Published online Mar 27, 2026. doi: 10.4240/wjgs.v18.i3.115621
Revised: December 25, 2025
Accepted: January 22, 2026
Published online: March 27, 2026
Processing time: 126 Days and 3.5 Hours
Pancreaticoduodenectomy (PD) is a key surgical procedure for treating pancreatic head and periampullary tumors. Postoperative anastomotic leakage (AL) is a common, yet serious, complication. However, the relationship between the timing of early enteral nutrition (EEN) and the risk for AL remains controversial, which prompted this single-center retrospective cohort study including data from patients who underwent PD at the authors’ institution between January 2023 and December 2024.
To investigate the correlation between the timing of initiating EEN and the in
Patients who underwent PD were divided into 2 groups (n = 100 each) based on initiation of postoperative EEN: Study (≤ 24 hours); and control (> 72 hours). The incidence of total postoperative AL, pancreatic, biliary, and gastrointestinal fistulas, serum albumin (ALB), prealbumin (pre-ALB), and transferrin (TF), time to recovery of bowel sounds, first flatus and defecation, and length of post
Serum ALB, pre-ALB, and TF levels in the study group were significantly higher, and times to recovery of bowel sounds, first flatus and defecation, and length of postoperative hospital stay were significantly shorter in the study group than those in the control group (P < 0.05). Furthermore, the total incidence of AL and grade B/C pancreatic fistulas were significantly lower (P < 0.05), with no statistical difference in the incidence of biliary and gastrointestinal fistulas between the 2 groups (P > 0.05). Multivariate logistic regression analysis revealed that initiating EEN ≤ 24 hours after surgery was an independent protective factor against AL, whereas soft pancreatic texture, pancreatic duct diameter < 3 mm, and prolonged operative duration were independent risk factors.
Initiating EEN ≤ 24 hours after PD reduces the incidence of total AL and pancreatic fistulas, effectively improving postoperative nutritional status, accelerating recovery of gastrointestinal function, and shortening hospital stay.
Core Tip: This retrospective cohort study, involving data from 200 patients who underwent pancreaticoduodenectomy, revealed that initiating early enteral nutrition (EEN) ≤ 24 hours versus > 72 hours postoperatively, was an independent protective factor against total anastomotic leakage and clinically relevant (grade B/C) pancreatic fistulas. EEN was also associated with significantly improved nutritional status (serum albumin, prealbumin, and transferrin levels), accelerated recovery of gastrointestinal function, and shortened hospital stay. These findings challenge traditional delayed feeding practices and support the integration of EEN into enhanced recovery pathways for this major surgery.
- Citation: Yan B, Zhang LQ, Shen YQ. Correlation between the initiation of early enteral nutrition and anastomotic fistula after pancreaticoduodenectomy. World J Gastrointest Surg 2026; 18(3): 115621
- URL: https://www.wjgnet.com/1948-9366/full/v18/i3/115621.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i3.115621
Pancreaticoduodenectomy (PD) is the preferred radical surgical procedure for pancreatic head, periampullary, distal common bile duct, and duodenal tumors. However, this procedure is characterized by a wide resection range, multiple anastomoses (pancreaticoenteric, biliary enteric, and gastroenteric), and severe trauma, leading to a persistently high incidence of postoperative complications. Among these complications, anastomotic leakage (AL) is the most common and consequential, and significantly prolongs hospital stay, increases medical costs, and impedes patient recovery[1].
The traditional view holds that the gastrointestinal tract is in a “paralytic” state in the early postoperative period. Thus, initiating enteral nutrition (EN) too early may increase anastomotic tension and stimulate digestive juice secretion, thereby inducing or exacerbating AL. Therefore, in clinical practice, EN is often delayed for 3-5 days postoperatively or even later, and parenteral nutrition support is preferentially adopted[2]. However, with the promotion of the enhanced recovery after surgery (ERAS) concept, the implementation of postoperative EN has been gradually optimized. Previous studies have suggested that early EN (EEN) can maintain the integrity of the intestinal mucosal barrier, reduce bacterial translocation, regulate immune function, and promote recovery from intestinal peristalsis, thereby lowering the incidence of infectious complications[3,4]. Nevertheless, for PD, a highly complex and high-risk abdominal surgery, considerable controversy remains regarding the safety of EEN, particularly its impact on the incidence of AL[5].
In view of this, the present study retrospectively analyzed data from 200 patients who underwent PD and systematically compared the effects of EN initiation within 24 hours postoperatively (study group) vs 72 hours postoperatively (control group) on AL and other selected postoperative outcomes. We aimed to provide evidence for optimizing postoperative nutritional management strategies for PD, reducing the incidence of complications, and improving patient prognosis.
This single-center retrospective cohort study included data from 200 patients who underwent PD (Child or Whipple procedure) at the authors’ hospital between January 2023 and December 2024. The inclusion criteria were as follows: (1) Age ≥ 18 years; (2) Successful indwelling of jejunal feeding tube after surgery and receiving EN support; (3) Complete clinical data; and (4) Informed consent obtained from patients and their family members. The exclusion criteria were as follows: (1) Combined resection of other organs during surgery (such as hepatic lobectomy, colectomy, etc.); (2) Preoperative severe malnutrition [body mass index (BMI) < 18.5 kg/m2 or serum albumin (ALB) < 30 g/L] without correction; (3) Inability to tolerate EN due to critical postoperative condition; and (4) Death within 24 hours after surgery.
Based on the above inclusion and exclusion criteria, 200 patients were ultimately enrolled and divided into 2 groups according to the time of postoperative initiation of EN: Study [≤ 24 hours (n = 100)]; and control [> 72 hours (n = 100)]. This study was approved by the ethics committee of the authors’ hospital.
All surgeries were performed by an experienced team of hepatobiliary and pancreatic surgeons using surgical approaches including the Child or Whipple methods[6]. Pancreaticoenteric anastomosis was mainly performed using the pancreatic duct-to-jejunal mucosa anastomosis technique, biliary-enteric anastomosis adopted the choledochojejunal end-to-side anastomosis, and gastroenteric anastomosis used either gastrojujenal end-to-side anastomosis or duodenojejunal end-to-side anastomosis. During surgery, abdominal drainage tubes were routinely placed (one near the pancreaticoenteric anastomosis and another near the biliary-enteric anastomosis). Postoperative management followed the ERAS principles, including multimodal analgesia, early ambulation, and goal-directed fluid therapy. Both groups received the same basic postoperative treatment(s), such as anti-infection and acid suppression therapies, and pancreatic enzyme secretion inhibition (using somatostatin analogs).
Patients in both groups received intervention with intact protein EN formulas (e.g., Nutrison, Nutricia/Danone, Hoofddorp, The Netherlands), which are rich in proteins, carbohydrates, lipids, vitamins, and minerals, and provide a complete spectrum of nutrients. The nutritional solution was continuously infused at a constant rate via a dedicated infusion pump to ensure stability and uniformity of the energy and nitrogen supply. If the target energy requirement could not be met through the enteral route, parenteral nutrition supplementation was initiated, and glucose, fat emulsions, and compound amino acid solutions were infused via the peripheral or central veins to ensure that patients obtained sufficient energy and nitrogen sources, maintaining anabolic metabolism and homeostasis of physiological functions. In the study group, within 24 hours after surgery (usually 6-12 hours postoperatively, after vital signs sta
The choice to initiate EN after > 72 hours in the control group reflects conventional postoperative nutritional practices that were still commonly adopted in the authors’ institution during the early study period, particularly in patients undergoing high-risk pancreatic surgery. At that time, concerns regarding anastomotic safety and gastrointestinal intolerance often led to delayed enteral feeding beyond 72 hours.
Incidence of AL[7] pancreatic, biliary, and gastrointestinal AL. The diagnostic criteria were based on international universal standards, as follows.
Pancreatic fistula: According to the 2016 International Study Group on Pancreatic Surgery criteria, starting from 3rd postoperative day 3, if the amylase concentration in the drainage fluid exceeded 3 times the upper limit of normal serum amylase, accompanied by clinically relevant impacts, defined as grade B or C pancreatic fistula. This study mainly focused on the occurrence of grade B/C pancreatic fistulas[8].
Biliary fistula: Defined as a bilirubin concentration in postoperative drainage fluid higher than 3 times the serum bilirubin level for > 3 consecutive days, or requiring intervention or surgical treatment.
Gastrointestinal AL: Contrast-medium extravasation at the anastomotic site detected by imaging examinations (such as computed tomography or contrast radiography) or confirmed AL during intraoperative exploration. Total AL incidence: Patients with any of the above types of AL after surgery were included in the total AL statistics.
Time to recovery of gastrointestinal function: Time to recovery of bowel sounds (first detection of bowel sounds after surgery), time to first flatus, and time to first defecation.
Hospital stay: Number of postoperative hospital days.
Changes in nutritional indicators: Serum ALB, pre-ALB, and transferrin (TF) levels were compared preoperatively vs postoperative day 7.
Statistical analysis was performed using SPSS version 26.0 (IBM Corporation, Armonk, NY, United States). Measurement data are expressed as mean ± SD and the independent samples t-test was used for between-group comparisons. Count data are expressed as n (%), and the χ2 test or Fisher’s exact test was used for between-group comparisons. Differences with P < 0.05 were considered to be statistically significant. Univariate and multivariate logistic regression analyses were used to calculate odds ratios (OR) and corresponding 95% confidence interval (CI) to identify risk factors for AL.
There were no statistically significant differences in baseline data between the 2 groups, including age, sex, BMI, preoperative comorbidities (diabetes mellitus, hypertension), type of primary disease, surgical method (Child/Whipple), operative duration, intraoperative blood loss, pancreatic texture (soft/hard), and pancreatic duct diameter (≥ 3 mm/< 3 mm) (all P > 0.05), thus confirming comparability between the 2 groups (Table 1).
| Characteristic | Group | χ2/t | P value | |
| Study (n = 100) | Control (n = 100) | |||
| Age, years | 52.36 ± 8.72 | 53.18 ± 9.21 | 0.647 | 0.519 |
| Sex (male/female) | 58/42 | 61/39 | 0.187 | 0.666 |
| Body mass index, kg/m2 | 23.57 ± 2.82 | 23.85 ± 3.19 | 0.658 | 0.512 |
| Diabetes mellitus | 28 (28.00) | 32 (32.00) | 0.381 | 0.537 |
| Hypertension | 35 (35.00) | 38 (38.00) | 0.194 | 0.660 |
| Primary diagnosis | 1.241 | 0.743 | ||
| Carcinoma of head of pancreas | 52 (52.00) | 55 (55.00) | ||
| Ampullary carcinoma | 25 (25.00) | 22 (22.00) | ||
| Distal cholangiocarcinoma | 15 (15.00) | 18 (18.00) | ||
| Others | 8 (8.00) | 5 (5.00) | ||
| Operative procedure | 0.500 | 0.480 | ||
| Child method | 78 (78.00) | 82 (82.00) | ||
| Whipple procedure | 22 (22.00) | 18 (18.00) | ||
| Operative duration, minute | 320.54 ± 55.27 | 325.84 ± 60.13 | 0.649 | 0.517 |
| Intraoperative blood loss, mL | 450.21 ± 80.58 | 465.33 ± 95.78 | 1.208 | 0.228 |
| Pancreatic texture | 0.325 | 0.569 | ||
| Soft | 42 (42.00) | 46 (46.00) | ||
| Firm | 58 (58.00) | 54 (54.00) | ||
| Main pancreatic duct diameter, mm | 0.202 | 0.653 | ||
| ≥ 3 | 65 (65.00) | 68 (68.00) | ||
| < 3 | 35 (35.00) | 32 (32.00) | ||
On postoperative day 7, serum levels of ALB, pre-ALB, and TF in both groups decreased by varying degrees compared with those before surgery. However, the decrease in amplitude in the study group was significantly lower than that in the control group, and serum levels of ALB, pre-ALB, and TF in the study group on postoperative day 7 were significantly higher than those in the control group (P < 0.05) (Table 2).
| Group | n | ALB (g/L) | Pre-ALB (mg/L) | TF (g/L) | |||
| Before | After | Before | After | Before | After | ||
| Study | 100 | 38.28 ± 4.10 | 37.80 ± 3.87 | 220.53 ± 55.32 | 215.68 ± 51.37 | 2.48 ± 0.43 | 2.43 ± 0.39 |
| Control | 100 | 37.85 ± 4.54 | 32.57 ± 3.76 | 215.86 ± 58.72 | 187.69 ± 50.12 | 2.46 ± 0.38 | 1.97 ± 0.45 |
| t | 0.703 | 9.693 | 0.579 | 3.900 | 0.349 | 7.725 | |
| P value | 0.483 | 0.000 | 0.563 | 0.000 | 0.728 | 0.000 | |
The time to recovery of bowel sounds, times to first flatus and defecation, and length of postoperative hospital stay in the study group were significantly shorter than those in the control group (P < 0.05) (Table 3).
| Index | Study group (n = 100) | Control group (n = 100) | t | P value |
| Return of bowel sounds, hour | 38.56 ± 12.32 | 62.88 ± 18.73 | 10.848 | < 0.001 |
| Time to first flatus, hour | 48.21 ± 15.65 | 78.56 ± 22.48 | 11.080 | < 0.001 |
| Time to first defecation, hour | 72.43 ± 20.10 | 105.38 ± 28.95 | 9.349 | < 0.001 |
| Postoperative hospital stay, days | 14.25 ± 3.83 | 18.77 ± 5.24 | 6.964 | < 0.001 |
The incidences of total AL and grade B/C pancreatic fistulas in the study group were lower than those in the control group (P < 0.05). There were no statistically significant differences in the incidence of biliary fistula, gastrointestinal AL, and the incidence of ≥ 2 types of fistulas between the 2 groups (P > 0.05) (Table 4).
| Type of AL | Study (n = 100) | Control (n = 100) | χ2 | P value | OR (95%CI) |
| Overall AL | 18 (18.00) | 32 (32.00) | 5.227 | 0.022 | 0.467 (0.241-0.904) |
| Pancreatic fistula (grade B/C) | 12 (12.00) | 25 (25.00) | 5.604 | 0.018 | 0.409 (0.192-0.870) |
| Bile leak | 6 (6.00) | 9 (9.00) | 0.617 | 0.432 | 0.652 (0.223-1.907) |
| Gastroenteric AL | 3 (3.00) | 6 (6.00) | - | 0.498a | 0.500 (0.129-1.950) |
| ≥ 2 concurrent fistulas | 2 (2.00) | 5 (5.00) | - | 0.445a | 0.400 (0.079-2.014) |
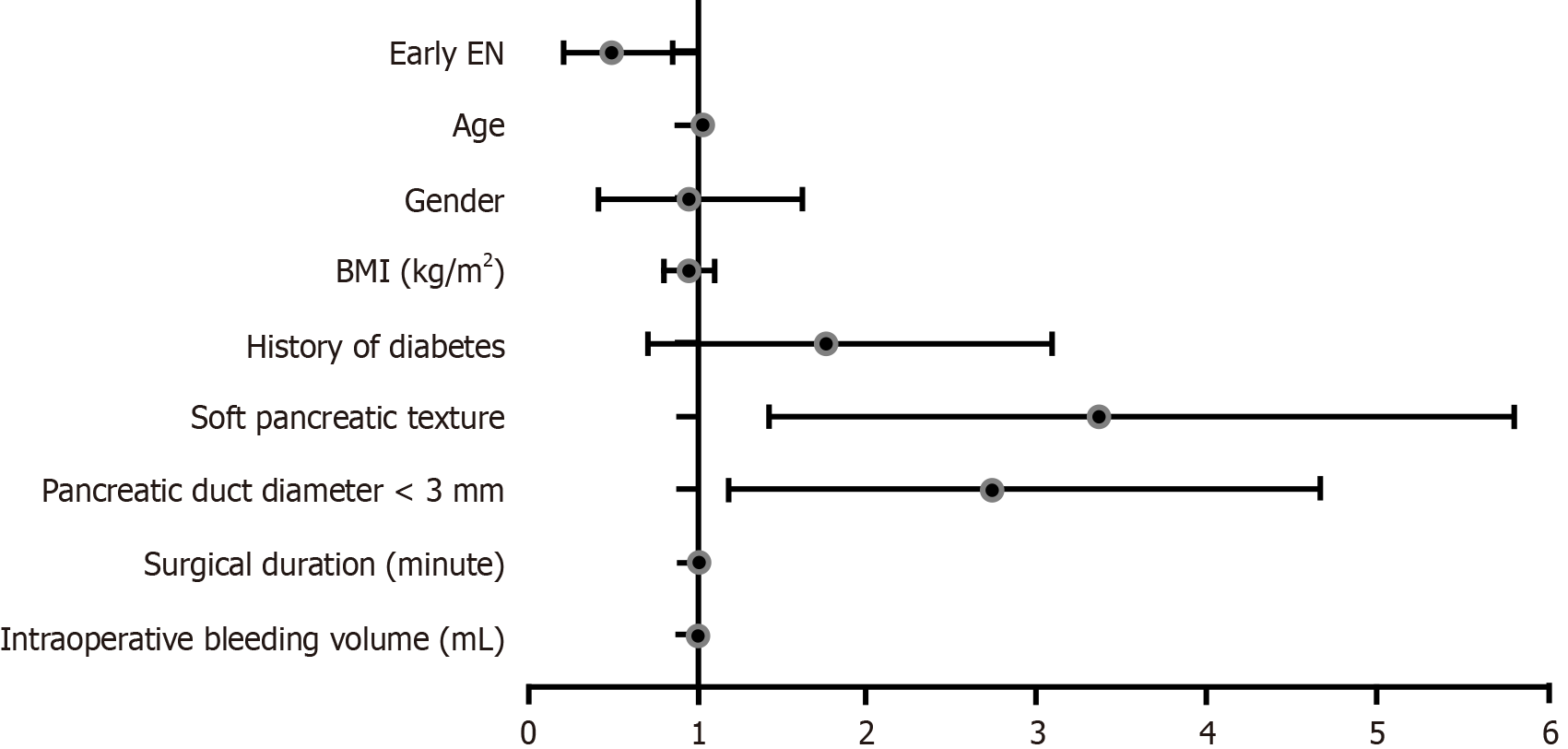
To confirm whether initiating EN ≤ 24 hours after surgery was an independent protective factor for AL and to control for the influence of confounding variables, multivariate logistic regression analysis on the occurrence of total AL. The occurrence of AL was set as the dependent variable (0 = no occurrence, 1 = occurrence), and the included variables were as follows: Sex (0 = female, 1 = male), history of diabetes mellitus (0 = no, 1 = yes), pancreatic texture (0 = hard pancreas, 1 = soft pancreas), pancreatic duct diameter (0 = pancreatic duct ≥ 3 mm, 1 = pancreatic duct < 3 mm), and timing of EN initiation (0 ≥ 72 hours, 1 ≤ 24 hours). Age, operative duration, and intraoperative blood loss were the continuous variables.
Results revealed that initiation of EEN was an independent protective factor for AL, whereas soft pancreatic texture, pancreatic duct diameter < 3 mm, and prolonged operative duration were independent risk factors for AL (Table 5 and Figure 1).
| Variable | β coefficient | SE | χ2 | P value | OR (95%CI) |
| Early enteral nutrition | -0.868 | 0.362 | 5.75 | 0.016 | 0.419 (0.210-0.850) |
| Age, years | 0.028 | 0.018 | 2.418 | 0.120 | 1.028 (0.993-1.065) |
| Sex | -0.204 | 0.351 | 0.339 | 0.561 | 0.815 (0.412-1.618) |
| Body mass index, kg/m2 | -0.041 | 0.062 | 0.439 | 0.507 | 0.959 (0.851-1.078) |
| History of diabetes | 0.387 | 0.378 | 1.050 | 0.307 | 1.472 (0.703-3.088) |
| Soft pancreatic texture | 1.055 | 0.361 | 8.539 | 0.003 | 2.871 (1.420-5.810) |
| Pancreatic duct < 3 mm | 0.854 | 0.359 | 5.659 | 0.015 | 2.348 (1.180-4.670) |
| Operative duration, minute | 0.008 | 0.003 | 7.111 | 0.008 | 1.008 (1.002-1.014) |
| Intraoperative blood loss, mL | 0.002 | 0.001 | 4.000 | 0.134 | 1.002 (1.000-1.004) |
AL is a common complication of PD that seriously affects postoperative recovery[9]. This study systematically evaluated the effect of EN initiation timing on the incidence of AL and overall postoperative rehabilitation through a retrospective cohort analysis. Results revealed that EEN significantly reduced the incidence of AL (18% vs 32%) and significantly reduced the incidence of pancreatic fistulas (grade B/C) (12% vs 25%). In addition, EEN significantly improved nutri
Traditional clinical practice generally suggests that the early postoperative period is a high-risk period for anastomotic complications. Initiating EN too early may increase anastomotic tension through mechanisms such as increasing intestinal mechanical stretch, stimulating hormone secretion, or enhancing intestinal peristalsis, thereby inducing AL[10]. More recent physiological studies have shown that the intestine, an important organ for maintaining immune homeostasis, can benefit from early nutritional intervention to exert its barrier function. Intestinal mucosal epithelial cells have a short renewal cycle (approximately 3-5 days), and the integrity of their structure highly depends on the continuous supply of nutritional substrates in the intestinal lumen, especially key nutrients such as glutamine and short-chain fatty acids[11,12]. Long-term postoperative fasting or delayed EN can lead to intestinal mucosal atrophy, downregulation of tight junction protein expression, and increased intestinal permeability, thereby promoting bacterial and endotoxin translocation, inducing a systemic inflammatory response, and indirectly destroying the microenvironment required for anastomotic healing[13,14]. Results of this study revealed that serum levels of ALB, pre-ALB, and TF in the study group on postoperative day 7 were significantly higher than those in the control group, suggesting that EEN can effectively alleviate postoperative negative nitrogen balance, reduce the consumption of acute-phase proteins, and provide the necessary metabolic support for tissue repair[15,16]. These nutritional and metabolic advantages may enhance the tensile strength of the anastomosis by inhibiting excessive inflammatory responses, improving local tissue oxygenation, and promoting collagen deposition, thereby reducing the risk for AL[16]. It should be noted that surgeons may have been more inclined to initiate EEN in patients with relatively stable intraoperative conditions. Although baseline characteristics, including known surgical risk factors, such as pancreatic texture, duct diameter, operative time, and blood loss, were well balanced between the groups, the influence of unmeasured confounders, such as subtle technical variations in anastomosis or intraoperative tissue perfusion, cannot be completely ruled out.
Multivariate logistic regression further confirmed that initiating EN within 24 hours after surgery was an independent protective factor for AL (OR = 0.419, 95%CI: 0.210-0.850; P = 0.016), indicating that EEN administration can significantly reduce the incidence of AL. The protective effect of EN can be achieved through multi-target regulation of the neuroendocrine-immune network. First, nutritional substrates directly stimulate the intestinal mucosa to release gastrointestinal hormones, such as cholecystokinin and motilin, promoting the rhythmic emptying of bile and pancreatic juice and reducing high pressure in the pancreatic duct[17]. Second, the oral or tube feeding route activates the gut–brain axis, and the vagal nerve reflex inhibits the release of pro-inflammatory factors, such as tumor necrosis factor-alpha and interleukin-6, creating an immune microenvironment conducive to anastomotic healing[18]. Finally, early recovery of intestinal peristalsis can accelerate the drainage and absorption of abdominal inflammatory exudates, reducing the chemical erosion and mechanical compression of local effusion on the anastomosis[19]. The synergistic effects of these mechanisms reduce the risk for AL.
In this study, there was no statistically significant difference in the incidence of gastrointestinal anastomotic and biliary fistulas between the 2 groups, which may have been limited by the sample size or related to differences in their pathophysiological mechanisms. In contrast, the occurrence of pancreatic fistula depends more significantly on the integrity of the pancreatic exocrine function and the accuracy of the anastomotic technique[20]. Biliary fistulas are closely related to factors, such as blood supply to the bile duct, anastomotic tension, and local infection[21]. Gastrointestinal anastomosis has a relatively low incidence of postoperative fistulas because of its abundant blood supply and strong tissue-healing ability[22]. EEN may have a specific protective effect on pancreatic fistulas, and its mechanism may involve the “physiological regulation” of pancreatic exocrine function[23]. More specifically, the intake of a small amount of nutrients in the early postoperative period can induce rhythmic secretion of pancreatic enzymes, preventing the accumulation of pancreatic juice in the pancreatic duct and formation of high pressure in the pancreatic duct[24]. In contrast, in the control group, due to delayed feeding, pancreatic juice may have accumulated in the pancreatic duct, and if there are minor defects in the anastomosis, leakage is more likely to occur under high pressure[25]. This result is consistent with the results of a multicenter randomized controlled trial reporting that initiating EN within 24 hours after surgery reduced the incidence of clinically relevant pancreatic fistula after PD by 40% without aggravating delayed gastric emptying[26].
This study has several limitations. First, it adopted a retrospective observational design, which is inherently subject to methodological constraints. Although multivariate logistic regression analysis was performed to adjust for known confounding variables, causal inferences between the timing of EN initiation and postoperative outcomes could not be definitively established, and residual confounding from unmeasured factors remained possible. Second, patient allocation to the early and delayed EN groups was not randomized. The timing of EN initiation was influenced by the evolution of institutional perioperative nutritional management strategies and clinical decision-making during different periods, rather than by strict random assignment. Consequently, selection bias cannot be completely excluded. Although the baseline characteristics and several well-established surgical risk factors, including pancreatic texture, pancreatic duct diameter, operative duration, and intraoperative blood loss, were comparable between the groups, unmeasured confounders, such as subtle intraoperative conditions, technical details of the anastomosis, tissue perfusion, and surgeon judgment, may have influenced both nutritional strategy selection and postoperative outcomes. Third, the control group was defined by the initiation of EN > 72 hours postoperatively, which represents a relatively conservative feeding strategy compared with current ERAS recommendations, in which EN is commonly initiated within 48-72 hours. This grouping strategy may have amplified the observed benefits of EEN and potentially overestimated the effect size. Nevertheless, delayed EN remains a common practice in many centers, particularly following high-risk pancreatic surgery and, therefore, retains its clinical relevance. Fourth, although this study focused on AL and nutritional and gastrointestinal recovery outcomes, some secondary safety endpoints, such as delayed gastric emptying, reoperation rates, and other postoperative complications, were not comprehensively evaluated. The absence of these data limited the ability to fully assess the overall safety profile of EEN in this cohort. Finally, this was a single-center study, and the findings may have been influenced by local surgical expertise, perioperative management protocols, and the degree of ERAS implementation. Therefore, the generalizability of our results to other institutions and healthcare settings may be limited. Future multicenter, prospective, randomized controlled trials with broader safety outcome assessments and refined comparisons of different early feeding windows are required to validate and extend these findings.
Initiating EN ≤ 24 hours after PD is not only safe and feasible, but also significantly reduces the incidence of total AL and clinically relevant pancreatic fistula (grade B/C), improves nutritional status, accelerates the rehabilitation process, and shortens the length of hospital stay. EEN was an independent protective factor against AL. These results provide a strong basis for nutritional management after PD, helps optimize perioperative management, improves patient prognosis, and has important clinical value.
| 1. | Russell TB, Labib PL, Denson J, Streeter A, Ausania F, Pando E, Roberts KJ, Kausar A, Mavroeidis VK, Marangoni G, Thomasset SC, Frampton AE, Lykoudis P, Maglione M, Alhaboob N, Bari H, Smith AM, Spalding D, Srinivasan P, Davidson BR, Bhogal RH, Croagh D, Dominguez I, Thakkar R, Gomez D, Silva MA, Lapolla P, Mingoli A, Porcu A, Shah NS, Hamady ZZR, Al-Sarrieh BA, Serrablo A; RAW Study Collaborators, Aroori S. Postoperative complications after pancreatoduodenectomy for malignancy: results from the Recurrence After Whipple's (RAW) study. BJS Open. 2023;7:zrad106. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (1)] |
| 2. | Triantafillidis JK, Papakontantinou J, Antonakis P, Konstadoulakis MM, Papalois AE. Enteral Nutrition in Operated-On Gastric Cancer Patients: An Update. Nutrients. 2024;16:1639. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 30] [Article Influence: 15.0] [Reference Citation Analysis (1)] |
| 3. | Hu MM, Ding YL, Li J. Effectiveness of early enteral nutrition support in patients undergoing gastrointestinal perforation repair surgery within the enhanced recovery. World J Gastrointest Surg. 2025;17:106384. [PubMed] [DOI] [Full Text] |
| 4. | Liu X, Chen Q, Fu Y, Lu Z, Chen J, Guo F, Li Q, Wu J, Gao W, Jiang K, Dai C, Miao Y, Wei J. Early Nasojejunal Nutrition Versus Early Oral Feeding in Patients After Pancreaticoduodenectomy: A Randomized Controlled Trial. Front Oncol. 2021;11:656332. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 5. | Jeganathan NNA, Koltun WA. Special Considerations of Anastomotic Leaks in Crohn's Disease. Clin Colon Rectal Surg. 2021;34:412-416. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 6. | Wang K, Dong SS, Zhang W, Ni YY, Xie F, Wang JC, Wang XH, Li YW. Surgical methods influence on the risk of anastomotic fistula after pancreaticoduodenectomy: a systematic review and network meta-analysis. Surg Endosc. 2023;37:3380-3397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 7. | Russell TB, Aroori S. Procedure-specific morbidity of pancreatoduodenectomy: a systematic review of incidence and risk factors. ANZ J Surg. 2022;92:1347-1355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 8. | Bassi C, Marchegiani G, Dervenis C, Sarr M, Abu Hilal M, Adham M, Allen P, Andersson R, Asbun HJ, Besselink MG, Conlon K, Del Chiaro M, Falconi M, Fernandez-Cruz L, Fernandez-Del Castillo C, Fingerhut A, Friess H, Gouma DJ, Hackert T, Izbicki J, Lillemoe KD, Neoptolemos JP, Olah A, Schulick R, Shrikhande SV, Takada T, Takaori K, Traverso W, Vollmer CM, Wolfgang CL, Yeo CJ, Salvia R, Buchler M; International Study Group on Pancreatic Surgery (ISGPS). The 2016 update of the International Study Group (ISGPS) definition and grading of postoperative pancreatic fistula: 11 Years After. Surgery. 2017;161:584-591. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3570] [Cited by in RCA: 3314] [Article Influence: 368.2] [Reference Citation Analysis (4)] |
| 9. | Labori KJ, Tholfsen T, Yaqub S, Lassen K, Kleive D, Waage A. Gastro- or Duodenojejunostomy Leaks After Pancreatoduodenectomy: Single Center Experience and Narrative Literature Review. J Gastrointest Surg. 2021;25:3130-3136. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 10. | Lee SY, Han EC. Impact of Early Oral Feeding on Postoperative Outcomes after Elective Colorectal Surgery: A Systematic Review and Meta-Analysis. Dig Surg. 2025;42:26-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 11. | Ma T, Xue X, Tian H, Zhou X, Wang J, Zhao Z, Wang M, Song J, Feng R, Li L, Jing C, Tian F. Effect of the gut microbiota and their metabolites on postoperative intestinal motility and its underlying mechanisms. J Transl Med. 2023;21:349. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 18] [Reference Citation Analysis (1)] |
| 12. | Nakamura A, Matsumoto M. Role of polyamines in intestinal mucosal barrier function. Semin Immunopathol. 2025;47:9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 16] [Article Influence: 16.0] [Reference Citation Analysis (1)] |
| 13. | Pan C, Zhang H, Zhang L, Chen L, Xu L, Xu N, Liu X, Meng Q, Wang X, Zhang ZY. Surgery-induced gut microbial dysbiosis promotes cognitive impairment via regulation of intestinal function and the metabolite palmitic amide. Microbiome. 2023;11:248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 40] [Reference Citation Analysis (1)] |
| 14. | Frizon E, Aguilar-Nascimento JE, Zanini JC, Roux MS, Schemberg BCL, Tonello PL, Dock-Nascimento DB. EARLY REFEEDING AFTER COLORECTAL CANCER SURGERY REDUCES COMPLICATIONS AND LENGTH OF HOSPITAL STAY. Arq Bras Cir Dig. 2025;37:e1854. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 15. | Dhiraaj S, Thimmappa L, Issac A, Halemani K, Mishra P, Mavinatop A. The Impact of Early Enteral Nutrition on Post-operative Hospital Stay and Complications in Infants Undergoing Congenital Cardiac Surgery: A Systematic Review and Meta-analysis. J Caring Sci. 2023;12:14-24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 16. | Okamoto Y, Sakaguchi T, Ikematsu Y, Kanai T, Hirayama K, Tamura H, Hayashi T, Nishiwaki Y, Konno H, Aoki K. Early enteral nutrition with arginine compensates for negative nitrogen balance in patients undergoing curative total gastrectomy. J Med Invest. 2023;70:325-333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 17. | Abouarab AG, Mohsen AH, Mosellhy NE, Mousa MI, Abbas O, Manea H. Evaluating the Impact of Enteral Nutrition vs Parenteral Nutrition on Postoperative Outcomes in Esophageal Cancer Patients After Esophagectomy: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Cureus. 2025;17:e92413. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (2)] |
| 18. | He C, Shi H, Yu Z, Ma C, Jiao Z, Li J, Yang F. The progress of the microbe-gut-brain axis in sepsis-associated encephalopathy. Front Cell Infect Microbiol. 2025;15:1587463. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 19. | Huang SQ, Wen Y, Sun HY, Deng J, Zhang YL, Huang QL, Wang B, Luo ZL, Tang LJ. Abdominal paracentesis drainage attenuates intestinal inflammation in rats with severe acute pancreatitis by inhibiting the HMGB1-mediated TLR4 signaling pathway. World J Gastroenterol. 2021;27:815-834. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 13] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 20. | Kato H, Asano Y, Ito M, Kawabe N, Arakawa S, Shimura M, Koike D, Hayashi C, Kamio K, Kawai T, Ochi T, Yasuoka H, Higashiguchi T, Tochii D, Kondo Y, Nagata H, Utsumi T, Horiguchi A. The usefulness of preoperative exocrine function evaluated by the (13)C-trioctanoin breath test as a significant physiological predictor of pancreatic fistula after pancreaticoduodenectomy. BMC Surg. 2022;22:49. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 21. | Yin R, Xu S, Gong Y, Zhu J, Zhu H, Tao Y, Li X. The risk factors of biliary fistula after radical resection of perihilar cholangiocarcinoma in elderly patients and its influence on prognosis: a retrospective cohort study. J Gastrointest Oncol. 2023;14:390-404. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (4)] |
| 22. | Wang J, Li MH. Risk factors for anastomotic fistula development after radical colon cancer surgery and their impact on prognosis. World J Gastrointest Surg. 2023;15:2470-2481. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 23. | Yuan J, Xiu D. Effects of early enteral nutrition on pancreatic fistula and long-term prognosis after distal pancreatectomy or enucleation of pancreatic tumours in a major academic university hospital in China: protocol for a single-centre randomised controlled trial. BMJ Open. 2023;13:e068469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 24. | Lakananurak N, Gramlich L. Nutrition management in acute pancreatitis: Clinical practice consideration. World J Clin Cases. 2020;8:1561-1573. [PubMed] [DOI] [Full Text] |
| 25. | Li JF, Xie MJ, Wei JX, Yang CN, Chen GW, Li LQ, Zhao YN, Liu LJ, Xie S. Common bile duct stump stones and recurrent acute pancreatitis after Roux-en-Y choledochojejunostomy in a child: A case report. World J Gastrointest Surg. 2025;17:102105. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 26. | Joliat GR, Martin D, Labgaa I, Melloul E, Uldry E, Halkic N, Fotsing G, Cristaudi A, Majno-Hurst P, Vrochides D, Demartines N, Schäfer M. Early enteral vs. oral nutrition after Whipple procedure: Study protocol for a multicentric randomized controlled trial (NUTRIWHI trial). Front Oncol. 2022;12:855784. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |