Published online Mar 27, 2026. doi: 10.4240/wjgs.v18.i3.115216
Revised: December 16, 2025
Accepted: January 21, 2026
Published online: March 27, 2026
Processing time: 167 Days and 7.1 Hours
Pleomorphic leiomyosarcoma (LMS) of gastric origin is extremely rare, especially when accompanied by retroperitoneal extension and hepatic metastasis. Since the introduction of KIT (cluster of differentiation 117) immunohistochemistry, many gastric mesenchymal tumors have been reclassified as gastrointestinal stromal tumors, making true gastric LMS unusual. We report a large pleomorphic gastric LMS initially misdiagnosed as retroperitoneal sarcoma to highlight diagnostic pitfalls and the central role of complete surgical resection.
A 58-year-old woman presented with a 1-month history of progressive abdominal distension and bilateral lower limb edema and had a 1-year history of a self-pal
Pleomorphic gastric LMS may mimic retroperitoneal sarcoma, and complete margin-negative (R0) resection remains the key treatment.
Core Tip: Pleomorphic leiomyosarcoma of gastric origin is an exceptionally rare and aggressive tumor, especially when accompanied by retroperitoneal invasion and hepatic metastasis. We report a unique case initially misdiagnosed as retroperitoneal sarcoma, later confirmed intraoperatively to arise from the stomach near the pylorus. Complete curative (R0) resection including distal pancreatectomy and partial hepatectomy achieved favorable short-term outcomes. The patient refused conventional adjuvant therapy and received Huaier granules, a traditional Chinese medicine with potential antitumor effects. This case emphasizes the diagnostic challenges of gastric leiomyosarcoma and suggests that Huaier granules may have a role in postoperative disease control.
- Citation: Huang K, Wang W, Xu ML, He YS, Zhao JY, Hu XF. Pleomorphic gastric leiomyosarcoma with retroperitoneal extension and hepatic metastasis: A case report. World J Gastrointest Surg 2026; 18(3): 115216
- URL: https://www.wjgnet.com/1948-9366/full/v18/i3/115216.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i3.115216
Soft tissue sarcomas are rare neoplasms, accounting for less than 1% of newly diagnosed adult cancers, and approximately 10%-15% of these tumors arise in the retroperitoneum[1,2]. Leiomyosarcoma (LMS) accounts for 10%-20% of retroperitoneal soft tissue sarcomas and is characterized by aggressive local invasion and a high rate of distant metastasis, most commonly to the liver[3]. The pleomorphic variant, marked by significant cellular atypia and high mitotic activity, constitutes about 8.6% of LMS cases[4]. In the post-KIT era, after the routine introduction of cluster of differentiation 117 (CD117) (KIT) and discovered on gastrointestinal stromal tumor 1 (DOG1) immunohistochemistry, most gastric mesen
Here, we report a case of pleomorphic gastric LMS with retroperitoneal extension and synchronous hepatic metastasis. The patient underwent complete (R0) surgical resection and subsequently chose Huaier granules, a traditional Chinese medicine with reported antitumor properties, as exploratory adjunctive therapy. We briefly review the relevant literature and discuss the diagnostic challenges and therapeutic considerations for this rare entity.
A 58-year-old woman presented on March 29, 2024, with a 1-month history of progressive abdominal distension and bilateral lower limb edema, on a background of an incidentally palpated abdominal mass noted approximately 1 year earlier.
One year before admission, the patient accidentally noticed a palpable abdominal mass that was initially asymptomatic and did not affect her daily life. During the month before presentation, she developed gradually worsening abdominal distension accompanied by bilateral lower limb edema. She sought care at a local hospital, where symptomatic treatment failed to relieve her symptoms. Abdominal ultrasound performed on March 28, 2024, revealed a hepatic space-occupying lesion, and she was referred to our institution on March 29, 2024, with a provisional diagnosis of “hepatic space-occupying lesion” for further evaluation and management.
Her past medical history was notable for hypertension since 2018, which was well controlled with oral amlodipine and bisoprolol. She also had a history of cerebellar hemorrhage in 2018, with no residual neurological deficits. No other significant chronic illnesses, previous abdominal surgeries, or known drug allergies were recorded.
There was no documented history of tobacco or excessive alcohol use. No family history of malignant tumors or here
On admission, the patient was in a generally good condition with obvious abdominal distension and bilateral lower limb edema. A large, ill-defined abdominal mass was palpable in the upper abdomen. No additional remarkable findings were documented on systemic examination.
Baseline laboratory examinations, including routine blood tests, serum biochemistry, coagulation profile, and tumor markers, were obtained as part of the preoperative assessment. The results did not provide specific clues beyond con
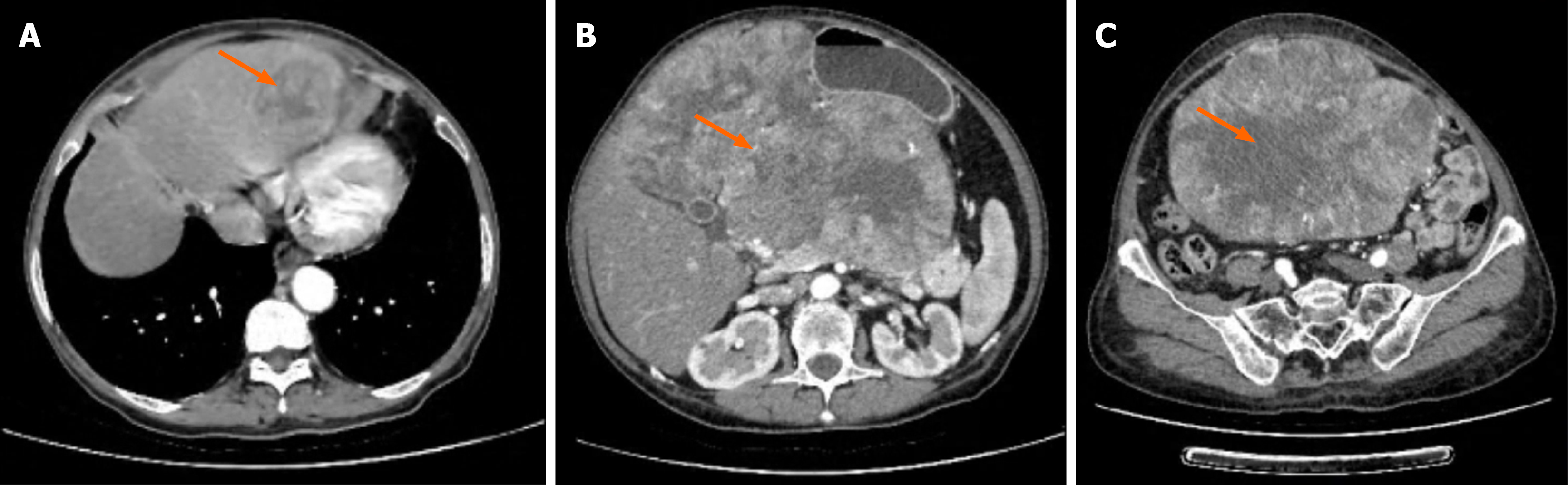
Preoperative contrast-enhanced computed tomography on March 29, 2024 demonstrated a 14.1 cm × 18.6 cm × 26.8 cm heterogeneous mass occupying the hepatogastric space, with patchy low-density areas and extension toward the pelvis. A 4.4 cm × 3.9 cm lesion consistent with hepatic metastasis was identified in the left liver lobe, showing uneven en
No formal multidisciplinary expert consultation was convened. The treatment strategy was developed through dis
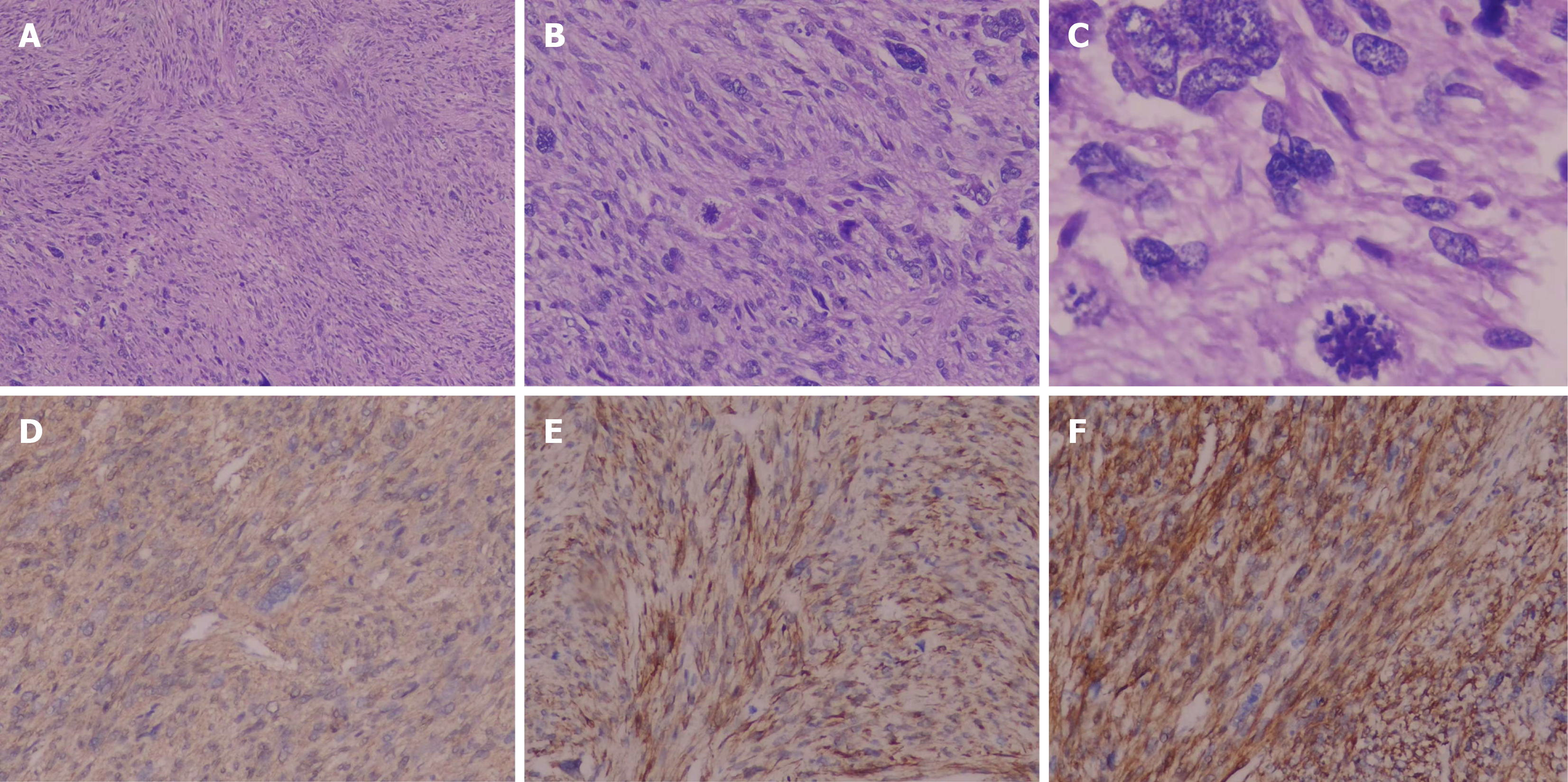
Postoperative histopathology of the resected specimen revealed spindle-shaped tumor cells with marked nuclear pleo
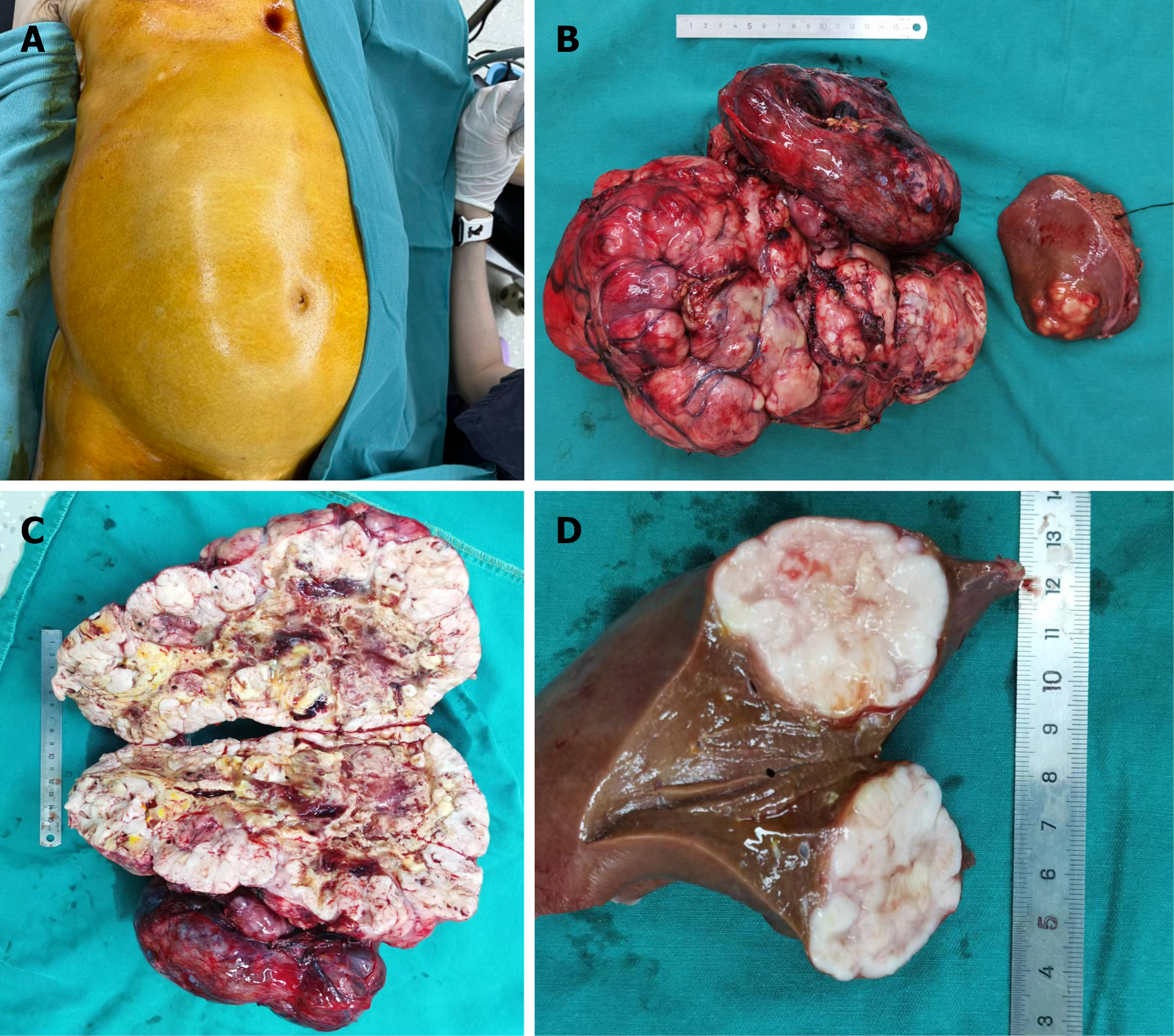
On April 9, 2024, the patient underwent open exploratory laparotomy. Intraoperative exploration confirmed a 14.1 cm × 18.6 cm × 26.8 cm mass originating from the gastric greater curvature near the pylorus, extending posteriorly into the retroperitoneal space and invading the pancreatic body and tail, which appeared atrophic. A 4.4 cm × 3.9 cm metastatic lesion was identified in the left liver lobe, and there was no evidence of peritoneal seeding or distant extra-abdominal metastases.
Complete margin-negative (R0) resection was achieved through en bloc removal of the primary tumor and involved structures, including partial gastrectomy, distal pancreatectomy, and partial hepatectomy. The resected specimen weighed approximately 8 kg, and intraoperative frozen-section analysis confirmed negative surgical margins. Representative intraoperative and specimen photographs are shown in Figure 3. No major intraoperative complications were encountered, and hemostasis and reconstruction were satisfactory.
The patient recovered well postoperatively and was discharged in stable condition. Her abdominal distension and bilateral lower limb edema resolved completely, and her performance status improved.
She was followed regularly in the outpatient clinic with clinical examinations, laboratory tests, and imaging studies. Follow-up abdominal contrast-enhanced computed tomography and chest imaging were performed at regular intervals to monitor for local recurrence or distant metastasis. At 17 months after surgery (September 14, 2025), no radiological or clinical evidence of tumor recurrence or new metastasis was detected, indicating favorable short-term disease control. The patient continues under active surveillance, and long-term outcomes remain to be determined.
Primary gastric LMS is an exceedingly rare malignancy, particularly in the era following the introduction of CD117 (KIT) and DOG1 immunohistochemistry, when many historical gastric “smooth muscle” tumors were reclassified as gast
Preoperative differentiation between gastric-origin LMS and retroperitoneal sarcoma can be difficult, as both may appear as large heterogeneous masses with poorly defined boundaries on imaging. In our patient, the loss of fat planes between the mass, pancreas, and liver led to the initial misdiagnosis of retroperitoneal sarcoma. Intraoperative findings confirming a gastric origin near the pylorus highlight the importance of comprehensive exploration and intraoperative pathological assessment.
Complete surgical resection with negative margins (R0) remains the mainstay and only potentially curative treatment for LMS, regardless of its site[18,19]. In gastric LMS, prognosis depends primarily on tumor size, grade, and resection status. Adjuvant chemotherapy or radiotherapy has limited efficacy, and no standard regimen has been established. An
Our patient declined systemic chemotherapy and instead received Huaier granules (Trametes robiniophila), a traditional Chinese medicine with reported antiangiogenic and immunomodulatory effects. Although no clinical data exist on its efficacy in LMS, studies in hepatocellular carcinoma and breast cancer have suggested that Huaier extracts can inhibit proliferation and metastasis through tumor microenvironment modulation. The patient remained recurrence-free at 17 months after complete resection; however, causality cannot be inferred, and longer follow-up is warranted.
Overall, the prognosis of gastric LMS remains poor, though some series (for example, Mayo Clinic data) report 5-year survival rates around 45%, especially in patients with low-grade tumors, early stage, and without metastasis at pre
Taken together, pleomorphic gastric LMS is an exceptionally rare and aggressive soft-tissue sarcoma that may mimic retroperitoneal sarcoma on imaging. Accurate diagnosis requires integration of radiological, intraoperative, and immunohistochemical findings to exclude gastrointestinal stromal tumor. Complete surgical excision remains the cornerstone of management. This case underscores the diagnostic challenges of pleomorphic gastric LMS and the importance of complete resection as the cornerstone of management. Longer follow-up and further clinical studies are needed to clarify any potential role of Huaier as an adjunct.
Pleomorphic LMS of gastric origin with retroperitoneal extension and synchronous hepatic metastasis is exceptionally rare and can mimic retroperitoneal sarcoma on preoperative imaging. Accurate diagnosis requires integration of radiologic findings, comprehensive intraoperative assessment, and immunohistochemistry to distinguish it from gastrointestinal stromal tumor and other sarcomas. Complete margin-negative resection of the primary tumor and metastatic lesions remains the key determinant of short-term disease control and survival. Complementary therapies such as Huaier granules should be considered exploratory and cannot replace standard oncologic treatments, but they may be discussed with patients in the context of shared decision-making and careful follow-up.
| 1. | Willburger JCF, von Strauss M, Peterson CJ, Glass TR, Kettelhack C. Incidence, Treatment and Outcome of Patients with Retroperitoneal Soft-Tissue Sarcoma in Switzerland 2005-2015: A Population-Based Analysis. World J Surg. 2022;46:461-468. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 2. | Álvarez Álvarez R, Manzano A, Agra Pujol C, Artigas Raventós V, Correa R, Cruz Jurado J, Fernandez JA, Garcia Del Muro X, Gonzalez JA, Hindi N, Lozano Lominchar P, Martínez-Trufero J, Méndez R, Muñoz M, Muñoz Casares C, Orbis Castellanos F, Orellana Fernandez R, Paniagua González M, Redondo A, Valverde Morales C, Asencio JM. Updated Review and Clinical Recommendations for the Diagnosis and Treatment of Patients with Retroperitoneal Sarcoma by the Spanish Sarcoma Research Group (GEIS). Cancers (Basel). 2023;15:3194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (1)] |
| 3. | de Bree E, Michelakis D, Heretis I, Kontopodis N, Spanakis K, Lagoudaki E, Tolia M, Zografakis-Sfakianakis M, Ioannou C, Mavroudis D. Retroperitoneal Soft Tissue Sarcoma: Emerging Therapeutic Strategies. Cancers (Basel). 2023;15:5469. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 21] [Reference Citation Analysis (1)] |
| 4. | Oda Y, Miyajima K, Kawaguchi K, Tamiya S, Oshiro Y, Hachitanda Y, Oya M, Iwamoto Y, Tsuneyoshi M. Pleomorphic leiomyosarcoma: clinicopathologic and immunohistochemical study with special emphasis on its distinction from ordinary leiomyosarcoma and malignant fibrous histiocytoma. Am J Surg Pathol. 2001;25:1030-1038. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 86] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 5. | Aggarwal G, Sharma S, Zheng M, Reid MD, Crosby JH, Chamberlain SM, Nayak-Kapoor A, Lee JR. Primary leiomyosarcomas of the gastrointestinal tract in the post-gastrointestinal stromal tumor era. Ann Diagn Pathol. 2012;16:532-540. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 66] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 6. | Miettinen M, Sobin LH, Lasota J. True smooth muscle tumors of the small intestine: a clinicopathologic, immunhistochemical, and molecular genetic study of 25 cases. Am J Surg Pathol. 2009;33:430-436. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 19] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 7. | Garg R, AlRajjal A, Berri R, Barawi M. Primary Gastric Leiomyosarcoma: a Case Report and Review of the Literature. J Gastrointest Cancer. 2020;51:335-340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 14] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 8. | Kang WZ, Xue LY, Tian YT. Leiomyosarcoma of the stomach: A case report. World J Clin Cases. 2019;7:3575-3582. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 19] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (4)] |
| 9. | Hasnaoui A, Jouini R, Haddad D, Zaafouri H, Bouhafa A, Ben Maamer A, Ben Brahim E. Gastric leiomyosarcoma and diagnostic pitfalls: a case report. BMC Surg. 2018;18:62. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 10. | Wang T, Zreik R, Leng B. Primary Gastric Leiomyosarcoma: A Rare Case. Cureus. 2023;15:e49510. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 11. | Miettinen M, Sarlomo-Rikala M, Sobin LH, Lasota J. Gastrointestinal stromal tumors and leiomyosarcomas in the colon: a clinicopathologic, immunohistochemical, and molecular genetic study of 44 cases. Am J Surg Pathol. 2000;24:1339-1352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 237] [Cited by in RCA: 173] [Article Influence: 6.7] [Reference Citation Analysis (2)] |
| 12. | Lee Y. Primary gastric leiomyosarcoma: a case report and literature review. Kosin Med J. 2024;39:60-65. [DOI] [Full Text] |
| 13. | Sagara K, Takayoshi K, Kusumoto E, Uchino K, Matsumura T, Kusaba H, Momosaki S, Ikejiri K, Baba E. Favorable control of rapidly progressive retroperitoneal pleomorphic leiomyosarcoma with multimodality therapy: a case report. BMC Res Notes. 2014;7:377. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (1)] |
| 14. | Okamoto M, Matsuoka M, Soma T, Arai R, Kato H, Harabayashi T, Adachi H, Shinohara T, Sagawa T, Nishiyama N, Nambu T, Sakai W, Suzuki H, Kato H, Hiraga H. Metastases of soft tissue sarcoma to the liver: A Historical Cohort Study from a Hospital-based Cancer Registry. Cancer Med. 2020;9:6159-6165. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 15. | Dalal KM, Kattan MW, Antonescu CR, Brennan MF, Singer S. Subtype specific prognostic nomogram for patients with primary liposarcoma of the retroperitoneum, extremity, or trunk. Ann Surg. 2006;244:381-391. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 274] [Cited by in RCA: 290] [Article Influence: 14.5] [Reference Citation Analysis (1)] |
| 16. | Kitasaki N, Abe T, Oshita A, Kobayashi T, Yonehara S, Ohdan H, Noriyuki T, Nakahara M. Long-term survival by repeat resection for metastases from primary retroperitoneal leiomyosarcoma: A case report. Int J Surg Case Rep. 2021;82:105891. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 17. | Mehta V, Rajawat M, Rastogi S, Phulware RH, Mezencev R. Leiomyosarcoma of the stomach with metastasis to the liver: a case report with review of the literature. Future Sci OA. 2018;4:FSO264. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 18. | Schmitz E, Nessim C. Retroperitoneal Sarcoma Care in 2021. Cancers (Basel). 2022;14:1293. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 33] [Article Influence: 8.3] [Reference Citation Analysis (1)] |
| 19. | Øines MN, Smith HG, Preisler L, Penninga L. Leiomyosarcoma of the abdomen and retroperitoneum; a systematic review. Front Surg. 2024;11:1375483. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (5)] |
| 20. | Farrugia G, Kim CH, Grant CS, Zinsmeister AR. Leiomyosarcoma of the stomach: determinants of long-term survival. Mayo Clin Proc. 1992;67:533-536. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 16] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 21. | Lindsay PC, Ordonez N, Raaf JH. Gastric leiomyosarcoma: clinical and pathological review of fifty patients. J Surg Oncol. 1981;18:399-421. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 35] [Article Influence: 0.8] [Reference Citation Analysis (1)] |