Published online Mar 27, 2026. doi: 10.4240/wjgs.v18.i3.116149
Revised: December 10, 2025
Accepted: January 23, 2026
Published online: March 27, 2026
Processing time: 129 Days and 3.5 Hours
Delayed gastrointestinal function recovery after laparoscopic colorectal cancer resection is an important factor affecting patient rehabilitation. Traditional peri
To investigate the impact of ERAS protocol on time to first flatus after laparoscopic colorectal cancer resection.
Eighty patients who underwent laparoscopic colorectal cancer resection at the Department of Colorectal Oncology Surgery, The First Affiliated Hospital of Xiamen University, from May 2023 to August 2024 were selected as study subjects and divided into conventional group (n = 40) and ERAS group (n = 40) according to different perioperative management protocols. The conventional group recei
Compared to the conventional group, the ERAS group showed significantly shortened time to first flatus (2.2 ± 0.8 days vs 3.5 ± 1.2 days, P < 0.001), earlier time to first defecation (3.3 ± 1.0 days vs 4.8 ± 1.4 days, P < 0.001), and reduced time to bowel sounds recovery (2.5 ± 0.8 days vs 4.2 ± 1.3 days, P < 0.001). Mean hospital stay was significantly shortened in the ERAS group (9.1 ± 2.4 days vs 12.3 ± 3.2 days, P < 0.001), and postoperative complication rate was significantly reduced (12.5% vs 32.5%, P < 0.05). Statistically significant differences existed between the two groups in the incidence of gastrointestinal discomfort symptoms including abdominal distension, nausea, and vomiting (P < 0.05).
Application of ERAS protocol in laparoscopic colorectal cancer resection can effectively shorten time to first flatus, promote gastrointestinal function recovery, reduce hospital stay, and lower complication rates, demonstrating good clinical application value.
Core Tip: Enhanced recovery after surgery (ERAS) may accelerate gastrointestinal recovery after laparoscopic colorectal cancer resection, but time-to-first-flatus is rarely treated as a time-to-event endpoint. In this single-center retrospective cohort (n = 80), an ERAS pathway emphasizing shortened fasting with preoperative carbohydrates, goal-directed fluid therapy, multimodal analgesia, early mobilization, and early enteral nutrition was associated with a markedly earlier first flatus (approximately 1.3 days sooner), faster first defecation and bowel-sound recovery, fewer gastrointestinal symptoms, shorter length of stay (approximately 3.2 days reduction), and fewer complications. Analyses should prioritize survival methods (Kaplan-Meier, Cox/accelerated failure time) and adjust for secular trends and confounding (e.g., calendar month, operative factors, opioid use), with propensity techniques as sensitivity checks. Findings support routine ERAS implementa
- Citation: Zhang XL, Hong QQ, Huang YQ. Influence of enhanced recovery after surgery protocol on first exhaust time after laparoscopic resection of colorectal cancer. World J Gastrointest Surg 2026; 18(3): 116149
- URL: https://www.wjgnet.com/1948-9366/full/v18/i3/116149.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i3.116149
Colorectal cancer, as one of the most common malignancies globally, ranks among the top in both incidence and mortality rates among malignant tumors[1]. According to the latest World Health Organization statistics, over 1.9 million new cases of colorectal cancer occur globally each year, with approximately 930000 deaths, ranking third in malignancy incidence and second in mortality[2]. In China, with accelerating population aging and lifestyle changes, the incidence of colorectal cancer shows an annual upward trend, becoming a major public health issue seriously threatening national health[3].
Laparoscopic surgery has become the primary surgical treatment modality for colorectal cancer due to its minimally invasive advantages and rapid recovery[4]. However, delayed postoperative gastrointestinal function recovery remains an important factor affecting patient rehabilitation. Clinical studies indicate that the incidence of gastrointestinal dys
The mechanisms of delayed postoperative gastrointestinal function recovery are complex, mainly including: Neural reflex inhibition caused by surgical trauma; direct inhibitory effects of anesthetic drugs on the gastrointestinal tract; intestinal inflammatory response caused by mechanical stimulation from intraoperative traction and manipulation; sympathetic nervous excitation caused by postoperative pain and stress; and the effects of prolonged fasting and activity restriction in traditional perioperative management[7]. These factors interact to collectively cause postoperative wea
The introduction of the enhanced recovery after surgery (ERAS) concept provides new insights and practical guidance for improving postoperative gastrointestinal function recovery[9]. The ERAS concept was initially proposed by Danish surgeon Henrik Kehlet in the 1990s. After more than 30 years of development and refinement, it has formed a comprehensive perioperative management system based on evidence-based medicine[10]. ERAS emphasizes multidisciplinary collaboration, reducing surgical stress response, maintaining homeostasis, and promoting rapid patient recovery through optimization of preoperative, intraoperative, and postoperative management[11].
In the field of gastrointestinal surgery, ERAS protocols have been proven to effectively promote postoperative gas
International authoritative organizations have established ERAS guidelines for colorectal surgery. Multiple studies have confirmed that perioperative management guided by ERAS concepts can shorten time to first flatus, reduce postoperative complication rates, shorten hospital stays, and improve patient satisfaction[14]. However, current domestic and international research on ERAS in laparoscopic colorectal cancer surgery mostly focuses on overall effectiveness evaluation, with relatively few specialized studies using time to first flatus as a key indicator. Additionally, different studies vary in ERAS protocol development and outcome indicator selection, affecting the comparability and generalizability of research results[15].
Furthermore, ERAS-related research in China started relatively late and lags behind international advanced levels, urgently requiring high-quality clinical studies to provide scientific evidence for clinical practice. Based on this back
This retrospective study collected and analyzed clinical data from 80 patients with colorectal cancer who underwent laparoscopic surgical treatment at the Department of Colorectal Oncology Surgery, The First Affiliated Hospital of Xiamen University, from May 2023 to August 2024. The study protocol was approved by the hospital Ethics Committee and strictly adhered to the ethical principles of the Declaration of Helsinki.
Inclusion criteria: (1) Age 18 to 80 years, regardless of gender; (2) Primary colorectal cancer confirmed by pathological examination; (3) First curative surgical treatment; (4) Laparoscopic surgical approach; (5) Complete clinical data, inclu
Exclusion criteria: (1) Severe cardiac, hepatic, or renal insufficiency; (2) Preoperative intestinal obstruction or perforation; (3) Concurrent other malignancies; (4) Previous radiotherapy or chemotherapy; (5) Psychiatric illness or cognitive dys
Patients were divided into two groups according to the perioperative management protocol received: Conventional group (n = 40) and ERAS group (n = 40). Grouping was determined by patient admission time and department perioperative management protocol implementation status. Patients receiving traditional perioperative management from May 2023 to December 2023 were included in the conventional group, while patients receiving ERAS protocol management from January 2024 to August 2024 were included in the ERAS group.
Conventional group patients received traditional standardized perioperative management: (1) Routine preoperative fasting for 12 hours and water restriction for 6 hours; (2) Routine mechanical bowel preparation the night before surgery with oral polyethylene glycol electrolyte solution; (3) Routine preoperative placement of nasogastric tube and urinary catheter; (4) Routine intraoperative fluid management; (5) Clear liquid diet starting on postoperative day 3, gradually transitioning to semi-liquid and regular diet; (6) Routine postoperative parenteral nutritional support; (7) Nasogastric tube removal after flatus; (8) Bedside mobilization starting on postoperative day 2; and (9) Perioperative health education according to standard departmental nursing procedures.
ERAS group patients received ERAS protocol management based on conventional treatment: (1) Optimized preoperative preparation. Preoperative health education, detailed introduction of ERAS concepts and specific measures to patients and families to enhance patient compliance; no routine mechanical bowel preparation, simplified bowel preparation only for low rectal cancer patients the night before surgery; cessation of solid food intake 6 hours before surgery, with 300 mL carbohydrate-containing clear liquid (5% glucose solution) allowed 2 hours before surgery; prophylactic antibiotic use 30 minutes before surgery; (2) Optimized intraoperative management. Multimodal analgesia combining general anesthesia with epidural anesthesia; implementation of goal-directed fluid therapy, precise control of fluid volume based on hemodynamic parameters to avoid fluid overload or insufficiency; use of warming blankets and warmed fluids to maintain intraoperative body temperature; no routine nasogastric tube placement, immediate removal if placed; selective urinary catheter placement, removal within 24 hours postoperatively; (3) Optimized postoperative management. Small amounts of water starting 6 to 12 hours postoperatively, clear liquid diet within 24 hours; semi-liquid diet starting on postoperative day 1, gradually transitioning to soft diet on day 2; assisted in-bed mobilization 6 hours postoperatively, assisted ambulation on postoperative day 1, gradually increasing daily activity time; multimodal analgesia management combining non-steroidal anti-inflammatory drugs and opioids, with patient-controlled analgesia when necessary; enteral nutritional support starting on postoperative day 1, gradually increasing intake based on patient tolerance; oral prokinetic agents starting on postoperative day 1 to promote gastrointestinal motility recovery; dynamic monitoring of gastrointestinal function recovery indicators, timely adjustment of management protocol; and (4) Discharge criteria. Normal body temperature, no signs of infection; flatus and defecation, able to tolerate soft or regular diet; good pain control, able to take oral analgesics; good wound healing, no discharge; patient and family understand post-discharge precautions and agree to discharge.
Primary observation indicators: (1) Time to first flatus. Time interval from surgery completion to first spontaneous flatus; (2) Time to first defecation. Time interval from surgery completion to first spontaneous defecation; (3) Time to bowel sounds recovery. Time for postoperative bowel sounds to return to normal (3 to 5 times per minute); and (4) Length of hospital stay. Total days from admission to discharge.
Secondary observation indicators: (1) Gastrointestinal discomfort symptoms. Including occurrence of abdominal distension, nausea, and vomiting; (2) Postoperative complication rate. Including wound infection, anastomotic leak, pulmonary infection, urinary tract infection, intestinal obstruction, etc.; (3) Severe complication rate. Complications of Clavien-Dindo grade III or above; (4) Postoperative food tolerance. Assessment of patient tolerance to different foods; (5) 30-day readmission rate. Proportion of patients readmitted within 30 days after discharge due to related complications; and (6) Patient satisfaction. Assessed using a 5-point scale, with 1 = very dissatisfied and 5 = very satisfied.
Laboratory indicators: (1) White blood cell count (WBC); (2) C-reactive protein (CRP); and (3) Serum albumin; detection time points were 1 day preoperatively, postoperative day 3, and postoperative day 7.
SPSS 26.0 statistical software was used for data analysis. Continuous variables were expressed as mean ± SD, with independent sample t-tests used for inter-group comparisons. Categorical variables were expressed as n (%), with χ2 test or Fisher’s exact test used for inter-group comparisons. Repeated measurement data were analyzed using repeated measures ANOVA. Statistical significance was set at P < 0.05. Missing data were handled using complete case analysis.
Baseline characteristic comparison showed that both groups were well-matched across all measured parameters, ensuring validity of subsequent comparative analyses. The study enrolled 80 patients with a mean age of approximately 65 years, with no significant age difference between the conventional group (65.1 ± 11.8 years) and ERAS group (64.3 ± 12.1 years; t = 0.387, P = 0.700). Gender distribution was similarly balanced, with males comprising 55.0% (22/40) in the conventional group and 57.5% (23/40) in the ERAS group, showing no statistically significant difference (χ2 = 0.089, P = 0.766).
Anthropometric and preoperative assessments further confirmed inter-group comparability. Body mass index values were nearly identical between the conventional and ERAS groups (23.5 ± 2.9 kg/m2 vs 23.4 ± 3.0 kg/m2, respectively), with no significant difference observed (t = 0.156, P = 0.876). Preoperative serum albumin levels, a key indicator of baseline nutritional status, showed no significant difference between groups (39.2 ± 3.9 g/L vs 39.4 ± 3.8 g/L, t = 0.215, P = 0.830), indicating both groups entered the study with comparable nutritional status. Regarding tumor characteristics, both groups showed no statistical differences in tumor location, tumor-node-metastasis staging, or tumor differentiation. Surgery-related indicators revealed no statistically significant differences between groups in operative time, intraoperative blood loss, or anesthesia time, ensuring comparability of surgical factors (Table 1).
| Characteristic | Conventional group (n = 40) | ERAS group (n = 40) | Test statistic | P value |
| Age (years) | 65.1 ± 11.8 | 64.3 ± 12.1 | t = 0.387 | 0.700 |
| Gender | χ2 = 0.089 | 0.766 | ||
| Male | 22 (55.0) | 23 (57.5) | ||
| Female | 18 (45.0) | 17 (42.5) | ||
| BMI (kg/m2) | 23.5 ± 2.9 | 23.4 ± 3.0 | t = 0.156 | 0.876 |
| Preoperative serum albumin (g/L) | 39.2 ± 3.9 | 39.4 ± 3.8 | t = 0.215 | 0.830 |
| Tumor location | χ2 = 0.082 | 0.775 | ||
| Colon | 23 (57.5) | 22 (55.0) | ||
| Rectum | 17 (42.5) | 18 (45.0) | ||
| TNM staging | χ2 = 0.456 | 0.800 | ||
| Stage I | 8 (20.0) | 7 (17.5) | ||
| Stage II | 19 (47.5) | 20 (50.0) | ||
| Stage III | 13 (32.5) | 13 (32.5) | ||
| Tumor differentiation | χ2 = 0.289 | 0.865 | ||
| Well differentiated | 12 (30.0) | 13 (32.5) | ||
| Moderately differentiated | 21 (52.5) | 20 (50.0) | ||
| Poorly differentiated | 7 (17.5) | 7 (17.5) | ||
| Operative time (minutes) | 164.8 ± 31.9 | 163.5 ± 30.2 | t = 0.218 | 0.828 |
| Intraoperative blood loss (mL) | 84.9 ± 27.8 | 83.1 ± 26.5 | t = 0.325 | 0.746 |
| Anesthesia time (minutes) | 194.5 ± 34.8 | 192.8 ± 33.5 | t = 0.237 | 0.813 |
| ASA score | χ2 = 0.000 | 1.000 | ||
| Grade I-II | 32 (80.0) | 32 (80.0) | ||
| Grade III | 8 (20.0) | 8 (20.0) |
Gastrointestinal function recovery assessment revealed significantly superior recovery in the ERAS group compared to the conventional group. Time to first flatus, the primary outcome of this study, was 2.2 ± 0.8 days in the ERAS group, significantly earlier than 3.5 ± 1.2 days in the conventional group (t = 5.672, P < 0.001), representing a mean reduction of 1.3 days. Time to first defecation was 3.3 ± 1.0 days in the ERAS group vs 4.8 ± 1.4 days in the conventional group (t = 5.231, P < 0.001), a mean reduction of 1.5 days. Time to bowel sounds recovery was 2.5 ± 0.8 days in the ERAS group vs 4.2 ± 1.3 days in the conventional group (t = 6.894, P < 0.001), with highly statistically significant differences.
Food tolerance assessment showed that on postoperative day 1, the proportion of patients tolerating clear liquids was 92.5% (37/40) in the ERAS group, significantly higher than 62.5% (25/40) in the conventional group (χ2 = 12.345, P < 0.001). On postoperative day 2, 85.0% (34/40) of ERAS patients tolerated semi-liquid diet, while only 52.5% (21/40) in the conventional group reached this level (χ2 = 13.128, P < 0.001). By postoperative day 5, 95.0% (38/40) of ERAS patients could eat normally, compared to 75.0% (30/40) in the conventional group (χ2 = 8.571, P < 0.01). These results demonstrate that the ERAS protocol effectively promotes postoperative gastrointestinal function recovery through early enteral nutri
| Recovery parameter | Conventional group (n = 40) | ERAS group (n = 40) | Test statistic | P value |
| Time to first flatus (days) | 3.5 ± 1.2 | 2.2 ± 0.8 | t = 5.672 | < 0.001 |
| Time to first defecation (days) | 4.8 ± 1.4 | 3.3 ± 1.0 | t = 5.231 | < 0.001 |
| Time to bowel sounds recovery (days) | 4.2 ± 1.3 | 2.5 ± 0.8 | t = 6.894 | < 0.001 |
| Clear liquid tolerance on POD 1 | 25 (62.5) | 37 (92.5) | χ2 = 12.345 | < 0.001 |
| Semi-liquid diet tolerance on POD 2 | 21 (52.5) | 34 (85.0) | χ2 = 13.128 | < 0.001 |
| Normal diet on POD 5 | 30 (75.0) | 38 (95.0) | χ2 = 8.571 | < 0.01 |
Postoperative gastrointestinal symptom assessment demonstrated significant advantages in the ERAS group. Abdominal distension occurred in 27.5% (11/40) of ERAS patients vs 55.0% (22/40) in the conventional group (χ2 = 7.892, P < 0.01). Nausea affected 22.5% (9/40) and 45.0% (18/40) of patients respectively (χ2 = 6.781, P < 0.01), while vomiting was re
By postoperative day 3, 77.5% (31/40) of ERAS patients reported good to fair appetite, compared to 47.5% (19/40) in the conventional group (χ2 = 11.234, P < 0.001). Duration of anorexia was shorter in the ERAS group at 2.3 ± 1.0 days vs 4.0 ± 1.5 days in the conventional group (t = 5.876, P < 0.001). Hiccup episodes were less frequent, affecting 7.5% (3/40) and 22.5% (9/40) of patients respectively (χ2 = 4.587, P < 0.05). The cumulative symptom burden score was lower in the ERAS group at 8.5 ± 3.1 vs 15.1 ± 5.0 in the conventional group (t = 7.012, P < 0.001; Table 3).
| Gastrointestinal symptom1 | Conventional group (n = 40) | ERAS group (n = 40) | Test statistic | P value |
| Abdominal distension | 22 (55.0) | 11 (27.5) | χ2 = 7.892 | < 0.01 |
| Nausea | 18 (45.0) | 9 (22.5) | χ2 = 6.781 | < 0.01 |
| Vomiting | 13 (32.5) | 5 (12.5) | χ2 = 6.234 | < 0.05 |
| Hiccup episodes | 9 (22.5) | 3 (7.5) | χ2 = 4.587 | < 0.05 |
| Abdominal distension severity score | 4.5 ± 2.0 | 2.7 ± 1.4 | t = 4.678 | < 0.001 |
| Bloating-induced abdominal pain | 5.1 ± 2.2 | 3.0 ± 1.7 | t = 4.892 | < 0.001 |
| Good to fair appetite on day 3 | 19 (47.5) | 31 (77.5) | χ2 = 11.234 | < 0.001 |
| Duration of anorexia (days) | 4.0 ± 1.5 | 2.3 ± 1.0 | t = 5.876 | < 0.001 |
| 24-hour gastric residual volume (mL) | 86.8 ± 34.9 | 47.9 ± 22.3 | t = 4.012 | < 0.001 |
| Cumulative symptom burden score | 15.1 ± 5.0 | 8.5 ± 3.1 | t = 7.012 | < 0.001 |
Hospital stay analysis demonstrated significant advantages for the ERAS group. Mean total hospital stay in the ERAS group was 9.1 ± 2.4 days, substantially reduced compared to 12.3 ± 3.2 days in the conventional group (t = 5.123, P < 0.001). This 3.2-day reduction represents a 26.0% decrease in total hospital stay. When specifically examining the postope
Economic burden assessment revealed potential cost savings from ERAS implementation. Direct medical costs, in
| Clinical outcome indicator1 | Conventional group (n = 40) | ERAS group (n = 40) | Test statistic | P value |
| Total hospital stay (days) | 12.3 ± 3.2 | 9.1 ± 2.4 | t = 5.123 | < 0.001 |
| Postoperative hospital stay (days) | 9.5 ± 2.7 | 6.7 ± 2.0 | t = 5.345 | < 0.001 |
| Preoperative hospital stay (days) | 2.8 ± 0.8 | 2.4 ± 0.7 | t = 2.345 | 0.021 |
| Patients meeting discharge criteria on day 7 | 19 (47.5) | 34 (85.0) | χ2 = 14.567 | < 0.001 |
| Prolonged hospitalization (> 14 days) | 8 (20.0) | 2 (5.0) | χ2 = 5.128 | < 0.05 |
| Time to resume normal activities (days) | 18.2 ± 4.6 | 13.1 ± 3.7 | t = 5.678 | < 0.001 |
| Direct medical costs (yuan) | 51200 ± 9300 | 48100 ± 7500 | t = 1.789 | 0.078 |
| Medical costs for uncomplicated cases (yuan) | 47600 ± 6800 | 43700 ± 5100 | t = 2.891 | < 0.01 |
Statistical analysis of postoperative complications revealed significantly lower complication rates in the ERAS group compared to the conventional group. Overall complication rate was 12.5% (5/40) in the ERAS group vs 32.5% (13/40) in the conventional group, with statistically significant difference (χ2 = 5.342, P < 0.05). Analysis of specific complication types showed: Wound infection rate was 5.0% (2/40) in the ERAS group vs 15.0% (6/40) in the conventional group (χ2 = 3.987, P = 0.046); anastomotic leak incidence was 2.5% (1/40) in the ERAS group vs 7.5% (3/40) in the conventional group (P = 0.258, Fisher’s exact test); pulmonary infection rate was 7.5% (3/40) in the ERAS group vs 17.5% (7/40) in the conventional group (χ2 = 3.456, P = 0.063).
Severe complication rate (Clavien-Dindo grade III or above) was 5.0% (2/40) in the ERAS group vs 12.5% (5/40) in the conventional group (P = 0.221, Fisher’s exact test). Thirty-day readmission rate was 2.5% (1/40) in the ERAS group vs 10.0% (4/40) in the conventional group (P = 0.178, Fisher’s exact test). Although differences in some individual complications did not reach statistical significance, the ERAS group showed downward trends in all complication types. The significant reduction in overall complication rate fully demonstrates the safety and effectiveness of the ERAS protocol (Table 5).
| Complication type | Conventional group (n = 40) | ERAS group (n = 40) | Test statistic | P value |
| Overall complications | 13 (32.5) | 5 (12.5) | χ2 = 5.342 | < 0.05 |
| Wound infection | 6 (15.0) | 2 (5.0) | χ2 = 3.987 | 0.046 |
| Anastomotic leak | 3 (7.5) | 1 (2.5) | 0.2581 | |
| Pulmonary infection | 7 (17.5) | 3 (7.5) | χ2 = 3.456 | 0.063 |
| Urinary tract infection | 3 (7.5) | 1 (2.5) | 0.2581 | |
| Intestinal obstruction | 2 (5.0) | 0 (0.0) | 0.1541 | |
| Severe complications (grade III or above) | 5 (12.5) | 2 (5.0) | 0.2211 | |
| 30-day readmission | 4 (10.0) | 1 (2.5) | 0.1781 |
Postoperative inflammatory marker monitoring revealed significantly lower inflammatory response in the ERAS group compared to the conventional group. On postoperative day 3, WBC in the ERAS group was 9.1 ± 2.5 × 109/L, lower than 11.4 ± 3.2 × 109/L in the conventional group (t = 3.678, P < 0.001). CRP level in the ERAS group was 14.2 ± 6.4 mg/L, significantly lower than 22.8 ± 8.7 mg/L in the conventional group (t = 5.123, P < 0.001). By postoperative day 7, CRP level in the ERAS group decreased to 7.8 ± 3.7 mg/L vs 13.6 ± 5.9 mg/L in the conventional group (t = 5.456, P < 0.001). Serum albumin level recovery showed that on postoperative day 7, the ERAS group reached 36.3 ± 3.1 g/L, higher than 33.8 ± 4.1 g/L in the conventional group (t = 3.012, P < 0.01). These results indicate that the ERAS protocol not only promotes gastrointestinal function recovery but also effectively controls postoperative inflammatory response and promotes physical recovery through measures such as reducing surgical stress and optimizing nutritional support (Table 6).
| Indicator | Time point | Conventional group (n = 40) | ERAS group (n = 40) | Test statistic | P value |
| WBC count (× 109/L) | Preoperative | 6.6 ± 2.1 | 6.5 ± 2.0 | t = 0.218 | 0.828 |
| POD 3 | 11.4 ± 3.2 | 9.1 ± 2.5 | t = 3.678 | < 0.001 | |
| POD 7 | 8.8 ± 2.7 | 7.0 ± 2.2 | t = 3.456 | < 0.01 | |
| CRP (mg/L) | Preoperative | 4.0 ± 1.9 | 3.8 ± 1.7 | t = 0.512 | 0.610 |
| POD 3 | 22.8 ± 8.7 | 14.2 ± 6.4 | t = 5.123 | < 0.001 | |
| POD 7 | 13.6 ± 5.9 | 7.8 ± 3.7 | t = 5.456 | < 0.001 | |
| Serum albumin (g/L) | Preoperative | 39.2 ± 3.9 | 39.4 ± 3.8 | t = 0.215 | 0.830 |
| POD 7 | 33.8 ± 4.1 | 36.3 ± 3.1 | t = 3.012 | < 0.01 |
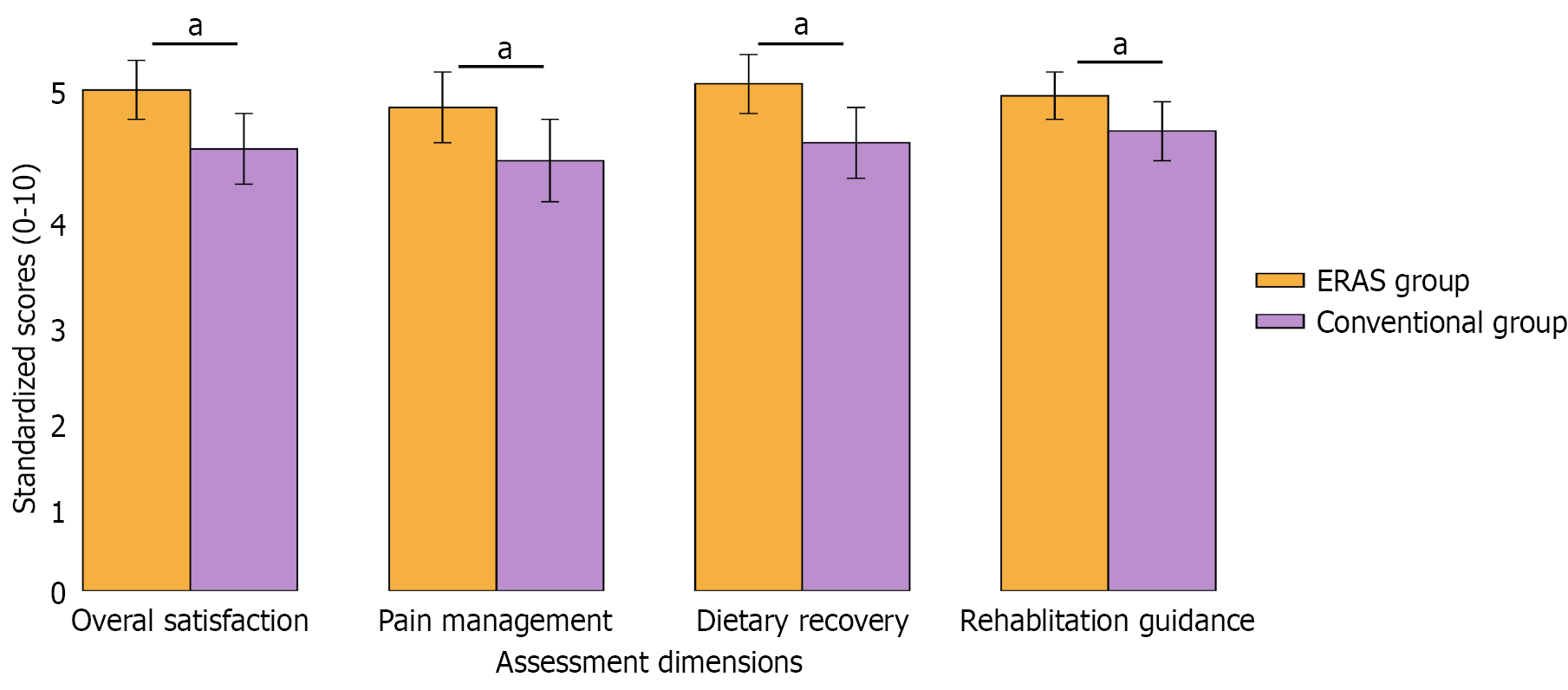
Patient satisfaction survey results demonstrated significantly higher overall satisfaction in the ERAS group compared to the conventional group. Using a 5-point scale (1 = very dissatisfied, 5 = very satisfied), overall satisfaction with perioperative management in the ERAS group scored 4.7 ± 0.4, significantly higher than 3.8 ± 0.8 in the conventional group (t = 5.678, P < 0.001). Specific dimension assessments showed: Pain management satisfaction scored 4.6 ± 0.5 in the ERAS group vs 3.6 ± 0.9 in the conventional group (t = 5.123, P < 0.001); dietary recovery satisfaction scored 4.8 ± 0.4 in the ERAS group vs 3.7 ± 0.8 in the conventional group (t = 6.894, P < 0.001); rehabilitation guidance satisfaction scored 4.7 ± 0.4 in the ERAS group vs 4.0 ± 0.7 in the conventional group (t = 4.987, P < 0.001). Patient acceptance survey of the ERAS protocol showed that 92.5% (37/40) of ERAS patients would recommend this management model to other patients, compared to 72.5% (29/40) in the conventional group (χ2 = 7.891, P < 0.01). These results indicate that the ERAS protocol not only improved objective clinical indicators but, more importantly, enhanced patients’ subjective experience and satisfaction (Figure 1).
Colorectal cancer represents one of the most significant global health challenges, with incidence rates continuing to rise in both developed and developing countries[16]. The World Health Organization’s latest epidemiological data emphasizes the severity of this issue, reporting over 1.9 million new cases globally annually, making colorectal cancer the third most common malignancy and second leading cause of cancer-related death[17]. In China, rapid socioeconomic development, population aging, and lifestyle changes have led to substantial increases in colorectal cancer incidence, making it a critical public health issue requiring immediate and comprehensive intervention strategies[18].
Laparoscopic surgery has become the primary treatment modality for colorectal cancer due to its minimally invasive advantages[19]. However, postoperative gastrointestinal function recovery remains a critical factor affecting patient rehabilitation. Time to first flatus, an important indicator for assessing gastrointestinal function recovery, not only affects patient comfort but delayed recovery may also increase complication risk and prolong hospital stay[20]. This study systematically evaluated the impact of the ERAS protocol on time to first flatus by comparing traditional perioperative management with ERAS protocol management.
Study results showed that time to first flatus in the ERAS group was 2.2 ± 0.8 days, significantly shorter than 3.5 ± 1.2 days in the conventional group (P < 0.001), representing a mean reduction of 1.3 days or 37.1%. These results are consistent with multiple domestic and international studies[21,22]. The observed 1.3-day reduction in first flatus time (37.1% improvement) can be mechanistically attributed to specific ERAS interventions implemented in our protocol. First, shortened preoperative fasting time combined with carbohydrate loading 2 hours before surgery directly reduced postoperative insulin resistance this metabolic optimization is reflected in our finding that 92.5% of ERAS patients tolerated clear liquids on postoperative day 1 vs only 62.5% in the conventional group (P < 0.001)[23]. Second, our goal-directed fluid therapy protocol, which maintained precise fluid balance based on hemodynamic parameters, prevented intestinal edema and maintained intestinal perfusion evidenced by the ERAS group’s significantly lower 24-hour gastric residual volume (47.9 ± 22.3 mL vs 86.8 ± 34.9 mL, P < 0.001)[24]. Third, early mobilization initiated 6 hours postoperatively promoted intestinal peristalsis through mechanical stimulation, contributing to the earlier bowel sounds recovery (2.5 ± 0.8 days vs 4.2 ± 1.3 days, P < 0.001). Finally, early enteral nutrition starting within 24 hours postoperatively directly stimulated intestinal hormone secretion and maintained gut barrier function, as demonstrated by our superior food tolerance rates throughout the postoperative period[25]. These mechanisms worked synergistically to achieve the signifi
This study also found that time to first defecation and bowel sounds recovery in the ERAS group were significantly earlier than the conventional group, further confirming the ERAS protocol’s promotion of overall gastrointestinal function recovery. The 1.5-day reduction in first defecation time (3.3 ± 1.0 days vs 4.8 ± 1.4 days) and 1.7-day reduction in bowel sounds recovery (2.5 ± 0.8 days vs 4.2 ± 1.3 days) represent clinically meaningful improvements that translate directly to reduced patient discomfort and earlier hospital discharge eligibility. More importantly, food tolerance in the ERAS group was significantly superior to the conventional group, with 92.5% of patients tolerating clear liquids on postoperative day 1, significantly higher than 62.5% in the conventional group. This improvement in early food tolerance not only reflects good gastrointestinal function recovery but also provides earlier nutritional support for patients, creating a virtuous cycle[26].
Postoperative gastrointestinal discomfort symptoms seriously affect patient comfort and satisfaction. This study demonstrated that incidence of abdominal distension, nausea, and vomiting in the ERAS group was significantly lower than the conventional group. These symptom reductions (abdominal distension: 27.5% vs 55.0%, P < 0.01; nausea: 22.5% vs 45.0%, P < 0.01; vomiting: 12.5% vs 32.5%, P < 0.05) can be directly linked to specific ERAS interventions: Avoiding routine nasogastric tubes eliminated a major source of gastric irritation[27]; goal-directed fluid therapy prevented the intestinal edema that contributes to nausea; early mobilization enhanced gastric emptying; and multimodal analgesia reduced opioid consumption, thereby minimizing opioid-induced constipation and nausea[28]. The cumulative symptom burden score (8.5 ± 3.1 vs 15.1 ± 5.0, P < 0.001) quantifies the overall improvement in patient wellbeing achieved through these comprehensive measures.
Shortened hospital stay represents one of the important clinical benefits of the ERAS protocol. In this study, mean hospital stay in the ERAS group was 9.1 ± 2.4 days, 3.2 days shorter than 12.3 ± 3.2 days in the conventional group, a 26.0% reduction. This substantial reduction in length of stay reflects the compounding effects of accelerated gastroin
Postoperative complication occurrence is an important factor affecting patient prognosis. This study demonstrated that overall complication rate in the ERAS group was 12.5%, significantly lower than 32.5% in the conventional group. The specific pathophysiological mechanisms underlying this 20% absolute risk reduction warrant detailed examination. The wound infection rate reduction (5.0% vs 15.0%, P = 0.046) can be attributed to optimized nutritional status (serum albumin 36.3 ± 3.1 g/L vs 33.8 ± 4.1 g/L at post-operative day 7, P < 0.01) and early mobilization reducing tissue hypoxia. The trend toward reduced anastomotic leak (2.5% vs 7.5%) likely reflects goal-directed fluid therapy’s prevention of intestinal edema and tissue hypoperfusion at the anastomotic site. Lower pulmonary infection rates (7.5% vs 17.5%) resulted from early mobilization, avoidance of routine nasogastric tubes that increase aspiration risk, and better pain control enabling effective coughing. The reduced inflammatory markers (CRP, 14.2 ± 6.4 mg/L vs 22.8 ± 8.7 mg/L at post-operative day 3, P < 0.001) provide objective evidence that ERAS protocols attenuate the surgical stress response, creating a more favorable milieu for healing and infection resistance. Degree of inflammatory response reflects severity of surgical trauma and bodily stress. This study demonstrated that postoperative WBC and CRP levels in the ERAS group were significantly lower than the conventional group, indicating that the ERAS protocol effectively reduced postoperative inflammatory response.
Patient satisfaction is an important indicator for evaluating medical service quality. This study demonstrated that overall satisfaction in the ERAS group was significantly higher than the conventional group, with 92.5% of patients willing to recommend this management model. Improved patient satisfaction not only reflects improvement in objective clinical indicators but also embodies the ERAS protocol’s emphasis on patient subjective experience. The ERAS protocol enhanced patient compliance and confidence through comprehensive preoperative health education; early removal of nasogastric tubes and urinary catheters improved patient comfort; multimodal analgesia effectively controlled postope
However, ERAS protocol implementation also faces some challenges. First, ERAS requires close collaboration of multidisciplinary teams, including surgeons, anesthesiologists, nurses, and nutritionists, necessitating establishment of good team building and communication mechanisms. Second, ERAS protocol implementation requires changing traditional medical concepts and habits, necessitating systematic training for medical personnel. Third, certain ERAS measures may increase medical personnel workload, requiring reasonable allocation of human resources. Additionally, patient compliance is an important factor affecting ERAS effectiveness, requiring enhanced preoperative education and postoperative guidance.
This study has some limitations. First, as a single-center retrospective study with relatively small sample size (40 cases per group), selection bias may exist. Second, grouping by time period rather than randomization, though baseline characteristics were comparable, may have some confounding factors (such as subtle differences in medical technology across time periods). Third, this study mainly focused on short-term clinical outcomes, lacking observation of long-term prognosis such as tumor recurrence and survival rates. Finally, this study did not analyze the individual effects of various ERAS protocol measures, unable to clarify which measures contribute most to gastrointestinal function recovery.
Despite the above limitations, this study still provides important evidence for ERAS protocol application in laparoscopic colorectal cancer surgery. Future research should conduct multicenter, large-sample randomized controlled trials to further validate ERAS protocol effectiveness; explore optimal combinations and implementation timing of various ERAS protocol measures; evaluate ERAS protocol impact on long-term prognosis; study ERAS protocol cost-effectiveness ratios; explore strategies for developing individualized ERAS plans. Additionally, attention should be paid to barrier factors in ERAS protocol implementation, developing effective implementation strategies to promote widespread clinical application of ERAS concepts.
This study, through retrospective analysis of 80 laparoscopic colorectal cancer resection patients (40 cases in conventional group, 40 cases in ERAS group), confirms that application of the ERAS protocol in laparoscopic colorectal cancer resection can significantly shorten time to first flatus, promote rapid gastrointestinal function recovery, while simultaneously shortening hospital stay, reducing postoperative complication rates, and improving patient satisfaction, demonstrating important clinical promotion value.
| 1. | Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76817] [Cited by in RCA: 69423] [Article Influence: 13884.6] [Reference Citation Analysis (40)] |
| 2. | Morgan E, Arnold M, Gini A, Lorenzoni V, Cabasag CJ, Laversanne M, Vignat J, Ferlay J, Murphy N, Bray F. Global burden of colorectal cancer in 2020 and 2040: incidence and mortality estimates from GLOBOCAN. Gut. 2023;72:338-344. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1885] [Cited by in RCA: 1483] [Article Influence: 494.3] [Reference Citation Analysis (3)] |
| 3. | Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F, Jemal A, Yu XQ, He J. Cancer statistics in China, 2015. CA Cancer J Clin. 2016;66:115-132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11304] [Cited by in RCA: 13209] [Article Influence: 1320.9] [Reference Citation Analysis (4)] |
| 4. | Jayne DG, Thorpe HC, Copeland J, Quirke P, Brown JM, Guillou PJ. Five-year follow-up of the Medical Research Council CLASICC trial of laparoscopically assisted versus open surgery for colorectal cancer. Br J Surg. 2010;97:1638-1645. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 856] [Cited by in RCA: 725] [Article Influence: 45.3] [Reference Citation Analysis (0)] |
| 5. | Vather R, Trivedi S, Bissett I. Defining postoperative ileus: results of a systematic review and global survey. J Gastrointest Surg. 2013;17:962-972. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 425] [Cited by in RCA: 382] [Article Influence: 29.4] [Reference Citation Analysis (1)] |
| 6. | Müller SA, Rahbari NN, Schneider F, Warschkow R, Simon T, von Frankenberg M, Bork U, Weitz J, Schmied BM, Büchler MW. Randomized clinical trial on the effect of coffee on postoperative ileus following elective colectomy. Br J Surg. 2012;99:1530-1538. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 82] [Article Influence: 5.9] [Reference Citation Analysis (1)] |
| 7. | Kehlet H, Holte K. Review of postoperative ileus. Am J Surg. 2001;182:3S-10S. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 271] [Cited by in RCA: 222] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 8. | Kalff JC, Schraut WH, Simmons RL, Bauer AJ. Surgical manipulation of the gut elicits an intestinal muscularis inflammatory response resulting in postsurgical ileus. Ann Surg. 1998;228:652-663. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 449] [Cited by in RCA: 397] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 9. | Ljungqvist O, Scott M, Fearon KC. Enhanced Recovery After Surgery: A Review. JAMA Surg. 2017;152:292-298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2935] [Cited by in RCA: 2493] [Article Influence: 277.0] [Reference Citation Analysis (1)] |
| 10. | Kehlet H. Multimodal approach to control postoperative pathophysiology and rehabilitation. Br J Anaesth. 1997;78:606-617. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2310] [Cited by in RCA: 1782] [Article Influence: 61.4] [Reference Citation Analysis (3)] |
| 11. | Greco M, Capretti G, Beretta L, Gemma M, Pecorelli N, Braga M. Enhanced recovery program in colorectal surgery: a meta-analysis of randomized controlled trials. World J Surg. 2014;38:1531-1541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 717] [Cited by in RCA: 645] [Article Influence: 53.8] [Reference Citation Analysis (1)] |
| 12. | Gustafsson UO, Scott MJ, Hubner M, Nygren J, Demartines N, Francis N, Rockall TA, Young-Fadok TM, Hill AG, Soop M, de Boer HD, Urman RD, Chang GJ, Fichera A, Kessler H, Grass F, Whang EE, Fawcett WJ, Carli F, Lobo DN, Rollins KE, Balfour A, Baldini G, Riedel B, Ljungqvist O. Guidelines for Perioperative Care in Elective Colorectal Surgery: Enhanced Recovery After Surgery (ERAS(®)) Society Recommendations: 2018. World J Surg. 2019;43:659-695. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1767] [Cited by in RCA: 1433] [Article Influence: 204.7] [Reference Citation Analysis (0)] |
| 13. | Zhuang CL, Ye XZ, Zhang XD, Chen BC, Yu Z. Enhanced recovery after surgery programs versus traditional care for colorectal surgery: a meta-analysis of randomized controlled trials. Dis Colon Rectum. 2013;56:667-678. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 402] [Cited by in RCA: 342] [Article Influence: 26.3] [Reference Citation Analysis (2)] |
| 14. | Spanjersberg WR, van Sambeeck JD, Bremers A, Rosman C, van Laarhoven CJ. Systematic review and meta-analysis for laparoscopic versus open colon surgery with or without an ERAS programme. Surg Endosc. 2015;29:3443-3453. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 168] [Cited by in RCA: 144] [Article Influence: 13.1] [Reference Citation Analysis (1)] |
| 15. | Pędziwiatr M, Mavrikis J, Witowski J, Adamos A, Major P, Nowakowski M, Budzyński A. Current status of enhanced recovery after surgery (ERAS) protocol in gastrointestinal surgery. Med Oncol. 2018;35:95. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 259] [Cited by in RCA: 230] [Article Influence: 28.8] [Reference Citation Analysis (7)] |
| 16. | Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53448] [Cited by in RCA: 56055] [Article Influence: 7006.9] [Reference Citation Analysis (6)] |
| 17. | Arnold M, Sierra MS, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global patterns and trends in colorectal cancer incidence and mortality. Gut. 2017;66:683-691. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3978] [Cited by in RCA: 3556] [Article Influence: 395.1] [Reference Citation Analysis (3)] |
| 18. | Feng RM, Zong YN, Cao SM, Xu RH. Current cancer situation in China: good or bad news from the 2018 Global Cancer Statistics? Cancer Commun (Lond). 2019;39:22. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1059] [Cited by in RCA: 1164] [Article Influence: 166.3] [Reference Citation Analysis (2)] |
| 19. | Lacy AM, García-Valdecasas JC, Delgado S, Castells A, Taurá P, Piqué JM, Visa J. Laparoscopy-assisted colectomy versus open colectomy for treatment of non-metastatic colon cancer: a randomised trial. Lancet. 2002;359:2224-2229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2003] [Cited by in RCA: 1771] [Article Influence: 73.8] [Reference Citation Analysis (2)] |
| 20. | Wolthuis AM, Bislenghi G, Fieuws S, de Buck van Overstraeten A, Boeckxstaens G, D'Hoore A. Incidence of prolonged postoperative ileus after colorectal surgery: a systematic review and meta-analysis. Colorectal Dis. 2016;18:O1-O9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 172] [Cited by in RCA: 159] [Article Influence: 15.9] [Reference Citation Analysis (2)] |
| 21. | Vlug MS, Wind J, Hollmann MW, Ubbink DT, Cense HA, Engel AF, Gerhards MF, van Wagensveld BA, van der Zaag ES, van Geloven AA, Sprangers MA, Cuesta MA, Bemelman WA; LAFA study group. Laparoscopy in combination with fast track multimodal management is the best perioperative strategy in patients undergoing colonic surgery: a randomized clinical trial (LAFA-study). Ann Surg. 2011;254:868-875. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 662] [Cited by in RCA: 588] [Article Influence: 39.2] [Reference Citation Analysis (2)] |
| 22. | Wang Q, Suo J, Jiang J, Wang C, Zhao YQ, Cao X. Effectiveness of fast-track rehabilitation vs conventional care in laparoscopic colorectal resection for elderly patients: a randomized trial. Colorectal Dis. 2012;14:1009-1013. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 125] [Article Influence: 8.9] [Reference Citation Analysis (1)] |
| 23. | Smith MD, McCall J, Plank L, Herbison GP, Soop M, Nygren J. Preoperative carbohydrate treatment for enhancing recovery after elective surgery. Cochrane Database Syst Rev. 2014;2014:CD009161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 152] [Article Influence: 12.7] [Reference Citation Analysis (1)] |
| 24. | Brandstrup B, Tønnesen H, Beier-Holgersen R, Hjortsø E, Ørding H, Lindorff-Larsen K, Rasmussen MS, Lanng C, Wallin L, Iversen LH, Gramkow CS, Okholm M, Blemmer T, Svendsen PE, Rottensten HH, Thage B, Riis J, Jeppesen IS, Teilum D, Christensen AM, Graungaard B, Pott F; Danish Study Group on Perioperative Fluid Therapy. Effects of intravenous fluid restriction on postoperative complications: comparison of two perioperative fluid regimens: a randomized assessor-blinded multicenter trial. Ann Surg. 2003;238:641-648. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1309] [Cited by in RCA: 1065] [Article Influence: 46.3] [Reference Citation Analysis (1)] |
| 25. | Lewis SJ, Andersen HK, Thomas S. Early enteral nutrition within 24 h of intestinal surgery versus later commencement of feeding: a systematic review and meta-analysis. J Gastrointest Surg. 2009;13:569-575. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 350] [Cited by in RCA: 278] [Article Influence: 16.4] [Reference Citation Analysis (3)] |
| 26. | Lassen K, Soop M, Nygren J, Cox PB, Hendry PO, Spies C, von Meyenfeldt MF, Fearon KC, Revhaug A, Norderval S, Ljungqvist O, Lobo DN, Dejong CH; Enhanced Recovery After Surgery (ERAS) Group. Consensus review of optimal perioperative care in colorectal surgery: Enhanced Recovery After Surgery (ERAS) Group recommendations. Arch Surg. 2009;144:961-969. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 912] [Cited by in RCA: 760] [Article Influence: 44.7] [Reference Citation Analysis (1)] |
| 27. | Nelson R, Edwards S, Tse B. Prophylactic nasogastric decompression after abdominal surgery. Cochrane Database Syst Rev. 2007;2007:CD004929. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 120] [Article Influence: 6.3] [Reference Citation Analysis (1)] |
| 28. | Apfelbaum JL, Chen C, Mehta SS, Gan TJ. Postoperative pain experience: results from a national survey suggest postoperative pain continues to be undermanaged. Anesth Analg. 2003;97:534-540. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1557] [Cited by in RCA: 1325] [Article Influence: 57.6] [Reference Citation Analysis (1)] |
| 29. | Spanjersberg WR, Reurings J, Keus F, van Laarhoven CJ. Fast track surgery versus conventional recovery strategies for colorectal surgery. Cochrane Database Syst Rev. 2011;CD007635. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 267] [Cited by in RCA: 306] [Article Influence: 20.4] [Reference Citation Analysis (1)] |
| 30. | Osland E, Yunus RM, Khan S, Memon MA. Early versus traditional postoperative feeding in patients undergoing resectional gastrointestinal surgery: a meta-analysis. JPEN J Parenter Enteral Nutr. 2011;35:473-487. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 186] [Cited by in RCA: 171] [Article Influence: 11.4] [Reference Citation Analysis (3)] |