Published online Mar 27, 2026. doi: 10.4240/wjgs.v18.i3.115910
Revised: December 26, 2025
Accepted: January 8, 2026
Published online: March 27, 2026
Processing time: 119 Days and 3.2 Hours
Gastrointestinal tumors are among the most common malignant tumors in older people. Surgical treatment for these tumors is characterized by extensive scope, severe trauma, and prolonged during of anesthesia, and it is considered to pose a high risk for post-operative cognitive dysfunction (POCD).
To retrospectively analyze the medical records of older patients with depression who developed POCD after surgery for a gastrointestinal tumor.
A total of 95 older patients with depression who underwent surgery. For a gastrointestinal tumor at the Second Affiliated Hospital of Soochow University between January 2021 and December 2024, were retrospectively selected. Patients were assigned to the POCD group or the non-POCD group based on the post-surgical presence of POCD. Indicators, including general data, peri-operative data, depressive status, cognitive function, and laboratory test results, were col
The incidence of POCD at seven days post-surgery was 36.84%. Univariate and multivariate logistic regression analyses showed that advanced age, high pre-operative Hamilton Depression Rating Scale (HAMD) score, lengthy duration of surgery, intra-operative administration of anticholinergic drugs, and high post-operative interleukin-6 (IL-6) change rate, were independent risk factors for POCD. Mediating effect analysis revealed that the pre-operative HAMD score had a significant positive predictive effect on post-operative IL-6, and post-operative IL-6 had a significant negative predictive effect on post-operative Montreal Cognitive Assessment (MoCA) score; the direct effect of HAMD on MoCA and the indirect effect mediated by IL-6 were significant, with the indirect effect accounting for 19.7% of the total effect. Additionally, the pre-operative HAMD score had a significant negative predictive effect on the post-operative cholinesterase (CHE) level, and the post-operative CHE level had a signi
The incidence of POCD is relatively high in older patients with depression after surgery for a gastrointestinal tumor. Advanced age, pre-operative depression severity, surgical trauma stress, and intra-operative administration of anticholinergic drugs were independent risk factors for POCD. The severity of pre-operative depression can upregulate the systemic inflammatory response, leading to cholinergic system dysfunction and increased risk of POCD.
Core Tip: Data from 95 older patients with depression who had gastrointestinal tumor surgery, were retrospectively analyzed, and there was an incidence of post-operative cognitive dysfunction (POCD) of 36.84%, seven days post-operatively. Advanced age, high pre-operative Hamilton Depression Rating Scale score, long duration of surgery, intra-operative anticholinergics, and high post-operative interleukin-6 (IL-6) change rate were independent risk factors for POCD. Mechanistically, pre-operative depression exacerbated systemic inflammation (via IL-6) and impaired cholinergic function (via reduced cholinesterase), jointly increasing the risk of POCD, with the two mediating effects accounting for 19.7% and 17.2% of the total effect, respectively. These findings provide a basis for the targeted prevention of POCD.
- Citation: Li W, Ye ZY, Zhao JQ. Retrospective analysis cognitive function changes in elderly depressive patients after gastrointestinal tumor surgery and exploration of cholinergic mechanism. World J Gastrointest Surg 2026; 18(3): 115910
- URL: https://www.wjgnet.com/1948-9366/full/v18/i3/115910.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i3.115910
Gastrointestinal tumors are among the most common malignant tumors in older people. Surgical treatment of these tumors is characterized by extensive scope, severe trauma, and prolonged duration of anesthesia, and it is regarded as posing a high-risk for post-operative cognitive dysfunction (POCD)[1,2]. POCD primarily manifests as impairments in multiple cognitive domains, including memory, attention, executive function, and speed of information processing. These impairments can persist for several weeks to months, and they may progress to permanent damage. This not only seriously affects the quality of post-operative recovery, ability for long-term self-care, and survival rate of patients, but it also imposes a heavy care and economic burden on families and society[3,4].
Older patients often have multiple underlying diseases, among which depression is relatively common. Depression is a systemic disease that is accompanied by significant neurobiological changes[5,6]. Studies have shown that patients with depression have various pathophysiological alterations, such as hyperfunction of the hypothalamic-pituitary-adrenal axis, activation of immune-mediated inflammation, and imbalance of neurotransmitter systems[7,8]. When older patients with depression experience major stress, such as surgical trauma, these changes significantly increase the vulnerability of their nervous system, resulting a greater likelihood of developing POCD[9,10].
Currently, there have been relatively few clinical studies on the correlation between depression in older people, surgery for gastrointestinal tumors, POCD, and cholinergic mechanisms. Therefore, in this retrospective study, the risk factors for changes in cognitive function in older patients with depression after surgery for a gastrointestinal tumor, were systematically analyzed, with a focus on exploring the potential mechanism of POCD from the perspective of inflammation-cholinergic system interactions. The aim was to provide a reference for pre-operative assessment, early risk warning, and the development of targeted neuroprotective strategies for high-risk cohorts.
A total of 95 older patients with depression who underwent surgery for a gastrointestinal tumor at the Second Affiliated Hospital of Soochow University between January 2021 and December 2024 were retrospectively selected.
Inclusion criteria: (1) Age ≥ 65 years; (2) Pre-operative diagnosis of depression by a psychiatrist in accordance with the International Classification of Diseases, 11th revision[11]; (3) Pre-operative American Society of Anesthesiologists (ASA) physical status classification of grade I-III; and (4) Complete clinical data, anesthesia records, and cognitive assessment data.
Exclusion criteria: (1) History of other psychiatric or neurological diseases, such as severe pre-operative cognitive impair
Data collection: Relevant clinical data were collected from the Hospital Information System, Anesthesia Information Management System, and Laboratory Information System, including: (1) General data: Age, sex, body mass index, years of education, smoking history, drinking history, ASA classification, and comorbidities; (2) Depression-related indicators: Trained psychiatric nurses assessed the 17-item Hamilton Depression Rating Scale (HAMD) score within one week pre-operatively (a higher score indicates more severe depressive symptoms); the type and duration of antidepressant use (e.g., selective serotonin reuptake inhibitors, serotonin and norepinephrine reuptake inhibitors) were recorded; (3) Tumor and surgery-related indicators: Tumor type, TNM staging, surgical method, duration of surgery, intra-operative blood loss, and intra-operative blood transfusion; (4) Anesthesia-related indicators: Anesthesia method, total dosage of anesthetic drugs, and intra-operative administration of anticholinergic drugs; (5) Post-operative pain intensity: The Visual Analog Scale (VAS) was used to assess the score at the peak of resting pain within 24 hours post-operatively; (6) Post-operative complications: The presence of complications such as infection (pulmonary, incisional) and delirium within seven days post-operatively, was recorded; (7) Laboratory indicators: Fasting venous blood was collected one day pre-operatively and on the first morning, post-operatively, to measure the serum interleukin-6 (IL-6), tumor necrosis factor-alpha
Grouping: Based on the criteria of the International Study of Post-operative Cognitive Dysfunction[12], patients were diagnosed with POCD if the MMSE score, seven days post-operatively, decreased by ≥ 2 points compared with the pre-operative score, or the MoCA score decreased by ≥ 3 points compared with the pre-operative score. Based on this, the patients were assigned to the POCD group or the non-POCD group.
Neuropsychological assessments (MMSE and MoCA) and evaluations of depression (HAMD) were performed by trained psychiatric nurses who were not involved in surgical or anesthesia management. Due to the retrospective nature of the study, complete assessor blinding of peri-operative clinical information could not be guaranteed, which may have introduced measurement bias.
SPSS software (version 26.0) was used for the data analysis. Data conforming to a normal distribution are expressed as mean ± SD and were compared using the independent samples t-test. Data not conforming to a normal distribution are expressed as median (interquartile range) and were compared using the Mann-Whitney U test (test statistic, Z value). Count data are expressed as n (%) and were compared using the χ2 test or Fisher’s exact test.
Indicators with statistical significance in the univariate analysis were included in the multivariate unconditional logi
Mediation analyses were performed using the PROCESS macro (model 4) with a bias-corrected bootstrap approach based on 5000 re-samples. The significance of indirect effects was determined using 95% bootstrap confidence intervals that did not include zero.
Model adequacy was further evaluated using commonly reported fit indices derived from a regression-based path analysis. The mediation models demonstrated acceptable goodness-of-fit, as indicated by a comparative fit index of 0.95, a Tucker-Lewis index of 0.93, a root-mean-square error of approximation of 0.056, and a standardized root mean square residual of 0.041. These values suggest that there is an overall satisfactory fit between the proposed mediation models and the observed data.
Among the 95 patients in this study, 35 were diagnosed with post-operative POCD, with an incidence of 36.8% (35/95). Univariate analysis revealed statistically significant differences (P < 0.05) between the POCD and non-POCD groups for age, pre-operative HAMD score, duration of surgery, intra-operative blood loss, post-operative VAS pain score, post-operative infection rate, percentage of patients administered anticholinergic drugs during surgery, and pre-operative/post-operative levels and change rates of the IL-6, TNF-α, and CHE levels (Table 1).
| Variable | POCD group (n = 35) | Non-POCD group (n = 60) | t/χ2/Z | P value |
| Age (years) | 75.2 ± 4.9 | 70.8 ± 5.6 | -3.892 | < 0.001 |
| Male sex | 18 (51.4) | 34 (56.7) | 0.256 | 0.613 |
| BMI (kg/m2) | 22.8 ± 3.5 | 23.3 ± 3.0 | 0.734 | 0.465 |
| Education (year) | 7.6 ± 3.5 | 9.0 ± 3.9 | 1.822 | 0.072 |
| ASA grade (II/III) | 20/15 | 43/17 | 1.438 | 0.230 |
| Pre-operative HAMD score | 21.1 ± 3.8 | 17.0 ± 3.9 | -5.063 | < 0.001 |
| Pre-operative MMSE score | 26.5 ± 1.6 | 27.0 ± 1.4 | 1.583 | 0.117 |
| Pre-operative MoCA score | 23.8 ± 2.3 | 24.5 ± 1.9 | 1.621 | 0.108 |
| Hypertension | 24 (68.6) | 34 (56.7) | 1.359 | 0.244 |
| Diabetes | 14 (40.0) | 17 (28.3) | 1.441 | 0.230 |
| Tumor site (gastric/colorectal) | 17/18 | 24/36 | 0.383 | 0.536 |
| TNM stage (I-II/III) | 19/16 | 40/20 | 1.186 | 0.276 |
| Laparoscopic approach | 22 (62.9) | 45 (75.0) | 1.655 | 0.198 |
| Duration of surgery (minute) | 205.7 ± 48.1 | 169.2 ± 38.5 | -4.052 | < 0.001 |
| Intra-operative blood loss (mL) | 200 (150, 250) | 120 (80, 180) | -4.125 | < 0.001 |
| Intra-operative anticholinergic use | 17 (48.6) | 11 (18.3) | 9.952 | 0.002 |
| 24-hour post-operative VAS score | 5.1 ± 1.1 | 4.2 ± 1.1 | -3.852 | < 0.001 |
| Post-operative infection | 9 (25.7) | 6 (10.0) | 4.183 | 0.041 |
| Pre-operative IL-6 (pg/mL) | 5.6 [4.1, 7.8] | 4.3 [2.9, 5.7] | -2.891 | 0.004 |
| Post-operative IL-6 (pg/mL) | 42.3 [31.5, 55.2] | 20.1 [15.3, 28.4] | -5.234 | < 0.001 |
| Pre-operative TNF-α (pg/mL) | 7.1 [5.6, 9.3] | 5.7 [4.3, 7.2] | -2.456 | 0.014 |
| Post-operative TNF-α (pg/mL) | 26.8 [19.4, 35.7] | 13.8 [9.6, 19.2] | -4.987 | < 0.001 |
| Pre-operative CHE (U/L) | 6 450 ± 1 380 | 7 080 ± 1 150 | 2.345 | 0.021 |
| Post-operative CHE (U/L) | 4 520 ± 1 280 | 5 820 ± 1 360 | 4.892 | < 0.001 |
| IL-6 change (%) | 655.4 ± 189.3 | 367.4 ± 120.6 | -8.123 | < 0.001 |
| TNF-α change (%) | 277.6 ± 95.8 | 142.1 ± 63.5 | -7.856 | < 0.001 |
| CHE change (%) | -29.8 ± 10.2 | -17.8 ± 6.9 | -6.234 | < 0.001 |
Using the presence of post-operative POCD (0, not present; 1, present) as the dependent variable and variables with P < 0.05 in univariate analysis as independent variables, a multivariate logistic regression model was established. The results indicated that age, pre-operative HAMD score, duration of surgery, intra-operative administration of anticholinergic drugs, and post-operative IL-6 change rate were independent risk factors for POCD in older patients with depression who had surgery for a gastrointestinal tumor (P < 0.05; Tables 2 and 3).
| Independent variable | Coding instruction | Variable type | Reference group/unit |
| Age | Actual value (years) | Continuous | Per 1-year increase |
| Pre-operative HAMD score | Actual value (points) | Continuous | Per 1-point increase |
| Operative time | Actual value (minute) | Continuous | Per 10-minute increase |
| Intra-operative blood loss | Actual value (mL) | Continuous | Per 50-mL increase |
| Intra-operative anticholinergic use | 0 = no, 1 = yes | Binary | Reference: No use (0) |
| 24-hour post-operative VAS score | Actual value (points) | Continuous | Per 1-point increase |
| Post-operative infection | 0 = no, 1 = yes | Binary | Reference: No infection (0) |
| Pre-operative IL-6 | Actual value (pg/mL) | Continuous | Per 1 pg/mL increase |
| Post-operative IL-6 | Actual value (pg/mL) | Continuous | Per 1 pg/mL increase |
| IL-6 change rate | (Post-operative - pre-operative)/pre-operative × 100% | Continuous | Per 10% increase |
| Pre-operative TNF-α | Actual value (pg/mL) | Continuous | Per 1 pg/mL increase |
| Post-operative TNF-α | Actual value (pg/mL) | Continuous | Per 1 pg/mL increase |
| TNF-α change rate | (Post-operative - pre-operative)/pre-operative × 100% | Continuous | Per 10% increase |
| Pre-operative CHE activity | Actual value (U/L) | Continuous | Per 100 U/L increase |
| Post-operative CHE activity | Actual value (U/L) | Continuous | Per 100 U/L increase |
| CHE change rate | (Post-operative - pre-operative)/pre-operative × 100% | Continuous | Per 1% increase |
| Variable (unit increment) | β | SE | Wald χ2 | P value | OR | 95%CI |
| Age (per 1 year) | 0.13 | 0.05 | 6.76 | 0.009 | 1.14 | 1.03-1.26 |
| Pre-operative HAMD score (per 1 point) | 0.18 | 0.06 | 9.00 | 0.003 | 1.20 | 1.07-1.35 |
| Duration of surgery (per 10 minutes) | 0.09 | 0.03 | 9.00 | 0.003 | 1.01 | 1.00-1.02 |
| Intra-operative anticholinergic use (yes vs no) | 1.18 | 0.42 | 7.88 | 0.005 | 3.25 | 1.43-7.38 |
| Post-operative IL-6 changes rate (per 10%) | 0.05 | 0.02 | 6.25 | 0.012 | 1.05 | 1.01-1.09 |
| Intra-operative blood loss (per 50 mL) | 0.08 | 0.05 | 2.56 | 0.110 | 1.08 | 0.98-1.19 |
| 24-hour post-operative VAS score (per 1 point) | 0.12 | 0.08 | 2.25 | 0.134 | 1.13 | 0.96-1.32 |
| Post-operative infection (yes vs no) | 0.65 | 0.48 | 1.83 | 0.176 | 1.92 | 0.75-4.91 |
| Pre-operative IL-6 (per 1 pg/mL) | 0.04 | 0.03 | 1.78 | 0.182 | 1.04 | 0.98-1.10 |
| Post-operative TNF-α change rate (per 10%) | 0.02 | 0.02 | 1.00 | 0.317 | 1.02 | 0.98-1.06 |
| Pre-operative CHE (per 100 U/L) | -0.01 | 0.01 | 1.00 | 0.317 | 0.99 | 0.97-1.01 |
| Post-operative CHE activity (per 100 U/L) | -0.02 | 0.02 | 1.00 | 0.317 | 0.98 | 0.94-1.02 |
| Post-operative TNF-α (per 1 pg/mL) | 0.01 | 0.02 | 0.25 | 0.617 | 1.01 | 0.97-1.05 |
| Pre-operative TNF-α (per 1 pg/mL) | 0.01 | 0.03 | 0.11 | 0.740 | 1.01 | 0.96-1.06 |
| CHE change rate (per 1%) | -0.01 | 0.03 | 0.11 | 0.740 | 0.99 | 0.94-1.04 |
| Constant | -20.85 | 5.23 | 15.89 | < 0.001 | 0.00 | - |
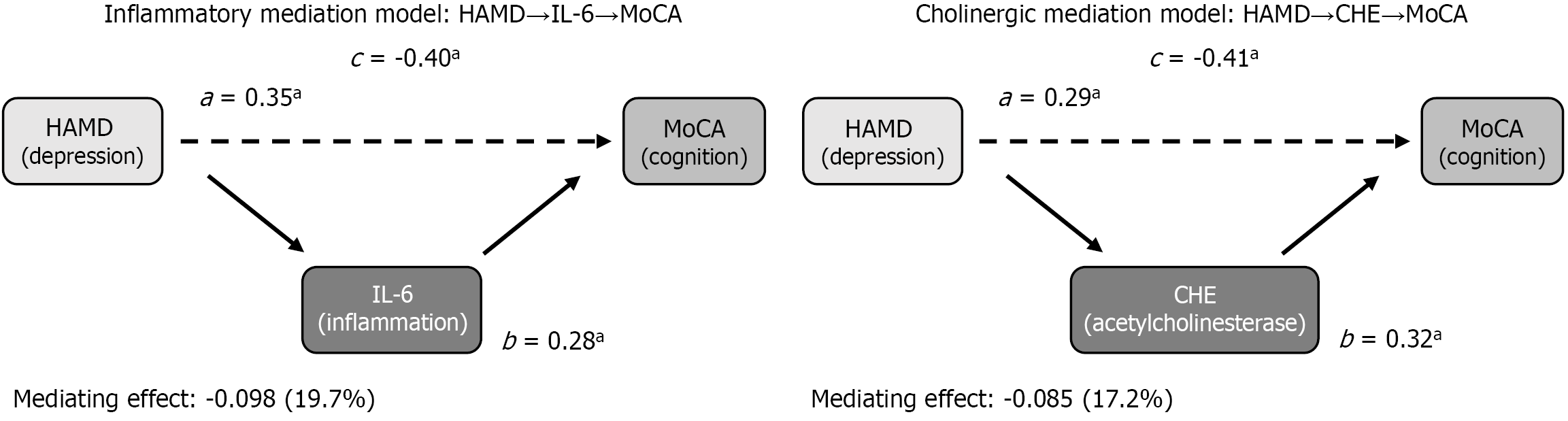
An inflammatory mediation model was constructed with the pre-operative HAMD score as the independent variable (X), post-operative MoCA score as the dependent variable (Y), and post-operative IL-6 level as the mediating variable (M). The results indicated that the pre-operative HAMD score significantly and positively predicted the post-operative IL-6 level (path a, β = 0.35, P < 0.01), and the post-operative IL-6 level significantly and negatively predicted the post-operative MoCA score (path b, β = -0.28, P < 0.01). The direct effect of the HAMD score on the MoCA score (path c) and the indirect effect mediated by the IL-6 level were significant, and the indirect effect accounted for 19.7% of the total effect. With the post-operative CHE level as the mediating variable (M), the cholinergic mediation model revealed that the pre-operative HAMD score significantly negatively predicted the post-operative CHE level (path a, β = -0.29, P < 0.01), and the post-operative CHE level significantly and positively predicted the post-operative MoCA score (path b, β = 0.32, P < 0.01); The indirect effect was significant, accounting for 17.2% of the total effect (Figure 1 and Table 4).
| Model | Pathway | Effect estimates | Boot SE | Boot 95%LLCI | Boot 95%ULCI | Percentage mediated (%) |
| Inflammatory mediator | HAMD to IL-6 to MoCA | -0.098 | 0.040 | -0.188 | -0.032 | 19.7 |
| Cholinergic mediator | HAMD to CHE to MoCA | -0.085 | 0.036 | -0.169 | -0.025 | 17.2 |
In recent years, as the population in China continues to age, and diagnostic and therapeutic techniques have advanced, a growing number of older patients have opted for surgical treatment. Resection of gastrointestinal tumors, one of the most common general surgeries, is frequently performed in this age group; however, it can trigger profound internal perturbations that severely challenge the physiological reserve of older people[13,14]. Simultaneously, older people are at high risk of depression. When the physiological decline of aging is compounded by poor psychological status and the intense stress of surgery, the likelihood of developing POCD increases sharply[15,16]. Once POCD is present, it prolongs hospitalization, increases medical costs, markedly impairs quality of life and long-term prognosis, and imposes a heavy burden on patients and their families. Clarifying the risk factors and pathogenic mechanisms underlying POCD in older patients with depression after surgery for a gastrointestinal tumor is of major clinical importance.
Among 95 older patients with pre-existing depression who underwent resection of a gastrointestinal tumor, the incidence of POCD on post-operative day 7 was 36.84%. Major gastrointestinal surgery is already recognized as a potent physiological and psychological stressor and an established precipitant of POCD[17,18]. The observed incidence was higher than those reported by Huang et al[19] and Wang et al[20] in unselected cohorts of older people undergoing similar procedures, indicating that comorbid depression significantly increases vulnerability to POCD. Depression is no longer viewed as a purely psychiatric disorder; it is a systemic illness accompanied by pronounced neurobiological alterations, such as hyperactivity of the hypothalamic-pituitary-adrenal axis, chronic immune-inflammatory activation, and an imbalance in multiple neurotransmitter systems. These changes place the brain in a “fragile” or “primed” state. When there are acute insults, such as surgical trauma, anesthesia, and pain, the compromised neuroendocrine and immune systems cannot adequately compensate; thus, precipitating or exacerbating acute deterioration in cognitive function[21]. Therefore, for older patients with depression, surgery is not merely a means of treating somatic disease but may it also act as a critical factor that worsens underlying psychopathology, consistent with the observation of Højris et al[22] that there is an association between depressive symptoms and cognitive changes in patients with testicular cancer.
Univariate and multivariate logistic regression analyses identified five independent predictors of POCD in older patients with pre-operative depression: Advanced age, high pre-operative HAMD score, prolonged duration of surgery, intra-operative administration of anticholinergics, and a large post-operative increase in serum the IL-6 level (all P < 0.05). The probable mechanisms are as follows: (1) Advanced age is accompanied by neuronal loss, decreased synaptic plasti
Regarding biomarker selection, IL-6 and TNF-α were chosen as representative pro-inflammatory cytokines because of their well-established roles in surgery-induced systemic inflammation and neuroinflammation. The serum CHE level has been used as an accessible peripheral indicator of cholinergic system function, and it has been shown to correlate with cognitive outcomes in older patients who have undergone surgery. However, other biomarkers related to neuroinflammation and cholinergic pathways, such as C-reactive protein, acetylcholinesterase subtypes, and specific neuroglial markers, were not included due to limitations in the available retrospective laboratory data. Therefore, the present biomarkers may not fully capture the complexity of the neuroinflammatory and cholinergic mechanisms. Future studies incorporating a broader biomarker panel are required to improve mechanistic interpretation.
Meanwhile, the inflammatory mediation pathway showed that a higher pre-operative HAMD score was associated with a higher post-operative IL-6 level (path a), and a higher IL-6 level predicted a poorer post-operative MoCA score (path b). The cholinergic mediation pathway showed that a higher pre-operative HAMD score was associated with a lower post-operative CHE level (path a), and that a lower post-operative CHE level predicted a poorer post-operative MoCA score (path b).
Depression is a state of chronic low-grade inflammation, characterized by elevated levels of pro-inflammatory cyto
In recent years, many investigators have proposed the “cholinergic anti-inflammatory pathway” theory, which holds that there is a vagus-nerve-mediated cholinergic anti-inflammatory pathway. By activating α7 nicotinic acetylcholine receptors in peripheral organs, such as the spleen, this pathway significantly inhibits the release of pro-inflammatory cytokines by macrophages and other cells, thereby reducing the inflammatory response. However, in older patients with depression, depression and surgical stress may inhibit the function of the cholinergic anti-inflammatory pathway, leading to the loss of inflammatory control, further aggravating the inflammatory response and cognitive impairment; thus, forming a vicious cycle[28,29]. A study by Deng et al[30] in aged rats showed that a stellate ganglion block can improve post-operative cognitive decline by regulating the stress neuroendocrine response, and its mechanism may involve regulation of the balance of cholinergic vagus nerves and other factors. This is inherently related to the results of the inflammatory and cholinergic mediation models used in this study, providing new targets and a theoretical basis for the clinical prevention and treatment of POCD.
This study has several limitations. First, this was a single-center retrospective study with a relatively small sample size (n = 95). Although the study cohort was representative of older patients with depression undergoing surgery for a gastrointestinal tumor in routine clinical practice, selection bias could not be completely avoided. Due to the retrospective design, the sample size was determined by the availability of eligible medical records rather than an a priori power calculation, which may have limited the statistical power to detect weaker associations. Second, retrospective studies rely on existing medical records and are inherently subject to information bias and unmeasured confounding factors. Va
In conclusion, the incidence of post-operative POCD is relatively high in older patients with depression after surgery for a gastrointestinal tumor. Advanced age, severity of pre-operative depression, surgical trauma stress, and intra-operative administration of anticholinergic drugs are independent risk factors for POCD. The severity of pre-operative depression can upregulate the systemic inflammatory response, trigger cholinergic system dysfunction, and increase the risk of POCD.
| 1. | Suskin JA, Paul SM, Stuckey AR, Conley YP, Levine JD, Hammer MJ, Miaskowski C, Dunn LB. Trajectories of Depressive Symptoms Among Patients Undergoing Chemotherapy for Breast, Gastrointestinal, Gynecological, or Lung Cancer. Cancer Nurs. 2026;49:E9-E19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 2. | Ding Y, Yu J, Cui F, Li J. Comparison of Intravenous and Inhalational Anesthetic on Postoperative Cognitive Outcomes in Elderly Patients Undergoing Cancer Surgery: Systematic Review and Meta-analysis. J Perianesth Nurs. 2022;37:683-690. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 3. | Xi L, Fang F, Yuan H, Wang D. Transcutaneous electrical acupoint stimulation for postoperative cognitive dysfunction in geriatric patients with gastrointestinal tumor: a randomized controlled trial. Trials. 2021;22:563. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 36] [Article Influence: 7.2] [Reference Citation Analysis (4)] |
| 4. | de Sain AM, van Zandvoort MJE, Mantione MHM, Huenges Wajer IMC, Willems PWA, Robe PA, Ruis C. A timeline of cognitive functioning in glioma patients who undergo awake brain tumor surgery: a response to Mahajan et al. and their letter to the editor. Acta Neurochir (Wien). 2023;165:2501-2502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 5. | Zhu M, Qi Y, He H, Zhang S, Mei Y. Effect of quadratus lumborum block on postoperative cognitive function in elderly patients undergoing laparoscopic radical gastrectomy: a randomized controlled trial. BMC Geriatr. 2021;21:238. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (1)] |
| 6. | Liu T, Yin C, Li Y, Gao F, Yu L, Wang Z, Wang Q. Effects of Transcutaneous Electrical Acupoint Stimulation on Postoperative Cognitive Decline in Elderly Patients: A Pilot Study. Clin Interv Aging. 2021;16:757-765. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 7. | Wei Y, Liu J, Gong X. Painless Gastrointestinal Endoscopy Assisted with Computed Tomography Image Information Data Monitoring in Postoperative Neurocognitive Dysfunction in Patients with Combined Anesthesia of Propofol and Butorphanol Tartrate under Electronic Health. Comput Math Methods Med. 2022;2022:7086472. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 8. | Ai Z. Effects of dexmedetomidine combined with dezocine on T lymphocytes, NK cells and cognitive function in elderly patients with gastrointestinal cancer after radical surgery. Cell Mol Biol (Noisy-le-grand). 2023;69:56-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 9. | Methods In Medicine CAM. Retracted: Magnetic Resonance Imaging Characteristic Evaluation of Dexmedetomidine on Neurocognitive Dysfunction in Elderly Patients with Colorectal Tumors after Laparoscopic Operation. Comput Math Methods Med. 2023;2023:9794158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 10. | Ding M, Xu X, Xia L, Cao Y. Magnetic Resonance Imaging Characteristic Evaluation of Dexmedetomidine on Neurocognitive Dysfunction in Elderly Patients with Colorectal Tumors after Laparoscopic Operation. Comput Math Methods Med. 2022;2022:1345695. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 11. | Robles R, de la Peña FR, Medina-Mora ME, de Los Dolores Márquez-Caraveo ME, Domínguez T, Juárez F, Rojas AG, Sarmiento-Hernández EI, Feria M, Sosa L, Aguerre RE, Ortiz S, Real T, Rebello T, Sharan P, Reed GM. ICD-11 Guidelines for Mental and Behavioral Disorders of Children and Adolescents: Reliability and Clinical Utility. Psychiatr Serv. 2022;73:396-402. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 12. | Hu J, Li CJ, Wang BJ, Li XY, Mu DL, Wang DX. The sensitivity and specificity of statistical rules for diagnosing delayed neurocognitive recovery with Montreal cognitive assessment in elderly surgical patients: A cohort study. Medicine (Baltimore). 2020;99:e21193. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 13. | Zhang H, Zhao L, Li M, Li X, Li R, Wu D. Efficacy and safety of intranasal insulin on postoperative cognitive dysfunction in elderly patients after laparoscopic radical resection of colorectal cancer: a double-blind pilot study. Front Aging Neurosci. 2024;16:1375841. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (1)] |
| 14. | Makkar M, Hunter R, Kulkarni A, Nguyen JMV. Postoperative Cognitive Decline in Patients Undergoing Major Gynecologic Oncology Surgery: A Pilot Prospective Study. J Obstet Gynaecol Can. 2024;46:102584. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 15. | Varpaei HA, Robbins LB, Farhadi K, Bender CM. Preoperative cognitive function as a risk factor of postoperative delirium in cancer surgeries: A systematic review and meta-analysis. J Surg Oncol. 2024;130:222-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (1)] |
| 16. | Larrabee K, Meeks N, Williams AM, Springer K, Siddiqui F, Chang SS, Ghanem T, Wu VF, Momin S, Tam S. Cognitive Function and Postoperative Outcomes in Patients with Head and Neck Cancer. Laryngoscope. 2023;133:2999-3005. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 17. | Wu Y, Yu C, Gao F. Risk factors for postoperative cognitive dysfunction in elderly patients undergoing surgery for oral malignancies. Perioper Med (Lond). 2023;12:42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (1)] |
| 18. | Janssen TL, de Vries J, Lodder P, Faes MC, Ho GH, Gobardhan PD, van der Laan L. The effects of elective aortic repair, colorectal cancer surgery and subsequent postoperative delirium on long-term quality of life, cognitive functioning and depressive symptoms in older patients. Aging Ment Health. 2021;25:896-905. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 19. | Huang H, Chou J, Tang Y, Ouyang W, Wu X, Le Y. Nomogram to predict postoperative cognitive dysfunction in elderly patients undergoing gastrointestinal tumor resection. Front Aging Neurosci. 2022;14:1037852. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 20. | Wang P, Yin X, Chen G, Li L, Le Y, Xie Z, Ouyang W, Tong J. Perioperative probiotic treatment decreased the incidence of postoperative cognitive impairment in elderly patients following non-cardiac surgery: A randomised double-blind and placebo-controlled trial. Clin Nutr. 2021;40:64-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 62] [Article Influence: 10.3] [Reference Citation Analysis (2)] |
| 21. | Chen W, Chen B, Wang F, Wu Q, Liu W, Wei H, Li C. Clinical Study of Stellate Ganglion Block Combined with General Anesthesia on Hemodynamics, Cognitive Function, and Gastrointestinal Function in Elderly Patients Undergoing Partial Hepatectomy. Evid Based Complement Alternat Med. 2021;2021:1426753. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 22. | Højris NF, Frederiksen Y, Agerbæk M, Nielsen SH, Holt M, Brand SL, Petersen NL, Knudsen UB, Amidi A. Longitudinal assessment of cognitive function in testicular cancer patients prior to orchiectomy and 9 months later and associations with tumor markers. Support Care Cancer. 2025;33:591. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 23. | Wang M, Wang J, Li X, Xu X, Zhao Q, Li Y. A predictive model for postoperative cognitive dysfunction in elderly patients with gastric cancer: a retrospective study. Am J Transl Res. 2022;14:679-686. [PubMed] |
| 24. | Priego-Parra BA, Remes-Troche JM. Bidirectional relationship between gastrointestinal cancer and depression: The key is in the microbiota-gut-brain axis. World J Gastroenterol. 2024;30:5104-5110. [PubMed] [DOI] [Full Text] |
| 25. | Potestio CP, Dibato J, Bolkus K, Awad A, Thayasivam U, Patel A, Bright A, Mitrev LV. Post-Operative Cognitive Dysfunction in Elderly Patients Receiving Propofol Sedation for Gastrointestinal Endoscopies: An Observational Study Utilizing Processed Electroencephalography. Cureus. 2023;15:e46588. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 26. | Yong R, Meng Y. Preoperative neutrophil-lymphocyte ratio, an independent risk factor for postoperative cognitive dysfunction in elderly patients with gastric cancer. Geriatr Gerontol Int. 2020;20:927-931. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 27. | Zhao J, Dai T, Ding L, Liang Y, Yuan W, Jiang Y, Zheng Y. Correlation between neutrophil/lymphocyte ratio, platelet/lymphocyte ratio and postoperative cognitive dysfunction in elderly patients with esophageal cancer. Medicine (Baltimore). 2023;102:e33233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 28. | Megari K, Thomaidou E, Kougioumtzis GA, Theodoratou M, Katsarou D, Karlafti E, Didaggelos M, Paramythiotis D, Argyriadou E. What Do Cancer Surgery and orthopedic Surgery Elderly Patients Have in Common? A Long-term Postoperative Cognitive Dysfunction in Orthopedic and Cancer Patients Original Research. Neurosci Insights. 2024;19:26331055231220906. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 29. | Li G, An C, Ren G, Xue D, Yan W. Association of anemia with postoperative cognitive decline and increased inflammatory markers in elderly gastrointestinal cancer patients. Am J Transl Res. 2024;16:4390-4402. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 30. | Deng X, Sun T, Zhao D, Sana SRGL, Li W. Stellate ganglion block potentially ameliorates postoperative cognitive decline in aged rats by regulating the neuroendocrine response to stress. Heliyon. 2023;9:e14337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (1)] |