Published online Mar 27, 2026. doi: 10.4240/wjgs.v18.i3.114661
Revised: November 19, 2025
Accepted: January 16, 2026
Published online: March 27, 2026
Processing time: 183 Days and 0.1 Hours
Although enhanced recovery after surgery (ERAS) protocols have significantly improved perioperative outcomes, opportunities for further optimization remain. The fastest recovery after surgery (FRAS) protocol was developed to address these gaps by integrating intensified multimodal interventions aimed at achieving ul
To evaluate the efficacy and safety of the FRAS protocol compared with those of conventional ERAS in patients who underwent elective gastrointestinal tumor surgery, with a focus on recovery speed, postoperative complications, patient-reported outcomes, and economic efficiency.
A retrospective cohort study involving 307 patients who underwent surgery between August 2021 and May 2025 was conducted. The FRAS group (n = 113) received an optimized protocol featuring ultra-early oral feeding (initiated at 1-2 hours after surgery), strict fluid restriction (< 500 mL/day), and early drain removal. The ERAS group (n = 194) received standard ERAS care. The primary outcomes included postoperative hospital stay and 30-day complication rates. Secondary outcomes included functional recovery metrics, inflammatory markers, quality of life, and patient satisfaction.
The FRAS group demonstrated a significantly shorter median postoperative hospital stay (22.4 hours vs 100.4 hours, P < 0.001) without exhibiting increased complication rates (4.42% vs 2.58%, P = 0.507). FRAS patients experienced earlier ambulation (4.23 hours vs 18.05 hours, P < 0.001), earlier oral intake (2.61 hours vs 11.88 hours, P < 0.001), and lower pain scores (4.85 vs 5.31, P = 0.007). The quality of life and satisfaction scores were sig
Compared with ERAS, the FRAS protocol significantly accelerates postoperative recovery while maintaining safety and improving early patient-reported outcomes. It represents a safe, efficient, and patient-centered advancement in perioperative care. Future studies should validate these findings in broader populations and explore individualized FRAS strategies for high-risk patients.
Core Tip: This study introduces fastest recovery after surgery as an optimized protocol building upon enhanced recovery after surgery. In patients who underwent gastrointestinal tumor surgery, fastest recovery after surgery achieved a remarkably short median postoperative stay of approximately 22.4 hours without increasing complication rates; moreover, it also significantly improved early quality of life and patient satisfaction. This effect represents a significant advancement in perioperative care, thus demonstrating that further acceleration of recovery beyond traditional enhanced recovery after surgery is both feasible and safe in carefully selected patients.
- Citation: Zhou DH, Li R, Xu DT, Zhang S, Zhang N, Ni YP, Liu HR, Chen Z, Huang ZX, Chang C, Shi ZH, Xie YX, Zhang SH, Shi RH, Ge TT, Zhou HM, Zhou MJ, Liu QZ, Tu XH. Evaluation of the recovery speed and safety of fastest recovery after surgery vs enhanced recovery after surgery in patients undergoing gastrointestinal tumor surgery. World J Gastrointest Surg 2026; 18(3): 114661
- URL: https://www.wjgnet.com/1948-9366/full/v18/i3/114661.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i3.114661
Gastrointestinal tumors are among the malignancies demonstrating the highest global incidence and mortality rates, and surgical intervention remains the primary curative approach[1,2]. However, there are numerous challenges associated with perioperative management. Traditional perioperative strategies often elicit surgical stress responses, intestinal functional inhibition, and postoperative pain, thus leading to prolonged recovery processes, increased complication risks, and significantly impaired postoperative quality of life[3,4].
To address these challenges, the enhanced recovery after surgery (ERAS) concept has emerged. ERAS employs a series of evidence-based perioperative optimization measures aimed at mitigating surgical stress, preserving physiological functions, and accelerating postoperative recovery[5,6]. In surgeries such as colorectal and gastric procedures, ERAS protocols have demonstrated substantial benefits, including reduced hospital stays, lower complication rates, and inte
Nonetheless, in clinical practice, traditional ERAS protocols still offer opportunities for further optimization in the pursuit of “accelerated” recovery. Variations in implementation across medical centers (such as differences in post
Despite the attractiveness of the FRAS concept, its efficacy and safety in gastrointestinal tumor surgery (particularly compared with conventional ERAS protocols), as well as whether it can significantly further accelerate recovery without increasing complication risks, currently lacks high-level evidence-based medical support. Most of the relevant studies are confined to small samples or single-center experiences, and there has been an insufficient focus on patient-reported out
Therefore, this study aimed to conduct a retrospective cohort analysis to systematically compare the effects of FRAS and ERAS protocols in patients who underwent gastrointestinal tumor surgery. This study emphasizes evaluating dif
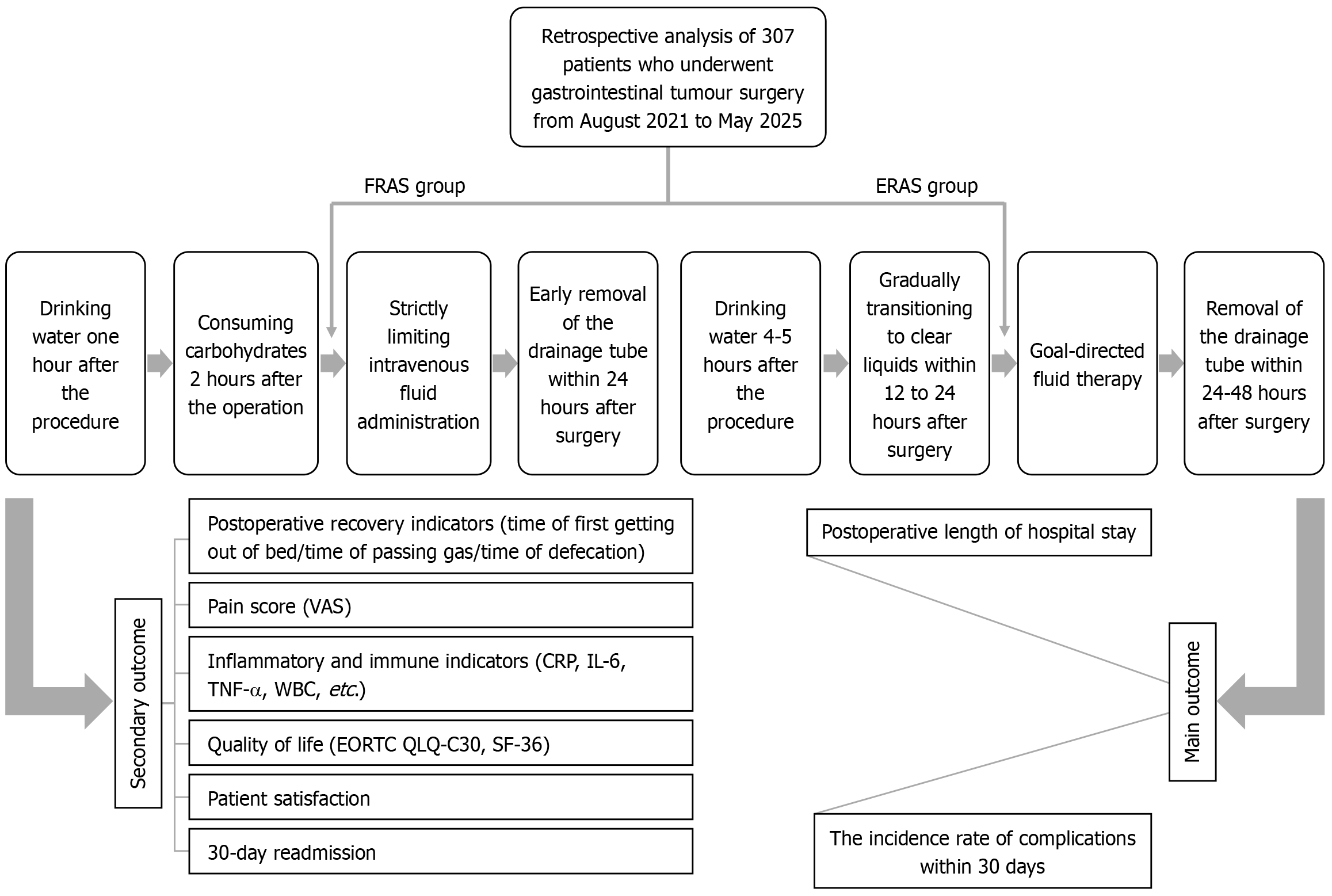
This retrospective cohort study aimed to compare the perioperative recovery speed and safety between the FRAS protocol and the ERAS protocol in patients who underwent gastrointestinal tumor surgery (Figure 1). The study protocol was approved by the Ethics Committee of Shanghai Fourth People’s Hospital (Approval No. SYLL2023013). Given that all of the clinical data were obtained from the electronic medical records system without involving human tissue or blood samples, the requirement for informed consent was waived.
Inclusion criteria: (1) Demographic characteristics; (2) Comorbidity profiles; (3) Tumor characteristics; (4) Preoperative assessment data; (5) Surgical details; (6) Postoperative recovery parameters; (7) Serial measurements of inflammatory markers, including C-reactive protein (CRP), interleukin (IL)-6, and tumor necrosis factor (TNF)-α, along with immune parameters, including white blood cell (WBC) count, neutrophil (NEUT) percentage, and lymphocyte (LYM) count; (8) Complication data; and (9) Follow-up data.
Exclusion criteria: (1) Metastatic disease (stage IV) or locally advanced tumors with extensive nodal involvement [clinical nodal stage 2-3 (cN2-3)]; (2) Severe organ dysfunction (cardiac dysfunction involving left ventricular ejection fraction < 40%, pulmonary dysfunction involving forced expiratory volume in 1 second < 50% predicted, hepatic dysfunction involving Child-Pugh class B or class C, or renal dysfunction involving estimated glomerular filtration rate < 30 mL/minute/1.73 m2); (3) American Society of Anesthesiologists classification ≥ grade III; (4) Emergency or palliative surgery; (5) History of major abdominal surgery or severe adhesions; (6) Active infection or inflammatory conditions; (7) Cog
The study population included 307 patients who underwent gastrointestinal tumor surgery at the Department of Gastrointestinal Surgery, Shanghai Fourth People’s Hospital, between August 2021 and May 2025. The inclusion criteria included patients aged 18-80 years with radiologically and pathologically confirmed primary gastrointestinal tumors who were scheduled for elective curative-intent surgery, who lacked evidence of gastrointestinal obstruction, who had well-controlled comorbidities (such as hypertension with blood pressure < 140/90 mmHg on medication, diabetes with glycated hemoglobin < 7.5%, and coronary heart disease with New York Heart Association class I-II), who had an adequate nutritional status [Nutritional Risk Screening (NRS) score < 3 and an album in concentration > 30 g/L], and who were willing to participate in the enhanced recovery program.
Both groups received standardized perioperative management, which was implemented by a unified multidisciplinary team and included preoperative nutritional risk assessment, a 6-minute walk test, psychological intervention, and respiratory training. The ERAS group followed traditional enhanced recovery protocols with 6 hours of solid food fasting and 2 hours of clear fluid fasting before surgery, along with propofol using short-acting opioids for anesthesia induction, intraoperative short-acting muscle relaxants, goal-directed fluid therapy, oral intake initiation at 4-5 hours after surgery, gradual transition to clear fluids within 12-24 hours, drain and urinary catheter removal within 24-48 hours, multimodal analgesia with non-steroidal anti-inflammatory drugs and local wound infiltration, and mobilization being encouraged on postoperative day 1 with discharge upon meeting standardized criteria. The FRAS group maintained identical preoperative fasting and anesthesia protocols but implemented accelerated postoperative care, including oral water intake at 1 hour and carbohydrate-rich drinks (50-100 mL/serving) at 2 hours after surgery, in addition to a clear fluid diet on postoperative day 1 with intravenous fluid restriction (< 500 mL), early drain removal (within 24 hours if output was < 30 mL), and discharge within 24-48 hours upon meeting recovery criteria, whereas identical analgesia and mobi
The primary outcomes included length of postoperative hospital stay (days from surgery to discharge) and incidence of complications within 30 days (assessed by using the Clavien-Dindo and Accordion classification systems). Secondary outcomes included postoperative recovery indicators (time to first mobilization, flatus, defecation, and oral intake); pain intensity assessed via Visual Analogue Scale (VAS) at 24 hours, 48 hours, and 72 hours after surgery; inflammatory mar
Statistical analyses were performed by using SPSS version 22.0 and R version 4.2.0. Continuous variables are presented as the means ± SD for normally distributed data or medians (interquartile ranges) for nonnormally distributed data, with normality being assessed by using the Shapiro-Wilk test. Categorical variables are expressed as n (%). Between-group comparisons employed the Student’s t test for normally distributed continuous variables, the Mann-Whitney U test for nonnormally distributed continuous variables, and the χ2 test or Fisher’s exact test for categorical variables. Primary outcomes were analyzed by using the Mann-Whitney U test for hospital stay and the χ2 test with Yates’ correction for complication rates, with noninferiority margins being set at 1 day and 5%, respectively. Secondary analyses included two-way repeated-measures ANOVA with Bonferroni correction for serial inflammatory markers, mixed-effects models for quality-of-life scores, and appropriate parametric or nonparametric tests for other secondary outcomes. All of the ana
The baseline characteristics of the patients in the FRAS and ERAS groups were comparable (Table 1). There were no significant differences detected in age, sex, body mass index, comorbidities (hypertension, diabetes, chronic obstructive pulmonary disease, and coronary heart disease), smoking history, alcohol consumption history, tumor location, NRS score, 6-minute walk test, or albumin levels (all P > 0.05). However, a trend towards a difference in the surgical method was observed (P = 0.085), with all of the patients in the FRAS group undergoing laparoscopic surgery, whereas in the ERAS group, 189 patients (97.42%) underwent laparoscopic surgery, and 5 patients (2.58%) underwent open surgery.
| Variable | Total | FRAS group (n = 113) | ERAS group (n = 194) | P value |
| Age (years) | 65.55 ± 10.57 | 65.60 ± 22.33 | 65.52 ± 22.32 | 0.945 |
| Sex (male) | 172 | 67 (38.95) | 105 (61.05) | 0.379 |
| BMI (kg/m2) | 22.32 ± 2.49 | 22.33 ± 2.48 | 22.32 ± 2.51 | 0.971 |
| Comorbidities | ||||
| Hypertension | 106 | 41 (38.68) | 65 (61.32) | 0.621 |
| Diabetes | 68 | 23 (33.82) | 45 (66.18) | 0.563 |
| COPD | 42 | 16 (38.10) | 26 (61.90) | 0.852 |
| CHD | 47 | 15 (31.91) | 32 (68.09) | 0.450 |
| Smoking history | 45 | 17 (37.78) | 28 (62.22) | 0.884 |
| Alcohol consumption history | 106 | 37 (34.91) | 69 (65.09) | 0.616 |
| Tumor location | 0.973 | |||
| Stomach | 90 | 34 (11.08) | 56 (18.24) | |
| Colorectal | 203 | 74 (24.10) | 129 (42.02) | |
| Others | 14 | 5 (1.63) | 9 (2.93) | |
| Surgical method | 0.085 | |||
| Laparoscopic surgery | 293 | 113 (36.81) | 189 (61.56) | |
| Open surgery | 14 | 0 | 5 (1.63) | |
| NRS score | 1.62 ± 1.44 | 1.48 ± 1.41 | 1.70 ± 1.45 | 0.201 |
| 6MWT | 422.02 ± 41.56 | 416.92 ± 41.95 | 424.98 ± 41.15 | 0.101 |
| Albumin | 40.09 ± 2.83 | 40.04 ± 2.87 | 40.12 ± 2.81 | 0.803 |
Comparative analysis of postoperative recovery outcomes revealed significant differences between groups (Table 2). The FRAS group demonstrated a significantly shorter time to first mobilization (4.23 ± 1.16 hours vs 18.05 ± 6.81 hours, P < 0.001), a shorter time to first oral intake (2.61 ± 0.88 hours vs 11.88 ± 4.28 hours, P < 0.001), a lower pain management VAS score (4.85 ± 1.36 vs 5.31 ± 1.46, P = 0.007), and a shorter length of hospital stay (22.40 ± 3.60 hours vs 100.43 ± 15.91 hours, P < 0.001). The FRAS group also demonstrated less intraoperative blood loss (55.33 ± 12.12 mL vs 59.20 ± 12.77 mL, P = 0.01). No significant differences were observed in operation time, time to first flatus, or time to first defecation (all P > 0.05).
| Variable | Total | FRAS group (n = 113) | ERAS group (n = 194) | P value |
| Operation time (minutes) | 143.34 ± 38.31 | 139.96 ± 37.43 | 145.30 ± 38.77 | 0.239 |
| Intraoperative blood loss (mL) | 57.77 ± 12.65 | 55.33 ± 12.12 | 59.20 ± 12.77 | 0.01 |
| Time to first flatus (hours) | 27.42 ± 8.43 | 27.41 ± 7.79 | 27.43 ± 8.80 | 0.979 |
| Time to first defecation (hours) | 40.63 ± 9.33 | 39.34 ± 8.07 | 41.39 ± 9.93 | 0.063 |
| Time to first mobilization | 12.99 ± 8.62 | 4.23 ± 1.16 | 18.05 ± 6.81 | < 0.001 |
| Time to first oral intake | 8.47 ± 5.64 | 2.61 ± 0.88 | 11.88 ± 4.28 | < 0.001 |
| Pain management score (VAS) | 5.14 ± 1.44 | 4.85 ± 1.36 | 5.31 ± 1.46 | 0.007 |
| Length of hospital stay (hours) | 5.14 ± 1.44 | 22.40 ± 3.60 | 100.43 ± 15.91 | < 0.001 |
The incidence and severity of postoperative complications within 30 days were similar between the groups (Table 3). The overall complication rate was 4.42% in the FRAS group and 2.58% in the ERAS group (P = 0.507). Specific complications included intestinal obstruction (3 cases in FRAS vs 4 cases in ERAS, P = 0.711), pulmonary infection (2 cases in FRAS vs 4 cases in ERAS, P = 0.859), and deep vein thrombosis (1 case in FRAS vs 2 cases in ERAS, P = 0.900). One complication with a Clavien-Dindo grade ≥ III occurred in the ERAS group. The reoperation rate was not significantly different (0% for FRAS vs 1.03% for ERAS, P = 0.279).
| Type of complications | Total | FRAS group (n = 113) | ERAS group (n = 194) | P value |
| Postoperative complications | 10 (3.26) | 5 (4.42) | 5 (2.58) | 0.507 |
| Anastomotic leakage | 1 | 0 | 1 (100) | 1 |
| Intra-abdominal infection | 1 | 0 | 1 (100) | 1 |
| Intestinal obstruction | 7 | 3 (42.9) | 4 (57.1) | 0.711 |
| Pulmonary infection | 6 | 2 (33.3) | 4 (66.7) | 0.859 |
| DVT | 3 | 1 (33.3) | 2 (66.7) | 0.900 |
| C-D Classification≥ grade III | 1 | 0 | 1 (100) | 1 |
| Reoperation rate | 2 | 0 | 2 (100) | 0.279 |
Repeated-measures ANOVA revealed no significant between-group differences (group P > 0.05) or time-by-group interactions (time × group P > 0.05) for any inflammatory or immune parameters (Table 4). Both groups exhibited peak CRP levels, IL-6 levels, TNF-α levels, WBC, and NEUT% at 24 hours after surgery, followed by a gradual decrease by 7 days after surgery. LYM remained stable across the time points. Although not statistically significant, the FRAS group exhibited lower CRP and IL-6 levels at postoperative day 7.
| Indicators (units) | Group | Pre-operative | Post-operative 24 hours | Post-operative 72 hours | Post-operative 7 days | Group P value | Time × P value |
| CRP (mg/L) | FRAS | 5.56 ± 5.15 | 58.69 ± 48.26 | 50.67 ± 40.57 | 42.77 ± 38.05 | 0.275 | 0.216 |
| ERAS | 5.67 ± 4.72 | 56.84 ± 48.01 | 41.07 ± 35.97 | 45.16 ± 41.52 | |||
| IL-6 (pg/mL) | FRAS | 5.55 ± 5.13 | 58.68 ± 48.24 | 50.66 ± 40.55 | 42.68 ± 37.93 | 0.274 | 0.252 |
| ERAS | 5.65 ± 4.70 | 56.83 ± 48.00 | 41.07 ± 35.96 | 45.06 ± 41.36 | |||
| TNF-α (pg/mL) | FRAS | 17.49 ± 16.66 | 79.30 ± 67.37 | 49.66 ± 42.80 | 34.75 ± 31.80 | 0.285 | 0.961 |
| ERAS | 17.90 ± 15.44 | 76.86 ± 67.23 | 39.77 ± 37.48 | 36.13 ± 33.58 | |||
| WBC (× 109/L) | FRAS | 7.52 ± 1.15 | 11.49 ± 1.10 | 9.89 ± 1.03 | 8.46 ± 0.86 | 0.427 | 0.717 |
| ERAS | 7.47 ± 1.25 | 11.62 ± 1.16 | 9.91 ± 1.03 | 8.56 ± 0.92 | |||
| NEUT% | FRAS | 64.00 ± 7.34 | 86.27 ± 5.93 | 78.82 ± 6.38 | 72.49 ± 8.01 | 0.617 | 0.923 |
| ERAS | 63.59 ± 7.65 | 86.30 ± 6.10 | 78.28 ± 6.35 | 72.61 ± 7.18 | |||
| LYM (× 109/L) | FRAS | 1.57 ± 0.50 | 1.57 ± 0.50 | 1.60 ± 0.49 | 1.57 ± 0.51 | 0.459 | 0.464 |
| ERAS | 1.58 ± 0.45 | 1.58 ± 0.49 | 1.59 ± 0.49 | 1.47 ± 0.51 |
The quality of life and satisfaction scores were significantly better in the FRAS group at the early time points (Table 5). The European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30 score was higher in the FRAS group at 1 week after surgery (66.01 ± 8.37 vs 58.61 ± 9.66, P < 0.001) and 1 month after surgery (78.17 ± 6.60 vs 71.94 ± 8.57, P < 0.001) but not at 3 months (P = 0.248). The Short Form 36 Health Survey Questionnaire physical functioning score did not significantly differ at 1 month (P = 0.555). VAS satisfaction scores were higher in the FRAS group at discharge (95.34 ± 3.50 vs 94.14 ± 5.35, P = 0.033) and 1 month after surgery (92.09 ± 5.23 vs 90.54 ± 6.30, P = 0.028). Satisfaction with dietary recovery was also higher in the FRAS group at 1 week after surgery (4.31 ± 0.57 vs 3.58 ± 0.83, P < 0.001). The readmission rate within 30 days was not significantly different (P = 0.122).
| Evaluation metrics | Time point | Total | ERAS group (n = 194) | FRAS group (n = 113) | P value |
| EORTC QLQ-C30 | 1-week post-operative | 61.33 ± 9.86 | 58.61 ± 9.66 | 66.01 ± 8.37 | < 0.001 |
| 1-month post-operative | 74.24 ± 8.46 | 71.94 ± 8.57 | 78.17 ± 6.60 | < 0.001 | |
| 3 months post-operative | 83.59 ± 7.32 | 83.22 ± 7.44 | 84.22 ± 7.10 | 0.248 | |
| SF-36 (PF score) | 1-month post-operative | 76.04 ± 9.63 | 75.79 ± 9.99 | 76.46 ± 9.01 | 0.555 |
| VAS satisfaction | At discharge | 94.58 ± 4.78 | 94.14 ± 5.35 | 95.34 ± 3.50 | 0.033 |
| 1-month post-operative | 91.11 ± 5.96 | 90.54 ± 6.30 | 92.09 ± 5.23 | 0.028 | |
| Satisfaction with dietary recovery | 1-week post-operative | 3.85 ± 0.82 | 3.58 ± 0.83 | 4.31 ± 0.57 | < 0.001 |
| Readmission rate | < 30 days | 10 | 4 (40) | 6 (60) | 0.122 |
This study represents the first systematic comparison between the FRAS protocol and the conventional ERAS protocol in patients who underwent gastrointestinal tumor surgery. Our primary finding is that, in rigorously selected patients, the FRAS protocol significantly accelerated postoperative recovery compared to ERAS; specifically, it reduced the median postoperative hospital stay to approximately 22.4 hours without increasing complication rates and simultaneously improved patient-reported early quality of life and satisfaction. Notably, our study provides more comprehensive and large-scale evidence within the context of gastrointestinal tumor surgery. For example, although Serra-Aracil et al[8] investigated the noninferiority of short-term urinary catheter management in colorectal procedures (reflecting a shared emphasis on accelerated recovery), our approach expands the optimization framework to encompass a broader spectrum of multimodal interventions, including early oral feeding, restrictive fluid management, and early removal of drainage tubes. Furthermore, our findings support the conceptual transition from ERAS to FRAS proposed by Wu et al[10], thereby offering robust clinical validation through detailed and systematic data. These results indicate important optimization and advancement in the philosophy of perioperative management, thus building upon the established achievements of ERAS.
The significantly shorter postoperative hospital stays in the FRAS group compared to the ERAS group represents a landmark finding. Although efforts to optimize the ERAS protocol have been performed in recent years, consistent re
Safety is the paramount prerequisite for evaluating any accelerated recovery protocol. This study observed that, despite the substantially accelerated recovery process in the FRAS group, there were no statistically significant differences observed in the overall complication rate or in the incidence of specific complication types compared with those in the ERAS group. These findings are consistent with conclusions obtained from recent studies on the safety of ultrashort-stay surgery over the past five years, thus indicating that, in carefully selected patients, accelerated recovery does not com
However, unlike studies focusing on isolated interventions (such as Imamura et al[13], who emphasized postoperative active diuresis, or MacVicar et al[9], who conducted a systematic review addressing postoperative oral fluid intake), this study provides innovative findings by evaluating the overall impact of a comprehensive and synergistic combination of interventions. This scenario suggests that the success of FRAS may not depend on a single breakthrough component; rather, it arises from the coordinated optimization of multiple evidence-based interventions in terms of timing and in
The dynamic monitoring of inflammatory markers revealed no significant differences in the trajectories of CRP, IL-6, or other indicators between the two groups. These findings suggest that the benefits of FRAS are primarily attributable to active management and early restoration of physiological function, rather than the mere attenuation of the magnitude of the surgical stress response[16]. Notably, although not statistically significant, the FRAS group tended to exhibit slightly lower levels of inflammatory markers on postoperative day 7, which may reflect faster functional recovery and an earlier transition out of the surgical stress state[17,18]. Future studies incorporating more sensitive inflammatory biomarkers or functional assessments may help to further elucidate the underlying involved mechanisms.
A notable strength of this study involves the comprehensive inclusion of patient-reported outcomes. The results revealed that the FRAS group achieved significantly better quality of life scores during the early postoperative period (at 1 week and 1 month) and higher satisfaction scores at multiple time points. This scenario strongly indicates that the benefits of FRAS extend beyond the objective metric of “length of stay” and translate into meaningful improvements in patients’ lived experiences[19,20]. These findings are consistent with recent research emphasizing the critical role of inte
In conclusion, although FRAS demonstrates significant advantages in gastrointestinal tumor surgery, future efforts should focus on high-risk patients. Given the notable recovery disparities observed in the retrospective data among specific subgroups (such as advanced age, high NRS, hypoalbuminemia, and multiple comorbidities), the development of risk prediction models to prospectively identify these populations for targeted intervention is essential. Transitioning FRAS from a standardized protocol to a dynamic, individualized strategy aligns with the current trend in gastrointestinal disease management towards personalized care. By integrating both physiological and psychological risk assessments, more precise perioperative management can be achieved, thereby ultimately optimizing patient-specific recovery trajectories.
The FRAS protocol serves as a safe, efficient, and patient-centered perioperative strategy that enhances early postope
The authors would like to extend their sincere gratitude to all of the individuals who contributed to the completion of this study. We gratefully acknowledge the dedicated contributions of the multidisciplinary perioperative team in the De
| 1. | Vilz TO, Post S, Langer T, Follmann M, Nothacker M, Willis MA; the POMGAT Guideline Group. Clinical Practice Guideline: Recommendations for the Perioperative Management of Pancreatic and Colorectal Cancer Patients. Dtsch Arztebl Int. 2024;121:681-687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 2. | Glover MJ, Bien J, Chen CT. Toward Precision Perioperative Therapy in GI Malignancies. JCO Precis Oncol. 2023;7:e2200381. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 3. | Peng LH, Wang WJ, Chen J, Jin JY, Min S, Qin PP. Implementation of the pre-operative rehabilitation recovery protocol and its effect on the quality of recovery after colorectal surgeries. Chin Med J (Engl). 2021;134:2865-2873. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 35] [Article Influence: 7.0] [Reference Citation Analysis (2)] |
| 4. | Lee HJ, Kim J, Yoon SH, Kong SH, Kim WH, Park DJ, Lee HJ, Yang HK. Effectiveness of ERAS program on postoperative recovery after gastric cancer surgery: a randomized clinical trial. Int J Surg. 2025;111:3306-3313. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 5. | Tian Y, Cao S, Liu X, Li L, He Q, Jiang L, Wang X, Chu X, Wang H, Xia L, Ding Y, Mao W, Hui X, Shi Y, Zhang H, Niu Z, Li Z, Jiang H, Kehlet H, Zhou Y. Randomized Controlled Trial Comparing the Short-term Outcomes of Enhanced Recovery After Surgery and Conventional Care in Laparoscopic Distal Gastrectomy (GISSG1901). Ann Surg. 2022;275:e15-e21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 51] [Article Influence: 12.8] [Reference Citation Analysis (2)] |
| 6. | Jeong O, Jang A, Jung MR, Kang JH, Ryu SY. The benefits of enhanced recovery after surgery for gastric cancer: A large before-and-after propensity score matching study. Clin Nutr. 2021;40:2162-2168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (1)] |
| 7. | Pagano E, Pellegrino L, Robella M, Castiglione A, Brunetti F, Giacometti L, Rolfo M, Rizzo A, Palmisano S, Meineri M, Bachini I, Morino M, Allaix ME, Mellano A, Massucco P, Bellomo P, Polastri R, Ciccone G, Borghi F; ERAS-colorectal Piemonte group. Implementation of an enhanced recovery after surgery protocol for colorectal cancer in a regional hospital network supported by audit and feedback: a stepped wedge, cluster randomised trial. BMJ Qual Saf. 2024;33:363-374. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 8. | Serra-Aracil X, Hidalgo J, Perucho NL, Montesinos CS, Diaz CG, Figueras RV, Rivilla SD, Romero Marcos JM, Gomez CC, Ferreres-Serafini J, Romaguera VP, Galvez A, Caro-Tarragó A; and on behalf of Vesicalcath-Study Group. Multicentre, prospective, randomized controlled non-inferiority trial of bladder catheter management in short-duration, minimally invasive colon surgery (The Vesicalcath I-Study). Int J Surg. 2025;111:8088-8098. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 9. | MacVicar E, Cullen F, Kastora SL, Parnaby C, Mackay C, Ramsay G. A systematic review of the impact of post-operative oral fluid intake on ileus following elective colorectal surgery. Int J Surg. 2022;103:106651. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 10. | Wu H, Liu Q, Zhang N, Chen J, Chen G, Xiong L, Tu X. Road of recovery in gastrointestinal surgery: From ERAS to FRAS. Gastroenterol Endosc. 2024;2:84-89. [DOI] [Full Text] |
| 11. | Han H, Wan R, Chen J, Fan X, Zhang L. Effects of the enhanced recovery after surgery (ERAS) protocol on the postoperative stress state and short-term complications in elderly patients with colorectal cancer. Cancer Rep (Hoboken). 2024;7:e1979. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 12. | Imran A, Ismail M, Raza AA, Gul T, Khan A, Shah SA. A Comparative Study Between the Early and Late Enteral Nutrition After Gastrointestinal Anastomosis Operations. Cureus. 2024;16:e52686. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 13. | Imamura H, Takahashi H, Wada H, Mukai Y, Asukai K, Hasegawa S, Yamamoto M, Takeoka T, Shinno N, Hara H, Kanemura T, Nakai N, Haraguchi N, Sugimura K, Nishimura J, Matsuda C, Yasui M, Omori T, Miyata H, Ohue M, Sakon M. Postoperative aggressive diuresis prevents postoperative tissue edema and complications in patients undergoing distal pancreatectomy. Langenbecks Arch Surg. 2022;407:645-654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (2)] |
| 14. | He J, Qian G, Mao Y, Gao L. Comparison of early vs. routine removal of abdominal drainage tube after laparoscopic appendectomy for perforated appendicitis: a retrospective cohort study. Front Surg. 2025;12:1617312. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 15. | Dong F, Li Y, Jin W, Qiu Z. Effect of ERAS pathway nursing on postoperative rehabilitation of patients undergoing gastrointestinal surgery: a meta-analysis. BMC Surg. 2025;25:239. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 16. | Liu G, Cao S, Liu X, Tian Y, Li Z, Sun Y, Zhong H, Wang K, Zhou Y. Short- and long-term outcomes following perioperative ERAS management in patients undergoing minimally invasive radical gastrectomy after neoadjuvant chemotherapy: A single-center retrospective propensity score matching study. Eur J Surg Oncol. 2025;51:109459. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 17. | Kifle F, Belay E, Kifleyohanes T, Demissie B, Galcha D, Mulye B, Presser E, Oodit R, Maswime S, Biccard B. Adherence to Enhanced Recovery After Surgery (ERAS) With Bellwether Surgical Procedures in Ethiopia: A Retrospective Study. World J Surg. 2025;49:1040-1050. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 18. | Yuan CD, Zhou BZ, Wang NY, Wan QQ, Hu ZZ. Evidence-based control of stress response on intraoperative physiological indexes and recovery of patients undergoing gastrointestinal surgery. World J Gastroenterol. 2025;31:102331. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 5] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (3)] |
| 19. | Kannan V, Ullah N, Geddada S, Ibrahiam A, Munaf Shakir Al-Qassab Z, Ahmed O, Malasevskaia I. Impact of "Enhanced Recovery After Surgery" (ERAS) protocols vs. traditional perioperative care on patient outcomes after colorectal surgery: a systematic review. Patient Saf Surg. 2025;19:4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 29] [Reference Citation Analysis (1)] |
| 20. | Bertocchi E, Brunelli D, Squaranti T, Campagnola D, Camparsi S, Tessari R, Menestrina N, Gentile I, Sanfilippo L, De Santis N, Guerriero M, Ruffo G. Cost Saving Analysis of an Enhanced Recovery After Surgery (ERAS) Program for Elective Colorectal Surgery in an ERAS Qualified and Training Center. World J Surg. 2025;49:850-858. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 21. | Wang J, Meng N, Chen K, Huang X, Feng L, Yang C, Li Z, Sun X. The Relationship Between Depressive Symptoms and Functional Gastrointestinal Disorders (FGIDs): The Chain Mediating Effect of Sleep Disorders and Somatic Symptom. Depress Anxiety. 2024;2024:5586123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |