Published online Mar 27, 2026. doi: 10.4240/wjgs.v18.i3.114683
Revised: November 29, 2025
Accepted: January 12, 2026
Published online: March 27, 2026
Processing time: 150 Days and 3.7 Hours
Postoperative recovery in hepatectomy patients is often prolonged; early mobi
To develop an early postoperative activity program for patients undergoing hepatectomy and to investigate its effect on rehabilitation quality.
Two hundred patients undergoing hepatectomy, between January 2022 and Janu
Patients in the intervention group achieved their first postoperative ambulation, passed flatus, completed hospitalization, and had drainage tubes removed earlier than those in the control group. Their International Classification of Functioning, Disability and Health - 20-item Functioning Scale fatigue scores on postoperative day 5 were lower than those of the control group, and their 6-minute walk test distances at discharge were longer. They demonstrated higher 36-Item Short Form Health Survey scores at discharge and 1 month postoperatively (P < 0.05). The 30-day postoperative complication rate (15.00%) was lower than that in the control group (31.00%), while overall nursing satisfaction (96.00%) exceeded that of the control group (82.00%), with all differences being statistically significant (P < 0.05). Patients in the intervention group exhibited lower Visual Analogue Scale pain scores and reduced C-reactive protein and interleukin-6 levels on postoperative days 3 and 5 compared to those in the control group, with a statistically significant interaction effect between the time and group (P < 0.05).
The early postoperative mobilization program is scientifically sound and effective. It significantly improves rehabilitation outcomes in hepatectomy patients, shortens the recovery time, reduces complication rates, alleviates pain, diminishes inflammatory and stress responses, enhances functional recovery and quality of life, and increases nursing satisfaction. This program demonstrates considerable value for clinical implementation.
Core Tip: An early postoperative activity program for hepatectomy patients significantly enhances recovery by accelerating ambulation, gastrointestinal function, and hospital discharge. It reduces pain, inflammatory and stress responses, and postoperative complications, while improving functional capacity, quality of life, and nursing satisfaction. This evidence-based program provides a practical and effective strategy for the perioperative care of hepatectomy patients.
- Citation: Xing YM, Zhang LQ, Wei MX, Shen YQ, Luan LL. Development of an early mobilization protocol for patients following hepatectomy and its effect on rehabilitation outcomes. World J Gastrointest Surg 2026; 18(3): 114683
- URL: https://www.wjgnet.com/1948-9366/full/v18/i3/114683.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i3.114683
Hepatic resection is the primary surgical intervention used for treating benign and malignant liver tumors and other hepatic pathologies. With continuous advancements in surgical techniques, anesthetic management, and perioperative nursing standards, the safety profile of this procedure has markedly improved[1,2]. Nevertheless, as a highly invasive and complex abdominal surgical procedure, hepatectomy inevitably induces severe physiological stress in patients. This results in protracted postoperative recovery and is associated with risks of complications, including pulmonary infection, deep vein thrombosis, intestinal obstruction, and delayed hepatic functional recovery, significantly compromising patients’ quality of life and long-term prognosis[3,4]. Accordingly, ways to promote rapid, high-quality recovery in hepatectomy patients through optimized perioperative management has become a key research focus and challenge for hepatobiliary surgery[5,6]. Using traditional nursing models, prolonged bed rest is often mandated owing to concerns over pain, drainage tube dislodgement, and bleeding risks. However, mounting clinical evidence indicates that prolonged bed rest induces adverse effects, including altered respiratory mechanics, pulmonary secretion retention, slowed lower limb blood flow, gastrointestinal motility suppression, and muscle disuse atrophy. This increases the risk of pulmonary infection, deep vein thrombosis, abdominal distension, and intestinal obstruction[7,8]. Therefore, establishing a specific, detailed, clearly structured, and safe early mobilization protocol for patients undergoing liver resection has significant clinical importance for enhancing the quality of recovery. Consequently, in this study, 200 liver resection patients admitted to the hospital between January 2022 and January 2023 were selected as subjects. The goal was to develop an early postoperative mobilization protocol and evaluate its efficacy, aiming to provide scientific evidence for clinical practice to improve rehabilitation outcomes in liver resection patients.
Following review and approval granted by the hospital ethics committee, 200 patients undergoing hepatectomy, admitted to the hospital between January 2022 and January 2023, were selected as the study subjects. The sample size was estimated as follows. Based on prior literature and pre-experimental results, a test level of α = 0.05 (two-tailed) was set, with a test power of 1-β = 0.90, a pooled standard deviation of σ ≈ 3.0, and an expected effect size of δ = 2.5. Considering the multi-criteria evaluation performed in this study and potential for up to 20% case attrition, exclusion, or incomplete data, the final sample size was determined as 100 subjects per group, totaling 200 subjects. The inclusion criteria were as follows: Age, 18-75 years; diagnosed via imaging and pathology, undergoing elective open or laparoscopic partial hepatectomy; American Society of Anesthesiologists physical status classification, I-III; informed consent and voluntary participation in the study. The exclusion criteria were as follows: Emergency surgery or combined multi-organ resection; severe cardiac, pulmonary, or renal insufficiency precluding early mobilization; pre-operative mobility impairment (e.g., hemiplegia, fractures) or cognitive dysfunction precluding cooperation; post-operative intensive care unit admission exceeding 24 hours. Patients were randomized, using a random number table, into intervention and control groups, each comprising 100 subjects. Baseline characteristics were comparable between the groups (P > 0.05). Table 1 provides this information.
| Group | n | Sex | Age (years) | BMI (kg/m2) | ASA classification II |
| Male | Female | ||||
| Intervention group | 100 | 58 (58.0) | 42 (42.0) | 57.32 ± 7.83 | 23.84 ± 2.26 |
| Control group | 100 | 60 (60.0) | 40 (40.0) | 56.62 ± 7.91 | 23.59 ± 2.34 |
| χ2/t value | - | 0.081 | 0.624 | 0.793 | 0.382 |
| P value | - | 0.776 | 0.533 | 0.429 | 0.826 |
The control group received conventional perioperative care for hepatectomy: Preoperative health education, routine bowel preparation, postoperative vital signs monitoring, fluid management, analgesia via pain pumps, on-demand antiemetic medication, care for abdominal drainage tubes, routine encouragement of bedside activities, and ambulation based on patient tolerance.
Intervention group: Please note that the in addition to standard care, the early postoperative mobilization protocol developed for this study was implemented; this included a multidisciplinary intervention team comprising specialists from Hepatobiliary Surgery, Anaesthesiology, Rehabilitation Medicine, and Nutrition, who developed an early mobilization protocol for hepatectomy based on enhanced recovery after surgery (ERAS) principles. This was achieved through a systematic review of domestic and international literature, combined with discussion tailored to our hospital’s context. All participating healthcare personnel underwent standardized training on the early mobilization protocol. Implementation commenced only after comprehensive assessment and certification, ensuring consistent execution. A detailed preoperative explanation of the content, objectives, and significance of the program was provided to patients and their families to ensure a full understanding and cooperation, accompanied by the distribution of illustrated activity guidance manuals. Multimodal precision analgesia was employed perioperatively to maintain Visual Analogue Scale (VAS) scores ≤ 4 in the patients. The thorough management of drainage tubes, urinary catheters, and other lines was implemented, utilizing shoulder-strap drainage bags to free patients’ hands and facilitate early mobilization. A “Postoperative Early Mobilisation Implementation Record Form” was developed for nurses to document each the execution, duration, and patient response associated with the activity session, with daily supervision performed by the head nurse.
Principles for individualized protocol adjustments: The early mobilization protocol served as a core framework. In practice, the assigned nurse and rehabilitation physician individualized the regimen based on the patient’s specific condition. Adjustments were guided by the following parameters.
Extent of resection: For patients undergoing major hepatic resection (e.g., hemihepatectomy or more), the initial activity intensity (e.g., timing of first ambulation, walking distance) was appropriately reduced, and the progression rate was decreased.
Surgical approach: For patients who underwent laparoscopic surgery, owing to lesser trauma and milder pain, the activity progression could be advanced accordingly - for example, attempting to sit on the edge of the bed or stand bedside within 24 hours postoperatively.
Patient’s baseline condition: For an advanced age (> 70 years) or frailty, gradual progression, prolonged bedside activity time, and required dedicated assistance during ambulation to prevent falls were emphasized.
Comorbidities such as diabetes or malnutrition: Close monitoring of activity tolerance and wound conditions was essential, ensuring adequate nutritional support to prevent increased energy expenditure from activities that would hinder recovery.
Poorly controlled pain (VAS > 4) or presence of significant dizziness, palpitations, or shortness of breath: Activities were paused or reduced, the underlying cause was actively addressed, and the protocol was reassessed and resumed only after symptom resolution. The early mobilization protocol for hepatectomy is detailed in Table 2.
| Postoperative time | Activity phase | Activity content and requirements | Person responsible for implementation | Adjustment principles and notes |
| 0-6 hours | Anesthesia recovery period | Supine position without pillow, head turned to one side; guide and assist patient in ankle pump exercises (flexion, extension, circular movements), 20 repetitions per set, one set every 2 hours | Assigned nurse | Applicable to all patients. Assess patient’s consciousness and vital signs |
| 6-24 hours | Bedside active movement period | Gradually raise head of bed to semi-recumbent position (30° → 45° → 60°) | Guided by nurse; performed by patient | For laparoscopic surgery patients or those in good general condition, attempting to sit on the edge of the bed with nurse assistance by the end of this phase can be considered. For elderly, frail, or patients with extensive resection, focus on bedside activities and prolong the adaptation time for semi-recumbent positions |
| Continue and intensify ankle pump exercises | ||||
| Guide patient in quadriceps isometric contractions and knee flexion/extension exercises | ||||
| Encourage deep breathing and effective coughing 5-10 times every hour | ||||
| 24-48 hours | Initial out-of-bed activity period | With nurse or family assistance, slowly sit on the edge of the bed, legs hanging, adapt for 3-5 minutes | Nurse/family assistance | These are standard recommendations. Laparoscopic surgery patients with rapid recovery may appropriately increase activity volume and frequency. For open surgery, extensive resection, or physically weak patients, strictly adhere to the “gradual progression” principle. The timing of first ambulation may be postponed as appropriate; the standing and walking duration should be shortened, based on patient tolerance and absence of discomfort |
| If no discomfort, stand with support for 1-2 minutes | ||||
| Walk slowly along bedside 5-10 m, 2-3 times daily | ||||
| > 48 hours | Progressive activity stage | Gradually increase distance, frequency, and duration according to patient tolerance | Supervised by nurse; performed by patient | The goal represents an ideal state and requires individualized assessment. For patients with slow recovery, the goal can be adjusted to walking with assistance or appropriately reducing the distance; the key is persistent daily activity. For patients with comorbid cardiovascular diseases, closely monitor heart rate and blood pressure changes during activity |
| Goal: By postoperative day 3, patient can walk independently > 100 m within the ward, 3-4 times daily |
The following protocols were implemented to compare differences between the two groups in terms of postoperative recovery indicators, complications, pain levels, inflammation and stress response indicators, functional recovery and quality of life levels, and nursing satisfaction.
Postoperative recovery indicators: The time of first ambulation, first passage of wind, length of hospital stay, and time of drainage tube removal for both groups were recorded.
Postoperative complications: The incidences of complications within 30 days postoperatively, including pulmonary infection, pleural effusion, deep vein thrombosis, intestinal obstruction, wound infection, bile leakage, and postoperative hemorrhage were documented.
Pain intensity: Pain levels at rest were assessed using the VAS at 6 hours, 24 hours, 3 days, and 5 days postoperatively.
Inflammatory and stress response markers: Fasting venous blood samples were collected preoperatively (day 1) and postoperatively (days 1, 3, and 5). Serum C-reactive protein (CRP) and interleukin-6 (IL-6) levels were measured using a fully automated biochemical analyzer.
Functional recovery and quality of life assessment: International Classification of Functioning, Disability and Health - 20-item Functioning Scale (ICF-20) was administered on postoperative day 5. This 20-item scale scores up to 100 points, with higher scores indicating greater fatigue severity. The 6-minute walk test (6MWT) was conducted at discharge, recording the maximum distance walked within 6 min to objectively evaluate the physical capacity and functional status. The 36-Item Short Form Health Survey (SF-36) was administered at discharge and 1 month postoperatively. Nursing satisfaction was assessed at discharge using our hospital’s proprietary nursing satisfaction questionnaire, scored out of 100. Scores ≥ 90 indicate “very satisfied”, 80-89 indicate “satisfied”, and < 80 indicate “dissatisfied”, calculated as follows: The overall satisfaction rate = (number of “very satisfied” + number of “satisfied”)/total number of cases × 100%.
Data were analyzed using SPSS 26.0. Count data, n (%) were subjected to χ2 tests; continuous data demonstrated a normal distribution based on Shapiro-Wilk tests. Between-group comparisons were assessed by performing t-tests, with P < 0.05 indicating statistically significant differences.
Patients in the intervention group demonstrated statistically significant reductions in the time to first ambulation, time to first passage of wind, length of hospital stay, and time to removal of drainage tubes compared with those in the control group (P < 0.05) (Table 3).
| Group | n | Time to first ambulation (hour) | Time to first flatus (hour) | Postoperative hospital stay (days) | Time to drain removal (hour) |
| Intervention group | 100 | 28.53 ± 6.27 | 45.66 ± 10.35 | 5.88 ± 1.57 | 9.21 ± 2.16 |
| Control group | 100 | 45.88 ± 9.79 | 62.43 ± 12.89 | 7.92 ± 2.23 | 12.50 ± 3.35 |
| t value | - | 15.207 | 10.432 | 7.894 | 8.563 |
| P value | - | < 0.001 | < 0.001 | < 0.001 | < 0.001 |
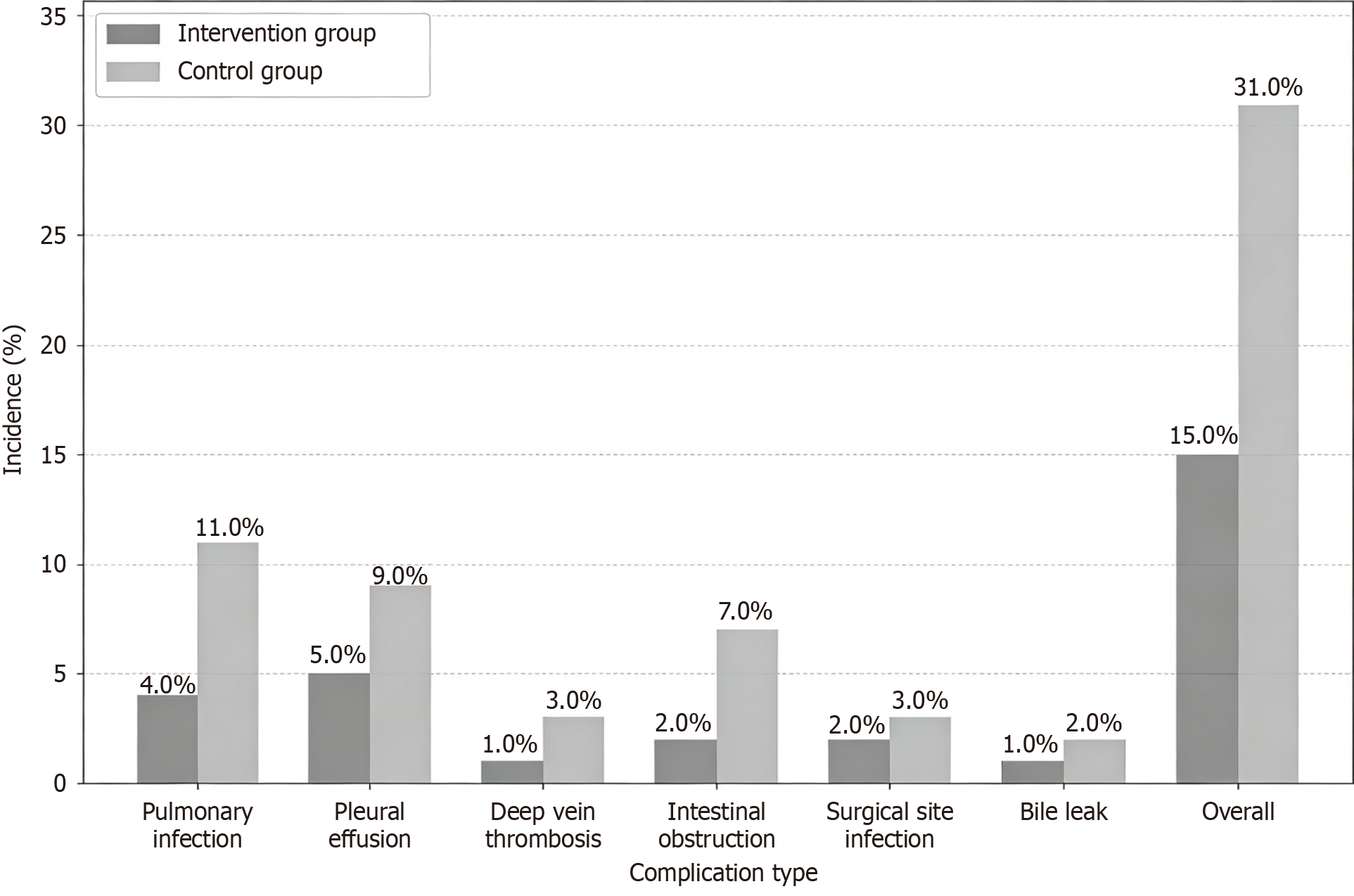
As illustrated in Figure 1, the incidence of complications, such as pulmonary infection, pleural effusion, deep vein thrombosis, intestinal obstruction, wound infection, and bile leakage within 30 days postoperatively was lower in the observation group (15.0%, 15/100) than in the control group (31.0%, 31/100), with this difference being statistically significant (χ2 = 7.440, P = 0.006).
Patients in the intervention group had significantly lower VAS pain scores on postoperative days 3 and 5 compared to those in the control group, with a statistically significant interaction effect detected between the time and group (P < 0.05) (Table 4).
| Group | n | Post-6 hours | Post-24 hours | Post-3 days | Post-5 days |
| Intervention group | 100 | 4.52 ± 1.23 | 3.87 ± 1.05 | 2.59 ± 0.82 | 1.64 ± 0.73 |
| Control group | 100 | 4.71 ± 1.36 | 3.93 ± 1.16 | 3.27 ± 1.05 | 2.32 ± 0.96 |
| t value | - | 1.052 | 0.387 | 5.432 | 5.876 |
| P value | - | 0.294 | 0.699 | < 0.001 | < 0.001 |
| Main effect of time | Wald χ2 = 215.36, P < 0.001 | ||||
| Main effect of group | Wald χ2 = 18.74, P < 0.001 | ||||
| Time × group interaction | Wald χ2 = 25.83, P < 0.001 | ||||
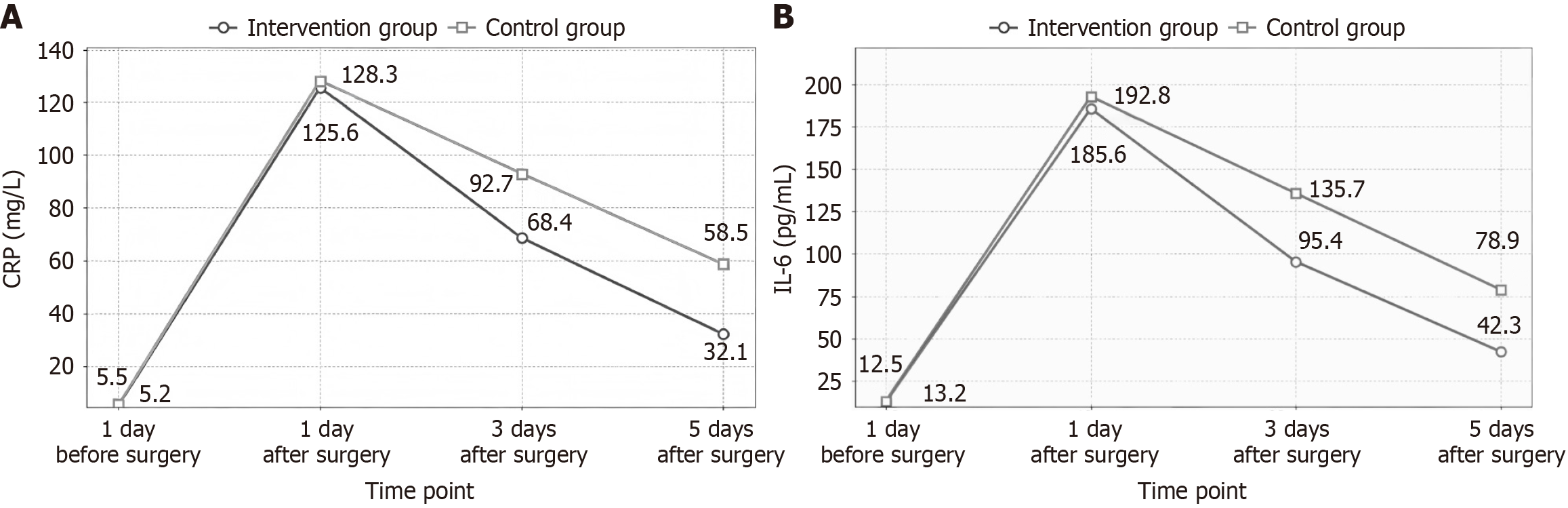
One day preoperatively, CRP and IL-6 levels showed no statistically significant differences between the groups (P > 0.05). On postoperative day 1, both groups exhibited significantly elevated CRP and IL-6 levels compared to preoperative values, though no intergroup difference was observed (P > 0.05). On postoperative days 3 and 5, CRP and IL-6 levels in the intervention group were significantly lower than those in the control group (P < 0.05). The interaction effect between the time and group was also statistically significant (P < 0.05) (Figure 2).
Patients in the intervention group had lower ICF-20 fatigue scores on postoperative day 5 compared to those in the control group. Moreover, at discharge, they achieved longer distances in the 6MWT than those in the control group. Both at discharge and 1 month postoperatively, their SF-36 scores were also significantly higher than those in the control group (P < 0.05) (Table 5).
| Group | n | ICF-20 | 6MWT (m) | SF-36 | |
| Pre-1 day | Post-7 days | ||||
| Intervention group | 100 | 48.59 ± 12.37 | 285.63 ± 45.88 | 70.54 ± 11.26 | 78.96 ± 10.55 |
| Control group | 100 | 62.87 ± 15.62 | 228.45 ± 52.34 | 62.18 ± 12.83 | 70.24 ± 11.82 |
| t | - | 7.123 | 8.324 | 5.236 | 6.457 |
| P value | - | < 0.001 | < 0.001 | < 0.001 | < 0.001 |
The overall satisfaction rate in the intervention group (96.0%) was significantly higher than that in the control group (82.0%) (P < 0.05) (Table 6).
| Group | n | Very satisfied | Satisfied | Dissatisfied | Overall satisfaction |
| Intervention group | 100 | 65 (65.0) | 31 (31.0) | 4 (4.0) | 96 (96.0) |
| control group | 100 | 48 (48.0) | 34 (34.0) | 18 (18.0) | 82 (82.0) |
| χ2 value | - | - | - | - | 10.256 |
| P value | - | - | - | - | 0.001 |
Currently, clinical practice regarding postoperative mobilization following liver resection varies considerably across China. The timing, intensity, and methods of mobilization often rely on the personal experience of healthcare professionals or patients themselves, with high-level evidence-based guidance and systematic protocols lacking. This results in conservative, delayed, or inadequate implementation, hindering the maximization of clinical benefits from early mobilization. In recent years, the ERAS protocol has been widely adopted and recognized globally within the surgical field. Its core principle involves implementing a series of evidence-based perioperative optimization measures to mitigate the patient’s surgical trauma stress response. This approach reduces complications, shortens hospital stays, and accelerates patient recovery[9,10]. Early postoperative mobilization, by challenging the conventional mindset of bed rest, significantly improves multiple systemic functions including respiration, circulation, and digestion. It also enhances metabolic capacity and immune functions, thereby accelerating the recovery process. This approach is widely recognized as a crucial and highly effective component of the ERAS multimodal protocol[11,12]. In this study, an early postoperative activity program based on ERAS principles within the context of postoperative rehabilitation care for liver resection patients was established, achieving favorable clinical outcomes.
Patients in the intervention group achieved earlier postoperative ambulation, earlier passage of wind, shorter postoperative hospital stays, and earlier removal of drainage tubes than the control group (P < 0.05). This outcome aligns closely with the core principles of the ERAS protocol, specifically “early ambulation, early oral intake, and early removal of tubes”[13,14]. Early ambulation activates skeletal muscle pump functions, promoting venous return in the lower limbs and reducing blood stasis, thereby lowering the risk of deep vein thrombosis[15,16]. Concurrently, upright activity facilitates diaphragmatic descent and increased lung volumes, promoting alveolar re-expansion and sputum clearance, effectively preventing atelectasis and pulmonary infection[17,18]. Moreover, early mobilization stimulates intestinal peristalsis, accelerates gastrointestinal functional recovery, and reduces the incidence of intestinal obstruction. This aligns with the intervention group’s significantly earlier anal flatus time, fully demonstrating the positive effect of early mobilization on promoting gastrointestinal functional recovery in patients.
Concurrently, patients in the intervention group exhibited lower VAS pain scores on postoperative days 3 and 5, alongside reduced CRP and IL-6 levels, compared to those in the control group. Furthermore, the interaction effect between the time and group demonstrated statistical significance (P < 0.05). Pain constitutes a common postoperative discomfort, not only disrupting rest and sleep but also intensifying the body’s stress response. Therefore, it represents one of the primary obstacles to early mobilization, thereby impeding the rehabilitation process[19,20]. When patients engage in early activity, stimulation of the nervous system promotes the release of endogenous analgesic substances, such as endorphins, effectively reducing pain perception[21,22]. Conversely, elevated levels of CRP and IL-6, key markers of inflammation and the stress response, often indicate the presence of inflammatory and stress states within the body[23,24]. The lower pain scores and decreased levels of inflammatory stress markers observed in the intervention group may be attributed to early mobilization promoting blood circulation. This, in turn, accelerates the metabolism and clearance of inflammatory mediators by modulating immune responses and mitigating the inflammatory cascade following tissue injury, thereby lowering systemic inflammation levels. Steinthorsdottir et al[25] similarly demonstrated that combined preoperative glucocorticoid intervention with early postoperative mobilization significantly reduces inflammatory markers in hepatectomy patients, consistent with the present findings.
The findings further revealed that patients in the intervention group had lower ICF-20 fatigue scores on postoperative day 5 than the control group, achieved greater 6MWT distances at discharge, and demonstrated superior SF-36 scores both at discharge and 1 month postoperatively (P < 0.05). This indicates that early mobilization not only shortens the physiological recovery time but also has positive effects on psychological and social functioning. Early mobilization disrupts the vicious cycle of “bed rest-fatigue-bed rest”, enhancing patients’ muscle strength, endurance, and balance while boosting their self-efficacy and confidence in recovery[26,27]. The improvement in the 6MWT results also reflects enhanced cardiopulmonary functions and overall physical fitness, consistent with the findings by Olivares et al[28] based on cardiac surgery patients. The elevation in SF-36 scores further indicates that early mobilization not only accelerates short-term recovery but may also exert sustained positive effects on patients’ long-term quality of life. Consequently, early mobilization holds significant value for the postoperative rehabilitation of liver resection patients, demonstrating marked promotional effects across physiological and psychological domains, from immediate recovery to long-term quality of life.
Furthermore, the incidence of complications within 30 days post-surgery was 15.00% in the intervention group, lower than the 31.00% observed in the control group. In addition, overall nursing satisfaction was 96.00% in the intervention group, higher than the 82.00% value noted for the control group, and this difference was statistically significant (P < 0.05). Notably, the intervention group demonstrated markedly superior control over common complications, including pul
However, this study had some limitations. First, as a single-center investigation, its sample representativeness is constrained. We included patients undergoing elective surgery, classified as American Society of Anesthesiologists I-III, with relatively stable conditions, excluding high-risk cohorts that would potentially most require the early mobilization intervention (such as emergency surgery, multi-organ resection, or those requiring prolonged post-operative intensive care unit monitoring). This may affect the generalizability of findings to complex real-world cases. In future studies, sample sizes should be expanded, multicenter collaborations should be conducted, and the inclusion of these high-risk subgroups for targeted analysis should be considered. Second, the current follow-up period, ending 1 month postoperatively, remains insufficient for assessing the long-term benefits of early mobilization protocols (such as sustained effects on the hepatic reserve, long-term complications, and quality of life). Although we plan to extend the follow-up to 6 months and supplement the protocol with long-term endpoints, such as liver function markers and readmission rates, the existing data remain insufficient to fully reflect the long-term effects of the intervention. Extending the follow-up to 1 year or beyond would enable a more comprehensive assessment of the overall value of the program.
In summary, the early postoperative activity program established in this study is scientifically sound and effective. Moreover, it significantly improves the rehabilitation quality of hepatectomy patients, shortens postoperative recovery times, reduces the incidence of complications, alleviates pain levels, diminishes inflammatory and stress responses, enhances functional recovery and quality of life, and increases nursing satisfaction. Consequently, it holds considerable value for clinical implementation.
| 1. | Ravenet A, Rhaiem R. Implications of the surgical approach for complex liver resection in the era of enhanced recovery programs: a critical appraisal of the "Orange Segments" randomized controlled trial. Hepatobiliary Surg Nutr. 2025;14:634-638. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 2. | Jongkatkorn C, Luvira V, Suwanprinya C, Piampatipan K, Leeratanakachorn N, Tipwaratorn T, Titapun A, Srisuk T, Theeragul S, Jarearnrat A, Thanasukarn V, Pugkhem A, Khuntikeo N, Pairojkul C, Kamsa-Ard S, Bhudhisawasdi V. Compliance with enhanced recovery after surgery predicts long-term outcome after hepatectomy for cholangiocarcinoma. World J Gastrointest Surg. 2023;15:362-373. [PubMed] [DOI] [Full Text] |
| 3. | Bernardi L, Joliat GR, Sasaki S, Schneider M, Labgaa I, Roulin D, Addor V, Halkic N, Uldry E, Fuks D, Melloul E. Challenges in the implementation of Enhanced Recovery After Surgery (ERAS) pathway for major hepatectomy: A retrospective cohort study. Surgery. 2025;186:109569. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 4. | Berardi G, Cucchetti A, Colasanti M, Angrisani M, Moschetta G, Chiappori D, Marini A, Antonelli G, Ferretti S, Meniconi RL, Guglielmo N, Mariano G, Usai S, Ettorre GM. Prehabilitation With Exercise and Nutrition to Reduce Morbidity of Major Hepatectomy in Patients With Sarcopenia: The PREHEP Randomized Clinical Trial. JAMA Surg. 2025;160:1068-1075. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 19] [Article Influence: 19.0] [Reference Citation Analysis (1)] |
| 5. | Bhat MY, Ali S, Gupta S, Ahmad Y, Lattoo MR, Ansari MJ, Patel A, Haq MFU, Parveen S. Feasibility, safety and effectiveness of the enhanced recovery after surgery protocol in patients undergoing liver resection. Ann Hepatobiliary Pancreat Surg. 2024;28:344-349. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 6. | Toh EQ, Wong HPN, Wang JDJ, Liau MYQ, Tan YF, Shelat VG. Prehabilitation programs in liver resection: a narrative review. Chin Clin Oncol. 2024;13:9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 7. | Pradhan A, Sarkar A, Haldar S, Chakraborty A, Pal AR. Compliance to enhanced recovery program in liver resection surgery: A retrospective cohort study. J Anaesthesiol Clin Pharmacol. 2025;41:318-322. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 8. | Arfa S, Turco C, Lakkis Z, Bourgeois S, Fouet I, Evrard P, Sennegon E, Roucoux A, Paquette B, Devaux B, Rietsch-Koenig A, Heyd B, Doussot A. Delayed return of gastrointestinal function after hepatectomy in an ERAS program: incidence and risk factors. HPB (Oxford). 2022;24:1560-1568. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (1)] |
| 9. | Mingaud H, de Guibert JM, Garnier J, Chow-Chine L, Gonzalez F, Bisbal M, Alisauskaite J, Sannini A, Léone M, Tezier M, Tourret M, Cambon S, Ewald J, Pouliquen C, Nguyen Duong L, Ettori F, Turrini O, Faucher M, Mokart D. Incidence and Predictive Factors of Acute Kidney Injury After Major Hepatectomy: Implications for Patient Management in Era of Enhanced Recovery After Surgery (ERAS) Protocols. J Clin Med. 2025;14:5452. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 10. | Joliat GR, Delabays C, Uldry E, Addor V, Fuks D, Melloul E. Specific Items of Enhanced Recovery After Surgery for Liver Surgery in Cirrhotic Patients: A Systematic Review. World J Surg. 2025;49:2125-2143. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 11. | Jones CN, Morrison BL, Kelliher LJ, Dickinson M, Scott M, Cecconi Ebm C, Karanjia N, Quiney N. Hospital Costs and Long-term Survival of Patients Enrolled in an Enhanced Recovery Program for Open Liver Resection: Prospective Randomized Controlled Trial. JMIR Perioper Med. 2021;4:e16829. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 12. | Nari GA, Gutierrez EC, Layun JL, Falgueras L, Mariot D, Ferret G, Caula C, Góngora J. THERE ARE NO ADVANTAGES BETWEEN LAPAROSCOPIC AND OPEN LIVER RESECTIONS WITHIN AN ENHANCED RECOVERY PROGRAM (ERAS). Arq Bras Cir Dig. 2021;34:e1593. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 13. | Vercauteren J, De Meyere C, Parmentier I, Steelant PJ, D'Hondt M. Finetuning and optimizing an enhanced recovery protocol for liver surgery in the era of minimally invasive liver surgery: a single centre experience with 630 liver resections. HPB (Oxford). 2025;27:1214-1222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 14. | Jitsumura M, Sethi P, Welsh FKS, Chandrakumaran K, Rees M. Model for safe elective liver resection during the SARS-CoV-2 (COVID-19) pandemic: lessons for enhanced recovery. Br J Surg. 2021;108:e189-e190. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 15. | Hao S, Reis HL, Quinn AW, Snyder RA, Parikh AA. Prehabilitation for Older Adults Undergoing Liver Resection: Getting Patients and Surgeons Up to Speed. J Am Med Dir Assoc. 2022;23:547-554. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 16. | Yu K, Yin B, Zhu Y, Meng H, Zhu W, Lu L, Wang J, Chen S, Ni J, Lin Y, Jia J. Efficacy of a Digital Postoperative Rehabilitation Intervention in Patients With Primary Liver Cancer: Randomized Controlled Trial. JMIR Mhealth Uhealth. 2025;13:e59228. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 17. | Nakanishi W, Miyagi S, Tokodai K, Fujio A, Sasaki K, Shono Y, Unno M, Kamei T. Effect of enhanced recovery after surgery protocol on recovery after open hepatectomy: a randomized clinical trial. Ann Surg Treat Res. 2020;99:320-328. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 18. | Huang H, Zhou P, Li J, Luo H, Yu L. Enhanced recovery after surgery in primary liver cancer patients undergoing hepatectomy: experience from a large tertiary hospital in China. BMC Surg. 2023;23:185. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 19. | Qiu J, Wang Y. Effect of accelerated rehabilitation combined with enteral nutrition on gastrointestinal function recovery after hepatectomy. Support Care Cancer. 2022;30:8927-8933. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 20. | Li DX, Ye W, Yang YL, Zhang L, Qian XJ, Jiang PH. Enhanced recovery nursing and mental health education on postoperative recovery and mental health of laparoscopic liver resection. World J Gastrointest Surg. 2023;15:1728-1738. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 21. | Zhou J, He X, Wang M, Zhao Y, Wang L, Mao A, Wang L. Enhanced Recovery After Surgery in the Patients With Hepatocellular Carcinoma Undergoing Hemihepatectomy. Surg Innov. 2022;29:752-759. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 22. | Feng J, Li K, Xu R, Feng H, Han Q, Ye H, Li F. Association between compliance with enhanced recovery after surgery (ERAS) protocols and postoperative outcome in patients with primary liver cancer undergoing hepatic resection. J Cancer Res Clin Oncol. 2022;148:3047-3059. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 26] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 23. | Fu XT, Tang Z, Chen JF, Shi YH, Liu WR, Gao Q, Ding GY, Song K, Wang XY, Zhou J, Fan J, Ding ZB. Laparoscopic hepatectomy enhances recovery for small hepatocellular carcinoma with liver cirrhosis by postoperative inflammatory response attenuation: a propensity score matching analysis with a conventional open approach. Surg Endosc. 2021;35:910-920. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 24. | Fung AKY, Chong CCN, Lai PBS. ERAS in minimally invasive hepatectomy. Ann Hepatobiliary Pancreat Surg. 2020;24:119-126. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 25. | Steinthorsdottir KJ, Awada HN, Schultz NA, Larsen PN, Hillingsø JG, Jans Ø, Kehlet H, Aasvang EK. Preoperative high-dose glucocorticoids for early recovery after liver resection: randomized double-blinded trial. BJS Open. 2021;5:zrab063. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 26. | Zhou X, Zhou X, Cao J, Hu J, Topatana W, Li S, Juengpanich S, Lu Z, Zhang B, Feng X, Shen J, Chen M. Enhanced Recovery Care vs. Traditional Care in Laparoscopic Hepatectomy: A Systematic Review and Meta-Analysis. Front Surg. 2022;9:850844. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 27. | Lv Q, Xiang YC, Qiu YY, Xiang Z. Safety and efficacy of the enhanced recovery after surgery protocol in hepatectomy for liver cancer. Clin Res Hepatol Gastroenterol. 2024;48:102493. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 28. | Olivares A, Paneroni M, Comini L, Zanelli E, Corica G, Tarro Genta F, Scalvini S. Six-minute walking test in post-cardiac surgery versus chronic heart failure patients attending cardiac rehabilitation: a measure of exercise tolerance and association with patients functioning by ICF. Eur J Phys Rehabil Med. 2024;60:889-892. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 29. | Magnin J, Bernard A, Cottenet J, Lequeu JB, Ortega-Deballon P, Quantin C, Facy O. Impact of hospital volume in liver surgery on postoperative mortality and morbidity: nationwide study. Br J Surg. 2023;110:441-448. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 24] [Reference Citation Analysis (0)] |
| 30. | Ciulli C, Fogliati A, Scacchi A, Scotti MA, Aprigliano M, Braga M, Romano F, Garancini M. Early compliance to enhanced recovery protocol as a predictor of complications after liver surgery. Updates Surg. 2025;77:697-703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |