Published online Mar 27, 2026. doi: 10.4240/wjgs.v18.i3.115972
Revised: January 1, 2026
Accepted: January 19, 2026
Published online: March 27, 2026
Processing time: 115 Days and 3.4 Hours
Severe acute pancreatitis (SAP) is linked to substantial morbidity and mortality. Early enteral nutrition (EEN) has emerged as a potential therapeutic approach to preserve gut barrier function and decrease complications.
To compare the efficacy and safety of enteral nutrition (EN) initiated within vs after 48 hours in SAP.
This prospective cohort study was conducted from January 2024 to December 2025, involving 100 patients diagnosed with SAP. The patients were segregated into two groups: The EEN group (n = 52), who received EN within 48 hours, and the delayed EN (DEN) group (n = 48), who received EN after 48 hours. The primary outcomes assessed were mortality, infectious complications, length of hospital stay, and organ failure. Secondary outcomes encompassed feeding intolerance, markers of intestinal permeability, and nutritional status.
The EEN group exhibited significantly lower mortality (5.8% vs 18.8%, P < 0.05), decreased infectious complications (17.3% vs 37.5%, P < 0.01), and shorter hospital stay (18.6 ± 6.2 days vs 26.4 ± 8.5 days, P < 0.001) compared to the DEN group. The EEN group also had a significantly lower incidence of organ failure (23.1% vs 45.8%, P < 0.01). Intestinal permeability markers indicated a quicker recovery in the EEN group. The rates of feeding intolerance were similar between the groups (13.5% vs 16.7%, P > 0.05).
EEN initiated within 48 hours significantly enhances clinical outcomes in patients with SAP by decreasing mortality, infectious complications, and organ failure, and facilitating quicker recovery of gut barrier function.
Core Tip: This prospective cohort study evaluated the effects of early enteral nutrition (EEN), which was initiated within 48 hours, on clinical outcomes in patients with severe acute pancreatitis (SAP). In comparison to delayed enteral nutrition (EN), EEN notably decreased mortality, infectious complications, and organ failure, while also reducing hospitalization duration and enhancing the restoration of gut barrier function. The incidence of feeding intolerance did not rise with the prompt initiation of therapy. These results underscore the importance of timely EN as a secure and efficient therapeutic approach to enhance the prognosis of SAP patients, emphasizing its significance in clinical care.
- Citation: Jiang ML, Bao JP, Jiang SC, Xu FG. Efficacy of early enteral nutrition in patients with severe acute pancreatitis: A prospective cohort study. World J Gastrointest Surg 2026; 18(3): 115972
- URL: https://www.wjgnet.com/1948-9366/full/v18/i3/115972.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i3.115972
Acute pancreatitis (AP) is a prevalent gastrointestinal condition with a rising global incidence, affecting approximately 34 per 100000 individuals annually[1]. Of these instances, 15%-20% advance to severe AP (SAP). SAP is defined by enduring organ failure lasting over 48 hours and either local or systemic complications[2]. SAP is linked to mortality rates varying from 15%-30%, with recent population-based research revealing an approximately threefold higher long-term mortality rate compared to the general population[3].
The pathophysiology of SAP involves a complex cascade of inflammatory responses, with gut barrier dysfunction playing a pivotal role in disease progression[4]. During SAP, the intestinal mucosa undergoes significant structural and functional alterations, including increased intestinal permeability, disruption of tight junction proteins, and dysbiosis of the intestinal microbiota[5,6]. Studies have shown that about 59% of patients with AP develop intestinal barrier injury within the first two weeks of disease onset, characterized by elevated lactulose/mannitol ratios and increased systemic endotoxemia[7]. Gut barrier failure facilitates bacterial translocation from the intestinal lumen to the systemic circulation, contributing to infected pancreatic necrosis, which accounts for up to 80% of deaths in SAP[8].
Nutritional support has become a cornerstone in the management of SAP with the primary goals of maintaining gut integrity, preventing bacterial translocation, and supporting the hypermetabolic state characteristic of critical illness[9]. Historically, parenteral nutrition has been considered the standard approach for avoiding pancreatic stimulation. However, accumulating evidence from multiple randomized controlled trials and meta-analyses has shown that enteral nutrition (EN) is superior to parenteral nutrition in reducing mortality, infectious complications, and hospital costs[10,11]. These benefits are attributed to the protective effects of EN on gut barrier function, including the preservation of intestinal epithelial integrity and prevention of intestinal mucosal atrophy[12].
Timing of EN initiation has emerged as a critical factor influencing SAP outcomes. Early EN (EEN), typically defined as starting EN within 24-48 hours of hospital admission, is believed to enhance gut barrier function by intervening before significant intestinal damage occurs[13]. Meta-analyses have demonstrated that initiating EEN within 48 hours is linked to lower rates of pancreatic infection, shorter hospital stays, and reduced healthcare costs compared to delayed feeding[14]. Despite increasing evidence supporting EEN, controversies persist regarding its clinical implementation, with current guidelines offering conflicting recommendations[15]. This prospective cohort study aims to evaluate the efficacy and safety of initiating EEN within 48 hours compared with delayed EN (DEN) in a well-defined cohort of SAP patients. The study includes a comprehensive evaluation of clinical outcomes, gut barrier function, and nutritional parameters.
This prospective observational cohort study was conducted at the Gaoxin Branch of the First Affiliated Hospital of Nanchang University, China, from January 2024 to December 2025. All patients or their legal representatives provided written informed consent before enrollment. The study aimed to compare outcomes between patients receiving EEN initiated within 48 hours of hospital admission (EEN group) and those receiving DEN after 48 hours (DEN group). The decision on the timing of EN initiation was based on the clinical judgment of the attending physician, patient he
Inclusion criteria: Patients were eligible for enrollment if they met the following criteria: (1) Aged 18-75 years; (2) Diagnosed with AP based on the presence of at least two of three criteria: Characteristic abdominal pain, serum amylase or lipase levels greater than three times the upper limit of normal, and characteristic findings on contrast-enhanced computed tomography (CT); (3) Classified as SAP according to the Revised Atlanta Classification, defined by persistent organ failure lasting more than 48 hours and/or presence of local or systemic complications; (4) Admitted within 72 hours of symptom onset; (5) Expected need for EN support for at least 7 days; and (6) Able to provide informed consent or have a legal representative available for consent.
Exclusion criteria: Patients were excluded if they met any of the following criteria: (1) A previous episode of AP within the past 6 months; (2) Known chronic pancreatitis with pancreatic exocrine insufficiency; (3) Malignant disease of the pancreas or other organs with an expected survival of less than 6 months; (4) Pregnancy or lactation; (5) Contraindications to EN including complete bowel obstruction, intestinal perforation, or active gastrointestinal bleeding; (6) A requirement for immediate surgical intervention; (7) Pre-existing severe liver disease with Child-Pugh class C cirrhosis; (8) Chronic kidney disease requiring dialysis; (9) Immunosuppressive therapy including corticosteroids, chemotherapy, or immunomodulatory agents; (10) Inability to place an enteral feeding tube; and (11) Withdrawal of consent or transfer to another facility within 7 days of enrollment.
EEN group: Patients in the EEN group received EN within 48 hours of hospital admission. Following initial fluid resuscitation and hemodynamic stabilization, a nasogastric or nasojejunal feeding tube was placed with fluoroscopic or endoscopic guidance. The tube placement was confirmed radiographically before initiating feeding. A semi-elemental formula with a caloric content of 1.0 kcal/mL was administered to all patients, providing 40 g/L of protein, 35 g/L of fat, and 125 g/L of carbohydrates. Feeding commenced at a rate of 20 mL/hour, incrementally increasing by 20 mL every 6-8 hours as tolerated, aiming for a rate of 80-100 mL/h to achieve 80%-100% of the calculated energy needs within 72-96 hours. Energy requirements were determined using the Harris–Benedict equation, applying a stress factor of 1.3-1.5 for critically ill patients. Gastric residual volumes were assessed every 4-6 hours, and feeding continued unless the residual volume exceeded 250 mL or signs of intolerance emerged. Prokinetic agents such as metoclopramide and erythromycin were administered as needed to enhance gastric emptying.
DEN group: Patients in the DEN group received EN 48 hours after admission, usually starting between 3 and 7 days based on clinical status and oral intake tolerance. The EEN group followed the same feeding formula, tube placement technique, and advancement protocol. Prior to starting EN, patients were given intravenous crystalloid and colloid solutions to ensure proper hydration and hemodynamic stability. Par EN was not given as a standard practice unless EN could not be started within 7 days due to ongoing contraindications.
All patients received standardized supportive care in accordance with institutional protocols and international guidelines. This care encompassed goal-directed fluid resuscitation using lactated Ringer’s solution at a rate of 5-10 mL/kg/hour for the initial 24 hours, with subsequent adjustments guided by hemodynamic parameters, urine output, and clinical assessment. Adequate analgesia was provided through intravenous opioids or patient-controlled analgesia. Prophylactic antibiotics were not routinely administered but were initiated when infection was confirmed, such as in cases of infected pancreatic necrosis as confirmed by imaging or culture, extrapancreatic infections, or signs of systemic sepsis. Patients with organ failure received appropriate organ support, including mechanical ventilation for respiratory failure, vasopressor support for circulatory failure, and renal replacement therapy for acute kidney injury. Abdominal imaging with contrast-enhanced CT was conducted at baseline and repeated at 5-7 days or sooner if clinical deterioration was observed.
Primary outcomes: (1) In-hospital mortality within 30 days of admission; (2) Infectious complications, which were defined as culture-proven infected pancreatic necrosis, bacteremia, pneumonia, urinary tract infection, or catheter-related bloodstream infection; (3) Length of hospital stay, calculated from admission to discharge or death; and (4) Persistent organ failure lasting more than 48 hours, such as respiratory failure necessitating mechanical ventilation, cardiovascular failure necessitating vasopressor support, or renal failure necessitating dialysis.
Secondary outcomes: (1) Feeding intolerance, defined as the inability to achieve 50% of target energy requirements within 7 days due to high gastric residuals, abdominal distension, vomiting, or diarrhea; (2) Time to achieve full nutritional support, defined as the delivery of 80% or more of calculated energy requirements; (3) Local complications including peripancreatic fluid collections, pancreatic pseudocysts, and pancreatic necrosis; (4) The need for interventional procedures including percutaneous drainage, endoscopic necrosectomy, or surgical debridement; (5) Intensive care unit admission and duration; (6) Nutritional status assessed by serum albumin, prealbumin, and transferrin levels at baseline, day 7, and day 14; (7) Inflammatory markers including C-reactive protein (CRP), procalcitonin, and white blood cell (WBC) count; and (8) Intestinal permeability assessed by the lactulose/mannitol ratio at baseline and day 7.
Demographic data, such as age, sex, body mass index, and comorbidities, were documented upon enrollment. The etiology of pancreatitis was ascertained through clinical history, laboratory tests, and imaging findings. Disease severity was evaluated using the Revised Atlanta Classification, Acute Physiology and Chronic Health Evaluation (APACHE II) score, and Balthazar CT severity index. Laboratory parameters, encompassing complete blood count, serum biochemistry, liver function tests, and levels of lipase, amylase, CRP, and procalcitonin, were assessed at admission and monitored sequentially during hospitalization. Nutritional parameters were evaluated at baseline, day 7, and day 14. Patients were monitored daily throughout their hospital stay, and all clinical occurrences were systematically documented in a standardized case report form. Follow-up was maintained until hospital discharge or death.
The sample size was determined based on the primary outcome of infectious complications, aiming to reduce the rate from 40% in the DEN group to 20% in the EEN group. With a two-sided alpha of 0.05 and 80% power, a minimum of 86 patients (43 per group) was necessary. Allowing for a 15% dropout rate, the target enrollment was set at 100 patients. Continuous variables were reported as mean ± SD for normally distributed data and as median with interquartile range for non-normally distributed data. Categorical variables were presented as n (%). Group comparisons utilized the student’s t-test or Mann-Whitney U test for continuous variables and the χ2 test or Fisher’s exact test for categorical variables. Survival analysis employed Kaplan-Meier curves and the log-rank test. Multivariate logistic regression analysis was conducted to identify independent predictors of mortality and complications, adjusting for potential confounders such as age, sex, APACHE II score, and etiology. All statistical tests were two-tailed, and P value below 0.05 were deemed statistically significant. Statistical analyses were carried out using SPSS version 26.0 software.
In total, 142 patients with SAP were screened during the study period. Forty-two patients were excluded due to failure to meet the inclusion criteria, refusal to participate, or the presence of exclusion criteria. The final cohort consisted of 100 patients, with 52 and 48 patients in the EEN and DEN groups, respectively. The baseline demographic and clinical characteristics are detailed in Table 1. The mean age was 48.3 ± 14.2 years in the EEN group and 50.1 ± 13.8 years in the DEN group (P = 0.512). A male predominance was noted in both groups, with 65.4% and 62.5% of the patients, respectively (P = 0.756). The most common etiology was biliary pancreatitis (46.2% in the EEN group vs 43.8% in the DEN group), followed by alcohol-induced pancreatitis (32.7% vs 35.4%). There were no significant differences between the groups in terms of comorbidities, including diabetes mellitus, hypertension, hyperlipidemia, or coronary artery disease. Severity scores, such as the APACHE II score and Balthazar CT severity index, were similar between the groups, indicating adequate matching at baseline. In the DEN group, EN was initiated at a median of 4.2 days (interquartile range: 3.5-5.8 days) after admission.
| Variable | EEN group (n = 52) | DEN group (n = 48) | P value |
| Age (years), mean ± SD | 48.3 ± 14.2 | 50.1 ± 13.8 | 0.512 |
| Male gender | 34 (65.4) | 30 (62.5) | 0.756 |
| BMI (kg/m²), mean ± SD | 24.8 ± 3.6 | 25.3 ± 3.9 | 0.493 |
| Etiology | 0.897 | ||
| Biliary | 24 (46.2) | 21 (43.8) | |
| Alcoholic | 17 (32.7) | 17 (35.4) | |
| Hypertriglyceridemia | 6 (11.5) | 5 (10.4) | |
| Idiopathic | 5 (9.6) | 5 (10.4) | |
| Comorbidities | |||
| Diabetes mellitus | 12 (23.1) | 10 (20.8) | 0.776 |
| Hypertension | 18 (34.6) | 16 (33.3) | 0.889 |
| Hyperlipidemia | 9 (17.3) | 8 (16.7) | 0.930 |
| APACHE II score, mean ± SD | 12.4 ± 3.8 | 12.8 ± 4.1 | 0.603 |
| Balthazar CT severity index, mean ± SD | 6.2 ± 1.8 | 6.5 ± 1.9 | 0.415 |
| WBC count (× 109/L), mean ± SD | 14.2 ± 4.3 | 14.8 ± 4.6 | 0.499 |
| Serum amylase (U/L), mean ± SD | 862 ± 346 | 894 ± 372 | 0.651 |
| C-reactive protein (mg/L), mean ± SD | 186 ± 72 | 192 ± 78 | 0.684 |
Table 2 summarizes the primary outcomes in the EEN and DEN groups. The EEN group showed significantly lower in-hospital mortality than the DEN group [3 patients (5.8%) vs 9 patients (18.8%), P = 0.038]. The incidence of infectious complications was notably reduced in the EEN group, with nine patients (17.3%) developing infections compared to 18 patients (37.5%) in the DEN group (P = 0.019). The specific types of infectious complications are detailed in Table 2; infected pancreatic necrosis was the most common infection in both groups, but occurred less frequently in the EEN group (7.7% vs 22.9%, P = 0.028). The mean length of hospital stay was significantly shorter in the EEN group (18.6 ± 6.2 days vs 26.4 ± 8.5 days, P < 0.001). Persistent organ failure occurred in 12 patients (23.1%) in the EEN group compared with 22 patients (45.8%) in the DEN group (P = 0.014), representing a 49.6% relative risk reduction with early feeding.
| Outcome | EEN group (n = 52) | DEN group (n = 48) | P value |
| Mortality | 3 (5.8)a | 9 (18.8)b | 0.038 |
| Infectious complications | 9 (17.3)a | 18 (37.5)b | 0.019 |
| Infected pancreatic necrosis | 4 (7.7)a | 11 (22.9)b | 0.028 |
| Bacteremia | 2 (3.8) | 4 (8.3) | 0.335 |
| Pneumonia | 2 (3.8) | 5 (10.4) | 0.191 |
| Urinary tract infection | 1 (1.9) | 2 (4.2) | 0.515 |
| Length of hospital stay (days) | 18.6 ± 6.2a | 26.4 ± 8.5b | < 0.001 |
| Persistent organ failure | 12 (23.1)a | 22 (45.8)b | 0.014 |
| Respiratory failure | 6 (11.5)a | 14 (29.2)b | 0.022 |
| Cardiovascular failure | 5 (9.6)a | 12 (25.0)b | 0.036 |
| Renal failure | 4 (7.7) | 8 (16.7) | 0.162 |
| ICU admission | 18 (34.6) | 24 (50.0) | 0.113 |
| ICU length of stay (days) | 6.8 ± 3.4a | 10.2 ± 5.1b | 0.006 |
The secondary outcomes and nutritional parameters are detailed in Table 3. Feeding intolerance rates were similar between the groups, with seven patients (13.5%) in the EEN group and eight patients (16.7%) in the DEN group experiencing intolerance (P = 0.645). The time to achieve full nutritional support was significantly shorter in the EEN group (4.2 ± 1.6 days vs 8.5 ± 2.8 days, P < 0.001). Local complications, such as peripancreatic fluid collection and pancreatic necrosis, occurred at comparable frequencies in both groups. However, the requirement for interventional procedures was lower in the EEN group, with eight patients (15.4%) needing percutaneous drainage or surgical intervention compared to 16 patients (33.3%) in the DEN group (P = 0.031). Nutritional status, as indicated by serum albumin and prealbumin levels, was better maintained in the EEN group on days 7 and 14 of hospitalization. Albumin levels on day 14 were 33.2 ± 4.8 g/L in the EEN group compared to 28.6 ± 5.4 g/L in the DEN group (P < 0.001). Prealbumin levels exhibited a similar pattern, with consistently higher values in the EEN group throughout the study period.
| Outcome | EEN group (n = 52) | DEN group (n = 48) | P value |
| Feeding intolerance | 7 (13.5) | 8 (16.7) | 0.645 |
| Time to full nutrition (days) | 4.2 ± 1.6a | 8.5 ± 2.8b | < 0.001 |
| Local complications | |||
| Pancreatic necrosis | 16 (30.8) | 18 (37.5) | 0.473 |
| Peripancreatic fluid collections | 22 (42.3) | 24 (50.0) | 0.435 |
| Pancreatic pseudocyst | 5 (9.6) | 7 (14.6) | 0.434 |
| Interventional procedures | 8 (15.4)a | 16 (33.3)b | 0.031 |
| Albumin (g/L) | |||
| Baseline | 36.4 ± 5.2 | 35.8 ± 5.6 | 0.579 |
| Day 7 | 34.8 ± 4.6a | 31.2 ± 5.8b | 0.001 |
| Day 14 | 33.2 ± 4.8a | 28.6 ± 5.4b | < 0.001 |
| Prealbumin (mg/L) | |||
| Baseline | 186 ± 42 | 182 ± 46 | 0.646 |
| Day 7 | 168 ± 38a | 142 ± 44b | 0.002 |
| Day 14 | 172 ± 36a | 136 ± 48b | < 0.001 |
| C-reactive protein day 7 (mg/L) | 82 ± 34a | 126 ± 52b | < 0.001 |
| Procalcitonin day 7 (ng/mL) | 1.8 ± 0.9a | 3.6 ± 1.8b | < 0.001 |
Table 4 presents markers of intestinal permeability and inflammation. The lactulose/mannitol ratio, a validated indicator of intestinal permeability, was notably higher at baseline in both groups compared to normal values, confirming the presence of gut barrier dysfunction. By day 7, the EEN group exhibited significantly lower lactulose/mannitol ratios compared to the DEN group (0.082 ± 0.028 vs 0.124 ± 0.042, P < 0.001), indicating a quicker recovery of intestinal barrier function. Serum levels of diamine oxidase, another marker of intestinal mucosal damage, followed a similar trend with lower values in the EEN group at day 7 (8.6 ± 3.2 U/L vs 12.4 ± 4.8 U/L, P < 0.001). Inflammatory markers, such as CRP and procalcitonin, decreased more rapidly in the EEN group, reaching significantly lower levels by day 7 of hospitalization. The WBC count normalized faster in the EEN group, with mean values of 9.2 ± 2.8 × 109/L at day 7 compared to 12.6 ± 4.2 × 109/L in the DEN group (P < 0.001).
| Parameter | EEN group (n = 52) | DEN group (n = 48) | P value |
| Lactulose/mannitol ratio | |||
| Baseline | 0.186 ± 0.062 | 0.192 ± 0.068 | 0.641 |
| Day 7 | 0.082 ± 0.028a | 0.124 ± 0.042b | < 0.001 |
| Diamine oxidase (U/L) | |||
| Baseline | 18.4 ± 5.6 | 19.2 ± 6.2 | 0.509 |
| Day 7 | 8.6 ± 3.2a | 12.4 ± 4.8b | < 0.001 |
| C-reactive protein (mg/L) | |||
| Baseline | 186 ± 72 | 192 ± 78 | 0.684 |
| Day 3 | 142 ± 56a | 178 ± 68b | 0.004 |
| Day 7 | 82 ± 34a | 126 ± 52b | < 0.001 |
| Procalcitonin (ng/mL) | |||
| Baseline | 4.8 ± 2.2 | 5.2 ± 2.6 | 0.414 |
| Day 7 | 1.8 ± 0.9a | 3.6 ± 1.8b | < 0.001 |
| WBC count (× 109/L) | |||
| Baseline | 14.2 ± 4.3 | 14.8 ± 4.6 | 0.499 |
| Day 7 | 9.2 ± 2.8a | 12.6 ± 4.2b | < 0.001 |
| Endotoxin (EU/mL) | |||
| Baseline | 0.42 ± 0.16 | 0.45 ± 0.18 | 0.384 |
| Day 7 | 0.18 ± 0.08a | 0.32 ± 0.14b | < 0.001 |
Table 5 presents results categorized by the etiology of pancreatitis. In patients with biliary pancreatitis, the EEN group exhibited significantly lower mortality (4.2% vs 19.0%, P = 0.041) and a shorter hospital stay (17.2 ± 5.8 days vs 24.6 ± 7.9 days, P = 0.002) compared to the DEN group. Similar advantages were noted in alcohol-induced pancreatitis, with decreased mortality (5.9% vs 17.6%, P = 0.042) and fewer infectious complications (11.8% vs 35.3%, P = 0.016) in the EEN group. Although the sample size was smaller for hypertriglyceridemia-induced pancreatitis, trends favored the EEN group for most outcomes. Subgroup analysis indicated that the positive effects of EEN were consistent across various etiologies, with no significant interaction between etiology and treatment effect (P for interaction = 0.682).
| Etiology and outcome | EEN group | DEN group | P value |
| Biliary pancreatitis | n = 24 | n = 21 | |
| Mortality | 1 (4.2)a | 4 (19.0)b | 0.041 |
| Infectious complications | 4 (16.7)a | 8 (38.1)b | 0.039 |
| Hospital stay (days) | 17.2 ± 5.8a | 24.6 ± 7.9b | 0.002 |
| Organ failure | 5 (20.8)a | 10 (47.6)b | 0.038 |
| Alcoholic pancreatitis | n = 17 | n = 17 | |
| Mortality | 1 (5.9)a | 3 (17.6)b | 0.042 |
| Infectious complications | 2 (11.8)a | 6 (35.3)b | 0.016 |
| Hospital stay (days) | 19.8 ± 6.4a | 27.2 ± 8.8b | 0.006 |
| Organ failure | 4 (23.5) | 7 (41.2) | 0.274 |
| Hypertriglyceridemia | n = 6 | n = 5 | |
| Mortality | 0 (0.0) | 1 (20.0) | 0.455 |
| Infectious complications | 1 (16.7) | 2 (40.0) | 0.524 |
| Hospital stay (days) | 20.5 ± 7.2 | 28.8 ± 9.6 | 0.178 |
| Organ failure | 2 (33.3) | 3 (60.0) | 0.524 |
| Idiopathic pancreatitis | n = 5 | n = 5 | |
| Mortality | 1 (20.0) | 1 (20.0) | 1.000 |
| Infectious complications | 2 (40.0) | 2 (40.0) | 1.000 |
| Hospital stay (days) | 18.6 ± 6.8 | 26.4 ± 10.2 | 0.246 |
| Organ failure | 1 (20.0) | 2 (40.0) | 0.524 |
Table 6 presents the results of a multivariate logistic regression analysis that identified the independent predictors of mortality. After adjusting for age, gender, APACHE II score, etiology, and the presence of organ failure, DEN remained an independent risk factor for mortality [odds ratio (OR) 3.84, 95%CI: 1.12-13.16, P = 0.032]. Other significant predictors included an APACHE II score greater than 15 (OR 5.23, 95%CI: 1.68-16.28, P = 0.004), the presence of organ failure at admission (OR 4.76, 95%CI: 1.42-15.94, P = 0.011), and age greater than 60 years (OR 2.98, 95%CI: 0.94-9.45, P = 0.064). The model demonstrated good discriminatory ability with an area under the receiver operating characteristic curve of 0.846 (95%CI: 0.764-0.928).
| Variable | Odds ratio | 95%CI | P value |
| Delayed enteral nutrition | 3.84 | 1.12-13.16 | 0.032 |
| APACHE II score > 15 | 5.23 | 1.68-16.28 | 0.004 |
| Organ failure at admission | 4.76 | 1.42-15.94 | 0.011 |
| Age > 60 years | 2.98 | 0.94-9.45 | 0.064 |
| Male gender | 1.42 | 0.48-4.21 | 0.527 |
| Biliary etiology | 0.86 | 0.28-2.64 | 0.793 |
| Alcoholic etiology | 1.12 | 0.34-3.68 | 0.853 |
| BMI > 30 kg/m2 | 1.76 | 0.52-5.93 | 0.365 |
| Diabetes mellitus | 1.58 | 0.48-5.21 | 0.451 |
| Pancreatic necrosis > 30% | 2.34 | 0.76-7.21 | 0.138 |
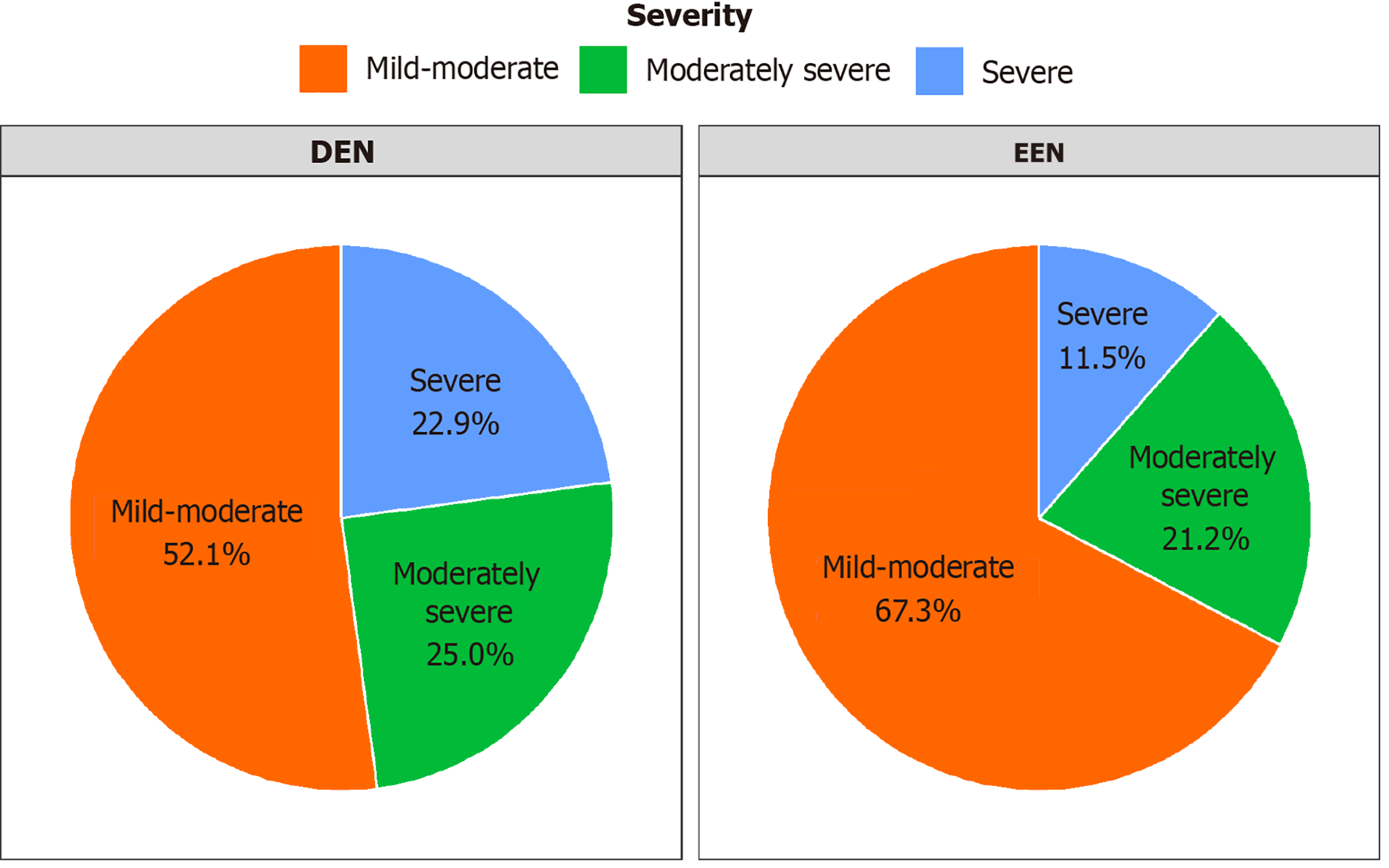
Figure 1 illustrates the distribution of SAP severity according to the Revised Atlanta Classification for both groups. The proportion of mild-to-moderate SAP was higher in the EEN group than in the DEN group, while severe SAP was more prevalent in the DEN group. This distribution pattern correlated with the observed differences in the clinical outcomes between the groups.
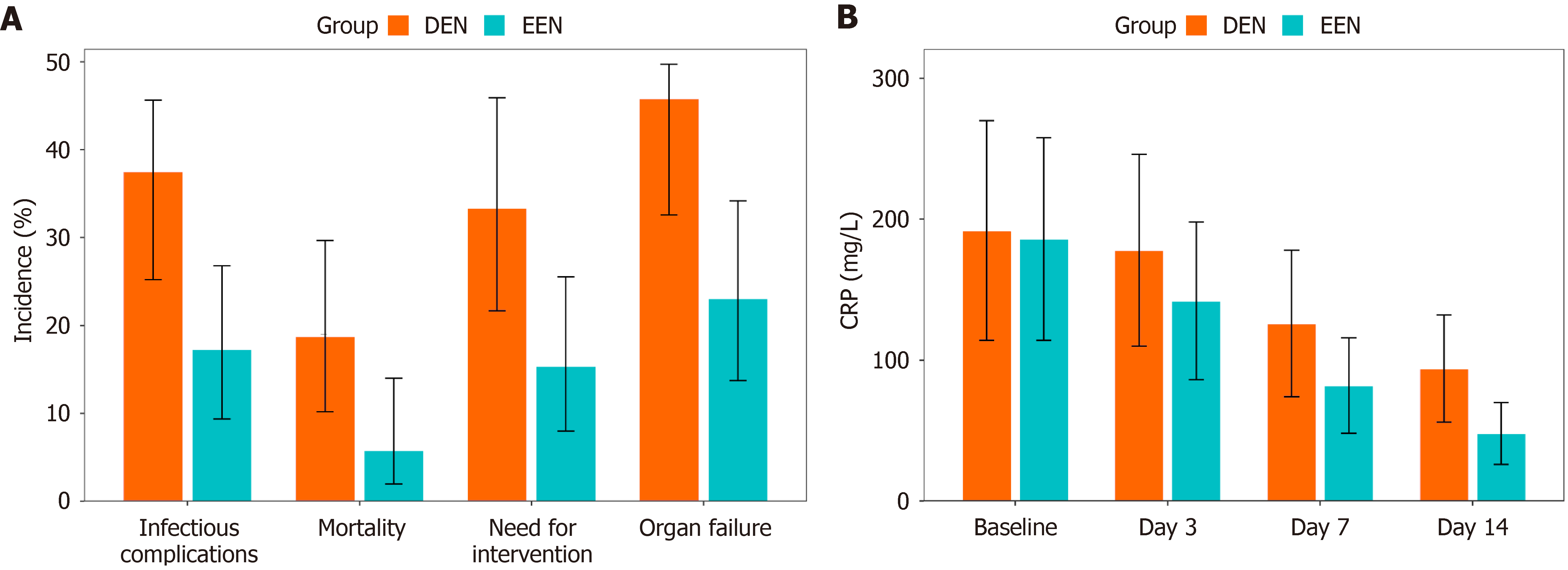
Figure 2A shows a bar graph comparing the incidence of major complications between the two groups. The EEN group exhibited consistently lower rates of all complication categories, with the most significant variances seen in infectious complications and organ failure. In particular, infected pancreatic necrosis was present in 7.7% of patients in the EEN group compared to 22.9% of patients in the DEN group, indicating a 66.4% decrease in relative risk.
Figure 2B illustrates the temporal progression of CRP levels over the initial 14 days of hospitalization. Both groups exhibited an initial increase in CRP; however, the EEN group displayed a swifter and more marked decrease, maintaining notably lower levels starting from day 3. This pattern suggests more effective control of systemic inflammation in patients who received prompt nutritional support.
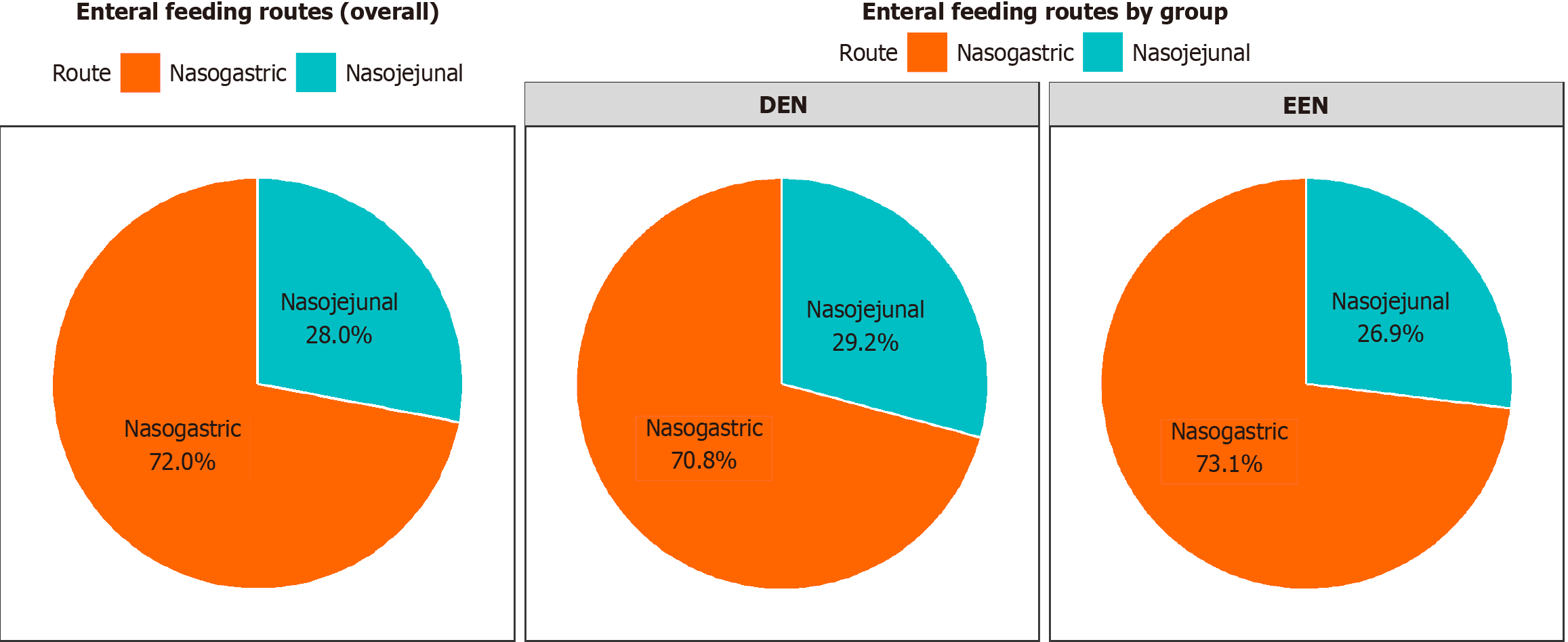
Figure 3 shows a pie chart depicting the distribution of feeding routes utilized in this study. Nasogastric feeding was the primary route in both groups, representing 73.1% of patients in the EEN group and 70.8% of patients in the DEN group. Nasojejunal feeding was employed in 26.9% and 29.2% of patients in the EEN and DEN groups, respectively, with no notable variance in the distribution of feeding routes between the groups (P = 0.791).
This prospective cohort study demonstrated that EEN, started within 48 hours of admission, significantly enhanced clinical outcomes in patients with SAP. The key findings revealed significant decreases in mortality, infectious complications, and organ failure, as well as a quicker restoration of gut barrier function and improved nutritional status. These results offer strong evidence endorsing the adoption of early feeding protocols in SAP management and add to the increasing literature supporting timely nutritional intervention in critically ill patients.
The observed decrease in mortality from 18.8% in the DEN group to 5.8% in the EEN group signifies a clinically significant improvement consistent with prior meta-analyses investigating EN timing in patients with SAP[16]. Our study’s mortality rates were similar to those documented in recent European cohort studies, which showed hospital mortality rates ranging from 5%-20% in SAP patients, depending on disease severity and center experience[17]. The survival advantage linked with EEN persisted even after adjusting for baseline disease severity, comorbidities, and other potential confounders in multivariate analysis, indicating an independent protective effect of early feeding. The mechanism behind this mortality reduction likely involves various pathways, such as maintaining gut barrier integrity, reducing the systemic inflammatory response, and preventing infectious complications, which are the primary causes of late mortality in SAP[18].
The 53.6% relative reduction in infectious complications observed in the EEN group is a significant finding, given the established role of infected pancreatic necrosis as a major determinant of adverse outcomes in SAP. The protective effects of EEN against infection can be attributed to multiple mechanisms related to gut barrier function. During SAP, intestinal permeability increases significantly within the first 72 hours due to intestinal ischemia, oxidative stress, and the disruption of tight junction proteins[19]. Gut barrier failure facilitates the translocation of enteric bacteria from the intestinal lumen to the pancreatic necrotic tissue via lymphatic or hematogenous routes[20]. By initiating EN early, luminal nutrients directly nourish the intestinal epithelial cells, maintain mucosal integrity, preserve gut-associated lymphoid tissue function, and prevent microbial dysbiosis, which characterizes SAP[21].
Our study presents direct evidence of enhanced gut barrier function in the EEN group by assessing validated biomarkers, such as the lactulose/mannitol ratio and diamine oxidase levels. The significantly lower lactulose/mannitol ratio recorded on day 7 in patients in the EEN group signifies a quicker restoration of intestinal permeability compared to delayed feeding. This result aligns with prior studies indicating that EEN sustains the integrity of intestinal epithelial tight junctions and diminishes paracellular permeability in experimental models and critical illness clinical trials[22]. The decrease in serum endotoxin and diamine oxidase concentrations further validates that EEN effectively alleviates in
The differential effects of EEN on organ failure represent another clinically relevant finding. Persistent organ failure was less common in the EEN group compared to the DEN group, with notable benefits observed for respiratory and cardiovascular failure. This observation is consistent with the understanding that gut-derived inflammatory mediators and bacterial products play a significant role in the systemic inflammatory response syndrome and multiple organ dysfunction syndrome, which are typical of severe critical illness[23]. Recent studies employing shotgun metagenomics have shown that SAP is linked to significant changes in gut microbiota composition, including a reduction in beneficial short-chain fatty acid-producing bacteria and an increase in potentially pathogenic species[24]. EEN may help maintain the diversity and metabolic functions of the intestinal microbiome, which are essential for immune homeostasis and the prevention of excessive systemic inflammation.
The similar rates of feeding intolerance between the two groups challenge previous concerns regarding the tolerance of early feeding in critically ill patients with SAP. Only 13.5% of the EEN patients experienced significant feeding in
The nutritional benefits of EEN extend beyond the prevention of complications and include better preservation of lean body mass and faster recovery of nutritional status. The significantly higher albumin and prealbumin levels observed in the EEN group on days 7 and 14 reflected improved protein synthesis and reduced catabolism compared to delayed feeding. Early provision of protein-calorie nutrition helps counteract the hypercatabolic state, maintain skeletal muscle mass, support immune function, and accelerate recovery[26].
Our subgroup analysis, stratified by etiology, indicated that the advantages of EEN were generally consistent across various causes of pancreatitis, such as biliary, alcoholic, and hypertriglyceridemic etiologies. This discovery implies that the protective mechanisms of early feeding function autonomously from the triggering factors and are mainly associated with the systemic inflammatory response and gut barrier dysfunction common to all forms of SAP[27]. Nonetheless, some variation in treatment effects has been noted, with notably significant benefits observed in biliary and alcoholic pancreatitis in comparison to other etiologies.
Multivariate analysis revealed that DEN emerged as an independent risk factor for mortality, even after adjusting for established predictors such as the APACHE II score and organ failure, providing compelling evidence for the pivotal role of early feeding in enhancing outcomes[28]. The substantial effect size (OR, 3.84) indicates a clinically significant impact that stands out when compared to other therapeutic interventions for SAP. The coherence of benefits across various outcome measures and the biologically plausible pathways connecting nutritional timing to gut barrier function further bolster the argument for a causal relationship between early feeding and improved outcomes, rather than mere con
This study holds significant clinical implications. First, the results support the feasibility and safety of initiating early feeding protocols within 48 hours of admission for patients with SAP. While personalized treatment is crucial and absolute contraindications to EN must be adhered to, our data indicate that concerns regarding early feeding tolerance should not routinely postpone nutritional support in hemodynamically stable patients without specific contraindications. Second, the benefits of EEN were observed without the need for specialized feeding routes, as nasogastric feeding was predominantly successful[29]. This discovery is particularly relevant in settings with limited resources where the placement of nasojejunal tubes via endoscopy or fluoroscopy may not be readily accessible. Third, the rapid decline in inflammatory markers and gut permeability indices in the EEN group implies that the optimal therapeutic window for maximizing the advantages of early feeding occurs within the initial days of hospitalization, underscoring the sig
Our study had several limitations that require consideration. First, the observational cohort design, while enabling pragmatic evaluation in real-world clinical practice, introduces the potential for selection bias and confounding effects, despite our efforts to adjust for measured covariates. Although a multivariate analysis was conducted to control for potential confounders, residual confounding from unmeasured variables could not be entirely excluded. A randomized controlled trial would offer definitive evidence of causality. Second, the study was conducted at a single tertiary referral center with established expertise in managing SAP, potentially limiting its generalizability to community hospitals and resource-limited settings. Third, our definition of EEN as feeding within 48 hours encompasses a relatively broad timeframe. Future studies should explore more detailed timing considerations. Fourth, we did not systematically assess the long-term outcomes beyond hospital discharge, including quality of life and pancreatic function. Subsequent studies should integrate an extended follow-up period to assess the durability of the benefits of early nutritional support.
This prospective cohort study showed that EEN initiated within 48 hours of admission significantly enhanced clinical outcomes in patients with SAP. Compared to delayed feeding, EEN was associated with reduced mortality, fewer infectious complications, decreased organ failure, shorter hospital stays, and faster recovery of gut barrier function. These advantages were achieved with acceptable tolerance and without increased complications, thus supporting the safety of early feeding protocols. These results offer compelling evidence for incorporating EEN as a standard component of SAP management. Future multicenter, large-sample randomized controlled trials are needed to provide higher-level evidence and validate these findings.
| 1. | Petrov MS, Yadav D. Global epidemiology and holistic prevention of pancreatitis. Nat Rev Gastroenterol Hepatol. 2019;16:175-184. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 737] [Cited by in RCA: 618] [Article Influence: 88.3] [Reference Citation Analysis (1)] |
| 2. | Banks PA, Bollen TL, Dervenis C, Gooszen HG, Johnson CD, Sarr MG, Tsiotos GG, Vege SS; Acute Pancreatitis Classification Working Group. Classification of acute pancreatitis--2012: revision of the Atlanta classification and definitions by international consensus. Gut. 2013;62:102-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5667] [Cited by in RCA: 4804] [Article Influence: 369.5] [Reference Citation Analysis (7)] |
| 3. | Czapári D, Váradi A, Farkas N, Nyári G, Márta K, Váncsa S, Nagy R, Teutsch B, Bunduc S, Erőss B, Czakó L, Vincze Á, Izbéki F, Papp M, Merkely B, Szentesi A, Hegyi P; Hungarian Pancreatic Study Group. Detailed Characteristics of Post-discharge Mortality in Acute Pancreatitis. Gastroenterology. 2023;165:682-695. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 48] [Article Influence: 16.0] [Reference Citation Analysis (1)] |
| 4. | Li XY, He C, Zhu Y, Lu NH. Role of gut microbiota on intestinal barrier function in acute pancreatitis. World J Gastroenterol. 2020;26:2187-2193. [PubMed] [DOI] [Full Text] |
| 5. | Zhang C, Li G, Lu T, Liu L, Sui Y, Bai R, Li L, Sun B. The Interaction of Microbiome and Pancreas in Acute Pancreatitis. Biomolecules. 2023;14:59. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 37] [Article Influence: 12.3] [Reference Citation Analysis (1)] |
| 6. | Agarwal S, Goswami P, Poudel S, Gunjan D, Singh N, Yadav R, Kumar U, Pandey G, Saraya A. Acute pancreatitis is characterized by generalized intestinal barrier dysfunction in early stage. Pancreatology. 2023;23:9-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 21] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 7. | Singh N, Sonika U, Moka P, Sharma B, Sachdev V, Mishra SK, Upadhyay AD, Saraya A. Association of endotoxaemia & gut permeability with complications of acute pancreatitis: Secondary analysis of data. Indian J Med Res. 2019;149:763-770. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 8. | Beyer G, Hoffmeister A, Lorenz P, Lynen P, Lerch MM, Mayerle J. Clinical Practice Guideline—Acute and Chronic Pancreatitis. Dtsch Arztebl Int. 2022;119:495-501. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 28] [Article Influence: 7.0] [Reference Citation Analysis (2)] |
| 9. | Lakananurak N, Gramlich L. Nutrition management in acute pancreatitis: Clinical practice consideration. World J Clin Cases. 2020;8:1561-1573. [PubMed] [DOI] [Full Text] |
| 10. | Al-Omran M, Albalawi ZH, Tashkandi MF, Al-Ansary LA. Enteral versus parenteral nutrition for acute pancreatitis. Cochrane Database Syst Rev. 2010;2010:CD002837. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 118] [Article Influence: 7.4] [Reference Citation Analysis (1)] |
| 11. | Guo YB, Liu Y, Ma J, Cai Y, Jiang XM, Zhang H. Effect of early enteral nutrition support for the management of acute severe pancreatitis: A protocol of systematic review. Medicine (Baltimore). 2020;99:e21569. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 12. | McClave SA, Martindale RG, Vanek VW, McCarthy M, Roberts P, Taylor B, Ochoa JB, Napolitano L, Cresci G; A. S.P.E.N. Board of Directors; American College of Critical Care Medicine; Society of Critical Care Medicine. Guidelines for the Provision and Assessment of Nutrition Support Therapy in the Adult Critically Ill Patient: Society of Critical Care Medicine (SCCM) and American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.). JPEN J Parenter Enteral Nutr. 2009;33:277-316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1378] [Cited by in RCA: 925] [Article Influence: 54.4] [Reference Citation Analysis (1)] |
| 13. | Li JY, Yu T, Chen GC, Yuan YH, Zhong W, Zhao LN, Chen QK. Enteral nutrition within 48 hours of admission improves clinical outcomes of acute pancreatitis by reducing complications: a meta-analysis. PLoS One. 2013;8:e64926. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 80] [Cited by in RCA: 73] [Article Influence: 5.6] [Reference Citation Analysis (3)] |
| 14. | Li X, Ma F, Jia K. Early enteral nutrition within 24 hours or between 24 and 72 hours for acute pancreatitis: evidence based on 12 RCTs. Med Sci Monit. 2014;20:2327-2335. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 26] [Article Influence: 2.2] [Reference Citation Analysis (2)] |
| 15. | Working Group IAP/APA Acute Pancreatitis Guidelines. IAP/APA evidence-based guidelines for the management of acute pancreatitis. Pancreatology. 2013;13:e1-15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1350] [Cited by in RCA: 1099] [Article Influence: 84.5] [Reference Citation Analysis (7)] |
| 16. | de-Madaria E, Buxbaum JL, Maisonneuve P, García García de Paredes A, Zapater P, Guilabert L, Vaillo-Rocamora A, Rodríguez-Gandía MÁ, Donate-Ortega J, Lozada-Hernández EE, Collazo Moreno AJR, Lira-Aguilar A, Llovet LP, Mehta R, Tandel R, Navarro P, Sánchez-Pardo AM, Sánchez-Marin C, Cobreros M, Fernández-Cabrera I, Casals-Seoane F, Casas Deza D, Lauret-Braña E, Martí-Marqués E, Camacho-Montaño LM, Ubieto V, Ganuza M, Bolado F; ERICA Consortium. Aggressive or Moderate Fluid Resuscitation in Acute Pancreatitis. N Engl J Med. 2022;387:989-1000. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 262] [Cited by in RCA: 193] [Article Influence: 48.3] [Reference Citation Analysis (2)] |
| 17. | Wolbrink DRJ, van de Poll MCG, Termorshuizen F, de Keizer NF, van der Horst ICC, Schnabel R, Dejong CHC, van Santvoort HC, Besselink MG, van Goor H, Bouwense SAW, van Bussel BCT; Dutch Pancreatitis Study Group and the Dutch National Intensive Care Evaluation (NICE) Collaborators. Trends in Early and Late Mortality in Patients With Severe Acute Pancreatitis Admitted to ICUs: A Nationwide Cohort Study. Crit Care Med. 2022;50:1513-1521. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 25] [Reference Citation Analysis (0)] |
| 18. | Leppäniemi A, Tolonen M, Tarasconi A, Segovia-Lohse H, Gamberini E, Kirkpatrick AW, Ball CG, Parry N, Sartelli M, Wolbrink D, van Goor H, Baiocchi G, Ansaloni L, Biffl W, Coccolini F, Di Saverio S, Kluger Y, Moore E, Catena F. 2019 WSES guidelines for the management of severe acute pancreatitis. World J Emerg Surg. 2019;14:27. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 661] [Cited by in RCA: 502] [Article Influence: 71.7] [Reference Citation Analysis (2)] |
| 19. | Ammori BJ, Leeder PC, King RF, Barclay GR, Martin IG, Larvin M, McMahon MJ. Early increase in intestinal permeability in patients with severe acute pancreatitis: correlation with endotoxemia, organ failure, and mortality. J Gastrointest Surg. 1999;3:252-262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 260] [Cited by in RCA: 231] [Article Influence: 8.6] [Reference Citation Analysis (5)] |
| 20. | Hollemans RA, Timmerhuis HC, Besselink MG, Bouwense SAW, Bruno M, van Duijvendijk P, van Geenen EJ, Hadithi M, Hofker S, Van-Hooft JE, Kager LM, Manusama ER, Poley JW, Quispel R, Römkens T, van der Schelling GP, Schwartz MP, Spanier BWM, Stommel M, Tan A, Venneman NG, Vleggaar F, van Wanrooij RLJ, Bollen TL, Voermans RP, Verdonk RC, van Santvoort HC; Dutch Pancreatitis Study Group. Long-term follow-up study of necrotising pancreatitis: interventions, complications and quality of life. Gut. 2024;73:787-796. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 21. | Liu J, Yan Q, Li S, Jiao J, Hao Y, Zhang G, Zhang Q, Luo F, Zhang Y, Lv Q, Zhang W, Zhang A, Song H, Xin Y, Ma Y, Owusu L, Ma X, Yin P, Shang D. Integrative metagenomic and metabolomic analyses reveal the potential of gut microbiota to exacerbate acute pancreatitis. NPJ Biofilms Microbiomes. 2024;10:29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 31] [Reference Citation Analysis (1)] |
| 22. | Liu F, Xiao Z, Zeng H, Li J, Ai F, Qi J. Early enteral nutrition with fructooligosaccharides improves prognosis in severe acute pancreatitis. Sci Rep. 2025;15:5267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (5)] |
| 23. | Zheng Z, Ding YX, Qu YX, Cao F, Li F. A narrative review of acute pancreatitis and its diagnosis, pathogenetic mechanism, and management. Ann Transl Med. 2021;9:69. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 48] [Article Influence: 9.6] [Reference Citation Analysis (1)] |
| 24. | Yu S, Xiong Y, Fu Y, Chen G, Zhu H, Mo X, Wu D, Xu J. Shotgun metagenomics reveals significant gut microbiome features in different grades of acute pancreatitis. Microb Pathog. 2021;154:104849. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 25. | O'keefe SJ, Graham T, Coté GA, Whitcomb DC, Evans A, Soni D. Early Enteral Feeding in Severe Acute Pancreatitis: A Randomized Clinical Trial Between Gastric vs Distal Jejunal Feeding. Tech Innov Gastrointest Endosc. 2023;25:337-346. [DOI] [Full Text] |
| 26. | Cederholm T, Jensen GL, Correia MITD, Gonzalez MC, Fukushima R, Higashiguchi T, Baptista G, Barazzoni R, Blaauw R, Coats AJS, Crivelli AN, Evans DC, Gramlich L, Fuchs-Tarlovsky V, Keller H, Llido L, Malone A, Mogensen KM, Morley JE, Muscaritoli M, Nyulasi I, Pirlich M, Pisprasert V, de van der Schueren MAE, Siltharm S, Singer P, Tappenden K, Velasco N, Waitzberg D, Yamwong P, Yu J, Van Gossum A, Compher C; GLIM Core Leadership Committee, GLIM Working Group. GLIM criteria for the diagnosis of malnutrition - A consensus report from the global clinical nutrition community. J Cachexia Sarcopenia Muscle. 2019;10:207-217. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 795] [Cited by in RCA: 746] [Article Influence: 106.6] [Reference Citation Analysis (1)] |
| 27. | Hidalgo NJ, Pando E, Mata R, Fernandes N, Villasante S, Barros M, Herms D, Blanco L, Balsells J, Charco R. Impact of comorbidities on hospital mortality in patients with acute pancreatitis: a population-based study of 110,021 patients. BMC Gastroenterol. 2023;23:81. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 28. | Wiley MB, Mehrotra K, Bauer J, Yazici C, Bialkowska AB, Jung B. Acute Pancreatitis: Current Clinical Approaches, Molecular Pathophysiology, and Potential Therapeutics. Pancreas. 2023;52:e335-e343. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 20] [Article Influence: 6.7] [Reference Citation Analysis (1)] |
| 29. | Singh N, Sharma B, Sharma M, Sachdev V, Bhardwaj P, Mani K, Joshi YK, Saraya A. Evaluation of early enteral feeding through nasogastric and nasojejunal tube in severe acute pancreatitis: a noninferiority randomized controlled trial. Pancreas. 2012;41:153-159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 103] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 30. | Wereszczynska-Siemiatkowska U, Swidnicka-Siergiejko A, Siemiatkowski A, Dabrowski A. Early enteral nutrition is superior to delayed enteral nutrition for the prevention of infected necrosis and mortality in acute pancreatitis. Pancreas. 2013;42:640-646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 68] [Article Influence: 5.2] [Reference Citation Analysis (0)] |