: Feasibility study. Medicine (Baltimore). 2018;97:e11570. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 1.3] [Reference Citation Analysis (0)] |