Published online Mar 27, 2026. doi: 10.4240/wjgs.v18.i3.115906
Revised: December 1, 2025
Accepted: January 14, 2026
Published online: March 27, 2026
Processing time: 150 Days and 7.4 Hours
A nomogram model predictive of pancreatic ductal adenocarcinoma (PDAC) out
To determine the prognostic value of a nomogram model combining the SII and PNI in patients with PDAC after radical surgery.
One hundred sixty-four patients who underwent radical surgery for PDAC be
An SII > 335.82, a PNI ≤ 44.45, poorly differentiated PDAC, no postoperative adjuvant chemotherapy, and a carbohydrate antigen 19-9 level > 37 U/mL were independent risk factors for poor prognosis of patients with PDAC after radical surgery. These five factors were included in the nomogram model. The internal validation C-index of the nomogram model was 0.691 (95% confidence interval: 0.626-0.755). The area under the ROC curve values for the 1-year, 2-year, and 3-year postoperative survival rates in the training cohort were 0.684, 0.762, and 0.822, respectively; the area under the ROC curve values for the external validation cohort were 0.772, 0.755, and 0.796, respectively.
The nomogram model based on the SII and PNI accurately predicted the survival risk and prognosis in patients with PDAC after surgery, providing a quantitative tool for individualized treatment decisions.
Core Tip: A nomogram prediction model that was constructed based on the systemic immune inflammation index and prognostic nutrition index accurately assessed the comprehensive preoperative inflammation-immune-nutrition status of patients with pancreatic cancer. The convenience and simplicity of the model, especially when combined with the online calculator, enhanced the predictive potential for the prognosis of patients with pancreatic cancer after surgery. This model is particularly suitable for primary hospitals with limited resources. However, the results of this study need to be verified by prospective samples from multiple centers with large samples.
- Citation: Tang J, Ding SL, Wu Y, Wu YP, Yuan XB, Wu PF, Sha DS. Prognostic value of a nomogram model for pancreatic cancer incorporating the systemic immune-inflammation and prognostic nutritional indices. World J Gastrointest Surg 2026; 18(3): 115906
- URL: https://www.wjgnet.com/1948-9366/full/v18/i3/115906.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i3.115906
Pancreatic cancer is a highly aggressive malignant tumor of the digestive tract with the following clinical features: Insi
Inflammation promotes the occurrence, progression, and metastasis of tumors by regulating the tumor microenvi
Therefore, this study aimed to develop a nomogram model based on the SII and PNI for predicting 1-year, 2-year, and 3-year survival rates in patients with PDAC who have undergone radical surgery. The efficacy and clinical significance of the nomogram model were also determined.
This study was approved by the Institutional Ethical Committee of Rugao People’s Hospital, approval No. KY202211013 and proceeded in compliance with the Declaration of Helsinki. Each participant signed a written informed consent for treatment and the use and publication of their clinical data and information. The confidentiality agreement has been honored, and private patient information has been protected.
Patients with PDAC who were treated at the Affiliated Hospital of Nantong University from January 2017 to December 2020 were designated as the training cohort. The patients with PDAC who were treated at Rugao People’s Hospital between January 2019 and December 2023 were designated as the external validation cohort. The inclusion criteria were as follows: (1) PDAC diagnosis according to the 2021 Guidelines for the Diagnosis and Treatment of Pancreatic Cancer in China[12] and the postoperative pathologic diagnosis was PDAC; (2) Underwent radical surgery for PDAC; (3) Preopera
Data collection included general clinical information, routine serum biochemical test results, and intraoperative and postoperative details. The general clinical information included age, gender, contact number, and a history of chronic diseases (hypertension, diabetes, chronic bronchitis emphysema, and coronary atherosclerotic heart disease). The following routine serum biochemical test results within 1 week preoperatively were included: Platelet count; lymphocyte count; neutrophil count; hemoglobin concentration; total bilirubin level; aspartate aminotransferase level; alanine aminotransferase level; albumin level; carcinoembryonic antigen level; and carbohydrate antigen 19-9 (CA19-9) level. Intraoperative and postoperative details included the following: Surgical method; intraoperative tumor location; postoperative pathologic report (tumor size, lymph node metastases, degree of tumor degree, surgical margins, vascular tumor thrombus, and presence or absence of vascular and nerve invasion); tumor stage according to the 8th edition of the American Joint Committee on Cancer Tumor-node-metastasis Staging Manual; postoperative adjuvant chemotherapy; recent postoperative complications; and other indicators. The postoperative short-term complications were defined as any of the following complications that occurred within 1 month after surgery: Gastrointestinal anastomotic fistula; pancreatic fistula; biliary fistula; delayed gastric emptying; postoperative massive hemorrhage of the pancreas; abdominal cavity infection; pulmonary infection; and surgical incision infection.
The SII and PNI values were calculated based on the biochemical and serum routine data 1 week preoperatively as follows: SII = platelet count (109/L) × neutrophil count (109/L)/Lymphocyte count (109/L)[8]; and PNI = serum albumin concentration (g/L) + lymphocyte count (109/L) × 5[9].
Follow-up data, including patient mortality, the time of death, and the postoperative adjuvant therapy, were recorded through telephone interviews and outpatient follow-up evaluation. The follow-up period ended on December 31, 2024. The end of the follow-up period or patient death was the endpoint event. Patient survival status during the follow-up period was recorded, and the survival time was recorded monthly.
Statistical analysis was performed using SPSS 25.0 and R 4.2.2 software. The mean ± SD was applied to continuous variables with a normal distribution. Continuous variables with a non-normal distribution are represented by the median [lower and upper quartiles (median (P25, P75)]. Counting variables are expressed in terms of frequency and 100% [n (%)]. The optimal SII and PNI cutoff values were determined using the surv-cutpoint function in the R language surv-miner package based on the maximum selection test method. The patients were divided into high and low groups according to the SII and PNI levels. A χ2 or Fisher’s exact test was used to analyze the correlation of clinicopathologic characteristics between the training and external validation cohorts. The independent risk factors influencing overall survival (OS) in patients after radical resection of PDAC were determined using the Cox regression model; a P value < 0.05 identified significant factors.
The forestplot, rms, survival receiver operating characteristic (ROC), ggplot2, and other packages in R language were applied to establish the nomogram model of the 1-year, 2-year, and 3-year postoperative survival rates of patients with PDAC, including the independent risk factors with statistical differences in the multivariable Cox regression analysis (SII, PNI, degree of tumor differentiation, postoperative adjuvant chemotherapy, and CA19-9 level). Internal validation was performed using the bootstrap resampling method. The efficacy of the nomogram model was evaluated and verified through a time-dependent ROC curve, C-index, calibration curve, and decision curve analysis (DCA). A web calculator was developed using the “shiny: Web Application Framework for R package.” Results were considered significant if the P value was < 0.05.
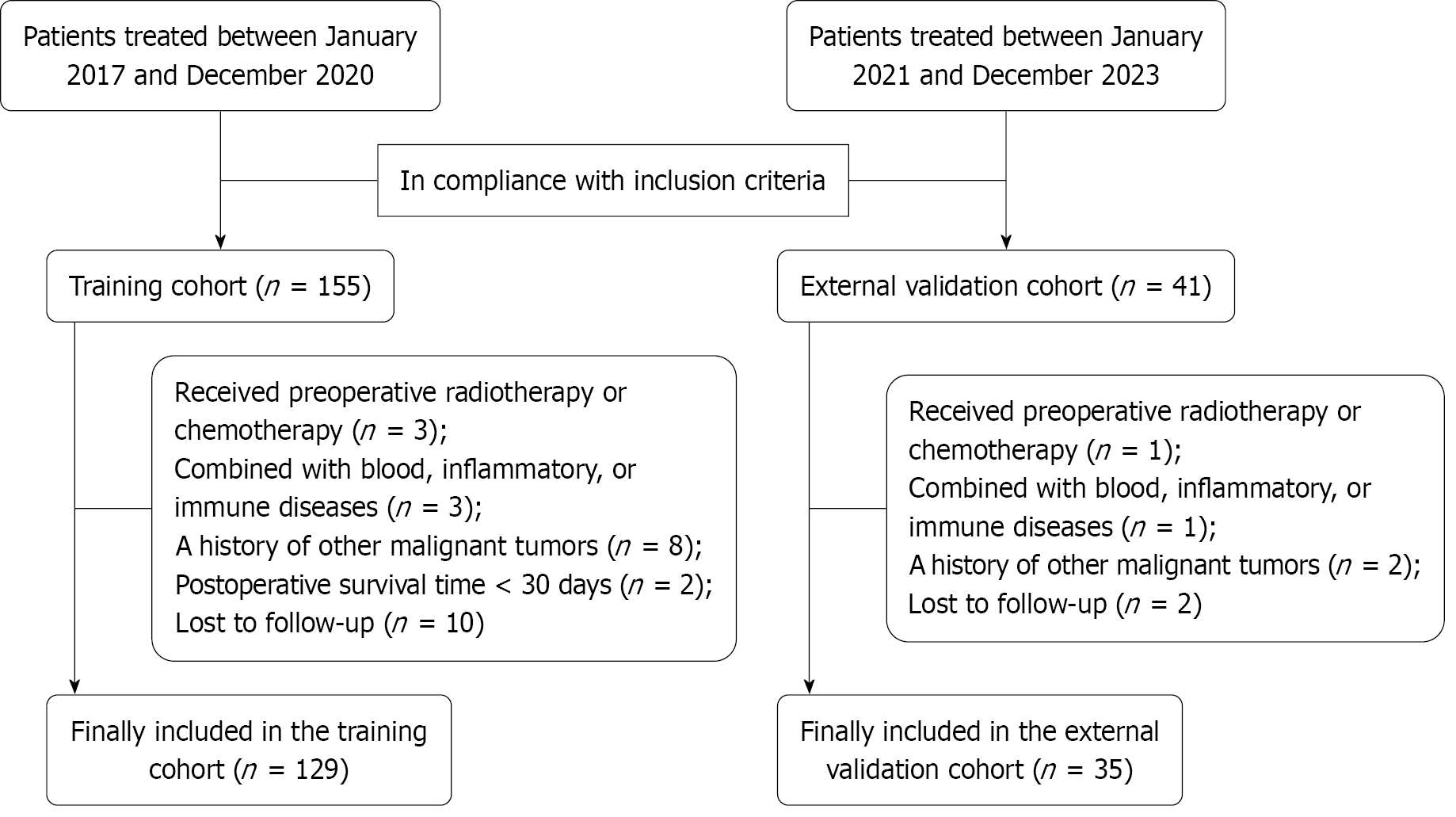
The training cohort was initially comprised of 155 patients. However, 3 patients received preoperative radiotherapy or chemotherapy, 3 patients had blood, inflammatory, or immune diseases, 8 patients had other coexisting malignant tu
The baseline data from patients with PDAC are shown in Table 1. With the exception of gender, surgical methods, and the SII, no significant differences were detected between the training and external validation cohorts.
| Clinicopathologic factors | Total number (n = 164) | Training cohort (n = 129) | External validation cohort (n = 35) | P value |
| Gender | 0.022a | |||
| Male | 89 (54.3) | 76 (58.9) | 13 (37.1) | |
| Female | 75 (45.7) | 53 (41.1) | 22 (62.9) | |
| Age (years) | 0.624 | |||
| ≤ 65 | 81 (49.4) | 65 (50.4) | 19 (54.3) | |
| > 65 | 83 (50.6) | 64 (49.6) | 16 (45.7) | |
| Underlying diseases | 0.341 | |||
| No | 82 (50) | 67 (51.9) | 15 (42.9) | |
| Yes | 82 (50) | 62 (48.1) | 20 (57.1) | |
| Surgical method | < 0.001a | |||
| Endoscopic surgery | 32 (19.5) | 16 (12.4) | 16 (45.7) | |
| Open surgery | 132 (80.5) | 113 (87.6) | 19 (54.3) | |
| Tumor location | 0.804 | |||
| Pancreatic body/tail | 58 (35.4) | 45 (34.9) | 13 (37.1) | |
| Pancreatic head | 106 (64.6) | 84 (65.1) | 22 (62.9) | |
| Tumor maximum diameter (cm) | 0.301 | |||
| ≤ 3 | 81 (49.6) | 61 (47.3) | 20 (57.1) | |
| > 3 | 83 (50.6) | 68 (52.7) | 15 (42.9) | |
| T staging | 0.543 | |||
| T1-T2 | 120 (73.2) | 93 (72.1) | 27 (77.1) | |
| T3-T4 | 42 (26.9) | 36 (27.9) | 8 (22.9) | |
| N grouping | 0.192 | |||
| N0 | 100 (61.0) | 82 (63.6) | 18 (51.4) | |
| N1-N2 | 64 (39.0) | 47 (36.4) | 17 (48.6) | |
| TNM staging | 0.771 | |||
| I-II | 144 (87.8) | 114 (88.4) | 30 (85.7) | |
| III | 20 (12.2) | 15 (11.6) | 5 (14.3) | |
| Degree of tumor differentiation | 0.871 | |||
| Moderately or well-differentiated | 105 (64.0) | 83 (64.3) | 22 (62.9) | |
| Poorly differentiated | 59 (36.0) | 46 (35.7) | 13 (37.1) | |
| Surgical margins | 0.343 | |||
| Negative | 158 (96.3) | 123 (95.3) | 35 (100.0) | |
| Positive | 6 (3.7) | 6 (4.65) | 0 (0.0) | |
| Vascular tumor thrombus | 0.072 | |||
| No | 81 (49.4) | 59 (45.7) | 22 (62.9) | |
| Yes | 83 (50.6) | 70 (54.3) | 13 (37.1) | |
| Vascular/nerve invasion | 0.381 | |||
| No | 20 (12.2) | 14 (10.9) | 6 (17.1) | |
| Yes | 144 (87.8) | 115 (89.1) | 29 (82.9) | |
| Lymph node metastasis | 0.139 | |||
| No | 102 (62.2) | 84 (65.1) | 18 (51.4) | |
| Yes | 62 (37.8) | 45 (34.9) | 17 (48.6) | |
| Postoperative adjuvant chemotherapy | 0.847 | |||
| No | 110 (67.1) | 42 (32.6) | 12 (34.3) | |
| Yes | 54 (32.9) | 87 (67.4) | 23 (65.7) | |
| Recent postoperative complications | 0.960 | |||
| No | 126 (76.8) | 99 (76.7) | 27 (77.1) | |
| Yes | 38 (23.2) | 30 (23.3) | 8 (22.9) | |
| CEA (ng/mL) | 0.322 | |||
| ≤ 5 | 123 (75.0) | 99 (76.7) | 24 (68.6) | |
| > 5 | 41 (25.0) | 30 (23.3) | 11 (31.4) | |
| CA19-9 (U/mL) | 0.274 | |||
| ≤ 37 | 40 (24.4) | 29 (22.5) | 11 (31.4) | |
| > 37 | 124 (75.6) | 100 (77.5) | 24 (68.6) | |
| SII | 0.011a | |||
| ≤ 335.82 | 58 (35.4) | 52 (40.3) | 6 (17.1) | |
| > 335.82 | 106 (64.6) | 77 (59.7) | 29 (82.9) | |
| PNI | 0.331 | |||
| ≤ 44.45 | 68 (41.5) | 56 (43.4) | 12 (34.3) | |
| > 44.45 | 96 (58.5) | 73 (56.6) | 23 (65.7) | |
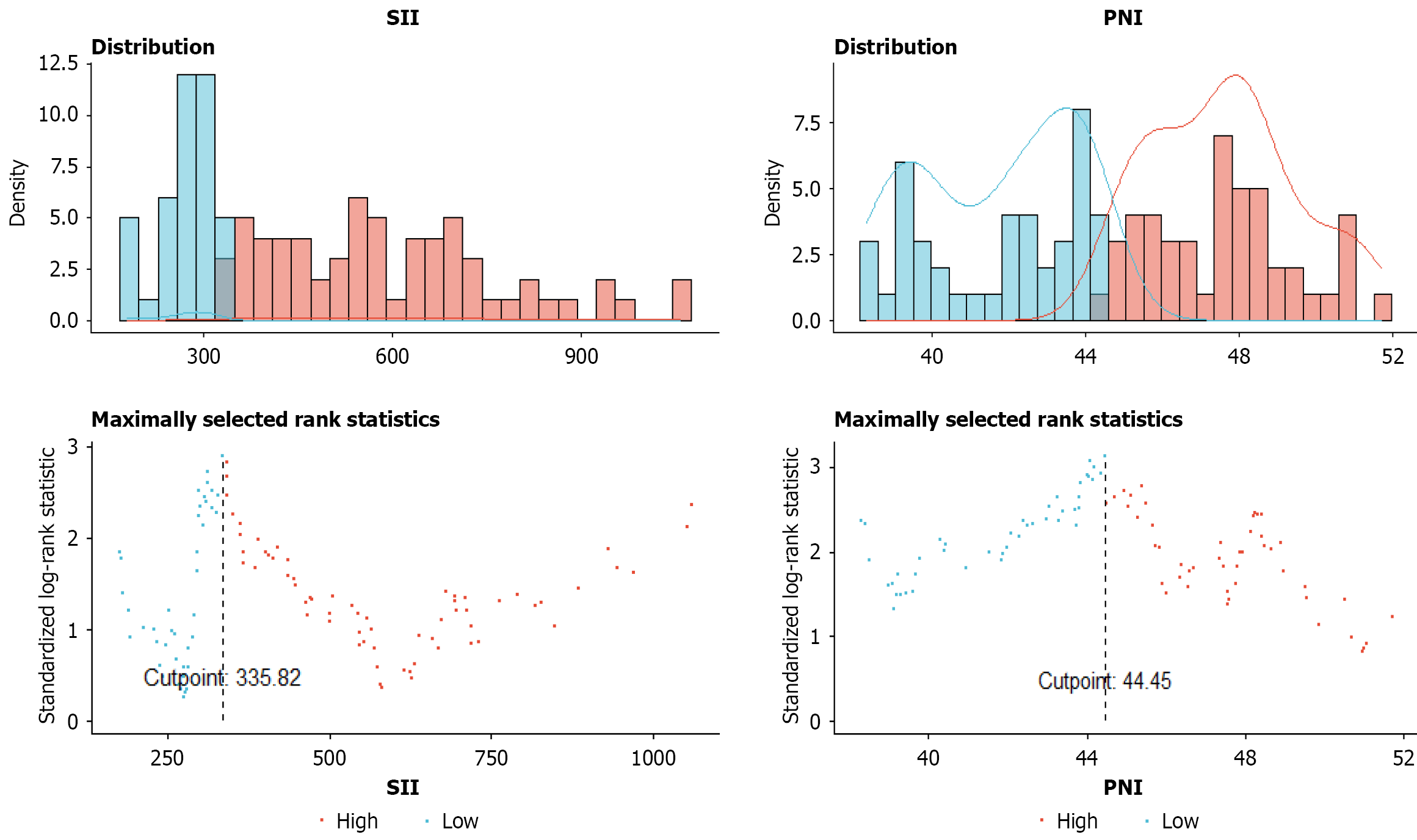
The SII and PNI of the patients in the training cohort were 412.9 (278.0, 686.7) and 45.23 ± 5.06, respectively. The surv-cutpoint function in the R language surv-miner package was used to determine the partitioning threshold and the opti
Univariable Cox regression analyses showed that an SII > 335.82 [hazard ratio (HR) = 1.844, 95% confidence interval (CI): 1.194-2.849; P = 0.006], a PNI ≤ 44.45 (HR = 1.990, 95%CI: 1.305-3.034; P = 0.001), a maximum tumor diameter > 3 cm (HR = 1.816, 95%CI: 1.194-2.762; P = 0.005), T3-T4 staging (HR = 1.568, 95%CI: 1.013-2.427; P = 0.043), a poorly differentiated tumor (HR = 1.607, 95%CI: 1.057-2.442; P = 0.026), no postoperative adjuvant chemotherapy (HR = 0.575, 95%CI: 0.373-0.885; P = 0.012), and a CA19-9 level > 37 U/mL (HR = 2.021, 95%CI: 1.142-3.576; P = 0.016) were significantly related to poor prognosis in patients with PDAC after radical surgery (Table 2).
| Clinicopathologic factors | Univariable analysis | P value | Multivariable analysis | P value | ||
| HR | 95%CI | HR | 95%CI | |||
| Gender | ||||||
| Female | 1 (reference) | - | - | - | - | |
| Male | 0.955 | 0.634-1.438 | 0.824 | - | - | - |
| Age (years) | ||||||
| ≤ 65 | 1 (reference) | - | - | - | - | |
| > 65 | 1.107 | 0.736-1.665 | 0.626 | - | - | - |
| Underlying diseases | ||||||
| No | 1 (reference) | - | - | - | - | |
| Yes | 0.992 | 0.661-1.491 | 0.970 | - | - | - |
| Surgical methods | ||||||
| Endoscopic surgery | 1 (reference) | - | - | - | - | |
| Open surgery | 1.101 | 0.570-2.128 | 0.774 | - | - | - |
| Tumor location | ||||||
| Pancreatic body/tail | 1 (reference) | - | - | - | - | |
| Pancreatic head | 0.754 | 0.495-1.147 | 0.187 | - | - | - |
| Tumor maximum diameter (cm) | ||||||
| ≤ 3 | 1 (reference) | - | - | - | - | |
| > 3 | 1.816 | 1.194-2.762 | 0.005a | 1.592 | 0.962-2.633 | 0.070 |
| T staging | ||||||
| T1-T2 | 1 (reference) | - | - | - | - | - |
| T3-T4 | 1.568 | 1.013-2.427 | 0.043a | 1.061 | 0.624-1.802 | 0.827 |
| N grouping | ||||||
| N0 | 1 (reference) | - | - | - | - | - |
| N1-N2 | 1.325 | 0.877-2.002 | 0.181 | - | - | |
| TNM staging | ||||||
| I-II | 1 (reference) | - | - | - | - | - |
| III | 1.129 | 0.601-2.120 | 0.707 | - | - | - |
| Degree of tumor differentiation | ||||||
| Moderately or well-differentiated | 1 (reference) | - | - | - | - | - |
| Poorly differentiated | 1.607 | 1.057-2.442 | 0.026a | 1.614 | 1.036-2.515 | 0.034a |
| Surgical margins | ||||||
| Negative | 1 (reference) | - | - | - | - | - |
| Positive | 1.033 | 0.451-2.371 | 0.938 | - | - | - |
| Vascular tumor thrombus | ||||||
| No | 1 (reference) | - | - | - | - | - |
| Yes | 1.058 | 0.700-1.599 | 0.790 | - | - | - |
| Vascular/nerve invasion | ||||||
| No | 1 (reference) | - | - | - | - | - |
| Yes | 1.715 | 0.859-3.426 | 0.126 | - | - | - |
| Lymph node metastasis | ||||||
| No | 1 (reference) | - | - | - | - | - |
| Yes | 1.251 | 0.825-1.897 | 0.292 | - | - | - |
| Postoperative adjuvant chemotherapy | ||||||
| No | 1 (reference) | - | - | - | - | - |
| Yes | 0.575 | 0.373-0.885 | 0.012a | 0.561 | 0.360-0.874 | 0.011a |
| Recent postoperative complications | ||||||
| No | 1 (reference) | - | - | - | - | - |
| Yes | 1.218 | 0.763-1.945 | 0.408 | - | - | - |
| CEA (ng/mL) | ||||||
| ≤ 5 | 1 (reference) | - | - | - | - | - |
| > 5 | 0.917 | 0.562-1.495 | 0.728 | - | - | - |
| CA19-9 (U/mL) | ||||||
| ≤ 37 | 1 (reference) | - | - | - | - | - |
| > 37 | 2.021 | 1.142-3.576 | 0.016a | 1.844 | 1.021-3.330 | 0.043a |
| SII | ||||||
| ≤ 335.82 | 1 (reference) | - | - | - | - | - |
| > 335.82 | 1.844 | 1.194-2.849 | 0.006a | 1.891 | 1.217-2.938 | 0.005a |
| PNI | ||||||
| ≤ 44.45 | 1 (reference) | - | - | - | - | - |
| > 44.45 | 1.990 | 1.305-3.034 | 0.001a | 2.149 | 1.381-3.343 | 0.001a |
The factors with statistical differences (P < 0.05) based on univariable Cox analysis were further analyzed by multivaria
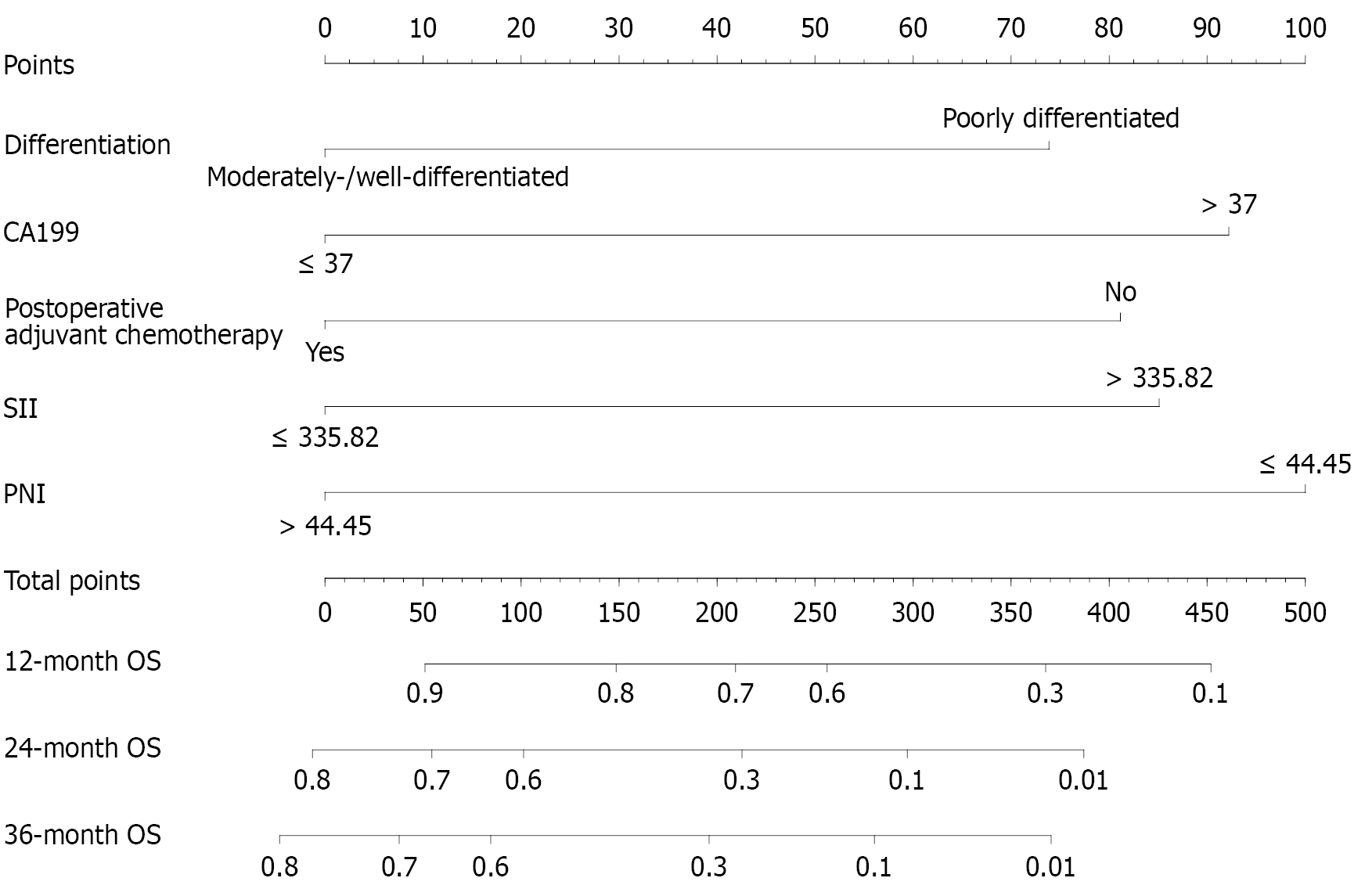
The predictive nomogram model was established using the rms package in R language software, including the inde
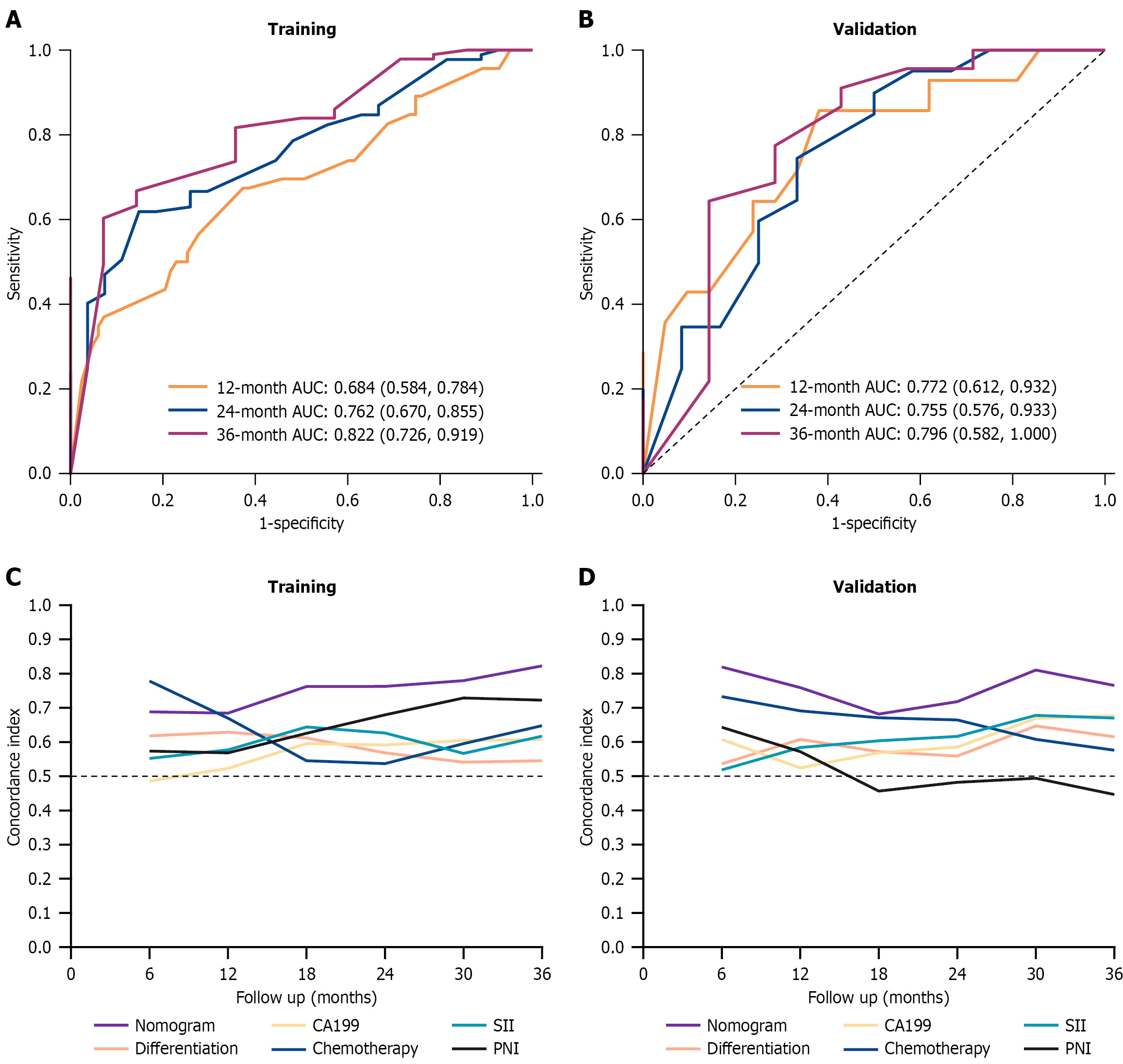
The bootstrap resampling method verified the model in the internal dataset using the survival ROC and ggplot2 packages in the R language software. The calculated C-index was 0.691 (95%CI: 0.626-0.755), indicating that the prediction model had good discrimination. The ROC curves of the nomogram prediction model in the training and external validation cohorts 1 year, 2 years, and 3 years after surgery were plotted and the area under the ROC curve (AUC) performance were analyzed. The results suggested that AUC values of the 1-year, 2-year, and 3-year postoperative survival rates in the training cohort were 0.684, 0.762, and 0.822, respectively; the 1-year, 2-year, and 3-year postoperative survival rates in the external validation cohort were 0.772, 0.755, and 0.796, respectively (Figure 4A and B). Time-dependent C-index analysis revealed that the nomogram model had better prognostic accuracy in predicting the clinical outcomes of OS compared with any other single prognostic marker (Figure 4C and D). The nomogram had a higher time-dependent C-index for the OS of patients with pancreatic cancer compared with the degree of tumor differentiation, chemotherapy, CA19-9 level, SII, and PNI.
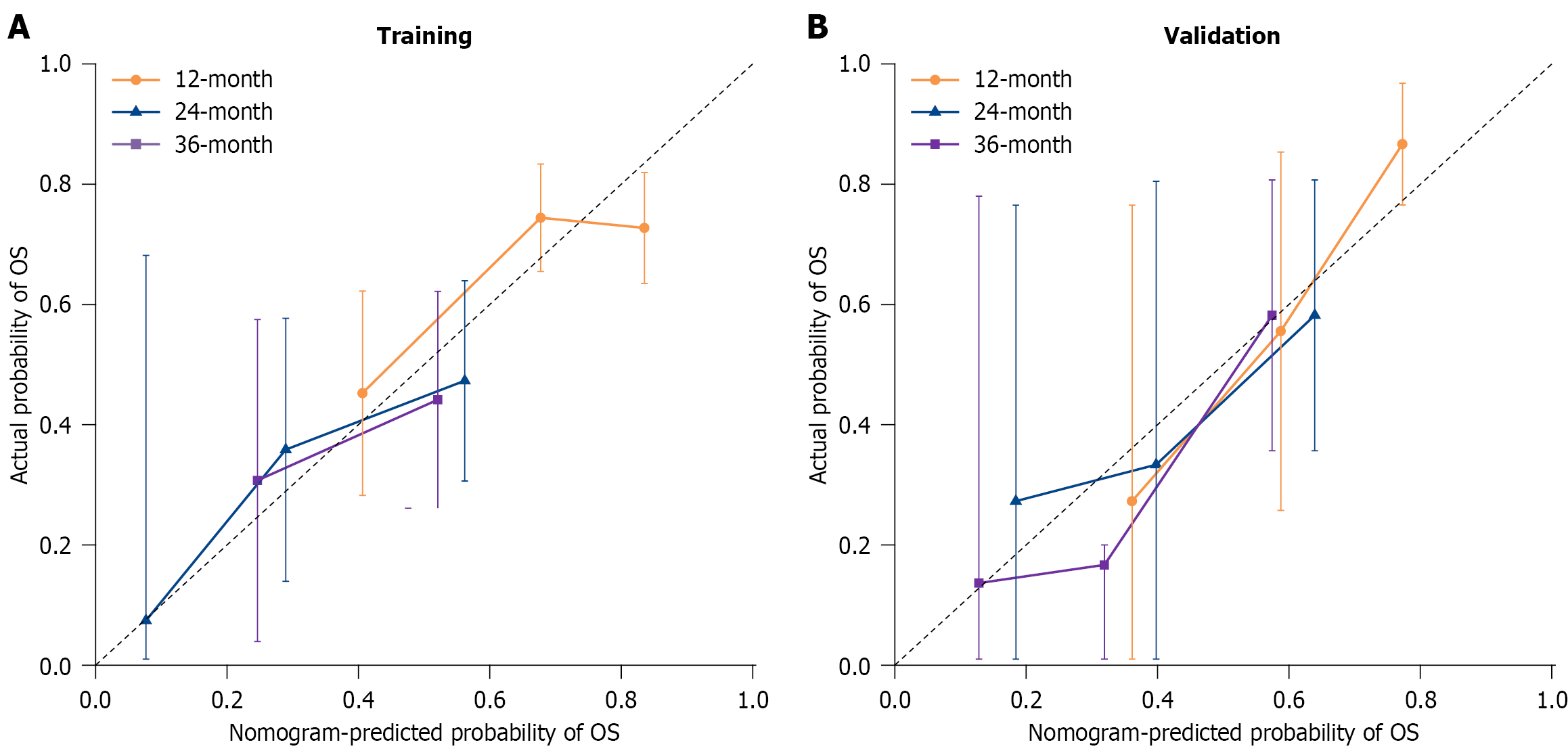
The calibration curves of the 1-year, 2-year, and 3-year survival rates of patients with pancreatic cancer in the training (Figure 5A) and external validation cohorts (Figure 5B) were plotted using the rms package of the R language software. The horizontal axis represents the predicted probability of the model, and the vertical axis represents the actual pro
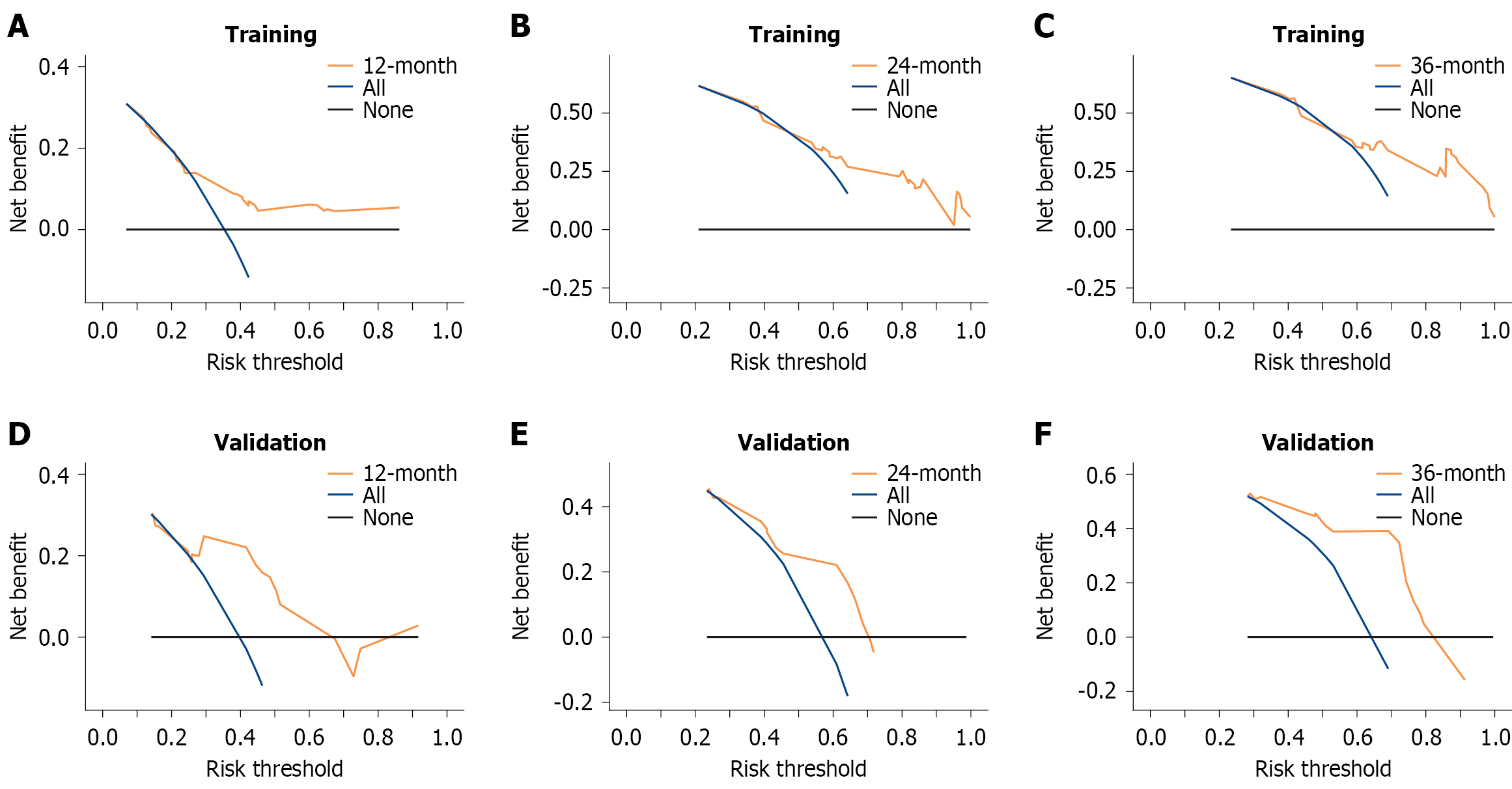
The DCA curves of the training cohort (Figure 6A-C) and the external validation cohort (Figure 6D-F) showed that the nomogram provided a moderate additional net benefit in terms of survival probabilities 1 year, 2 years, and 3 years after surgery. The DCA curve indicated that the model has potential clinical practicability.
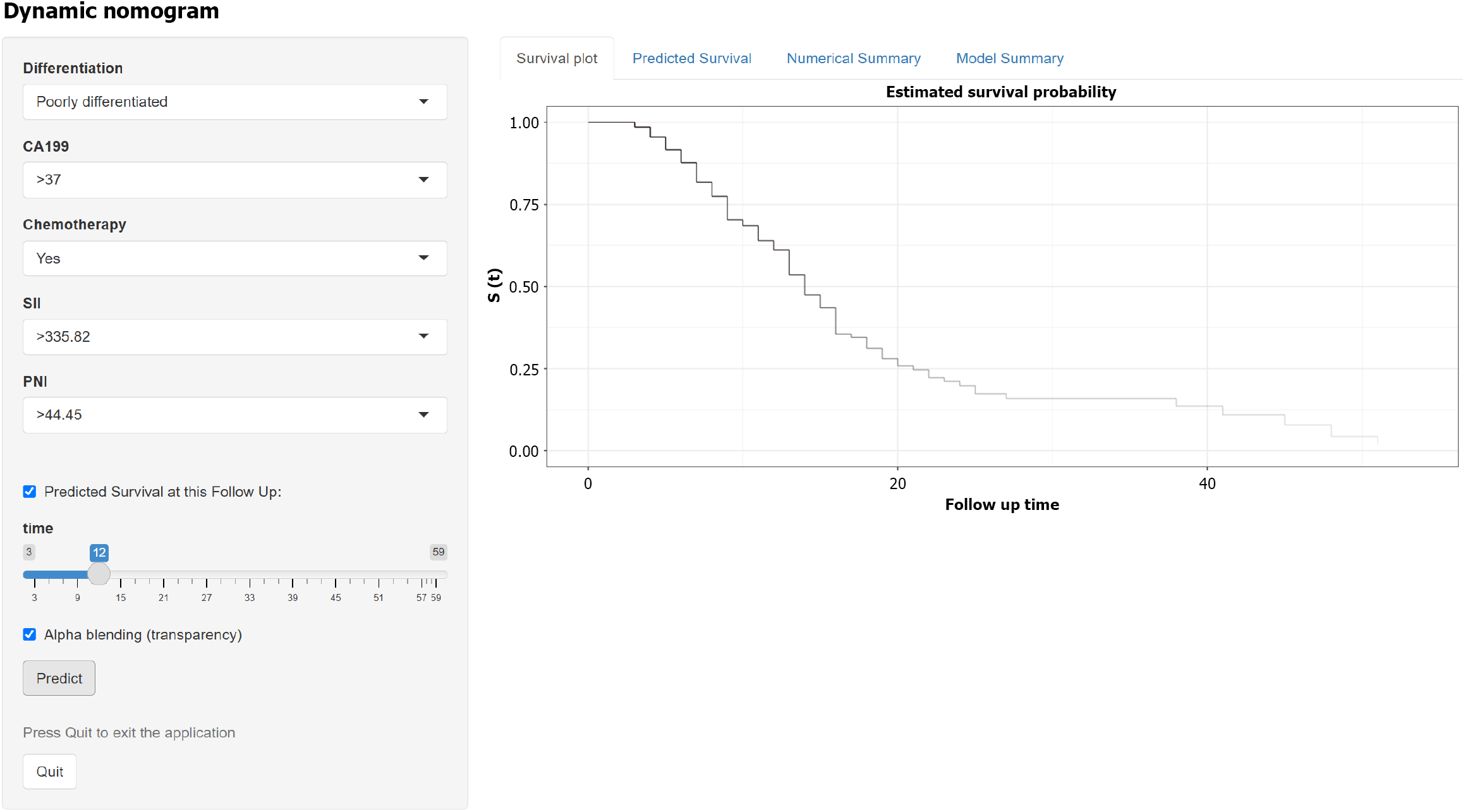
A web calculator was used to further enhance the practicality of the nomogram prediction model. Users can visit https://tj190019.shinyapps.io/DynNomapp/ directly to implement the dynamic model of the nomogram in Figure 7 and then input the clinical pathologic characteristics. The network server will automatically generate the survival probability curve and 95%CI. The website easily predicts patient OS.
A multidimensional integrated nomogram model, including inflammation, immunity, and nutrition, was developed. The model, which consisted of SII and PNI indicators, was confirmed to have good predictive value for the postoperative survival of patients with PDAC.
Most studies have used the ROC curve method to determine the best SII and PNI cutoff values for postoperative survival of patients with PDAC, but the ROC curve method has limitations. Specifically, the ROC curve method only considers disease outcomes and ignores survival time and missing data. The current study determined the cutoff value using the maximum selection test method with the Kaplan-Meier curve and the R language surv-miner package. This method fully incorporates survival time and is more in line with the characteristics of cancer prognosis research than the ROC curve method.
Patients with an elevated SII before surgery usually have an increased platelet count, increased neutrophil count, or decreased lymphocyte count, suggesting the coexistence of inflammation and weakened immunity[8]. Platelets can disguise tumor cells[13-15], and low lymphocyte counts can impair the immune response[16]. Inflammation and malnu
The current study fully referred to the results of previous studies and developed the nomogram model that consisted of the degree of tumor differentiation, the CA19-9 level, chemotherapy status, the SII, and the PNI. In the current study the AUC values of the 1-year, 2-year, and 3-year survival rates after surgery in the training cohort were 0.684, 0.762, and 0.822, respectively. The AUC values of the 1-year, 2-year, and 3-year survival rates after surgery in the external validation cohort were 0.772, 0.755, and 0.796, respectively. Furthermore, the calibration curve analysis confirmed that there was good consistency between the predicted probability of the model and the actual observed probability. These results indicate that the nomogram model has good discrimination value.
Xu et al[24] developed a nomogram model to predict survival of patients with PDAC using magnetic resonance imaging and clinical data with a predictive efficacy (C-index) of 0.78. Huang et al[25] developed a model using the microRNA-24 level with a predictive efficacy of 0.82. However, due to the high cost and professional skills required for these models involving magnetic resonance imaging radiomics[24] and RNA sequencing[25], the clinical applicability is limited. The model developed in the current study was based on routine hematologic indicators and can be directly obtained from routine blood and biochemical tests. The core advantage of this model lies in the fact that the hematologic indicators are noninvasive, easy to obtain, and low cost. The AUC results showed good predictive value of the nomogram model, and the DCA showed significant clinical benefits. Combined with the developed online prognostic risk calculator, the visualization and convenience of prognostic assessment have been achieved. Therefore, this model may have greater application potential in resource-limited environments, such as grassroots hospitals.
The previous PNI model had AUC values of 0.826, 0.798, and 0.846 for predicting 1-year, 3-year, and 5-year OS[10]. Sun et al[11] reported an AUC of 0.689 for SII. The current study combined the SII and PNI to predict OS in PDAC, unlike previous reports that used the PNI[10] or SII[11]. The AUC in the training cohort of the current study was lower than the AUC reported by Yang et al[10] but was higher than the AUC reported by Sun et al[11]. Moreover, the current study validated the prediction value in the external cohort and achieved moderately good AUC values (0.772, 0.755, and 0.796 for 1-year, 2-year, and 3-year, respectively, survival rates).
This study had some limitations that should be acknowledged. First, this study focused solely on the patients with PDAC after radical resection and did not explore the prognostic predictive value of the SII and PNI for patients with other types of pancreatic cancer, including unresectable pancreatic cancer. Second, there were regional and population biases, and the sample size of the external validation cohort in this study was relatively small. Third, existing studies show variability in the optimal SII and PNI cutoff values. This heterogeneity may stem from differences in study populations, detection methods, or statistical models, potentially limiting the clinical application and promotion of research outcomes. Therefore, further multicenter, large-sample, and prospective studies need to be conducted to de
The nomogram prediction model presented herein was based on the SII and PNI and accurately assessed the comprehensive preoperative inflammation-immune-nutrition status of patients with pancreatic cancer, demonstrating significant postoperative prognostic significance for patients with PDAC. Due to the convenience and simplicity of this model, the nomogram is more likely to be applicable in primary hospitals with limited resources. The results of this study need to be verified by prospective samples from multiple centers with large samples given the population limitations of this study and the failure to standardize key parameters.
| 1. | Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16785] [Cited by in RCA: 15069] [Article Influence: 7534.5] [Reference Citation Analysis (23)] |
| 2. | Zhao C, Gao F, Li Q, Liu Q, Lin X. The Distributional Characteristic and Growing Trend of Pancreatic Cancer in China. Pancreas. 2019;48:309-314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 47] [Article Influence: 6.7] [Reference Citation Analysis (1)] |
| 3. | Stoop TF, Javed AA, Oba A, Koerkamp BG, Seufferlein T, Wilmink JW, Besselink MG. Pancreatic cancer. Lancet. 2025;405:1182-1202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 250] [Cited by in RCA: 227] [Article Influence: 227.0] [Reference Citation Analysis (1)] |
| 4. | Chen Q, Ren S, Cui S, Huang J, Wang D, Li B, He Q, Lang R. Prognostic and recurrent significance of SII in patients with pancreatic head cancer undergoing pancreaticoduodenectomy. Front Oncol. 2023;13:1122811. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 5. | Lu JN, Zhou LS, Zhang S, Li JX, Xu CJ. Performance of nutritional and inflammatory markers in patients with pancreatic cancer. World J Clin Oncol. 2024;15:1021-1032. [PubMed] [DOI] [Full Text] |
| 6. | Greten FR, Grivennikov SI. Inflammation and Cancer: Triggers, Mechanisms, and Consequences. Immunity. 2019;51:27-41. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3199] [Cited by in RCA: 2830] [Article Influence: 404.3] [Reference Citation Analysis (5)] |
| 7. | Teja M, Garrido MI, Ocanto A, Couñago F. Prognostic impact of inflammatory and nutritional biomarkers in pancreatic cancer. World J Clin Oncol. 2025;16:101191. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (4)] |
| 8. | Han R, Tian Z, Jiang Y, Guan G, Wang X, Sun X, Yu Y, Jing X. Prognostic significance of the systemic immune inflammation index in patients with metastatic and unresectable pancreatic cancer. Front Surg. 2022;9:915599. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (2)] |
| 9. | Zhao P, Wu Z, Wang Z, Wu C, Huang X, Tian B. Prognostic role of the prognostic nutritional index in patients with pancreatic cancer who underwent curative resection without preoperative neoadjuvant treatment: A systematic review and meta-analysis. Front Surg. 2022;9:992641. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 26] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 10. | Yang J, Zhou H, Li H, Zhao F, Tong K. Nomogram incorporating prognostic immune-inflammatory-nutritional score for survival prediction in pancreatic cancer: a retrospective study. BMC Cancer. 2024;24:193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (1)] |
| 11. | Sun Y, Hu J, Wang R, Du X, Zhang X, E J, Zheng S, Zhou Y, Mou R, Li X, Zhang H, Xu Y, Liao Y, Jiang W, Liu L, Wang R, Zhu J, Xie R. Meaningful nomograms based on systemic immune inflammation index predicted survival in metastatic pancreatic cancer patients receiving chemotherapy. Cancer Med. 2024;13:e7453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 12. | Chinese Pancreatic Surgery Association; Chinese Society of Surgery; Chinese Medical Association. [Guidelines for the diagnosis and treatment of pancreatic cancer in China(2021)]. Zhonghua Wai Ke Za Zhi. 2021;59:561-577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 13. | Geranpayehvaghei M, Dabirmanesh B, Khaledi M, Atabakhshi-Kashi M, Gao C, Taleb M, Zhang Y, Khajeh K, Nie G. Cancer-associated-platelet-inspired nanomedicines for cancer therapy. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2021;13:e1702. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 27] [Article Influence: 5.4] [Reference Citation Analysis (1)] |
| 14. | Liu Y, Zhang Y, Ding Y, Zhuang R. Platelet-mediated tumor metastasis mechanism and the role of cell adhesion molecules. Crit Rev Oncol Hematol. 2021;167:103502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 67] [Article Influence: 13.4] [Reference Citation Analysis (7)] |
| 15. | Placke T, Örgel M, Schaller M, Jung G, Rammensee HG, Kopp HG, Salih HR. Platelet-derived MHC class I confers a pseudonormal phenotype to cancer cells that subverts the antitumor reactivity of natural killer immune cells. Cancer Res. 2012;72:440-448. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 402] [Cited by in RCA: 355] [Article Influence: 25.4] [Reference Citation Analysis (4)] |
| 16. | Ogino S, Nosho K, Irahara N, Meyerhardt JA, Baba Y, Shima K, Glickman JN, Ferrone CR, Mino-Kenudson M, Tanaka N, Dranoff G, Giovannucci EL, Fuchs CS. Lymphocytic reaction to colorectal cancer is associated with longer survival, independent of lymph node count, microsatellite instability, and CpG island methylator phenotype. Clin Cancer Res. 2009;15:6412-6420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 357] [Cited by in RCA: 350] [Article Influence: 20.6] [Reference Citation Analysis (4)] |
| 17. | Jabłońska B, Pawlicki K, Mrowiec S. Associations between Nutritional and Immune Status and Clinicopathologic Factors in Patients with Pancreatic Cancer: A Comprehensive Analysis. Cancers (Basel). 2021;13:5041. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 18. | Okadome K, Baba Y, Yagi T, Kiyozumi Y, Ishimoto T, Iwatsuki M, Miyamoto Y, Yoshida N, Watanabe M, Baba H. Prognostic Nutritional Index, Tumor-infiltrating Lymphocytes, and Prognosis in Patients with Esophageal Cancer. Ann Surg. 2020;271:693-700. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 310] [Cited by in RCA: 289] [Article Influence: 48.2] [Reference Citation Analysis (2)] |
| 19. | Soeters PB, Wolfe RR, Shenkin A. Hypoalbuminemia: Pathogenesis and Clinical Significance. JPEN J Parenter Enteral Nutr. 2019;43:181-193. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1096] [Cited by in RCA: 927] [Article Influence: 132.4] [Reference Citation Analysis (5)] |
| 20. | Cheng L, Zhang Y. Cell death, IL-1 cytokines, and tumor progression. Cancer Cell. 2025;43:817-819. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 21. | Luo G, Jin K, Deng S, Cheng H, Fan Z, Gong Y, Qian Y, Huang Q, Ni Q, Liu C, Yu X. Roles of CA19-9 in pancreatic cancer: Biomarker, predictor and promoter. Biochim Biophys Acta Rev Cancer. 2021;1875:188409. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 323] [Cited by in RCA: 264] [Article Influence: 52.8] [Reference Citation Analysis (1)] |
| 22. | Grünwald BT, Devisme A, Andrieux G, Vyas F, Aliar K, McCloskey CW, Macklin A, Jang GH, Denroche R, Romero JM, Bavi P, Bronsert P, Notta F, O'Kane G, Wilson J, Knox J, Tamblyn L, Udaskin M, Radulovich N, Fischer SE, Boerries M, Gallinger S, Kislinger T, Khokha R. Spatially confined sub-tumor microenvironments in pancreatic cancer. Cell. 2021;184:5577-5592.e18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 319] [Article Influence: 63.8] [Reference Citation Analysis (1)] |
| 23. | Hajatdoost L, Sedaghat K, Walker EJ, Thomas J, Kosari S. Chemotherapy in Pancreatic Cancer: A Systematic Review. Medicina (Kaunas). 2018;54:48. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 54] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 24. | Xu X, Qu J, Zhang Y, Qian X, Chen T, Liu Y. Development and validation of an MRI-radiomics nomogram for the prognosis of pancreatic ductal adenocarcinoma. Front Oncol. 2023;13:1074445. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 25. | Huang J, Zhang Q, Ge Y, Zheng R, Yang M, Sun Y, Go VLW, Zhang Z, Fang H, Liu J, Guo J, Xiao GG. Serum microRNA-24-based nomogram predicts prognosis for patients with resected pancreatic cancer. Sci Rep. 2025;15:8159. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |