Published online Mar 27, 2026. doi: 10.4240/wjgs.v18.i3.115191
Revised: November 13, 2025
Accepted: January 15, 2026
Published online: March 27, 2026
Processing time: 168 Days and 10.1 Hours
Incisional hernia (IH) is one of the most common complications after liver trans
To identify the independent risk factors for postoperative IH following liver transplantation and to develop a Cox regression-based nomogram for individualized risk prediction.
This single-center retrospective cohort study included 511 liver transplant re
Univariate analysis showed that age, hepatitis, chronic obstructive pulmonary disease, ascites, malignancy as the transplant indication, history of abdominal surgery, red blood cell count, white blood cell count, serum albumin, total bilirubin, alanine aminotransferase, aspartate aminotransferase, prothrombin time, and international nor
Hepatitis, ascites, hypoalbuminemia, impaired liver function, and coagulation abnormalities are key risk factors for IH following liver transplantation. The nomogram developed in this study demonstrated high accuracy and clinical utility, providing a valuable tool for individualized postoperative management and preventive strategies.
Core Tip: This study developed and validated a nomogram that incorporates key factors, including age, hepatitis, ascites, hypoalbuminemia, elevated alanine aminotransferase, and prolonged prothrombin time, to accurately predict the risk of incisional hernia following liver transplantation, facilitating the identification of high-risk patients for targeted preventive management. The nomogram developed in this study provides a valuable tool for individualized postoperative management and preventive strategies.
- Citation: Liu SY, Xie XY, Zhang KN, Ma YQ, Ababakri A, Hao HZ, Zhang L, Guo QJ, Yu XH, Xie Y, Jiang WT. Risk factors and nomogram prediction of postoperative incisional hernia following liver transplantation: A retrospective cohort study. World J Gastrointest Surg 2026; 18(3): 115191
- URL: https://www.wjgnet.com/1948-9366/full/v18/i3/115191.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i3.115191
Liver transplantation represents a vital therapeutic option for a wide range of severe liver diseases, including hepatocellular carcinoma, primary biliary cirrhosis, and acute liver failure, and remains the only curative treatment for end-stage liver disease. With continuous advances in surgical techniques and optimization of immunosuppressive regimens, long-term survival and quality of life among liver transplant recipients have improved significantly. However, post
Previous studies suggest that IH is associated with multiple factors, including age, smoking history, corticosteroid use, operative duration, and emergency surgery[4,5]. In the liver transplantation population, management of IH is more com
However, few studies have systematically explored predictive tools for postoperative IH following liver trans
This retrospective cohort study included patients who underwent liver transplantation at Tianjin First Central Hospital between January 2019 and December 2021. Patients were categorized into the IH group and control group based on postoperative occurrence of IH. Patients with a preoperative history of abdominal wall hernia were excluded. The final diagnosis of IH was confirmed during tension-free hernia repair surgery.
All patients underwent orthotopic liver transplantation performed by the same surgical team, using ABO-compatible grafts from deceased donors. Postoperative immunosuppressive therapy consisted of a triple regimen including tac
The diagnosis of IH was based on physical examination and imaging evaluation, and confirmed intraoperatively during hernia repair. Follow-up was conducted through outpatient visits and telephone interviews: Every 2 months during the first 2 years post-transplant, and every 6 months thereafter. The primary endpoint was IH status within 2 years after repair. Follow-up was completed by December 31, 2024. Missing data were minimal (< 5%) and handled by complete-case analysis.
Descriptive statistics were used to summarize baseline characteristics. Categorical variables were compared using the χ² test or Fisher’s exact test, and continuous variables were analyzed with Student’s t-test. Univariate and multivariate Cox regression analyses were performed to identify risk factors for IH. A nomogram was constructed using the rms package in R software, and predictive performance was assessed using the concordance index (C-index). A two-tailed P < 0.05 was considered statistically significant.
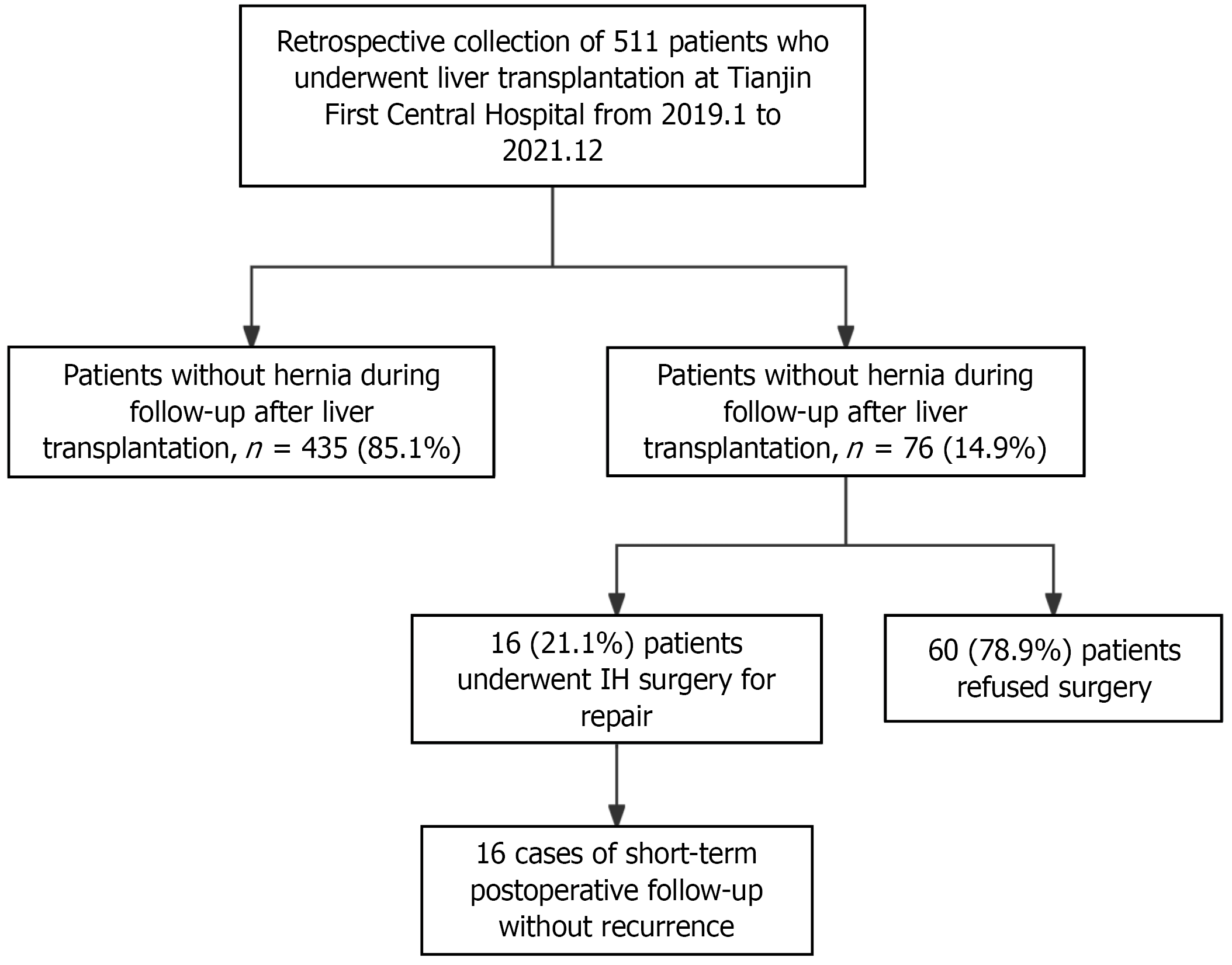
This study included clinical data of patients who underwent liver transplantation at Tianjin First Central Hospital between January 2019 and December 2021. Both patients who developed IH after transplantation and those who did not were enrolled. The screening process of eligible patients according to the inclusion criteria is shown in Figure 1. During the study period, a total of 511 liver transplantations were performed. Among them, 435 patients (85.1%) did not develop IH during follow-up, while 76 patients (14.9%) developed IH, of whom 16 underwent hernia repair surgery. In the IH group, there were 55 male (72.4%) and 21 female (27.6%) patients, with an age range of 40-73 years. The median time to IH occurrence after liver transplantation was 12.5 months (interquartile range: 6.5-38 months).
Baseline characteristics of the patients are summarized in Table 1. There were no significant differences between the groups in terms of sex distribution and smoking history (P > 0.05). Compared with patients without IH, those who developed IH were significantly older (median age 66 years vs 51 years, P < 0.001). Regarding comorbidities, patients in the IH group had a markedly higher prevalence of hepatitis and chronic obstructive pulmonary disease (COPD; both P < 0.001), as well as a higher incidence of ascites (P < 0.001). In addition, a history of prior abdominal surgery was strongly associated with the development of IH (P < 0.001), suggesting that surgical manipulation and tissue healing may play important roles in postoperative complications.
| Characteristics | No incisional hernia | Developed incisional hernia | P value |
| n | 435 | 76 | |
| Gender | 0.287 | ||
| Male | 339 (66.3) | 55 (10.8) | |
| Female | 96 (18.8) | 21 (4.1) | |
| Age | 51 (44, 57) | 66 (64, 67) | < 0.001 |
| Smoking | 0.379 | ||
| No | 297 (58.1) | 48 (9.4) | |
| Yes | 138 (27) | 28 (5.5) | |
| Hepatitis | < 0.001 | ||
| No | 435 (85.1) | 56 (11) | |
| Yes | 0 (0) | 20 (3.9) | |
| COPD | < 0.001 | ||
| No | 435 (85.1) | 69 (13.5) | |
| Yes | 0 (0) | 7 (1.4) | |
| Ascites | < 0.001 | ||
| No | 429 (84) | 67 (13.1) | |
| Yes | 6 (1.2) | 9 (1.8) | |
| Other1 | 0.106 | ||
| No | 402 (78.7) | 66 (12.9) | |
| Yes | 33 (6.5) | 10 (2) | |
| Abdominal surgeries history2 | < 0.001 | ||
| No | 435 (85.1) | 73 (14.3) | |
| Yes | 0 (0) | 3 (0.6) | |
| Cancer3 | < 0.001 | ||
| No | 339 (66.3) | 37 (7.2) | |
| Yes | 96 (18.8) | 39 (7.6) | |
| Red blood cell counts, 1 × 1012/L | 4.42 (3.925, 4.875) | 4.15 (3.4075, 4.5875) | < 0.001 |
| White blood cell counts, 1 × 109/L | 5.81 (4.31, 7.76) | 5.095 (3.7025, 6.545) | 0.013 |
| PLT (125-350), 1 × 109/L | 172 (120, 238.5) | 181 (103.75, 234.25) | 0.539 |
| ALB, g/L | 41.8 (37.7, 44.7) | 35.3 (32.275, 40.375) | < 0.001 |
| Total bilirubin, μmol/L | 20 (14, 32) | 25.5 (14, 50) | 0.021 |
| ALT, U/L | 19.7 (14.2, 30.95) | 34.9 (22.45, 53.725) | < 0.001 |
| AST, U/L | 23.7 (17.8, 33.55) | 44.8 (26.175, 71.5) | < 0.001 |
| PT (8.8-13.8), seconds | 15.3 (14.2, 16.5) | 12.9 (11.875, 15.05) | < 0.001 |
| INR (0.8-1.2) | 1.05 (1, 1.16) | 1.16 (1.0675, 1.3425) | < 0.001 |
In terms of laboratory findings, patients with IH had abnormal postoperative red blood cell and white blood cell counts (P < 0.05), along with significantly lower serum albumin (ALB) levels (35.3 g/L vs 41.8 g/L, P < 0.001). Liver function markers, including total bilirubin, alanine aminotransferase (ALT), and aspartate aminotransferase (AST), were significantly elevated in the IH group (P < 0.05). Coagulation parameters revealed an increased international normalized ratio in patients with IH (P < 0.001). Overall, hepatitis, hypoalbuminemia, impaired liver function, coagulation abnormalities, and comorbidities (such as COPD, ascites, and history of abdominal surgery) were closely associated with the occurrence of IH following liver transplantation.
Univariate Cox regression analysis: As shown in Table 2, univariate Cox regression analysis identified several significant prognostic factors: Age [hazard ratio (HR) = 1.191, 95% confidence interval (CI): 1.148-1.235, P < 0.001], hepatitis (HR = 15.240, 95%CI: 8.958-25.926, P < 0.001), COPD (HR = 5.032, 95%CI: 2.291-11.054, P < 0.001), ascites (HR = 6.538, 95%CI: 3.242-13.185, P < 0.001), prior abdominal surgery (HR = 25.832, 95%CI: 7.797-85.579, P < 0.001), tumor (HR = 2.810, 95%CI: 1.779-4.437, P < 0.001), abnormal red blood cell count (HR = 0.503, 95%CI: 0.373-0.678, P < 0.001), abnormal white blood cell count (HR = 0.887, 95%CI: 0.803-0.981, P = 0.020), ALB (HR = 0.895, 95%CI: 0.864-0.927, P < 0.001), total bilirubin (HR = 1.002, 95%CI: 1.000-1.004, P = 0.031), ALT (HR = 1.005, 95%CI: 1.003-1.007, P < 0.001), AST (HR = 1.002, 95%CI: 1.001-1.003, P < 0.001), prothrombin time (PT; HR = 0.803, 95%CI: 0.750-0.861, P < 0.001), and international normalized ratio (HR = 2.915, 95%CI: 1.616-5.257, P < 0.001).
| Characteristics | Total (n) | Univariate analysis | Multivariate analysis | ||
| Hazard ratio (95%CI) | P value | Hazard ratio (95%CI) | P value | ||
| Gender | 487 | ||||
| Male | 376 | Reference | |||
| Female | 111 | 1.225 (0.736-2.037) | 0.435 | ||
| Age | 487 | 1.191 (1.148-1.235) | < 0.001 | 1.131 (1.075-1.191) | < 0.001 |
| Smoking | 487 | ||||
| No | 328 | Reference | |||
| Yes | 159 | 1.147 (0.711-1.852) | 0.574 | ||
| Hepatitis | 487 | ||||
| No | 468 | Reference | Reference | ||
| Yes | 19 | 15.240 (8.958-25.926) | < 0.001 | 2.225 (1.058-4.682) | 0.035 |
| COPD | 487 | ||||
| No | 480 | Reference | Reference | ||
| Yes | 7 | 5.032 (2.291-11.054) | < 0.001 | 0.266 (0.089-0.792) | 0.017 |
| Ascites | 487 | ||||
| No | 472 | Reference | Reference | ||
| Yes | 15 | 6.538 (3.242-13.185) | < 0.001 | 5.687 (1.925-16.802) | 0.002 |
| Other1 | 487 | ||||
| No | 446 | Reference | |||
| Yes | 41 | 1.396 (0.691-2.818) | 0.353 | ||
| Abdominal surgeries history2 | 487 | ||||
| No | 484 | Reference | Reference | ||
| Yes | 3 | 25.832 (7.797-85.579) | < 0.001 | 2.182 (0.338-14.068) | 0.412 |
| Cancer3 | 487 | ||||
| No | 359 | Reference | Reference | ||
| Yes | 128 | 2.810 (1.779-4.437) | < 0.001 | 1.116 (0.651-1.916) | 0.689 |
| Red blood cell counts, 1 × 1012/L | 487 | 0.503 (0.373-0.678) | < 0.001 | 0.829 (0.592-1.163) | 0.277 |
| White blood cell counts, 1 × 109/L | 487 | 0.887 (0.803-0.981) | 0.020 | 0.902 (0.796-1.023) | 0.108 |
| PLT (125-350), 1 × 109/L | 487 | 0.999 (0.996-1.002) | 0.425 | ||
| ALB, g/L | 487 | 0.895 (0.864-0.927) | < 0.001 | 0.933 (0.886-0.982) | 0.008 |
| Total bilirubin, μmol/L | 487 | 1.002 (1.000-1.004) | 0.031 | 1.000 (0.997-1.002) | 0.775 |
| ALT, U/L | 487 | 1.005 (1.003-1.007) | < 0.001 | 1.013 (1.004-1.021) | 0.003 |
| AST, U/L | 487 | 1.002 (1.001-1.003) | < 0.001 | 0.996 (0.993-0.999) | 0.023 |
| PT (8.8-13.8), seconds | 486 | 0.803 (0.750-0.861) | < 0.001 | 0.903 (0.816-0.999) | 0.047 |
| INR (0.8-1.2) | 487 | 2.915 (1.616-5.257) | < 0.001 | 2.042 (0.735-5.674) | 0.171 |
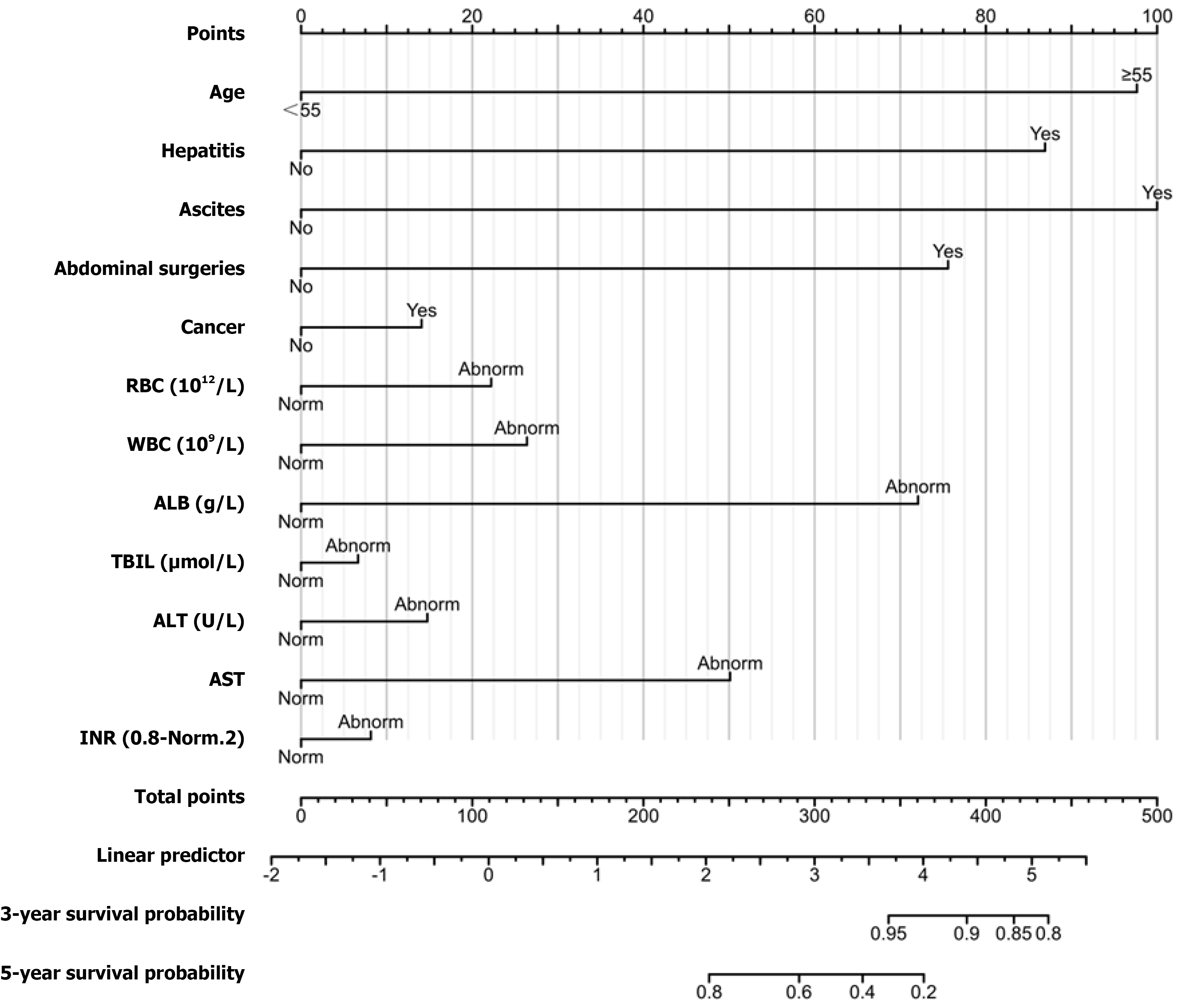
Multivariate Cox regression analysis: In multivariate Cox regression analysis, several variables remained independent prognostic factors. These included age (HR = 1.131, 95%CI: 1.075-1.191, P < 0.001), hepatitis (HR = 2.225, 95%CI: 1.058-4.682, P = 0.035), ascites (HR = 5.687, 95%CI: 1.925-16.802, P = 0.002), serum ALB (HR = 0.933, 95%CI: 0.886-0.982, P = 0.008), ALT (HR = 1.013, 95%CI: 1.004-1.021, P = 0.003), AST (HR = 0.996, 95%CI: 0.993-0.999, P = 0.023), and PT (HR = 0.903, 95%CI: 0.816-0.999, P = 0.047). Among these, older age, hepatitis, ascites, and elevated ALT were identified as adverse prognostic factors, whereas higher serum ALB levels were associated with favorable prognosis. Notably, the HRs for AST and PT were less than 1, which may reflect complex interactions among hepatic function markers and other covariates in the multivariate model, rather than indicating a strictly protective effect.
Based on the results of multivariate Cox regression analysis, a nomogram model for predicting the risk of IH after liver transplantation was developed (Figure 2), incorporating significantly associated variables. Each predictor was assigned a score according to its regression coefficient, and the cumulative score was used to estimate the probability of IH occur
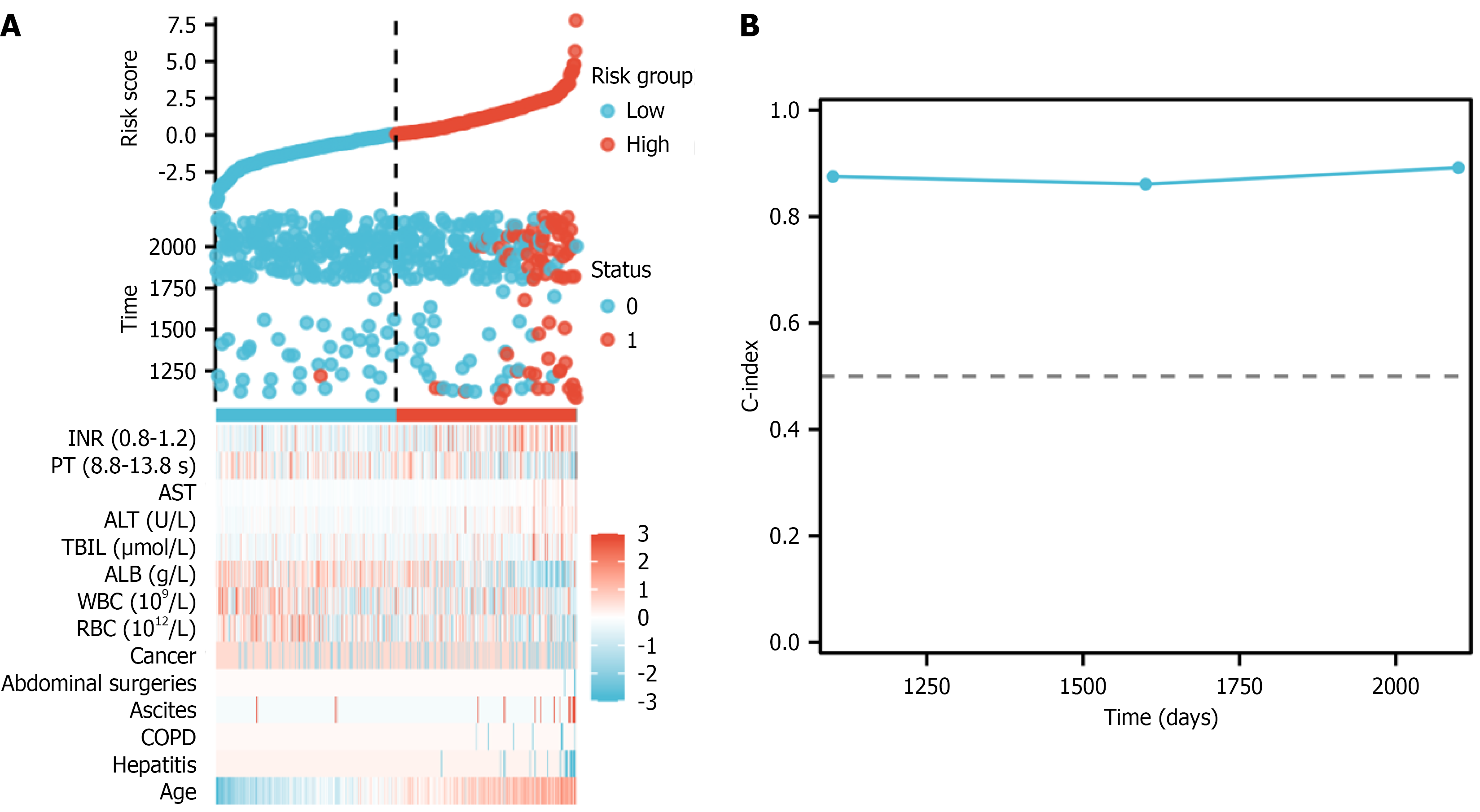
This study is the first to establish a validated nomogram specifically predicting postoperative IH following liver transplantation, providing an individualized approach to risk assessment. The findings demonstrated that hepatitis, ascites, abnormal serum ALB levels, liver function impairment (ALT, AST), and coagulation dysfunction (prolonged PT) were closely associated with IH occurrence. Among these, hepatitis, ascites, and hypoalbuminemia were confirmed as independent risk factors in multivariate Cox analysis, underscoring their clinical importance in risk assessment and the development of preventive strategies. The nomogram derived from these factors achieved a C-index of 0.874, indicating strong predictive accuracy and good discrimination. Risk stratification further validated its clinical utility, as high-risk patients exhibited significantly higher IH incidence compared with low-risk patients, suggesting that this model could serve as a practical tool for individualized risk assessment and follow-up management. Consistent with previous lite
The occurrence of IH after laparotomy is influenced by multiple factors, including obesity, smoking, chronic lung disease, ascites or hyperbilirubinemia, anemia, malnutrition, connective tissue disorders, immunosuppressive therapy, age > 70 years, and male sex[14-16]. Liver transplant recipients are particularly vulnerable, not only due to these factors but also due to transplant-specific risks such as prolonged immunosuppression, hepatic dysfunction, and coagulation abnormalities. In this study, hepatitis significantly increased IH risk, potentially through mechanisms related to coagulopathy, hypoalbuminemia, and malnutrition, all of which impair wound healing. Hypoalbuminemia was also identified as an independent risk factor, consistent with its pivotal role in collagen synthesis and wound repair[17,18]. These findings highlight the need for clinicians to emphasize perioperative nutritional assessment and active correction of hypoalbuminemia. Furthermore, ascites is a well-recognized risk factor for IH, likely contributing through increased abdominal wall tension and delayed wound healing[19-21]. In other transplant fields, such as kidney transplantation, the incidence of IH is relatively lower, with major risk factors including overweight, pulmonary complications, and lymphocele[22-24], suggesting organ-specific differences in IH risk profiles.
Regarding hernia repair, the literature reports an overall IH incidence of approximately 15.1% following liver transplantation, typically occurring in the intermediate to late postoperative period, with recurrence rates around 12.4% after initial repair[25,26]. In our study, follow-up of patients who underwent hernia repair revealed favorable short-term quality of life outcomes, with no complications or recurrences observed, indicating that surgical repair in stable patients can achieve good short-term efficacy. Additionally, the nomogram established in this study provides a quantitative tool to estimate individual IH risk, aiding in early identification of high-risk patients and guiding personalized management strategies. For example, perioperative measures for high-risk patients could include reinforced incision protection, optimized immunosuppressive regimens, strict intra-abdominal pressure control, and long-term dynamic monitoring to reduce IH incidence. The strengths of this study include: (1) A relatively adequate sample size covering multiple clinical indicators, allowing for a comprehensive analysis; and (2) The first construction of a predictive nomogram for IH risk specifically in liver transplant recipients, providing an intuitive tool for clinical risk prediction. Nonetheless, several limitations should be noted. First, this was a single-center retrospective cohort study, with potential selection and in
In conclusion, this study identified hepatitis and hypoalbuminemia as independent risk factors for IH after liver transplantation and established a feasible predictive model. Future research should integrate prospective multicenter data to further investigate transplant-specific IH risk factors and refine risk stratification tools, thereby guiding precision prevention and targeted interventions.
| 1. | Mudge M, Hughes LE. Incisional hernia: a 10 year prospective study of incidence and attitudes. Br J Surg. 1985;72:70-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 717] [Cited by in RCA: 633] [Article Influence: 15.4] [Reference Citation Analysis (0)] |
| 2. | Höer J, Lawong G, Klinge U, Schumpelick V. [Factors influencing the development of incisional hernia. A retrospective study of 2,983 laparotomy patients over a period of 10 years]. Chirurg. 2002;73:474-480. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 195] [Cited by in RCA: 227] [Article Influence: 9.5] [Reference Citation Analysis (1)] |
| 3. | van Ramshorst GH, Eker HH, Hop WC, Jeekel J, Lange JF. Impact of incisional hernia on health-related quality of life and body image: a prospective cohort study. Am J Surg. 2012;204:144-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 207] [Cited by in RCA: 303] [Article Influence: 21.6] [Reference Citation Analysis (0)] |
| 4. | Parker SG, Mallett S, Quinn L, Wood CPJ, Boulton RW, Jamshaid S, Erotocritou M, Gowda S, Collier W, Plumb AAO, Windsor ACJ, Archer L, Halligan S. Identifying predictors of ventral hernia recurrence: systematic review and meta-analysis. BJS Open. 2021;5:zraa071. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 97] [Article Influence: 19.4] [Reference Citation Analysis (1)] |
| 5. | Mavros MN, Athanasiou S, Alexiou VG, Mitsikostas PK, Peppas G, Falagas ME. Risk factors for mesh-related infections after hernia repair surgery: a meta-analysis of cohort studies. World J Surg. 2011;35:2389-2398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 107] [Article Influence: 7.6] [Reference Citation Analysis (1)] |
| 6. | Kroese LF, Gillion JF, Jeekel J, Kleinrensink GJ, Lange JF; Hernia-Club Members. Primary and incisional ventral hernias are different in terms of patient characteristics and postoperative complications - A prospective cohort study of 4,565 patients. Int J Surg. 2018;51:114-119. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 45] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 7. | Stabilini C, Cavallaro G, Dolce P, Capoccia Giovannini S, Corcione F, Frascio M, Sodo M, Merola G, Bracale U. Pooled data analysis of primary ventral (PVH) and incisional hernia (IH) repair is no more acceptable: results of a systematic review and metanalysis of current literature. Hernia. 2019;23:831-845. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 45] [Article Influence: 6.4] [Reference Citation Analysis (1)] |
| 8. | Köckerling F, Schug-Paß C, Adolf D, Reinpold W, Stechemesser B. Is pooled data analysis of ventral and incisional hernia repair acceptable? Front Surg. 2015;2:15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 32] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 9. | Fikatas P, Schoening W, Lee JE, Chopra SS, Seehofer D, Guckelberger O, Puhl G, Neuhaus P, Schmidt SC. Incidence, risk factors and management of incisional hernia in a high volume liver transplant center. Ann Transplant. 2013;18:223-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 42] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 10. | Cos H, Ahmed O, Garcia-Aroz S, Vachharajani N, Shenoy S, Wellen JR, Doyle MM, Chapman WC, Khan AS. Incisional hernia after liver transplantation: Risk factors, management strategies and long-term outcomes of a cohort study. Int J Surg. 2020;78:149-153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (2)] |
| 11. | Ayvazoglu Soy EH, Kirnap M, Yildirim S, Moray G, Haberal M. Incisional Hernia After Liver Transplant. Exp Clin Transplant. 2017;15:185-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 16] [Article Influence: 1.8] [Reference Citation Analysis (2)] |
| 12. | Hegab B, Abdelfattah MR, Azzam A, Al Sebayel M. The usefulness of laparoscopic hernia repair in the management of incisional hernia following liver transplantation. J Minim Access Surg. 2016;12:58-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 1.4] [Reference Citation Analysis (4)] |
| 13. | Bosanquet DC, Ansell J, Abdelrahman T, Cornish J, Harries R, Stimpson A, Davies L, Glasbey JC, Frewer KA, Frewer NC, Russell D, Russell I, Torkington J. Systematic Review and Meta-Regression of Factors Affecting Midline Incisional Hernia Rates: Analysis of 14,618 Patients. PLoS One. 2015;10:e0138745. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 260] [Cited by in RCA: 298] [Article Influence: 27.1] [Reference Citation Analysis (1)] |
| 14. | Maki H, Kim BJ, Kawaguchi Y, Fernandez-Placencia R, Haddad A, Panettieri E, Newhook TE, Baumann DP, Santos D, Tran Cao HS, Chun YS, Tzeng CD, Vauthey JN, Vreeland TJ. Incidence of and Risk Factors for Incisional Hernia After Hepatectomy for Colorectal Liver Metastases. J Gastrointest Surg. 2023;27:2388-2395. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 15. | Barranquero AG, Molina JM, Gonzalez-Hidalgo C, Porrero B, Blázquez LA, Ocaña J, Gandarias Zúñiga C, Fernández Cebrián JM. Incidence and risk factors for incisional hernia after open abdominal aortic aneurysm repair. Cir Esp (Engl Ed). 2022;100:684-690. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 16. | Gianchandani R, Pérez E, Moneva E, Menéndez A, Sánchez JM, Díaz C, Concepción V, Barrera MA. Laparoscopic Incisional Hernia Repair After Liver Transplantation: Long-Term Series and Literature Review. Transplant Proc. 2020;52:1514-1517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 17. | Alhambra-Rodríguez de Guzmán C, Morandeira-Rivas AJ, Herrero-Bogajo ML, Moreno-Sanz C. Incidence and Risk Factors of Incisional Hernia After Single-Incision Endoscopic Surgery. J Laparoendosc Adv Surg Tech A. 2020;30:251-255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 13] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 18. | Ortega-Deballon P, Renard Y, de Launay J, Lafon T, Roset Q, Passot G. Incidence, risk factors, and burden of incisional hernia repair after abdominal surgery in France: a nationwide study. Hernia. 2023;27:861-871. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 27] [Reference Citation Analysis (1)] |
| 19. | Schaffellner S, Sereinigg M, Wagner D, Jakoby E, Kniepeiss D, Stiegler P, Haybäck J, Müller H. Ventral incisional hernia (VIH) repair after liver transplantation (OLT) with a biological mesh: experience in 3 cases. Z Gastroenterol. 2016;54:421-425. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 7] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 20. | Kang BM. Risk of incisional hernia after laparoscopic colorectal surgery: surgeon's worries and challenges. J Minim Invasive Surg. 2022;25:9-10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 21. | Mao Y, Xi L, Lu C, Miao J, Li Q, Shen X, Yu C. Incidence, risk factors, and predictive modeling of stoma site incisional hernia after enterostomy closure: a multicenter retrospective cohort study. BMC Gastroenterol. 2023;23:201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 22. | Costa L, Martin D, Zingg T, Venetz JP, Demartines N, Golshayan D, Matter M. Incidence, Risk Factors, and Management of Incisional Hernias After Kidney Transplant: A 20-Year Single Center Experience. Transplant Proc. 2023;55:337-341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (1)] |
| 23. | Solano QP, Thumma JR, Mullens C, Howard R, Ehlers A, Delaney L, Fry B, Shen M, Englesbe M, Dimick J, Telem D. Variation of ventral and incisional hernia repairs in kidney transplant recipients. Surg Endosc. 2023;37:3173-3179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 24. | Cassese G, Castaldi A, Al Taweel B, Le Quintrec M, Thuret R, Navarro F, Panaro F. Incisional hernia repair after kidney transplantation in a tertiary high-volume center: outcomes from a 10-year retrospective cohort study. Int Urol Nephrol. 2022;54:525-531. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 25. | Butler JR, O'Brien DC, Kays JK, Kubal CA, Ekser B, Fridell JA, Mangus RS, Powelson JA. Incisional Hernia After Orthotopic Liver Transplantation: A Systematic Review and Meta-analysis. Transplant Proc. 2021;53:255-259. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 26. | Frountzas M, Nikolaou C, Maris S, Stavrou E, Giannopoulos P, Schizas D, Stergios K, Toutouzas K. Open or laparoscopic mesh repair of incisional hernia in patients that underwent liver transplantation: A systematic review and proportional meta-analysis. Clin Transplant. 2020;34:e14103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (1)] |