Published online Mar 27, 2026. doi: 10.4240/wjgs.v18.i3.116006
Revised: November 21, 2025
Accepted: January 19, 2026
Published online: March 27, 2026
Processing time: 147 Days and 21.9 Hours
We read with great interest the study by Zhang et al in the World Journal of Gastrointestinal Surgery which presents the first human evidence linking intestinal obstruction to gut-vascular barrier (GVB) injury, as demonstrated by elevated levels of plasmalemma vesicle associated protein 1 (PV1). This letter proposes that autonomic imbalance - specifically, suppressed vagal tone, reflected by reduced heart rate variability (HRV) - may represent a key mechanistic link between in
Core Tip: This article highlights a potentially overlooked mechanism - the role of the autonomic nervous system, particularly vagal tone, which may serve as a critical connection between intestinal obstruction and gut-vascular barrier disruption. Future research should investigate the link between heart rate variability and plasmalemma vesicle associated protein 1 expression, as integrating this non-invasive measure with a molecular marker could improve prognostication and reveal novel therapeutic avenues, such as vagus nerve stimulation.
- Citation: Dong MQ, Wu J. Letter to the Editor: Gut-vascular barrier dysfunction in intestinal obstruction: The role of vagal suppression and heart rate variability. World J Gastrointest Surg 2026; 18(3): 116006
- URL: https://www.wjgnet.com/1948-9366/full/v18/i3/116006.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i3.116006
Intestinal obstruction (IO) is one of the most common medical emergencies encountered in emergency departments and surgical practice. This condition is characterized by a partial or complete blockage of the passage of gastrointestinal contents, mainly attributed to mechanical factors such as tumors, adhesions, volvulus, or hernia[1]. The classic clinical presentation of IO encompasses abdominal pain, vomiting, abdominal distension, and cessation of defecation and flatus. The intestine acts as a critical frontline defense system, with its barrier comprising the mucus layer, the epithelial barrier, and the innermost gut-vascular barrier (GVB). The GVB is composed of endothelial cells interconnected by tight and adherens junctions, supported by enteric glial cells, pericytes, and fibroblasts. This complex structure regulates the exchange of substances between the intestinal lumen and the circulatory system[2]. Previous research on IO predominantly focused on the mechanisms underlying epithelial barrier injury[3]. For example, Wang et al[4] reported that neurokinin B exacerbates intestinal inflammation and compromises epithelial barrier integrity by activating the neurokinin 3 receptor, ultimately leading to increased serum levels of interleukin-6 and tumor necrosis factor-α, However, the specific role of the GVB in IO and its clinical significance in disease progression remains unclear. Thus, the study by Zhang et al[5] in the World Journal of Gastrointestinal Surgery represents a significant breakthrough. They provided the first systematic human evidence that IO induces substantial GVB damage, as demonstrated by a significant increase in the expression of plasmalemma vesicle associated protein 1 (PV1). PV1 is an endothelial-specific type II transmembrane glycoprotein encoded by the plas
This case-control study, which integrated molecular biology techniques with systematic statistical analysis, demonstrated that IO induces GVB disruption, the extent of which is closely associated with clinical outcomes. Reverse transcription quantitative polymerase chain reaction and immunofluorescence analyses revealed significantly increased mRNA and protein expression levels of PV1, a specific biomarker of GVB damage, in the intestinal tissues of IO patients compared with those of the non-obstructed control group. Linear regression analysis revealed that PV1 expression in IO patients was positively correlated with perioperative infection markers (procalcitonin, C-reactive protein, and white blood cell count), conventional serum indicators of hepatic and renal function (alanine aminotransferase, aspartate aminotransferase, and creatinine), and additional clinical management indices, including length of hospitalization (though this may be influenced by non-medical factors) and duration of antibiotic therapy (which reflects the clinical response to suspected or confirmed infection). Stratified analysis based on PV1 expression levels revealed that the high-expression subgroup exhibited significantly worse clinical parameters compared with the low-expression subgroup. Logistic regression analysis identified high PV1 expression as an independent risk factor for postoperative complications in IO patients after adjusting for age, sex, and body mass index. These findings indicate that the extent of GVB damage is directly associated with clinical deterioration, supporting PV1 as a potential prognostic biomarker for IO.
The key innovation of this study lies in being the first to systematically demonstrate in humans that IO directly results in GVB disruption and to establish its correlation with clinical outcomes. Moving beyond the traditional focus on the epi
However, this study presents several limitations. First, as a single-center, cross-sectional observational study with a limited sample size and no a priori power calculation, its statistical power, particularly in the PV1 high/Low subgroup analyses with further reduced sample numbers, may be insufficient. This limitation possibly explains the lack of significant differences in key outcomes such as severe complications and mortality between subgroups. Second, the inclusion of non-obstructed patients with intestinal tumors as controls can introduce potential bias, as their baseline PV1 Levels may be inherently high, potentially underestimating the actual impact of IO on the GVB. Third, the assessment of GVB damage relied solely on postoperative intestinal tissue PV1 measurements, an invasive approach that did not include simultaneous evaluation of the epithelial barrier. This approach limited the clinical translational potential of the study. Finally, although an association was observed between PV1 expression and adverse outcomes, the observational design of the study does not allow for causal inference. Moreover, the clinical cutoff value for PV1 was not defined, and its prognostic significance requires validation in future studies.
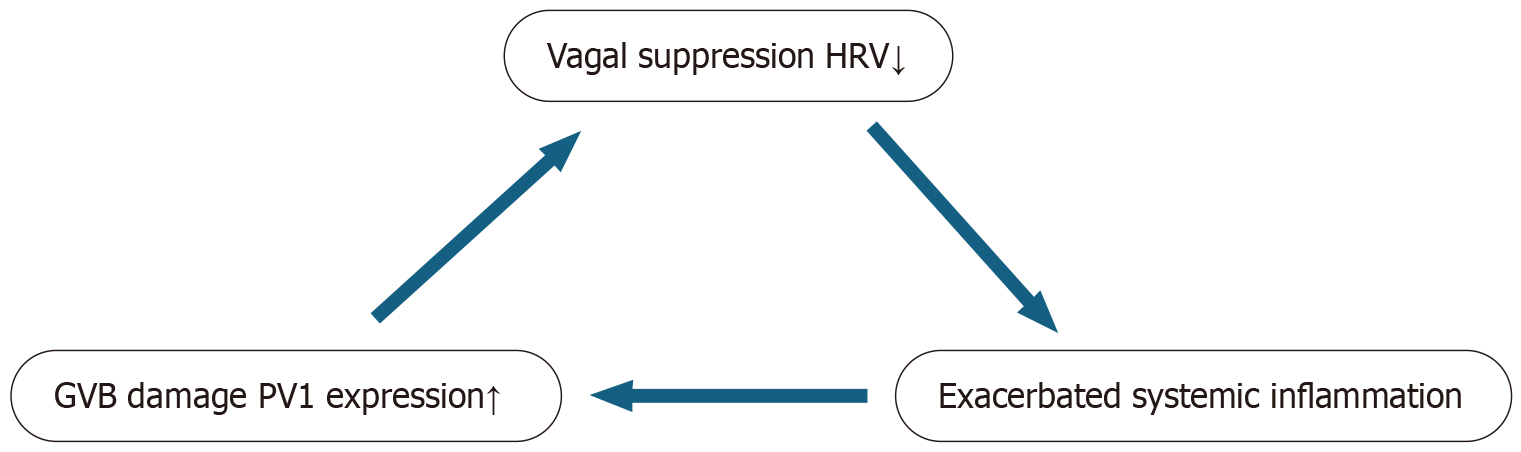
The increased resting heart rate observed in patients with IO may reflect more than a general stress response. On a deeper physiological level, this observation indicates reduced heart rate variability (HRV) and impaired vagal nerve activity. This imbalance within the autonomic nervous system potentially establishes a mechanistic connection between the primary pathology of IO and secondary GVB damage.
HRV, defined as the variation in time intervals between consecutive heartbeats, serves as a key non-invasive indicator for assessing autonomic nervous system function, particularly vagal tone[7,8]. The vagus nerve (especially its right branch) is the primary physiological modulator of HRV. Intact vagal activity is associated with high HRV, whereas vagal suppression leads to a substantial reduction in HRV. Therefore, HRV levels can be directly interpreted as a reflection of central efferent vagal tone. The vagus nerve plays a crucial role in regulating intestinal inflammation and maintaining barrier integrity through mechanisms such as the “cholinergic anti-inflammatory pathway”[9]. Previous studies have demonstrated that vagal stimulation and cholinergic signaling can suppress inflammatory responses, reduce the de
Future research should focus on elucidating the causal role of GVB injury in IO and advancing the clinical translation of PV1 in combination with non-invasive indicators such as HRV. The primary objective is to conduct large-scale, multicenter prospective cohort studies to systematically assess the dynamic relationships between HRV, PV1 expression, and GVB integrity. Research efforts should also aim to identify soluble forms of PV1 to support the development of blood-based assays and to directly validate the proposed pathway linking reduced HRV to GVB disruption (Figure 1). Such investigations would help establish an integrated prognostic model that incorporates functional, molecular, and inflammatory indicators, while also critically assessing the predictive utility of clinical parameters such as length of hospital stay.
At the mechanistic level, further elucidation of the molecular pathways underlying GVB disruption is essential. Employing animal models of IO and conditional gene knockout technologies, future studies should clarify the core signaling networks responsible for PV1 upregulation and barrier failure. Future research should focus on the regulatory role of the cholinergic anti-inflammatory pathway in maintaining GVB integrity and evaluate the therapeutic potential of vagus nerve stimulation or selective receptor agonists. Furthermore, the research scope should extend beyond the GVB alone by simultaneously assessing the functional status of the epithelial barrier (e.g., zonula occludens-1 and occludin) to comprehensively delineate the broader landscape of GVB dysfunction in IO and its internal interactions.
Ultimately, all research efforts should be directed toward clinical application. The major future challenge involves designing and implementing interventional clinical trials to evaluate the efficacy of GVB-protective strategies (e.g., HRV-based biofeedback, vagus nerve stimulation, or drug repurposing). Studies must also determine the optimal timing for these interventions (preoperative, intraoperative, or postoperative) and integrate real-world data with advanced an
This study provides the first human evidence that IO induces GVB damage, as indicated by elevated PV1 expression. The extent of this damage positively correlates with perioperative infection, hepatocellular injury, and adverse outcomes, establishing PV1 as a potential prognostic biomarker. Future research should translate this finding by developing non-invasive biomarkers and exploring GVB-targeted therapies to improve clinical outcomes.
The authors are grateful to the investigators of the original study for their valuable contribution, which served as the basis for this commentary.
| 1. | Catena F, De Simone B, Coccolini F, Di Saverio S, Sartelli M, Ansaloni L. Bowel obstruction: a narrative review for all physicians. World J Emerg Surg. 2019;14:20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 92] [Article Influence: 13.1] [Reference Citation Analysis (1)] |
| 2. | Spadoni I, Zagato E, Bertocchi A, Paolinelli R, Hot E, Di Sabatino A, Caprioli F, Bottiglieri L, Oldani A, Viale G, Penna G, Dejana E, Rescigno M. A gut-vascular barrier controls the systemic dissemination of bacteria. Science. 2015;350:830-834. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 607] [Cited by in RCA: 559] [Article Influence: 50.8] [Reference Citation Analysis (4)] |
| 3. | Bertocchi A, Carloni S, Ravenda PS, Bertalot G, Spadoni I, Lo Cascio A, Gandini S, Lizier M, Braga D, Asnicar F, Segata N, Klaver C, Brescia P, Rossi E, Anselmo A, Guglietta S, Maroli A, Spaggiari P, Tarazona N, Cervantes A, Marsoni S, Lazzari L, Jodice MG, Luise C, Erreni M, Pece S, Di Fiore PP, Viale G, Spinelli A, Pozzi C, Penna G, Rescigno M. Gut vascular barrier impairment leads to intestinal bacteria dissemination and colorectal cancer metastasis to liver. Cancer Cell. 2021;39:708-724.e11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 453] [Cited by in RCA: 407] [Article Influence: 81.4] [Reference Citation Analysis (4)] |
| 4. | Wang F, Jiang X, Zhao Z, Wang Y, Jiang H, Gao Y, Wang H, Li Z. Neurokinin B is a potential target for treating disruption of intestinal mucosal barrier in acute mechanical intestinal obstruction. Peptides. 2025;191:171419. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 5. | Zhang HF, Guo Y, Chen XJ, Zhang YN, Peng H, Liu ZM, Zhang XY. Associations of clinical indexes and prognosis with gut-vascular barrier damage in patients with intestinal obstruction. World J Gastrointest Surg. 2025;17:111041. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 6. | Yang-Jensen SK, Nägele NS, Jensen BAH. From gut to blood: barrier dysfunction as a driver of systemic low-grade inflammation in cardiometabolic disease. Am J Physiol Cell Physiol. 2025;329:C1723-C1741. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (3)] |
| 7. | Stein PK, Kleiger RE. Insights from the study of heart rate variability. Annu Rev Med. 1999;50:249-261. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 336] [Cited by in RCA: 307] [Article Influence: 11.4] [Reference Citation Analysis (1)] |
| 8. | Ashinze P, Gaur A, Banerjee S, Olaniyan OM, Akande E, Idris-Agbabiaka A, Aboderin C, Bonu IS, Adeniyi SA, Eriobuna E, Adeyemo A, Lawal F, Ayodeji O. The role of heart rate variability in cardiac surgery: applications and innovations. Cardiothorac Surg. 2025;33:34. [DOI] [Full Text] |
| 9. | Matteoli G, Boeckxstaens GE. The vagal innervation of the gut and immune homeostasis. Gut. 2013;62:1214-1222. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 220] [Cited by in RCA: 218] [Article Influence: 16.8] [Reference Citation Analysis (1)] |
| 10. | Ishioh M, Nozu T, Miyagishi S, Funayama T, Ueno N, Takakusaki K, Okumura T. Carnosine improves colonic hyperpermeability through the brain histamine H1 receptor, basal forebrain cholinergic neurons, adenosine A2B receptors and vagus nerve in rats. Eur J Pharmacol. 2025;1002:177844. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 11. | Berntson GG, Bigger JT Jr, Eckberg DL, Grossman P, Kaufmann PG, Malik M, Nagaraja HN, Porges SW, Saul JP, Stone PH, van der Molen MW. Heart rate variability: origins, methods, and interpretive caveats. Psychophysiology. 1997;34:623-648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2974] [Cited by in RCA: 2421] [Article Influence: 83.5] [Reference Citation Analysis (1)] |