Published online Mar 27, 2026. doi: 10.4240/wjgs.v18.i3.115497
Revised: December 12, 2025
Accepted: January 22, 2026
Published online: March 27, 2026
Processing time: 140 Days and 3.2 Hours
Biliary atresia is a progressive fibro-obliterative cholangiopathy of infancy treated primarily by Kasai portoenterostomy (KPE) to restore bile flow. Postoperative pain control remains challenging and may affect recovery and prognosis.
To assess whether an early, structured pain management protocol reduces com
This study included 120 infants with biliary atresia who underwent KPE at The First Affiliated Hospital of the University of South China from March 2022 to March 2025. Using random allocation, infants were divided into an observation group (n = 60) receiving multimodal analgesia (ultrasound-guided transversus abdominis plane block plus patient-controlled intravenous analgesia), and a control group (n = 60) receiving on-demand opioid analgesia. Groups were com
The observation group showed lower rates of postoperative cholangitis, pulmonary infection, and total complications than the control group (P < 0.05). Face, Legs, Activity, Cry, Consolability scores at 6-72 hours postoperatively were significantly lower in the observation group, with a marked group-time interaction. Ramsay scores were higher at 6 hours and 12 hours but lower at 24 hours and 48 hours, also demonstrating significant group-time interaction (P < 0.05). The observation group required less opioid use within 48 hours and had shorter times to first defecation, Pediatric Intensive Care Unit (PICU) stay, and total hospitalization (P < 0.05). Six-month native liver survival was higher in the observation group (76.7%, 46/60) than in the control group (61.7%, 37/60; P < 0.05).
Early multimodal analgesia combining ultrasound-guided transversus abdominis plane block and patient-controlled intravenous analgesia provides safer, more effective analgesia, reduces complications, and enhances postoperative recovery and native liver survival after KPE.
Core Tip: This study demonstrates that an early multimodal analgesia protocol, utilizing ultrasound-guided transversus abdominis plane block combined with patient-controlled intravenous analgesia, is superior to conventional on-demand opioid analgesia for infants after Kasai surgery. This approach not only provides superior and safer pain control but is also associated with reduced postoperative complications (particularly cholangitis and pulmonary infection), faster recovery, and significantly improved 6-month native liver survival.
- Citation: Yan ZJ, Guo XH, Huang ZX, Huang J, Liu WJ. Impact of early postoperative pain management strategies on complication rates in infants with biliary atresia after Kasai portoenterostomy. World J Gastrointest Surg 2026; 18(3): 115497
- URL: https://www.wjgnet.com/1948-9366/full/v18/i3/115497.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i3.115497
Biliary atresia is one of the most severe diseases of the digestive system in the neonatal and infant periods. Characterized by progressive inflammation and fibrotic obstruction of the intrahepatic and extrahepatic bile ducts, it results in obstructive jaundice and cholestatic liver cirrhosis. Without timely intervention, affected infants may die of liver failure within a short period[1,2]. Currently, hepatoportoenterostomy (Kasai procedure) is the preferred surgical treatment for biliary atresia. By reconstructing the bile drainage pathway, this procedure slows the progression of liver cirrhosis, thereby providing infants the opportunity for native liver survival or creating the conditions for future liver trans
As the Kasai procedure is a complex and invasive abdominal surgery involving extensive hilar dissection and jejunal anastomosis, severe acute postoperative pain is an inevitable clinical concern[5,6]. Historically, postoperative pain management in children - especially after complex abdominal operations - has not received the same level of attention as in adults. The common practice has been an “on-demand” analgesia model in which opioids are administered intramuscularly by nurses only when infants exhibit severe crying. This approach has notable drawbacks, including delayed analgesia, inconsistent efficacy, and large fluctuations in plasma drug concentrations, which easily lead to a vicious cycle of “pain-drug administration-sedation-recurrence of pain”[7,8].
Infants with biliary atresia undergoing Kasai portoenterostomy commonly experience several postoperative complications that substantially affect both short- and long-term outcomes. Among these, postoperative cholangitis is the most frequent and life-threatening complication, often associated with impaired bile drainage, bacterial translocation, and postoperative immunosuppression. Incisional and pulmonary infections may also occur, partly due to reduced mobility, inadequate airway clearance, and stress-induced immune dysfunction. In addition, some infants develop portal hypertension-related events, reflecting progressive hepatic fibrosis and impaired liver recovery.
Growing evidence indicates that severe early postoperative pain may exacerbate stress responses, weaken immune function, impair respiratory effort, reduce effective coughing, and delay gastrointestinal recovery, thereby increasing the risk of cholangitis, pulmonary infection, and other complications. Therefore, optimizing early postoperative analgesia may play a crucial role not only in enhancing patient comfort but also in preventing complications and improving outcomes. Accordingly, this study aimed to determine whether an early, structured multimodal pain management strategy could reduce complication rates and improve short-term postoperative outcomes - including native liver survival - compared with traditional on-demand analgesia in infants undergoing the Kasai procedure.
This study was approved by the Ethics Committee of the First Affiliated Hospital of the University of South China. A total of 120 infants with biliary atresia who underwent the Kasai procedure at the same institution from March 2022 to March 2025 were enrolled. Sample size estimation was based on data from preliminary experiments. With α set at 0.05 (two-tailed), β at 0.2, and a statistical power of 80%, it was calculated that each group required at least 55 participants. Considering a 10% attrition rate, the final sample size was determined to be 60 infants per group, totaling 120 par
Inclusion criteria: Diagnosed with type III biliary atresia based on clinical examination, laboratory findings, and intraoperative exploration; age ≤ 90 days; undergoing the Kasai procedure for the first time and meeting the relevant surgical indications; American Society of Anesthesiologists physical status classification II-III; legal guardians provided informed consent.
Exclusion criteria: Presence of severe congenital heart disease, severe pulmonary or neurological disease; preoperative severe infection or significant coagulation dysfunction; known allergy to any anesthetic or analgesic drug used in the study; coexistence of other congenital syndromes that could interfere with pain assessment; or withdrawal during the study.
A statistician who was not involved in the study generated a random numbered list, which was then sealed. The researchers sequentially numbered the enrolled infants and assigned them to either the observation group or the control group according to this list, with 60 participants in each group. Comparison of baseline characteristics - including sex, age, weight, American Society of Anesthesiologists classification, preoperative total bilirubin level, and operative duration - revealed no statistically significant differences between the two groups (P > 0.05), indicating good baseline comparability (Table 1).
| Variable | n | Male | Female | Age (days) | Weight (kg) | ASA II | ASA III | Preoperative total bilirubin (μmol/L) | Surgery time (minute) |
| Observation | 60 | 32 (53.3) | 28 (46.7) | 65.3 ± 12.5 | 5.2 ± 0.8 | 38 (63.3) | 22 (36.7) | 185.6 ± 45.2 | 215.4 ± 35.6 |
| Control | 60 | 30 (50.0) | 30 (50.0) | 63.8 ± 11.9 | 5.1 ± 0.7 | 35 (58.3) | 25 (41.7) | 192.1 ± 42.7 | 208.9 ± 38.1 |
| Statistical test value | - | χ2 = 0.134 | t = 0.685 | t = 0.721 | χ2 = 0.402 | t = 0.816 | t = 0.965 | ||
| P value | - | 0.714 | 0.495 | 0.472 | 0.526 | 0.416 | 0.337 | ||
Anesthesia method: (1) Upon entering the operating room, all infants were monitored using electrocardiography, noninvasive blood pressure, and pulse oxygen saturation; (2) Anesthesia induction: Midazolam (Jiangsu Nhwa Pharmaceutical Co., Ltd., China; National Drug Approval No. H10980025) was administered intravenously at 0.05 mg/kg, propofol emulsion (Sichuan Guorui Pharmaceutical Co., Ltd.; H20040079) at 3 mg/kg, fentanyl (Jiangsu Nhwa Pharmaceutical Co., Ltd., China; H20113509) at 5 μg/kg, and rocuronium bromide (Zhejiang Xianju Pharmaceutical Co., Ltd., China; H20123186) at 0.6 mg/kg; (3) After induction, endotracheal intubation was performed, and each infant was connected to a ventilator for mechanical ventilation; (4) Anesthesia maintenance: Anesthesia was maintained with inhalation of 1% sevoflurane (Shanghai Hengrui Pharmaceutical Co., Ltd., China; H20070172) combined with continuous intravenous infusion of remifentanil (Jiangsu Nhwa Pharmaceutical Co., Ltd., China; H20143315) at 0.1-0.3 μg/kg/minute; (5) Thirty minutes before the end of surgery, parecoxib sodium (Pfizer Pharmaceuticals Ltd., China; J20130049) was administered intravenously at 0.5 mg/kg for preemptive analgesia; and (6) After surgery, the endotracheal tube was removed once spontaneous breathing was adequately restored, and the infant was transferred to the PICU.
Study protocols: The observation group received a multimodal analgesia protocol primarily consisting of early pos
The control group received a conventional “on-demand” analgesia protocol involving intramuscular or intravenous bolus administration of opioids. When nurses determined that an infant’s Face, Legs, Activity, Cry, Consolability (FLACC) score was ≥ 4, pethidine hydrochloride (Northeast Pharmaceutical Group Shenyang No. 1 Pharmaceutical Co., Ltd., China; National Drug Approval No. H21022436) was administered intramuscularly at 0.5-1 mg/kg as prescribed, or morphine hydrochloride (Northeast Pharmaceutical Group Shenyang No. 1 Pharmaceutical Co., Ltd., China; H21022436) was administered intravenously at 0.05-0.1 mg/kg by slow injection. The interval between two doses was at least 4 hours.
Differences between the two groups were compared in terms of postoperative complications, FLACC scores at multiple time points within 72 hours after surgery, sedation effects, sedative drug dosages, postoperative recovery parameters, and native liver survival at 6 months postoperatively.
Postoperative complications: The occurrence of postoperative complications within 30 days after surgery was recorded, including cholangitis, incisional infection or dehiscence, pulmonary infection, and portal hypertension-related events.
FLACC score: Pain was evaluated by the same nurse, who was blinded to group allocation, at 6, 12, 24, 48, and 72 hours after surgery. The FLACC scale assesses five domains: Face, Legs, Activity, Cry, and Consolability, each scored from 0 to 2. The total score ranges from 0 to 10, with higher scores indicating greater pain intensity.
Sedation assessment and drug dosage: Sedation was evaluated using the Ramsay Sedation Scale by the same nurse, who was blinded to group allocation, at 6, 12, 24, 48, and 72 hours after surgery. The scale classifies the level of sedation from anxiety or restlessness to deep sleep into six grades, scored from 1 to 6. Higher scores indicate deeper sedation.
Sedative drug dosage and postoperative recovery parameters: The following parameters were recorded: Total opioid consumption within 48 hours after surgery (converted to the morphine-equivalent dose), time to recovery of bowel sounds, time to first defecation, time to initiation of oral feeding, length of stay in the PICU, and total hospital stay.
Native liver survival: Infants were followed up for 6 months after surgery, and their native liver survival status was documented, defined as the absence of liver transplantation and satisfactory resolution of jaundice.
Data were analyzed using SPSS version 26.0 (IBM Corp., Armonk, NY, Unites States). Categorical variables were expressed as frequencies and percentages, and intergroup comparisons were performed using the χ2 test or Fisher’s exact test as appropriate. Continuous variables were presented as mean ± SD. The normality of data distribution was assessed using the Shapiro-Wilk test. Differences between groups were analyzed using the independent-samples t-test, and data collected at multiple time points were evaluated using repeated-measures analysis of variance. A two-tailed P < 0.05 was considered statistically significant.
Within 30 days after surgery, the incidences of cholangitis, incisional infection, pulmonary infection, and portal hypertension-related events in the observation group were all lower than those in the control group. Among these, the differences in cholangitis and pulmonary infection rates were statistically significant (P < 0.05), whereas differences in incisional infection and portal hypertension-related events were not statistically significant. The overall complication rate in the observation group was significantly lower than that in the control group (31.7% vs 66.7%, P < 0.001) (Table 2).
| Complication | n | Cholangitis | Incision infection | Pulmonary infection | Portal hypertension-related events | Total complications |
| Observation | 60 | 11 (18.3) | 3 (5.0) | 3 (5.0) | 2 (3.3) | 19 (31.7) |
| Control | 60 | 21 (35.0) | 5 (8.3) | 10 (16.7) | 4 (6.7) | 40 (66.7) |
| χ2 | - | 4.183 | 0.782 | 4.227 | 0.811 | 14.286 |
| P value | - | 0.041 | 0.716 | 0.040 | 0.678 | < 0.001 |
The FLACC scores of the observation group at 6, 12, 24, 48, and 72 hours after surgery were significantly lower than those of the control group. Moreover, a significant group-time interaction was observed, indicating statistically significant differences between groups over time (P < 0.05) (Tables 3 and 4).
| Group | n | 6 hours post-surgery | 12 hours post-surgery | 24 hours post-surgery | 48 hours post-surgery | 72 hours post-surgery |
| Observation | 60 | 2.1 ± 0.8 | 1.9 ± 0.7 | 2.3 ± 0.9 | 2.0 ± 0.6 | 1.8 ± 0.5 |
| Control | 60 | 5.8 ± 1.2 | 4.9 ± 1.1 | 4.5 ± 1.0 | 3.2 ± 0.8 | 2.1 ± 0.7 |
| t value | - | 19.452 | 17.832 | 13.567 | 9.341 | 2.789 |
| P value | - | < 0.001 | < 0.001 | < 0.001 | < 0.001 | 0.006 |
| Source of variation | Sum of squares | Degrees of freedom | Mean square | F value | P value |
| Group | 350.247 | 1 | 350.247 | 185.632 | < 0.001 |
| Time | 45.892 | 4 | 11.473 | 35.417 | < 0.001 |
| Group × time | 37.456 | 4 | 9.364 | 28.915 | < 0.001 |
The Ramsay sedation scores in the observation group at 6 and 12 hours after surgery were higher than those in the control group, whereas scores at 24 and 48 hours were lower. A significant group-time interaction was also observed, with statistically significant differences between groups (P < 0.05) (Tables 5 and 6).
| Group | n | 6 hours post-surgery | 12 hours post-surgery | 24 hours post-surgery | 48 hours post-surgery | 72 hours post-surgery |
| Observation | 60 | 2.5 ± 0.5 | 2.6 ± 0.4 | 2.4 ± 0.5 | 2.3 ± 0.4 | 2.2 ± 0.3 |
| Control | 60 | 2.1 ± 0.6 | 2.2 ± 0.5 | 3.2 ± 0.7 | 2.5 ± 0.5 | 2.3 ± 0.4 |
| t value | - | 4.082 | 4.915 | 7.341 | 2.449 | 1.500 |
| P value | - | < 0.001 | < 0.001 | < 0.001 | 0.016 | 0.136 |
| Source of variation | Sum of squares | Degrees of freedom | Mean square | F value | P value |
| Group | 3.215 | 1 | 3.215 | 2.874 | 0.093 |
| Time | 18.942 | 4 | 4.736 | 15.328 | < 0.001 |
| Group × time | 15.507 | 4 | 3.877 | 12.546 | < 0.001 |
The opioid consumption of the observation group within 48 hours after surgery was lower than that of the control group. The time to first defecation, length of stay in the PICU, and total hospital stay were all shorter in the observation group than in the control group, with statistically significant differences (P < 0.05) (Table 7).
| Group | n | 48 hours opioid use (morphine-equivalent, mg) | Time to first defecation (hours) | PICU stay (days) | Total hospital stay (days) |
| Observation | 60 | 0.8 ± 0.3 | 38.5 ± 10.2 | 2.5 ± 0.8 | 14.6 ± 3.5 |
| Control | 60 | 1.6 ± 0.5 | 46.8 ± 12.5 | 3.2 ± 1.1 | 17.2 ± 4.8 |
| t value | - | 11.283 | 4.018 | 4.125 | 3.456 |
| P value | - | < 0.001 | < 0.001 | < 0.001 | 0.001 |
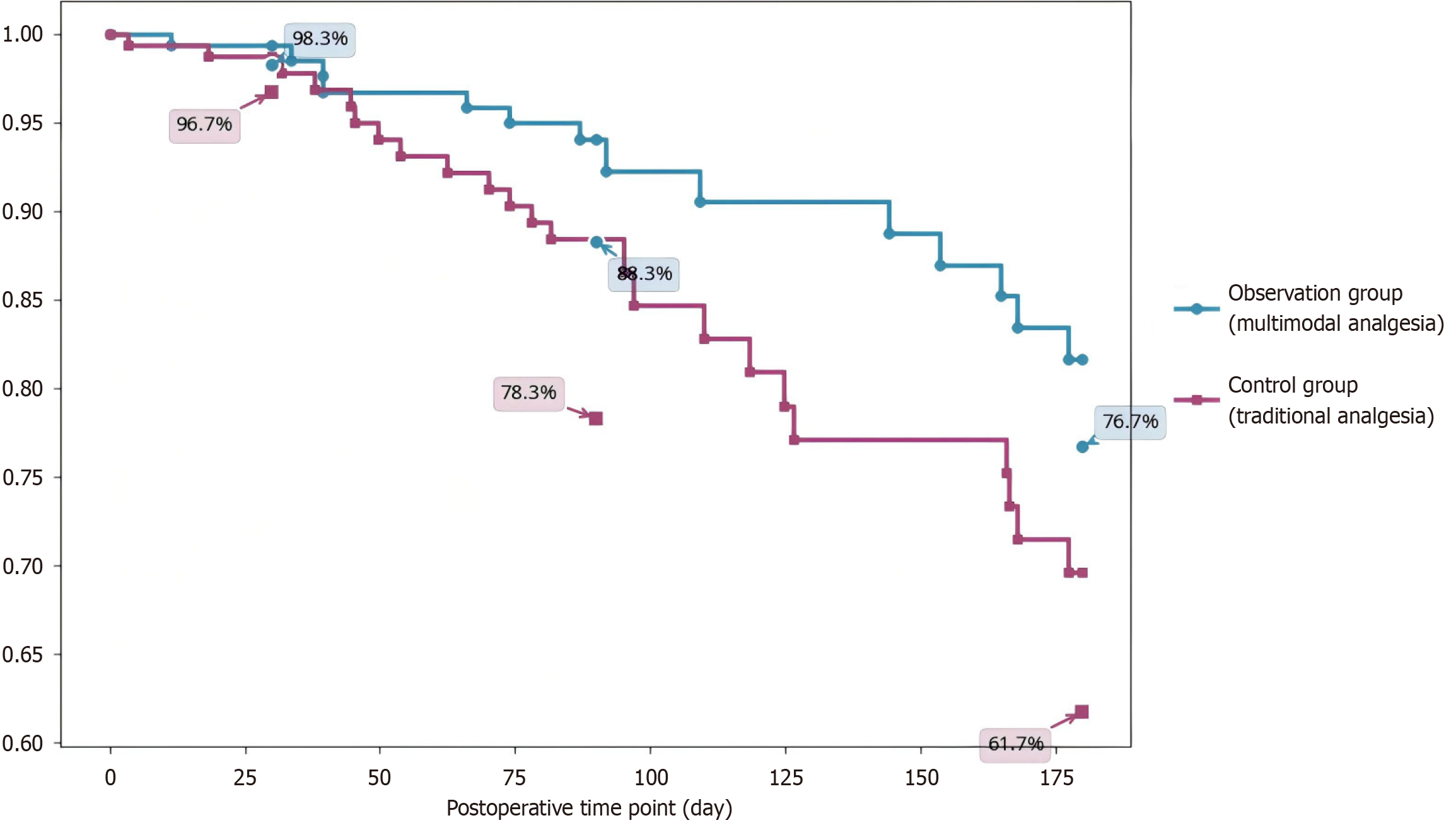
The 6-month postoperative native liver survival rate in the observation group was 76.7% (46/60), higher than 61.7% (37/60) in the control group, and the difference was statistically significant (P < 0.05) (Figure 1).
Pediatric biliary atresia is a severe hepatobiliary disorder in infancy, and the Kasai procedure remains the primary surgical intervention. However, postoperative pain management and recovery are key determinants of prognosis in affected infants[9,10]. The nociceptive system in infants is largely mature at birth, whereas pain modulation mechanisms are relatively underdeveloped, resulting in heightened pain sensitivity and reduced pain tolerance[11,12]. Severe postoperative pain not only causes crying and agitation, interfering with rest and wound healing, but also triggers a cascade of complex pathophysiological responses. The resulting stress state can activate the sympathetic nervous system, leading to tachycardia, hypertension, increased oxygen consumption, immunosuppression, and an elevated risk of infection[13,14]. Furthermore, pain-induced abdominal muscle tension and reduced activity can decrease tidal volume and predispose to atelectasis, thereby impairing respiratory function and delaying gastrointestinal recovery[15].
Notably, infants with biliary atresia often exhibit varying degrees of hepatic dysfunction, which compromises drug metabolism. Consequently, the use of conventional opioids requires caution because of their higher risk of respiratory depression and excessive sedation[16,17]. Therefore, identifying a safe and effective postoperative analgesic regimen is crucial for optimizing surgical outcomes in infants with biliary atresia.
Multimodal analgesia is a cornerstone of modern enhanced recovery after surgery protocols. This approach combines analgesic agents and techniques with different mechanisms of action, targeting multiple points along the pain pathway to achieve synergistic analgesic effects while minimizing the dosage and adverse effects of individual drugs[18,19]. Among these techniques, regional anesthesia methods such as the TAP block can effectively interrupt the transmission of incisional pain signals from the abdominal wall, thereby attenuating nociceptive input at its source. When combined with low-dose, background-infusion PCIA - administered by parents or nurses - this strategy maintains stable plasma drug concentrations and provides continuous, proactive pain control[20,21].
Whether this multimodal regimen can be safely implemented in infants undergoing the Kasai procedure and ultimately yield measurable clinical benefits remains a crucial question in pediatric anesthesia and perioperative medicine. The findings of this study demonstrated that the incidences of postoperative cholangitis, pulmonary infection, and overall complications were significantly lower in the observation group than in the control group (P < 0.05), highlighting the substantial advantage of multimodal analgesia in reducing postoperative complications.
Cholangitis is one of the most frequent and severe complications after the Kasai procedure, and its development is associated with multiple factors, including inadequate bile drainage, intestinal bacterial translocation, and immunosuppression[22,23]. Effective pain management may help reduce the risk of cholangitis by attenuating the stress response, enhancing immune function, and facilitating early mobilization. Aziz et al[24] reported that postoperative infectious complications are closely related to stress-induced immunosuppression and emphasized that optimizing analgesic strategies is an important measure for infection prevention. Furthermore, the lower incidence of pulmonary infection observed in the observation group may be attributed to effective pain control, which allowed infants to breathe deeply and expectorate sputum earlier, thereby reducing the risk of atelectasis and secondary pulmonary infection[25].
In this study, FLACC scores in the observation group at 6, 12, 24, 48, and 72 hours postoperatively were significantly lower than those in the control group. Ramsay sedation scores were higher in the observation group at 6 and 12 hours but lower at 24 and 48 hours. A significant group-time interaction was also observed (P < 0.05). These findings indicate that the multimodal analgesic regimen achieved precise modulation of sedation and analgesia levels according to infants’ physiological needs at different postoperative stages, thereby creating favorable conditions for recovery.
The higher Ramsay sedation scores observed in the observation group at 6 and 12 hours postoperatively suggest that infants experienced adequate analgesia and greater comfort, thereby reducing pain-induced crying and agitation. This promotes rest and recovery while mitigating the adverse physiological effects of excessive stress. At 24 and 48 hours postoperatively, the lower sedation scores in the observation group compared with the control group indicate that this regimen effectively prevents oversedation, allowing infants to move more freely and breathe more effectively, which facilitates the recovery of gastrointestinal and respiratory function.
Furthermore, the results demonstrated that the observation group had lower opioid consumption within 48 hours after surgery and shorter time to first defecation, PICU stay, and total hospital stay compared with the control group (P < 0.05). These findings further confirm the efficacy and safety of the multimodal analgesic regimen - it reduces opioid requirements and decreases the risk of opioid-related adverse events such as respiratory depression and oversedation, which is particularly important for infants with biliary atresia and impaired hepatic function. These outcomes are consistent with the findings of Phelps et al[26] and Bae et al[27], who reported that epidural or multimodal analgesia significantly improved pain control and reduced sedative requirements in infants following the Kasai procedure.
The shortened time to first defecation, PICU stay, and total hospital stay indicate that multimodal analgesia promotes early recovery of gastrointestinal function, reduces complications such as abdominal distension and intestinal obstruction, and accelerates overall rehabilitation. Madadi-Sanjani et al[28] emphasized that inadequate postoperative pain control inhibits gastrointestinal peristalsis through sympathetic activation, delays the initiation of oral feeding, and impairs nutrient absorption and hepatic functional recovery. The multimodal analgesic regimen used in this study provides more favorable conditions for early enteral nutrition by reducing opioid requirements - thereby minimizing gastrointestinal inhibition - and relieving abdominal incisional pain through regional blockade.
Furthermore, the study demonstrated that the 6-month postoperative native liver survival rate in the observation group was 76.7%, compared with 61.7% in the control group (P < 0.05). This finding suggests that early and effective pain management can significantly improve short-term prognosis in infants. A closer analysis indicates that multimodal analgesia substantially reduces surgical stress responses, decreases the incidence of severe complications such as cholangitis, and accelerates early feeding and functional recovery. These effects not only mitigate further hepatic injury but also help preserve residual hepatocyte function, establishing a positive cycle that supports hepatocyte regeneration and bile drainage. Findings by Udagawa et al[29] and Lemoine et al[30] likewise demonstrated that effective control of postoperative complications is closely associated with improved native liver survival rates.
This study has certain limitations. It was a single-center study with a relatively small sample size, short follow-up duration, and potential subjectivity in pain assessment. Future studies should employ multicenter, large-sample designs, extend the follow-up period, and develop more objective and accurate pain assessment tools to further verify the efficacy and safety of the multimodal analgesic regimen in larger populations and over longer observation periods.
In conclusion, for infants with biliary atresia undergoing the Kasai procedure, the implementation of a multimodal, protocolized early pain management strategy centered on ultrasound-guided TAP block combined with PCIA provides safer and more effective analgesia, facilitates postoperative recovery, reduces postoperative complication rates, and improves native liver survival. This strategy holds substantial clinical significance for enhancing the short-term prognosis of infants with biliary atresia.
| 1. | Singh SA, Balaraman KK, Johnson MI, Balaraman V, Puapong DP, Johnson SM, Tabak BD, Woo RK. The Assessment and Management of Biliary Atresia in Hawai'i, 2009-2023. Hawaii J Health Soc Welf. 2024;83:268-273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 2. | Nakayama DK. Morio Kasai Corrects the Uncorrectable: Hepatic Portoenterostomy for Biliary Atresia. J Pediatr Surg. 2024;59:161765. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 3. | Li Y, Jiang J, Wang H. Application value of ultrasound elastography in the diagnosis of pediatric surgical biliary atresia. Transl Pediatr. 2022;11:1433-1434. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 4. | Nakayama DK. Early Achievements in Pediatric Surgery at Vanderbilt University Hospital: Rollin Daniel, Jr., and James Kirtley, Jr. J Pediatr Surg. 2025;60:162190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 5. | O'Connor M, Martinez HR, Hoyos ME, Fraser CD, Well A. Outcomes of Kasai Portoenterostomy in Patients With Congenital Heart Disease: A Silent Comorbidity. J Pediatr Surg. 2025;60:162279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 6. | Tsung JW, Ramadurai S, Sosa P. Point-of-Care Ultrasound Evaluation of Biliary Atresia in the Pediatric Emergency Department. Pediatr Emerg Care. 2025;41:965-968. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 7. | Xing GD, Wang XQ, Duan L, Liu G, Wang Z, Xiao YH, Xia Q, Xie HW, Shen Z, Yu ZZ, Huang LM. Robotic-assisted Kasai portoenterostomy for child biliary atresia. World J Gastrointest Surg. 2024;16:3780-3785. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 8. | Li B, Chen BW, Xia LS. Laparoscopic Kasai Procedure of Treating Biliary Atresia with an Aberrant Right Hepatic Artery. J Laparoendosc Adv Surg Tech A. 2023;33:904-908. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 9. | Son TN, Mai DV, Tung PT, Liem NT. Laparoscopic versus open Kasai procedure for biliary atresia: long-term results of a randomized clinical trial. Pediatr Surg Int. 2023;39:111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 10. | Cho YJ, Kwon H, Kwon YJ, Ha S, Kim SC, Kim DY, Namgoong JM. Surgical treatment for intractable cholangitis with intrahepatic biliary cysts followed by Kasai operation in biliary atresia: a retrospective cohort study. Ann Surg Treat Res. 2024;107:363-368. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 11. | Cazares J, Koga H, Yamataka A. Advances in the surgical technique of Kasai portoenterostomy. Semin Pediatr Surg. 2024;33:151481. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 12. | Sanada Y, Sakuma Y, Onishi Y, Okada N, Hirata Y, Horiuchi T, Omameuda T, Lefor AK, Sata N. Long-term outcomes in pediatric patients who underwent living donor liver transplantation for biliary atresia. Surgery. 2022;171:1671-1676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 13. | Fitsiori A, McLin V, Toso S, Vargas MI. Reversible Brain Atrophy After Liver Transplantation for Biliary Atresia in Childhood. Neurol Clin Pract. 2021;11:e923-e925. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 14. | Orłowska E, Czubkowski P, Wołochowska K, Jarzębicka D, Motyl I, Socha P. Assessment of Lactobacillus casei rhamnosus (LGG) therapy in children with biliary atresia - Randomized placebo controlled trial. Clin Res Hepatol Gastroenterol. 2021;45:101753. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 15. | Thanh LN, Nguyen HP, Kieu TPT, Duy MN, Ha HTT, Thi HB, Nguyen TQ, Pham HD, Tran TD. Modified Kasai operation combined with autologous bone marrow mononuclear cell infusion for biliary atresia. BMC Surg. 2024;24:368. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 16. | Demir HB, Umman V, Gümüs T, Tunalı S, Barut D, Karakoyun M, Akarca US. Comparison of Liver Transplant Outcomes in Biliary Atresia Patients, Considering Whether They Underwent the Kasai Procedure Beforehand: A Single Center Analysis of 72 Patients. Transplant Proc. 2023;55:1199-1208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 17. | Shimizu T, Shun A, Thomas G. Portosystemic shunt for portal hypertension after Kasai operation in patients with biliary atresia. Pediatr Surg Int. 2021;37:101-107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 18. | Yassin NA, El-Tagy G, Abdelhakeem ON, Asem N, El-Karaksy H. Predictors of Short-Term Outcome of Kasai Portoenterostomy for Biliary Atresia in Infants: a Single-Center Study. Pediatr Gastroenterol Hepatol Nutr. 2020;23:266-275. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 19. | Feng Q, Li N, Ren Q, Zheng J, Li J. Does ultrasound elastography have high value in diagnosis of biliary atresia in pediatric surgery? Transl Pediatr. 2022;11:1431-1432. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 20. | Li Y, Ye H, Ding Y. Identification of Hub Genes and Immune Infiltration in Pediatric Biliary Atresia by Comprehensive Bioinformatics Analysis. Children (Basel). 2022;9:697. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 21. | Yoeli D, Mack CL, Luo Y, Chaidez A, De La Rosa NL, Wang Z, Cervantes-Alvarez E, Huang CA, Navarro-Alvarez N. Galectin-3 in biliary atresia and other pediatric cholestatic liver diseases. Hepatol Res. 2024;54:392-402. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 22. | Ptohis N, Kanna E, Batsari E, Lamprinou Z, Skondras I. Simultaneous Hepatic Artery and Portal Vein Thrombosis After Kasai Portoenterostomy: A Rare but Life-Threatening Complication. Cureus. 2025;17:e78855. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 23. | Yang C, Li T, Yu P, Zhan J. A nomogram model based on routine serum markers for predicting the occurrence of primary cholangitis after Kasai operation for biliary atresia. Transl Pediatr. 2025;14:1103-1116. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 24. | Aziz MA, Abdullatif HM, Soliman MS, Okasha S, Nabil N, Balah MM, El-Karaksy H. A comprehensive clinical and microbiological study on the diagnosis and management of cholangitis in patients with biliary atresia undergoing kasai portoenterostomy. Indian J Gastroenterol. 2025;44:506-516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 25. | Farooqui ZA, Hartman SJ, Stetson AE, Schepers EJ, Speck KE, Gadepalli SK, Van Arendonk KJ, Georgeades C, Lal DR, Deans KJ, Minneci PC, Apfeld JC, Saito JM, Mak GZ, Slidell MB, Lemoine C, Superina R, Wright TN, Downard CD, Devara LP, Hirschl RB, Landman MP, Leys CM, Markel TA, Rymeski B, Mullapudi B, Tiao GM; Midwest Pediatric Surgery Consortium. Real-world Multi-institutional Data From the Midwest Pediatric Surgery Consortium (MWPSC) to Assess the Effect of Delayed Kasai Procedure on Biliary Drainage in Patients With Biliary Atresia. J Pediatr Surg. 2025;60:162250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 26. | Phelps HM, Robinson JR, Chen H, Luckett TR, Conroy PC, Gillis LA, Hays SR, Lovvorn HN 3rd. Enhancing Recovery After Kasai Portoenterostomy With Epidural Analgesia. J Surg Res. 2019;243:354-362. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 11] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 27. | Bae E, Ganesh A, Flake AW, Gurnaney HG. Benefit of epidural analgesia for postoperative pain control after a Kasai Portoenterostomy: A ten-year retrospective cohort study. Paediatr Anaesth. 2023;33:154-159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 28. | Madadi-Sanjani O, Uecker M, Thomas G, Fischer L, Hegen B, Herrmann J, Reinshagen K, Tomuschat C. Optimizing Post-Kasai Management in Biliary Atresia: Balancing Native Liver Survival and Transplant Timing. Eur J Pediatr Surg. 2025;35:261-268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 29. | Udagawa D, Hasegawa Y, Obara H, Yamada Y, Shinoda M, Kitago M, Abe Y, Kuroda T, Kitagawa Y. Risk Assessment of Liver Transplantation After Kasai Portoenterostomy in Children and Adults. J Surg Res. 2023;290:109-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 30. | Lemoine CP, LeShock JP, Brandt KA, Superina R. Primary Liver Transplantation vs. Transplant after Kasai Portoenterostomy for Infants with Biliary Atresia. J Clin Med. 2022;11:3012. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (1)] |