Published online Mar 27, 2026. doi: 10.4240/wjgs.v18.i3.114720
Revised: November 30, 2025
Accepted: January 14, 2026
Published online: March 27, 2026
Processing time: 147 Days and 4 Hours
Endoscopic resection, encompassing both endoscopic mucosal resection and en
To evaluate the association between PAS and complication risk after endoscopic resection procedures to inform clinical decision-making.
This single-center retrospective cohort study included 80 patients who underwent endoscopic resection for colorectal polyps, stratified into PAS (n = 31) and non-PAS (n = 49) groups. Demographics, polyp characteristics, procedural variables, and post-procedural recovery indices were extracted. The primary endpoint was 30-day clinically significant complications (post-polypectomy bleeding, perfo
The overall complication rate was higher in the PAS group than in the non-PAS group (P < 0.05). Patients with PAS exhibited larger polyps, higher polyp multiplicity, a greater proportion of ESD, longer procedure times, more frequent hemoclip use, prolonged hospital stay, delayed resumption of oral intake and ambulation, and elevated post-procedural pain scores (P < 0.05). Multivariate analysis identified PAS, arterial hypertension, hyperlipidemia, anticoagulant/antiplatelet therapy, polyp diameter, polyp multiplicity, ESD as the resection modality, prolonged procedure time, and hemoclip deployment as independent risk factors for complications. Receiver operating cha
PAS independently predicts endoscopic resection complications, increasing technical difficulty, procedural time, and the risk of adverse events. Identifying pre-procedure PAS status should inform risk stratification and peri-ope
Core Tip: Prior abdominal surgery (PAS) is an independent risk factor for polypectomy complications after endoscopic mucosal resection and endoscopic submucosal dissection. This retrospective cohort study of 80 patients demonstrated that PAS increased technical difficulty, prolonged procedure time, and elevates rates of bleeding, perforation, infection, and post-procedural pain. Larger and multiple polyps, the endoscopic submucosal dissection modality, and hemoclip use further contribute to the risk of complications. Preoperative recognition of PAS status can guide individualized risk stratification, optimize procedural planning, and inform perioperative management strategies, ultimately improving patient safety and clinical outcomes in colorectal polypectomies.
- Citation: Wu QF, Huang ZQ, Yu ZH, Li JT, Huang L. Impact of prior abdominal surgery on post-polypectomy complications following endoscopic mucosal resection and endoscopic submucosal dissection. World J Gastrointest Surg 2026; 18(3): 114720
- URL: https://www.wjgnet.com/1948-9366/full/v18/i3/114720.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i3.114720
Adenomatous polyps, which are benign neoplasms of the gastrointestinal mucosa, are well-established premalignant lesions, whose timely removal reduces the incidence and mortality[1]. Endoscopic mucosal resection (EMR) and en
Prior abdominal surgery, particularly laparotomy, has been linked to longer procedural times, impaired luminal vi
Therefore, we conducted a retrospective cohort study to quantify the impact of prior abdominal surgery on com
This study retrospectively reviewed consecutive patients who underwent endoscopic resection of colorectal polyps at our center between January 2021 and December 2024. This study and the electronic medical records were validated.
Inclusion criteria: (1) Age ≥ 18 years; (2) Histologically verified colorectal adenomatous polyp[7]; (3) Treatment by EMR or ESD; (4) Written informed consent obtained before the procedure; and (5) Complete documentation.
Exclusion criteria: (1) Severe cardiac, hepatic, or renal failure; (2) Inherited coagulopathy or immunodeficiency; (3) Pregnancy or lactation; (4) Previous curative resection for colorectal cancer; and (5) Chemotherapy or radiotherapy within six months before index endoscopy.
Eighty patients fulfilled these criteria and were allocated to either the prior abdominal surgery (PAS) group (n = 31) or the non-PAS group (n = 49) according to surgical history.
Demographics and comorbidities: Age, sex, body-mass index, smoking (≥ 1 cigarette every day for ≥ 1 year), alcohol consumption (≥ 3 episodes every week for ≥ 1 year), arterial hypertension, type 2 diabetes mellitus, hyperlipidemia, and pre-procedural medications including anticoagulants (warfarin, direct oral anticoagulants) and antiplatelet agents (acetylsalicylic acid, clopidogrel).
Polyp characteristics: Location (left colon: Descending, sigmoid, rectum; right colon: Cecum, ascending, transverse), maximum diameter (mm), morphology according to the Paris classification[8] (pedunculated, semi-pedunculated, sessile), and multiplicity (single vs multiple).
Procedural variables: They included resection modality (EMR or ESD), procedure duration (min, from first submucosal injection to final hemostasis), use of hemoclips for defect closure, and immediate intraprocedural bleeding or perforation.
Recovery indices: Time to first oral fluid intake (hours), time to first ambulation (hours), and total length of hospital stay (days).
Bleeding: Hematochezia within 24 hours with a ≥ 2 g/dL drop in hemoglobin or requirement for endoscopic re-intervention or transfusion.
Perforation: Endoscopically visible pneumoperitoneum or free intraperitoneal air on plain abdominal radiography or computed tomography.
Infection: Core temperature > 38.5 °C for > 24 hours plus leukocytosis (> 12 × 109/L) with or without peritoneal signs.
Pain: Moderate-to-severe abdominal pain defined as a Visual Analog Scale score ≥ 4.
All analyses were performed using SPSS version 26.0. Continuous variables are expressed as mean ± SD and compared with the independent-samples t test. Categorical variables are presented as n (%) and compared with the χ2 test or Fisher’s exact test as appropriate. Independent predictors of post-procedural complications were identified using multivariate logistic regression analysis. Statistical significance was defined as a two-tailed P-value < 0.05.
No significant between-group differences were observed in sex, age, body-mass index, smoking status, alcohol con
| Variable | PAS (n = 31) | NPAS (n = 49) | χ2/t | P value |
| Sex | 0.024 | 0.877 | ||
| Male | 17 (54.84) | 26 (53.06) | ||
| Female | 14 (45.16) | 23 (46.94) | ||
| Ages (olds) | 56.73 ± 8.12 | 57.80 ± 8.09 | 0.576 | 0.567 |
| BMI (kg/m2) | 23.45 ± 2.10 | 22.70 ± 1.89 | 1.656 | 0.102 |
| Smoking history | 17 (54.84) | 19 (38.78) | 1.614 | 0.204 |
| Alcohol consumption | 9 (29.03) | 13 (26.53) | 0.060 | 0.807 |
| Comorbidities | ||||
| Arterial hypertension | 18 (58.07) | 16 (32.65) | 5.017 | 0.025 |
| Type 2 diabetes mellitus | 10 (32.26) | 8 (16.33) | 2.764 | 0.096 |
| Hyperlipidemia | 12 (38.71) | 7 (14.29) | 6.255 | 0.012 |
| Anticoagulant/antiplatelet therapy | 15 (48.39) | 9 (18.37) | 8.148 | 0.004 |
There were no significant intergroup differences in polyp location or Paris morphology (P > 0.05). In contrast, the ma
| Variable | PAS (n = 31) | NPAS (n = 49) | χ2/t | P value |
| Polyp location | 0.192 | 0.661 | ||
| Left colon | 18 (58.07) | 26 (53.06) | ||
| Right colon | 13 (41.93) | 23 (46.94) | ||
| Polyp size (mm) | 16.45 ± 4.98 | 13.20 ± 4.67 | 2.956 | 0.004 |
| Number of polyps | 1.87 ± 0.72 | 1.52 ± 0.39 | 2.818 | 0.006 |
| Polyp morphology | 0.298 | 0.585 | ||
| Pedunculated | 12 (38.71) | 22 (44.90) | ||
| Sessile | 19 (61.29) | 27 (55.10) | ||
| Resection modality | 6.140 | 0.013 | ||
| EMR | 15 (48.39) | 37 (75.51) | ||
| ESD | 16 (51.61) | 12 (24.49) | ||
| Procedure duration (minutes) | 45.29 ± 11.13 | 33.70 ± 10.57 | 4.681 | 0.000 |
| Prophylactic clip deployment | 25 (80.65) | 22 (44.90) | 10.010 | 0.002 |
Patients with PAS exhibited significantly longer hospital stays, delayed resumption of oral intake, later ambulation, and higher post-procedural Visual Analog Scale pain scores than those without a surgical history (P < 0.05, Table 3).
| Variable | PAS (n = 31) | NPAS (n = 49) | t | P value |
| Length of stay (days) | 8.19 ± 2.16 | 5.76 ± 1.78 | 5.472 | 0.000 |
| Time to resumption of oral intake (days) | 4.06 ± 1.35 | 2.69 ± 0.93 | 5.376 | 0.000 |
| Time to mobilization (days) | 5.38 ± 1.72 | 3.89 ± 1.35 | 4.319 | 0.000 |
| Post-operative VAS score | 5.24 ± 1.28 | 3.27 ± 1.06 | 7.467 | 0.000 |
The incidence of post-polypectomy complications was significantly higher in the PAS group than in the no prior surgery group (P < 0.05, Table 4). Specifically, the surgical-history group demonstrated markedly higher frequencies of hemorrhage (19.35% vs 10.20%), perforation (12.90% vs 4.08%), and post-procedural pain (35.48% vs 16.33%), collectively indicating that PAS was an independent risk factor for endoscopic complications.
| Groups | n | Hemorrhage | Perforation | Pyrexia | Infection | Abdominal pain | Complications |
| PAS | 31 | 6 (19.35) | 4 (12.90) | 3 (9.68) | 4 (12.90) | 8 (35.48) | 25 (80.65) |
| NPAS | 49 | 5 (10.20) | 2 (4.08) | 4 (8.16) | 3 (6.12) | 8 (16.33) | 22 (44.90) |
| χ2 | 10.010 | ||||||
| P value | 0.002 |
Using the occurrence of any complication as the binary dependent variable (1 = yes, 0 = no), we entered the following covariates into a multivariate logistic model: Arterial hypertension, hyperlipidemia, anticoagulant or antiplatelet therapy, PAS, polyp diameter, polyp multiplicity, ESD vs EMR resection modality, procedure duration, and the proportion of hemoclip deployment. All nine variables emerged as independent predictors of adverse events (P < 0.05; Table 5).
| Variable | β | SE | Wald χ2 | P value | OR | 95%CI |
| Hypertension | 1.049 | 0.475 | 4.888 | 0.027 | 2.856 | 1.126-7.240 |
| Hyperlipidemia | 1.332 | 0.550 | 5.865 | 0.015 | 3.790 | 1.289-11.139 |
| Anticoagulant/antiplatelet therapy | 1.427 | 0.515 | 7.678 | 0.006 | 4.167 | 1.518-11.434 |
| Prior abdominal surgery | 1.437 | 0.517 | 7.733 | 0.005 | 4.208 | 1.528-11.585 |
| Polyp size | 0.145 | 0.053 | 7.316 | 0.006 | 1.156 | 1.041-1.283 |
| Polyp number | 1.030 | 0.425 | 5.867 | 0.015 | 2.801 | 1.217-6.447 |
| Resection modality | 1.191 | 0.489 | 5.918 | 0.015 | 3.289 | 1.260-8.583 |
| Procedure duration | 0.104 | 0.028 | 13.896 | 0.000 | 1.110 | 1.051-1.172 |
| Prophylactic clip deployment | 1.632 | 0.538 | 9.210 | 0.002 | 5.114 | 1.782-14.671 |
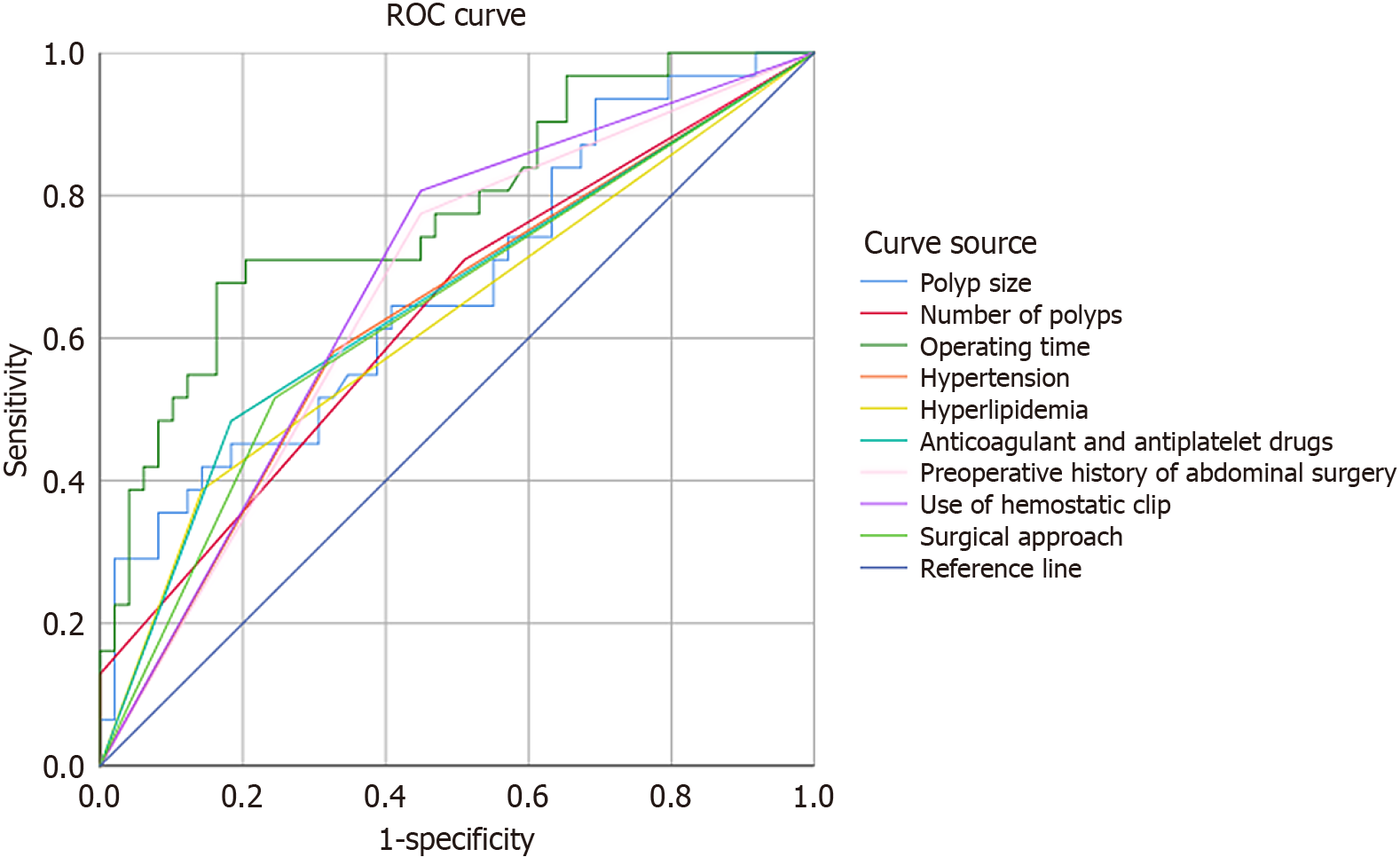
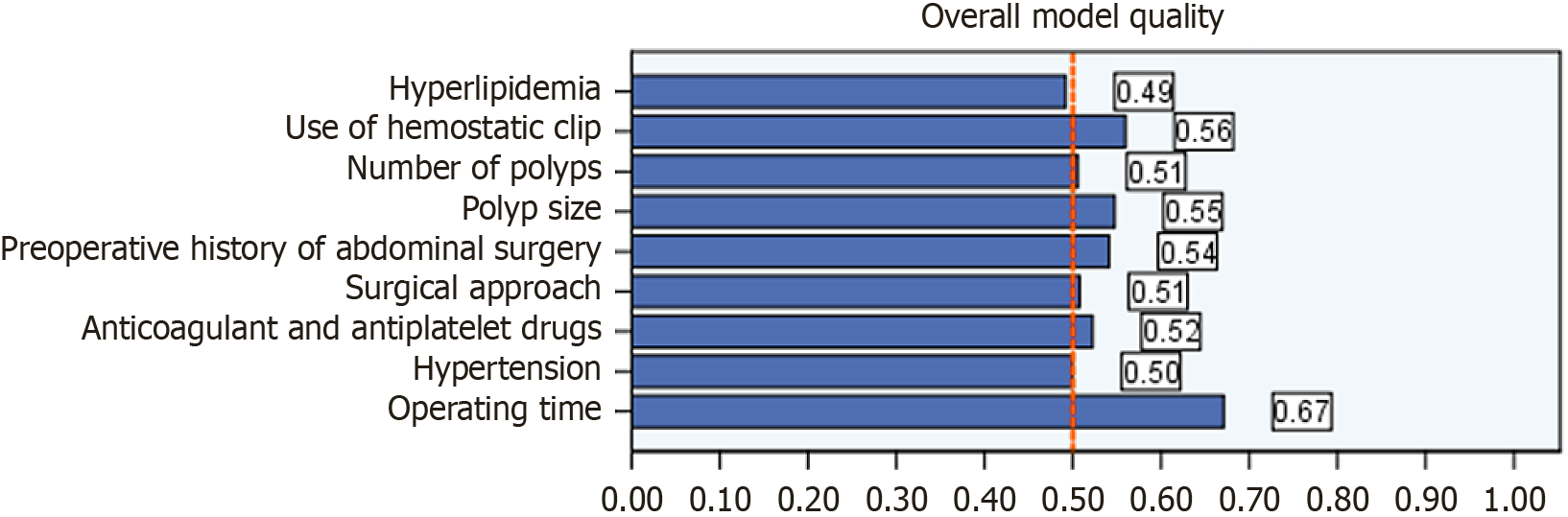
Receiver operating characteristic curves were constructed for each variable retained in the multivariate model. All nine factors demonstrated moderate discriminative capacity for postpolypectomy complications, with area under the curve values ranging from 0.63 to 0.81 (Figures 1 and 2; Table 6).
| Variable | AUC | 95%CI | Sensitivity (%) | Specificity (%) | Maximum Youden index |
| Hypertension | 0.627 | 0.500-0.754 | 73.53 | 90.00 | 0.635 |
| Hyperlipidemia | 0.622 | 0.492-0.752 | 70.59 | 80.00 | 0.506 |
| Anticoagulant/antiplatelet therapy | 0.650 | 0.522-0.778 | 67.65 | 95.36 | 0.630 |
| Prior abdominal surgery | 0.663 | 0.541-0.784 | 77.42 | 55.10 | 0.325 |
| Polyp size | 0.670 | 0.547-0.792 | 41.94 | 85.71 | 0.277 |
| Polyp number | 0.633 | 0.506-0.759 | 70.91 | 48.98 | 0.200 |
| Resection modality | 0.636 | 0.508-0.763 | 51.61 | 75.51 | 0.271 |
| Procedure duration | 0.777 | 0.671-0.883 | 67.74 | 83.67 | 0.514 |
| Prophylactic clip deployment | 0.679 | 0.560-0.798 | 80.65 | 55.10 | 0.358 |
Postoperative complications, ranging from bleeding and perforation to infection and abdominal pain, are major clinical challenges in endoscopic resection. Although polyp characteristics and procedural factors are well-established risk factors, the specific effect of patient history remains poorly defined. Endoscopic resection is the first-line curative modality for colorectal adenomatous polyps; however, procedure-related morbidity remains high. This study specifically addressed the knowledge gap regarding the influence of PAS influences the complication profiles. Using a stratified cohort design, we demonstrated that PAS independently doubled the risk of adverse events after EMR or ESD. This fin
Previous laparotomy precipitates extensive intraperitoneal adhesions that distort anatomical planes, tether and angulate bowel loops, and reduce mural compliance[9,10]. These alterations compress the luminal workspace, degrade endoscopic visualization, and impair the operator’s ability to delineate the lesion margins, thereby introducing pro
Consistent with this pathomechanism, patients with PAS required a mean procedure time of 45.29 ± 11.13 minutes - significantly longer than the 33.70 ± 10.57 minutes recorded in surgically naive controls - and underwent ESD in 51.61% of cases vs 24.49% in the control group. The preferential use of ESD presumably reflects deeper submucosal invasion or fibrosis within the adhesive segments, requiring en bloc excision for curative resection. Protracted operative duration, in turn, increases cumulative mucosal trauma, anesthetic exposure and the probability of adverse events[14-16]. In addition to mechanical impediments, ischemia-reperfusion injury within adhesive fields disrupts microvascular perfusion, whereas post-operative fibrosis and aberrant angiogenesis impair subsequent wound repair[17,18]. These biological sequelae manifest as attenuated epithelial proliferation and disorganized granulation tissue formation, providing a plausible mechanistic basis for the observed increase in delayed bleeding and intraluminal sepsis[19].
The four-fold increase in hemoclip deployment observed in patients with a previous laparotomy reflects heightened operator awareness of bleeding propensity. Although clips provide immediate hemostasis, their retention in the ulcer bed may perpetuate local inflammation and impair granulation tissue formation, paradoxically predisposing patients to late-onset hemorrhage[20,21]. This finding highlights the need for adjunctive pharmacological or mechanical hemostatic strategies that do not impede mucosal healing[22]. Multivariable modelling corroborated established risk modifiers - arterial hypertension, hyperlipidemia, antithrombotic therapy, large polyp diameter, multiplicity, ESD modality, and prolonged procedure time - and quantified their individual discriminative power (area under the curve of 0.63-0.81)[23-25]. Hypertension and hyperlipidemia promote arteriosclerotic fragility, whereas antithrombotic agents impair primary hemostasis. Large or multiple polyps necessitate wider resection fields that transect submucosal arteries, which inherently carry a higher vascular injury risk; and every additional 10 minutes of operative time incrementally increases exposure-related trauma and operator fatigue, compounding error probability.
The limitations of our study include its single-center retrospective design, which may have introduced a selection bias. We did not stratify the analyses by type, number, or time elapsed since previous abdominal operations, nor did we quantify the adhesion burden using preprocedural imaging or intraoperative scoring systems. Future prospective studies integrating radiographic adhesion scoring are needed to validate these findings and refine the risk stratification.
PAS is an independent modifiable predictor of morbidity after colorectal EMR/ESD. Surgeons should flag these patients for intensive pre-operative work-up, consider prophylactic hemostatic adjuncts, schedule procedures under optimal anticoagulation windows, and institute enhanced post-procedural surveillance to mitigate complications and safeguard patient safety.
| 1. | Wernly S, Datz C, Wernly B. RE: Long-Term Colorectal Cancer Incidence and Mortality After Colonoscopy Screening According to Individuals' Risk Profiles. J Natl Cancer Inst. 2022;114:779-780. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 2. | Arruda do Espirito Santo P, Meine GC, Baraldo S, Barbosa EC. Cold endoscopic mucosal resection versus cold snare polypectomy for colorectal lesions: a systematic review and meta-analysis of randomized controlled trials. Endoscopy. 2024;56:503-511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 3. | Jaruvongvanich V, Sempokuya T, Laoveeravat P, Ungprasert P. Risk factors associated with longer cecal intubation time: a systematic review and meta-analysis. Int J Colorectal Dis. 2018;33:359-365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 30] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 4. | He XJ, Chen ZP, Zeng XP, Jiang CS, Liu G, Li DL, Li DZ, Wang W. Gallbladder-preserving polypectomy for gallbladder polyp by embryonic-natural orifice transumbilical endoscopic surgery with a gastric endoscopy. BMC Gastroenterol. 2022;22:216. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 5. | Tsuruta A, Itoh T, Hirai T, Nakamura M. Multi-layered intra-abdominal adhesion prophylaxis following laparoscopic colorectal surgery. Surg Endosc. 2015;29:1400-1405. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 17] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 6. | Hayashi T, Kudo SE, Miyachi H, Sakurai T, Ishigaki T, Yagawa Y, Toyoshima N, Mori Y, Misawa M, Kudo T, Wakamura K, Katagiri A, Baba T, Ishida F. Management and risk factor of stenosis after endoscopic submucosal dissection for colorectal neoplasms. Gastrointest Endosc. 2017;86:358-369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 45] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 7. | Lieberman DA, Rex DK, Winawer SJ, Giardiello FM, Johnson DA, Levin TR. Guidelines for colonoscopy surveillance after screening and polypectomy: a consensus update by the US Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2012;143:844-857. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1565] [Cited by in RCA: 1457] [Article Influence: 104.1] [Reference Citation Analysis (2)] |
| 8. | The Paris endoscopic classification of superficial neoplastic lesions: esophagus, stomach, and colon: November 30 to December 1, 2002. Gastrointest Endosc. 2003;58:S3-S43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1503] [Cited by in RCA: 1357] [Article Influence: 59.0] [Reference Citation Analysis (3)] |
| 9. | Liakakos T, Thomakos N, Fine PM, Dervenis C, Young RL. Peritoneal adhesions: etiology, pathophysiology, and clinical significance. Recent advances in prevention and management. Dig Surg. 2001;18:260-273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 526] [Cited by in RCA: 471] [Article Influence: 18.8] [Reference Citation Analysis (1)] |
| 10. | Koo DC, Scalise PN, Ostertag-Hill CA, Naus AE, Durgin JM, Chiu MZ, Mejia Bautista M, Moskowitzova K, Staffa SJ, Gonzalez GR, Al-Ibraheemi A, Lee EJ, Demehri FR, Kim HB. Polyvinyl Alcohol Sponges Reduce Intraperitoneal Adhesions After Abdominal Surgery. J Surg Res. 2025;308:183-192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 11. | Schnüriger B, Barmparas G, Branco BC, Lustenberger T, Inaba K, Demetriades D. Prevention of postoperative peritoneal adhesions: a review of the literature. Am J Surg. 2011;201:111-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 171] [Article Influence: 11.4] [Reference Citation Analysis (2)] |
| 12. | Li C, Gao L, Liu H, Wang X, Luo H, Zhang C, Yu P, Tang B. Short-term outcomes of pelvic floor peritoneum closure in endoscopic low anterior resection of rectal cancer: A propensity score matching analysis. J Surg Oncol. 2021;123:271-277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (1)] |
| 13. | Akagi T, Shiraishi N, Hiroishi K, Etoh T, Yasuda K, Kitano S. Case series of intra-abdominal adhesions induced by artificial ulceration after endoscopic submucosal dissection before additional laparoscopic gastrectomy. Gastrointest Endosc. 2010;72:438-443. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 17] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 14. | Tanabe H, Higurashi T, Takatsu T, Misawa N, Yoshihara T, Goto S, Arimoto J, Ashikari K, Taniguchi L, Chiba H, Nakajima A. Effects of colorectal endoscopic submucosal dissection on postoperative abdominal symptoms: a prospective observational study. Surg Endosc. 2022;36:314-320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 15. | Lee SK, Kim TI, Shin SJ, Kim BC, Kim WH. Impact of prior abdominal or pelvic surgery on colonoscopy outcomes. J Clin Gastroenterol. 2006;40:711-716. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 40] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 16. | Fuccio L, Hassan C, Ponchon T, Mandolesi D, Farioli A, Cucchetti A, Frazzoni L, Bhandari P, Bellisario C, Bazzoli F, Repici A. Clinical outcomes after endoscopic submucosal dissection for colorectal neoplasia: a systematic review and meta-analysis. Gastrointest Endosc. 2017;86:74-86.e17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 266] [Cited by in RCA: 233] [Article Influence: 25.9] [Reference Citation Analysis (2)] |
| 17. | Takeuchi Y, Iishi H, Tanaka S, Saito Y, Ikematsu H, Kudo SE, Sano Y, Hisabe T, Yahagi N, Saitoh Y, Igarashi M, Kobayashi K, Yamano H, Shimizu S, Tsuruta O, Inoue Y, Watanabe T, Nakamura H, Fujii T, Uedo N, Shimokawa T, Ishikawa H, Sugihara K. Factors associated with technical difficulties and adverse events of colorectal endoscopic submucosal dissection: retrospective exploratory factor analysis of a multicenter prospective cohort. Int J Colorectal Dis. 2014;29:1275-1284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 94] [Article Influence: 7.8] [Reference Citation Analysis (1)] |
| 18. | ten Broek RP, Issa Y, van Santbrink EJ, Bouvy ND, Kruitwagen RF, Jeekel J, Bakkum EA, Rovers MM, van Goor H. Burden of adhesions in abdominal and pelvic surgery: systematic review and met-analysis. BMJ. 2013;347:f5588. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 506] [Cited by in RCA: 427] [Article Influence: 32.8] [Reference Citation Analysis (3)] |
| 19. | Jiang GL, Im WB, Donde Y, Wheeler LA. EP4 agonist alleviates indomethacin-induced gastric lesions and promotes chronic gastric ulcer healing. World J Gastroenterol. 2009;15:5149-5156. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 15] [Cited by in RCA: 19] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 20. | Yu Z, Albéniz E, Hu J, Li P, Li Q, Hu Y, Chen J, Wang J. Prevention of delayed post-polypectomy bleeding by prophylactic clipping after endoscopic colorectal polypectomy: a meta-analysis. Int J Colorectal Dis. 2022;37:2229-2236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 21. | Kim SH, Lee JK, Lim YJ, Kim JH. The risk factors for prolonged hemostatic clip retention after endoscopic submucosal dissection for gastric neoplasm. Surg Endosc. 2022;36:1123-1130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (2)] |
| 22. | He J, Zhang Z, Yang Y, Ren F, Li J, Zhu S, Ma F, Wu R, Lv Y, He G, Guo B, Chu D. Injectable Self-Healing Adhesive pH-Responsive Hydrogels Accelerate Gastric Hemostasis and Wound Healing. Nanomicro Lett. 2021;13:80. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 68] [Cited by in RCA: 148] [Article Influence: 29.6] [Reference Citation Analysis (1)] |
| 23. | Ninomiya Y, Oka S, Tanaka S, Nishiyama S, Tamaru Y, Asayama N, Shigita K, Hayashi N, Chayama K. Risk of bleeding after endoscopic submucosal dissection for colorectal tumors in patients with continued use of low-dose aspirin. J Gastroenterol. 2015;50:1041-1046. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 29] [Article Influence: 2.6] [Reference Citation Analysis (1)] |
| 24. | Yoshio T, Nishida T, Hayashi Y, Iijima H, Tsujii M, Fujisaki J, Takehara T. Clinical problems with antithrombotic therapy for endoscopic submucosal dissection for gastric neoplasms. World J Gastrointest Endosc. 2016;8:756-762. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 6] [Cited by in RCA: 9] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 25. | Azzolini F, Camellini L, Sassatelli R, Sereni G, Biolchini F, Decembrino F, De Marco L, Iori V, Tioli C, Cavina M, Bedogni G. Endoscopic submucosal dissection of scar-embedded rectal polyps: a prospective study (Esd in scar-embedded rectal polyps). Clin Res Hepatol Gastroenterol. 2011;35:572-579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (1)] |