Published online Feb 27, 2026. doi: 10.4240/wjgs.v18.i2.116100
Revised: November 18, 2025
Accepted: December 11, 2025
Published online: February 27, 2026
Processing time: 116 Days and 10.5 Hours
Obstructed defecation syndrome (ODS) is a heterogeneous pelvic floor disorder in which a persistent focus on “stapled transanal rectal resection (STARR) vs ventral rectopexy (VR)” has encouraged procedure-driven rather than phenotype-guided care. This opinion review synthesizes contemporary evidence on STARR and VR and proposes a phenotype-guided algorithm for surgical decision-making in ODS. We first summarize comparative outcome data, highlighting higher rates of late recurrence and urgency after STARR in contrast to more durable functional relief but greater invasiveness and mesh-related concerns after VR. We then outline a structured preoperative workup that integrates symptom scores, fluoroscopic or magnetic resonance defecography, and high-resolution anorectal manometry to discriminate between isolated structural disease, complex multilevel prolapse, functional defecatory disorders, mixed phenotypes, and ODS with impaired con
Core Tip: The management of obstructed defecation syndrome must evolve beyond the suboptimal dichotomy of stapled transanal rectal resection vs ventral rectopexy. This review advocates for a fundamental paradigm shift toward a phenotype-guided stratified treatment algorithm. Mandatory preoperative phenotyping, integrating advanced imaging and high-resolution anorectal manometry, is crucial for classifying patients into distinct structural and functional subtypes. This personalized approach optimizes patient selection and yields superior, durable functional outcomes, thereby establishing a new framework for precision pelvic floor surgery.
- Citation: Schiano di Visconte M, Sarnari S. Beyond stapled transanal rectal resection vs ventral rectopexy dichotomy: Toward a phenotype-guided surgical paradigm for obstructed defecation syndrome. World J Gastrointest Surg 2026; 18(2): 116100
- URL: https://www.wjgnet.com/1948-9366/full/v18/i2/116100.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i2.116100
Obstructed defecation syndrome (ODS) is a complex pelvic floor disorder. It is characterized by difficulty or inability to evacuate stools despite the urge to defecate. Clinical manifestations typically include excessive straining, incomplete rectal emptying, and the frequent need for manual assistance, such as perineal pressure or digital maneuvers, to facilitate evacuation[1,2]. ODS predominantly affects women and commonly coexists with anatomical abnormalities such as rectocele (an anterior rectal wall bulging into the vagina) or internal rectal intussusception. Functional disorders such as pelvic floor dyssynergia (paradoxical anal contraction) may also occur[2]. Initial management is conservative, including a high-fiber diet, adequate hydration, laxatives, pelvic floor exercises, and biofeedback. These non-surgical strategies alleviate symptoms in many patients[3,4]; however, a subset of patients with refractory ODS and structural anomalies may require surgical intervention[2].
Over the past two decades, two surgical approaches have dominated ODS management. Stapled transanal rectal resection (STARR) is a transanal procedure that resects redundant rectal tissue (e.g., rectocele or internal prolapse), whereas ventral rectopexy (VR) is typically performed laparoscopic ventral mesh rectopexy (LVMR) or robotic ventral mesh rectopexy (RVMR). VR suspends the anterior rectum to the sacrum with a mesh to correct prolapse and restore pelvic support[5]. These procedures address ODS from different perspectives: STARR removes the obstructing rectal fold, whereas VR repositions and stabilizes the rectum. Each possesses a distinct risk profile and set of indications. Historically, surgical decision-making has often framed STARR and VR as competing or mutually exclusive options, fueling debate over which procedure is “better” for ODS. Comparative outcomes highlighted a clear long-term divergence between STARR and VR, with higher late recurrence rates after STARR and more durable functional relief following VR (Table 1).
| Outcome | STARR | VR | Ref. |
| Symptomatic recurrence (5-10 years) | 20%-40% (up to about 40% at 10 years) | < 10% (about 8%-10% at 10 years) | Schiano di Visconte[5]; Mandovra et al[43]; Barra et al[44] |
| Symptom improvement at 12 months | 70%-90% (approximately 85% in one series) | 70%-90% (durable relief) | Schiano di Visconte[5]; Mandovra et al[43]; Mäkelä-Kaikkonen et al[45] |
| Anatomical recurrence | Approximately 20%-38% (rectocele/intussusception) | < 5%-8% | Mandovra et al[43]; Barra et al[44] |
| Surgical re-intervention rate | Up to about 20%-25% | < 10% | Mäkelä-Kaikkonen et al[45]; Gagliardi et al[46] |
| Quality-of-life improvement | Improves initially but declines at long-term follow-up | Stable/sustained improvement | Mandovra et al[43]; Mäkelä-Kaikkonen et al[45] |
| Procedure-specific complications | Urgency, pain/tenesmus (approximately 20%); rare anastomotic dehiscence or fistula | Mesh-related complications about 1%-2% (erosion about 0.9%); rare mesh detachment | Hess et al[47]; Ripamonti et al[18] |
| Patient satisfaction | About 70%-75% overall long-term satisfaction; about 20%-35% “very satisfied” at 10 years | About 70%-85% “high satisfaction” at 2 years | Mandovra et al[43]; Mäkelä-Kaikkonen et al[45]; Schiano di Visconte et al[6] |
| Mean operative time | Approximately 40 minutes | Approximately 115 minutes | Mandovra et al[43]; Mandovra et al[48] |
| Hospital stay (days) | Approximately 2.1 days | Approximately 3.3 days | Mandovra et al[43]; Mandovra et al[48] |
Crucially, the comparative data underscore a significant divergence in long-term durability, suggesting that a one-size-fits-all approach is inadequate. STARR is generally associated with a higher risk of late failure, with reported rates of recurrent obstructive symptoms ranging from 20% to 40% at 5-10 years and a higher incidence of reinterventions[6]. Conversely, VR tends to offer a more durable functional relief. For example, a randomized controlled trial (RCT) in elderly patients with ODS demonstrated a substantially lower mid-term symptom recurrence rate after VR than after STARR (7% vs 24%)[7]. Furthermore, a long-term series documented durable anatomical results following VR, with recurrence rates of approximately 8% at 10 years[8].
Early comparative studies and prevailing surgical preferences frequently positioned the transanal and transabdominal approaches as opposing paradigms[5,9]. This binary paradigm has fueled ongoing controversy in both literature and clinical practice, often portraying STARR and VR as competing techniques rather than complementary options for treating ODS.
In this opinion review, we argue that the conventional binary framing of STARR vs VR is outdated and overly simple. Rather than treating these operations as competitors, modern pelvic floor surgery should embrace a phenotype-guided approach in which STARR and VR are complementary tools within a stratified treatment algorithm. By tailoring the surgical strategy to the patient’s specific anatomic and functional ODS subtype, as defined through phenotypic evaluation (including imaging, manometry, and symptom profiles), we can optimize the outcomes and minimize the shortcomings of any single procedure. We examined the limitations of the current “either/or” paradigm, outline the rationale and evidence for a phenotype-based algorithm, discussed how this approach can be integrated into clinical practice, and proposed research priorities to validate stratified surgical decision-making for ODS.
The traditional dichotomy that positions STARR against VR represents a fundamental oversimplification that impedes optimal management of ODS. ODS is inherently multifactorial and heterogeneous, meaning that treating it as a uniform entity inevitably leads to a significant mismatch between the chosen surgical procedure and the patient’s specific under
Evidence from comparative studies highlights a clear trade-off between the two approaches, suggesting that an outright “winner” is unlikely when patient populations are unselected. Both STARR and VR achieve substantial early symptom relief in appropriately selected patients, with initial response rates ranging from 70% to 90%[5]. However, long-term follow-up revealed critical differences in durability. STARR procedures are associated with higher rates of relapse and symptom recurrence over time, with studies reporting recurrence of obstructive symptoms in 22%-40% of patients 5-10 years postoperatively[5]. Moreover, late sequelae such as persistent rectal urgency (occurring in approximately 12% of cases) and anatomical recurrence are frequently observed.
By contrast, VR results in sustained functional outcomes. For instance, a comparative randomized trial found that the mid-term recurrence of ODS symptoms was significantly lower after LVMR than after STARR (7% vs 24% at 2 years)[5]. Similarly, long-term follow-up studies have confirmed that VR patients maintain better constipation relief and require fewer subsequent reoperations[5]. Moreover, quality-of-life metrics often favor the rectopexy approach. One study demonstrated that although both procedures improved ODS symptom scores, only VR significantly enhanced the overall pelvic floor function and physical quality-of-life metrics, indicating a broader and more durable functional correction[1].
Despite its greater long-term durability, VR is a more extensive procedure requiring abdominal access, general ane
Conversely, the presumed advantage of STARR, its minimally invasive, perineal nature, is offset by a distinct spectrum of complications that underscore the danger of its indiscriminate use. Post-STARR issues can include chronic proctalgia, debilitating anorectal pain, new-onset fecal urgency, and incontinence[5]. Approximately 20% of patients report painful defecation 1 year after STARR[5], which can offset the perceived early benefits.
Perhaps the most significant limitation of the binary paradigm is its failure to account for underlying functional disorders. A key finding is that in up to 40% of patients with persistent ODS symptoms or incontinence following STARR, no residual anatomical defects can be identified[5]. This suggests that the primary cause of surgical failure was an unrecognized functional disorder, such as coexisting pelvic floor dyssynergia or occult pelvic floor laxity, for which transanal resection could not correct. This often leads to patient dissatisfaction and the subsequent need for a second, more complex operation.
This combination of technical success and symptomatic failure vividly illustrates the flaw inherent in the ‘either/or’ mentality: ODS patients who lack proper phenotyping may undergo an operation such as STARR, which addresses only a secondary anatomical defect while leaving the core functional issue uncorrected, ultimately failing to meet the patient’s clinical needs.
The necessity of a phenotype-guided approach is most evident in special populations, particularly in patients with impaired anal sphincter function (e.g., elderly multiparous women). Within an unstratified binary framework, a surgeon may perform STARR solely based on the presence of internal prolapse. However, transanal stapling can exacerbate incontinence in sphincter weakness, leading to iatrogenic morbidity. Recognizing this critical trade-off, expert consensus has shifted, with a European panel reaching unanimous agreement that for ODS patients with compromised sphincter control, VR (or alternative approaches that avoid additional anal trauma) should be the treatment of choice[9]. This clinical nuance, in which patient safety and functional preservation take precedence over a general preference for sim
In summary, there is no single solution for the ODS. The binary paradigm of “STARR vs VR” is an oversimplification that leads to surgical planning based on dogma rather than diagnostics. By focusing on a competitive contest, we risk losing sight of the primary goal of selecting the right surgery for the right patient through comprehensive phenotyping evaluation. This realization mandates a strategic shift from binary thinking to a stratified complementary treatment algorithm.
The future of ODS management lies in phenotype-based stratification, an individualized approach that categorizes patients according to their specific anatomical and functional pathologies, thereby guiding the development of tailored treatments. ODS is recognized not as a single disease but as a syndrome with diverse subtypes. Clinicians broadly distinguish between mechanical ODS (caused by structural abnormalities such as rectocele, rectal prolapse, or enterocele) and functional ODS (resulting from pelvic floor dyssynergia or anismus without significant mechanical blockage)[11]. In clinical practice, many patients present with a mixed ODS phenotype characterized by concurrent structural lesions (e.g., intussusception) and functional impairments (e.g., paradoxical puborectalis contraction). A phenotype-driven strategy acknowledges this heterogeneity by using objective diagnostics to match the patient to the intervention (surgical, behavioral, or pharmacological) most likely to correct their predominant defects.
Comprehensive preoperative evaluation forms the foundation of this management approach. It involves targeted history and validated symptom scoring (e.g., ODS score), as well as specialized testing.
High-quality imaging: Defecography [via fluoroscopy or dynamic magnetic resonance imaging (MRI)] is essential for delineating anatomical abnormalities, including rectocele depth, intussusception grade, and the presence of an enterocele[11]. If imaging reveals clear mechanical blockage, anatomical surgical correction is directly guided.
Functional assessment: When structural abnormalities are minimal or absent, the clinical focus shifts to functional evaluation. High-resolution anorectal manometry (HRAM) complemented by the balloon expulsion test is essential for characterizing functional defecatory disorders (FDD). Beyond measuring anal resting and squeeze pressures, HRAM quantifies rectal sensation, compliance, and propulsive pressure. These parameters enable the accurate subclassification of defecatory dysfunction according to the validated framework proposed by Ratuapli et al[12], which distinguishes coordination failure (types I-III) from pure propulsion failure (type IV). Type IV FDD is defined as normal or adequate anal relaxation with markedly reduced rectal propulsive pressure (< 40 mmHg), representing a distinct rectal inertia phenotype.
This distinction has direct therapeutic implications. HRAM-based phenotyping reveals that patients with type IV propulsion failure are unable to generate sufficient intrarectal pressure to expel stool despite exhibiting coordinated relaxation patterns[12,13]. In this context, applying a purely suspensive procedure to a patient with confirmed rectal inertia (< 40 mmHg) may not only fail to alleviate obstructed defecation but may also exacerbate symptoms by “suspending” the rectum that is intrinsically non-propulsive. Anchoring a functionally impaired rectum does not address the primary motility disorder and may eliminate compensatory straining mechanisms upon which the patient previously relied[13].
Management of type IV FDD should therefore prioritize motility-targeted strategies, including intensified conservative treatments and neuromodulation. In severe, refractory cases, particularly when rectal inertia coexists with colonic inertia, resectional procedures may represent definitive therapy[14]. In contrast, type I-III FDD are characterized by paradoxical contraction, inadequate relaxation, or combined coordination and propulsion defects. In these phenotypes, the presence of dyssynergia mandates pelvic floor biofeedback as first-line treatment, thereby avoiding inappropriate anatomical surgery and reducing postoperative functional failure[15].
Adjunctive botulinum toxin injections may aid in the management of pelvic floor dyssynergia in patients who remain symptomatic despite standard rehabilitative measures. Clinical studies have demonstrated that targeted intramuscular injections into the puborectalis muscle and/or internal anal sphincter effectively reduce paradoxical contractions and facilitate pelvic floor relaxation. This action subsequently improves balloon expulsion and the overall defecatory dynamics[16]. Although the therapeutic effect of botulinum toxin is transient, this period of reduced muscular hypertonicity can significantly enhance the efficacy of concurrent biofeedback therapy, enabling patients to practice coordinated evacuation patterns. Consequently, botulinum toxin should be considered as a second-line adjunct for carefully selected, refractory cases in a comprehensive, multimodal rehabilitation strategy.
Thus, HRAM is not merely a diagnostic but a critical decision-making tool that ensures precise phenotype procedure matching, particularly by identifying patients in whom suspensive procedures, such as VR, should be avoided due to underlying rectal inertia.
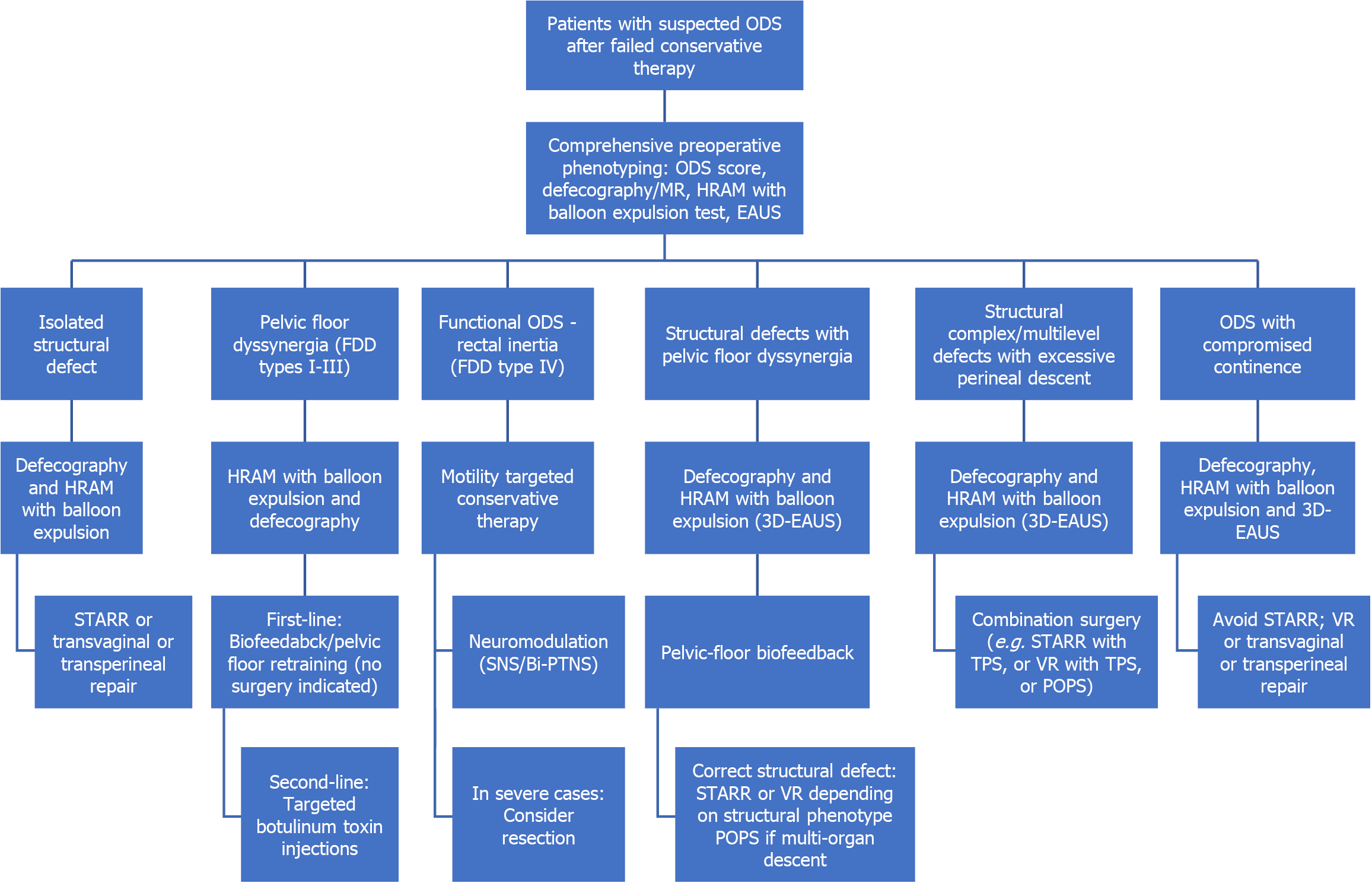
The first critical decision point of the algorithm separates patients into anatomical and functional ODS, with biofeedback being prioritized for the latter. The overall decision pathway is summarized in a practical flowchart that aligns diagnostic phenotyping with tailored interventions (Figure 1). Phenotype classification facilitates the use of surgical and non-surgical interventions as complementary rather than competing approaches.
The operational details of each phenotype, including the key features, diagnostic anchors, and primary recommended interventions, are summarized in Table 2.
| Phenotype | Key clinical features | Diagnostic findings (defecography/HRAM) | Recommended primary intervention | Rationale/clinical notes |
| Isolated structural ODS | Single anatomical defect (large rectocele or low-grade internal intussusception); preserved sphincter function and no major dyssynergia | Defecography: Solitary rectocele or low-grade internal prolapse without multi-compartment involvement; HRAM: Normal relaxation pattern and adequate rectal propulsive pressure | STARR or targeted local repair (e.g., site-specific rectocele repair) | Minimally invasive and directly addresses the mechanical obstruction; high symptom relief and satisfaction (> 80%) in appropriately selected “simple” ODS when continence and coordination are preserved |
| Complex structural ODS (multi-compartment) | Combined defects (e.g., rectocele with intussusception and/or enterocele and/or perineal descent) or high-grade internal prolapse approaching external prolapse | Defecography: Multi-compartment pelvic organ prolapse (anterior, apical, and posterior compartments) or high-grade internal prolapse; HRAM: Generally preserved coordination but anatomical obstruction predominates | Ventral rectopexy (LVMR/RVMR) and/or adjunct pelvic repairs; in selected women with multi-organ pelvic support defects, POPS with or without concomitant transanal repair | Provides comprehensive anatomical correction and pelvic support with long-term recurrence < 10%; VR addresses combined rectocele with intussusception and reduces perineal descent; in multi-compartment female prolapse, POPS offers comparable ODS relief while avoiding extensive sacral dissection, preserving sexual function, and remaining fully compatible with transanal procedures |
| Functional ODS with dyssynergia (types I-III FDD) | Outlet obstruction without structural lesion; paradoxical contraction or incomplete relaxation of the pelvic floor during attempted defecation | Defecography: Minimal or absent mechanical obstruction; HRAM: Types I-III coordination failure (paradoxical contraction, inadequate relaxation, or mixed pattern); Balloon expulsion test: Prolonged or failed | First-line: Pelvic-floor biofeedback and retraining; Second-line adjunct in refractory cases: Targeted botulinum toxin injection to puborectalis and/or internal anal sphincter | Corrects functional dyssynergia and avoids unnecessary surgery; success in up to 70% of patients; adjunctive botulinum toxin can transiently reduce hypertonicity, facilitate pelvic floor relaxation, and enhance the effectiveness of biofeedback in selected refractory cases within a multimodal rehabilitation strategy |
| Rectal inertia (type IV FDD) | Severe outlet obstruction with marked straining, sense of incomplete evacuation, often associated with slow-transit constipation; anal relaxation preserved but expulsive force inadequate | Defecography: Little or no fixed mechanical obstruction despite persistent incomplete emptying; HRAM: Type IV pattern with normal or adequate anal relaxation but markedly reduced rectal propulsive pressure (< 40 mmHg); Balloon expulsion: Markedly prolonged or failed | Motility-targeted therapies (intensified conservative measures, laxative optimization, and neuromodulation); in carefully selected severe cases with combined colonic/rectal inertia, consideration of resectional procedures; avoid suspensive operations such as STARR or ventral mesh rectopexy | Represents a distinct rectal inertia phenotype in which the primary defect is propulsion failure rather than obstruction; suspensive procedures may “anchor” a non-propulsive rectum and worsen symptoms without correcting motility; management should prioritize restoration of motility (including neuromodulation) and reserve resectional surgery for selected refractory patients, rather than applying standard suspensive ODS operations |
| Mixed ODS (structural with dyssynergia) | Coexistence of structural lesion (e.g., rectocele or internal intussusception) and functional defecatory disorder (paradoxical pelvic floor contraction) | Defecography: Demonstrable rectocele and/or internal prolapse; HRAM: Dyssynergic pattern (types I-III) | Stepwise multimodal therapy: Initial pelvic-floor physical therapy and biofeedback to correct dyssynergia, followed by anatomical correction (STARR, VR, or local repair as indicated by the dominant mechanical defect) | Surgery alone may only partially relieve symptoms when dyssynergia is ignored; treating the functional deficit first, then addressing the anatomical obstruction, reduces the risk of persistent ODS, postoperative urgency, and recurrence; staged, phenotype-guided management improves overall outcomes |
| ODS with compromised continence | ODS associated with weak sphincter, obstetric injury, or low anal pressures | HRAM: Low resting and/or squeeze pressures; Endoanal ultrasound or MRI may show sphincter defects; Clinical history of urge or passive incontinence | Avoid STARR; prefer VR, local rectocele repair; consider sacral neuromodulation when incontinence predominates and the structural component is minor | Preference for VR or local repairs mitigates the risk of continence deterioration and prevents iatrogenic anal sphincter injury |
| ODS with excessive perineal descent | Long-standing straining; perineal sagging or descent > 3 cm, often coexisting with rectocele or internal prolapse | Defecography or dynamic MRI: Perineal descent > 3 cm and pelvic floor laxity; Associated rectocele/intussusception may be present but does not fully explain outlet obstruction | Combined surgery: STARR with TPS or VR with TPS | Reinforces perineal body and prevents recurrence; emerging phenotype-guided strategy with improved long-term functional outcomes |
A phenotype-guided algorithm provides concrete guidance for complex scenarios, moving beyond the binary “STARR vs VR”: Predominantly structural ODS (isolated): These are patients whose diagnostic workup revealed a solitary anatomic defect (e.g., a large anterior rectocele or focal intussusception) with otherwise normal pelvic floor function. These patients are ideal candidates for targeted anatomical repairs. STARR or similar transanal stapled techniques can be highly effective in this case, as they directly remove the offending tissue that causes the outlet blockage. Indeed, evidence suggests that STARR provides excellent symptom relief in appropriately selected cases of isolated rectocele or intussusception, with a short-term success rate of 80% and high patient satisfaction when no other major dysfunction is present[10,17]. A long-term series reported 76% patient satisfaction at follow-up after STARR for internal prolapse, although some patients experienced recurrence, highlighting the importance of patient selection[18]. Thus, phenotype: “Simple” ODS (single-level anatomical), preferred approach: STARR, because of its minimally invasive nature and direct correction, as long as the anal sphincter is competent.
Predominantly structural ODS (complex/multilevel): Patients with multiple anatomical issues, such as combined moderate intussusception and a significant rectocele, possibly with descent of the perineum, or those with high-grade internal prolapse approaching external prolapse. In such cases, the abdominal approach is often more suitable for correcting the prolapse spectrum. LVMR is preferable for “complex” anatomical ODS phenotypes, as it allows simul
An additional suspensive option that merits consideration in female patients with multi-compartment pelvic organ descent is pelvic organ prolapse suspension (POPS)[20]. POPS provides a minimally invasive, one-stage repair that simultaneously addresses anterior, apical, and posterior defects. This makes it particularly suitable in cases where ODS coexists with uterine descent or stress urinary incontinence[21]. Comparative evidence demonstrates that POPS achieves anatomical correction and ODS symptom relief comparable to those of VMR. POPS avoids extensive sacral dissection and reduces the risk of hypogastric nerve traction injury[22]. Importantly, POPS is fully compatible with transanal procedures as originally described in combination with STARR. This allows tailored treatment of persistent rectocele or internal prolapse within the same operative session[20]. Early studies reported favorable safety, short operative times, and preserved sexual function. No increased incidence of dyspareunia has been reported with multi-compartment suspension[23]. In selected women with combined pelvic support defects, POPS is a valid alternative to VMR. It may be integrated into phenotype-guided algorithms when multi-organ descent contributes significantly to ODS.
Perineal resectional procedures also play an important role within a phenotype-based framework. Altemeier’s procedure and Delorme procedures are well-established options for full-thickness external rectal prolapse, particularly in elderly or high-risk patients. In cases of incarcerated or strangulated complete procidentia, a perineal approach under regional anesthesia may represent the safest or only feasible treatment[24-26]. These operations provide acceptable long-term control of prolapse with low perioperative mortality. In particular, Delorme’s procedure has been associated with improved continence and a low incidence of postoperative constipation. This finding supports its continued use in carefully selected patients[24,26]. Within our phenotype-guided algorithm, perineal resections are reserved for frail patients with external full-thickness prolapse or emergency presentations. These are conceptually distinct from the elective internal rectal prolapse related ODS phenotypes which represent the primary focus of this review.
Mixed ODS (structural + dyssynergia): A substantial proportion of patients exhibit both a structural lesion and a FDD. A classic example is internal rectal prolapse with paradoxical anal contractions (anismus). In such cases, when structural and functional issues coexist, surgery alone may only partially relieve symptoms, as shown by studies in which anato
Thus, the algorithm here becomes multimodal. First, the functional component is treated, typically with pelvic floor physical therapy and biofeedback to retrain proper relaxation during defecation[28]. Once the patient’s coordination improves (or if it proves refractory, but the anatomical blockage is clearly significant), proceed with anatomical correction using STARR or VR as indicated by the mechanical defect. Essentially, this phenotype may require combination therapy to address both behavioral and structural components. Failure to address the functional component is a well-recognized cause of anatomical surgical failure[11]. Finally, the phenotype-based approach emphasizes the identification and treat
ODS with compromised continence: For patients with low anal pressure or a weak sphincter, it is imperative to alleviate obstruction without compounding incontinence. Analyses of clinical practice show that, in cases of rectocele with poor sphincter function, surgeons consistently avoid STARR due to the risk of worsened continence, instead favoring less traumatic options such as VR or local perineal/transvaginal repairs[11,29,30]. When rectal intussusception coexisted with a poor sphincter, VR was favored approximately 30% of the time, with very few STARRs performed (3.8%)[9]. These real-world data exemplify a de facto phenotype-driven decision-making process: Phenotype: “ODS in incon
Furthermore, sacral neuromodulation (SNS), an established therapy for fecal incontinence that has also shown promise in improving constipation in selected patients, is now being considered for the “ODS in incontinent patient” phenotype, particularly in cases where prolapse is minor but functional inertia is significant[31].
ODS with excessive perineal descent: Long-standing ODS often leads to or exacerbates, descending perineum syn
In summary, the phenotype-guided algorithm uses a full armamentarium of interventions in a tailored manner. STARR and VR are viewed as complementary rather than competing, with each reserved for the context in which its benefits outweigh its disadvantages. Not every ODS patient should receive a mesh rectopexy, and not everyone is suited for a stapled resection; some may need neither (if conservative therapy suffices) or occasionally, both in sequence. The key is stratification: Incorporating the information from defecography, HRAM, anal sphincter evaluation, and patient-reported symptom profiles to categorize the patient, and then applying the surgical (or non-surgical) modality that aligns with that category. By doing so, we address the “right problem with the right solution”, potentially improving success rates and reducing complications. Importantly, this approach demands a multidisciplinary pelvic floor team mindset, often involving colorectal surgeons, gastroenterologists, radiologists, and physiotherapists working in concert to establish the phenotype and plan treatment.
Translating a phenotype-based algorithm into routine clinical practice requires both cultural and logistical shifts in ODS management. Clinically, adopting this stratified strategy means standardizing a thorough preoperative work-up for every ODS patient rather than jumping straight from symptoms to a favored operation. Practically, institutions should ensure that patients with constipation and outlet symptoms undergo objective diagnostic testing such as defecography and anorectal manometry prior to surgical referral[11,34-36]. Currently, this practice has not been universally adopted. A multicenter study revealed that although most specialists perform defecography for ODS, the use of standardized symptom scoring remains variable, with approximately 30% of centers not routinely employing any formal ODS scale[9,28,37]. To facilitate phenotype-driven care, pelvic floor units should implement protocols in which imaging and mano
Mandatory multidisciplinary review: All ODS work-ups should be discussed in a multidisciplinary pelvic floor meeting involving colorectal surgeons, gastroenterologists, radiologists, and specialized physiotherapists. Here, each case is formally “phenotype” (e.g., structural type I, mixed ODS) prior to initiating the treatment plan.
Broadening the skill set: Surgeons must broaden their expertise beyond a single technique. Clinicians must either broaden their surgical expertise to include both transanal (STARR, local repair) and abdominal (VR, resection rectopexy) procedures or collaborate formally within a dedicated pelvic floor team to ensure that patients are offered an optimal phenotype-matching operation[38].
Staged multimodal workflow: The gold-standard clinical workflow should involve: (1) Comprehensive phenotyping; (2) Initial non-operative treatment (e.g., biofeedback for all patients with a dyssynergia component; (3) Multidisciplinary agreement on the surgical plan; and (4) Executing the tailored intervention, which may involve a combination of techniques[2]. This ensures that coexisting issues such as anal sphincter weakness or slow-transit constipation have are not overlooked.
Educating patients is another critical integration step: Patient education is a crucial element of clinical integration. Individuals should understand why a specific surgical approach is recommended for their condition, and recognize that a different procedure, although effective in other cases, may not be appropriate for their phenotype. This helps to set appropriate expectations and shared decision-making. For instance, explaining to a patient that “because your tests show primarily a functional muscle problem, surgery won’t help and we should do therapy instead” can avoid unnecessary operations. Conversely, a patient with a clear mechanical obstruction can be counseled that targeted repair (via STARR or VR) has a high likelihood of improving symptoms based on their phenotype. As phenotype-based algorithms become standard, predictive tools such as risk calculators or decision aids may be developed to assist in clinical decision-making that inputs defecography and manometry findings to suggest an optimal treatment path.
The primary barriers include the limited availability and lack of standardization of advanced diagnostics, such as HRAM and MRI defecography, outside tertiary care centers, as well as institutional inertia and variability in clinician training[11,37]. Addressing these issues requires both infrastructure investment and formalized clinical education to ensure equitable access to this precision care model.
This shift away from the binary paradigm opens up critical avenues for generating highly stratified evidence. Traditional trials that randomized heterogeneous ODS populations to STARR or VR likely yielded inconclusive results because they failed to account for the underlying phenotypes[5]. There is an urgent need for prospective stratified trials to validate whether matching surgical interventions to the ODS subtype improves long-term outcomes. The research agenda should prioritize the following questions: (1) Validating phenotype-specific outcomes. Isolated structural ODS: RCTs should determine whether the less invasive STARR procedure is equivalent to VR in mid- and long-term functional outcomes for patients with the ideal phenotype, specifically isolated rectocele or low-grade intussusception and preserved function and continence (e.g., 5-year symptom recurrence)[1,5,9]. Optimal management of complex prolapse: Large comparative studies or RCTs are necessary to evaluate the necessity of combined procedures. For patients with a complex structural phenotype (e.g., high-grade intussusception combined with a large rectocele), RCTs should clarify whether LVMR alone is sufficient[38] or whether combining it with synchronous rectocele repair or resection (e.g., LVMR + STARR) leads to significantly better functional results or lower reoperation rates[10]; (2) Defining the role of combination and novel therapies. Therapy sequencing in mixed ODS: A trial is needed to compare outcomes of “surgery alone” vs “staged multimodal approach” (i.e., biofeedback followed by surgery) in the mixed ODS phenotype. This validates the intuitive but currently unproven notion that prehabilitation with biofeedback improves postoperative ODS scores and reduces recurrence in patients with an underlying functional deficit[39,40]. Perineal descent adjuncts: The integration of innovative techniques such as TPS, into existing procedures must be rigorously evaluated. A multicenter, prospective cohort study confirmed that combining STARR with TPS significantly improved long-term outcomes for patients with internal prolapse and excessive perineal descent compared to STARR alone, validating a phenotype-driven combination approach for this difficult subset[19]. Further research is required for VR + TPS. The role of neuromodulation: SNS, and bilateral percutaneous tibial nerve stimulation, which are effective for fecal incontinence and shown to improve constipation in selected cases, should be formally evaluated in ODS with high incontinence risk phenotype or in cases of prominent functional inertia[31,40]; and (3) Establishing long-term data infrastructure. Multicenter ODS registries: The foremost research priority is the establishment of prospective multicenter ODS surgical registries. These registries should index not only the procedural details and basic outcomes but also each patient’s preoperative phenotype (defecography and manometry findings)[11]. Over time, these long-term data (≥ 5-10 years) will allow researchers to identify which phenotypes are prone to specific failure modes after a given surgery, thus allowing for continuous refinement of the therapeutic algorithm. Patient-reported outcomes (PROMs): Future studies should heavily emphasize the use of validated PROMs (e.g., patient assessment of constipation quality of life and ODS scores) alongside objective anatomical success, as quality of life often favors VR in the long term[40,41]. Connecting specific functional phenotypes (e.g., rectal hyposensiti
By committing to stratified evidence generation, future clinical guidelines can transition from offering general sugges
The management of ODS is undergoing a fundamental paradigm shift, moving decisively beyond the restrictive dichotomy between STARR and VR. Considering the multifactorial heterogeneity of ODS, therapeutic strategies should be equally nuanced. The central clinical question is no longer which operation is inherently superior but rather which procedure is optimally suited to the patient’s specific defecatory phenotype. Embracing a phenotype-guided algorithm positions STARR and VR as complementary modalities, each indicated in distinct clinical contexts, and often partnered in multimodal approaches for complex presentations (e.g., STARR combined with TPS). This approach has the potential to improve patient outcomes by promoting durable symptom relief and reducing complications associated with phenotype-mismatched surgery. Realizing the benefits of this personalized approach requires immediate and decisive institutional commitment across the clinical spectrum. Clinically, this mandates a standardized and comprehensive preoperative workup for every patient presenting with outlet symptoms, ensuring the routine application of defecography or MRI for structural evaluation and HRAM for functional assessment prior to surgical consultation. The establishment of formal multidisciplinary pelvic floor conferences is essential to facilitate explicit phenotypic classification of each case (e.g., mixed ODS: Structural intussusception with functional dyssynergia) before treatment initiation. Surgeons should develop expertise in both transanal and abdominal techniques or establish robust collaborative pathways, thereby ensuring that patients receive the procedure aligned with their phenotypic classification rather than the surgeon’s preferred modality. To formalize this transition, the research agenda must prioritize the generation of evidence that is stratified and targeted. This requires the immediate launch of prospective, stratified randomized trials that compare interventions only within well-defined, homogeneous phenotypic subgroups (e.g., comparing STARR vs VR solely in the isolated structural ODS cohort) to generate the necessary level I evidence for algorithmic validation. Equally imperative is the creation of long-term multicenter ODS surgical registries designed to meticulously correlate long-term (≥ 5 years) functional outcomes and recurrence patterns with each patient’s initial preoperative phenotypic classification. This individualized, precision-based methodology aligns ODS management with a broader shift toward personalized surgical care. While the foundational literature largely originates from established European units, emerging data from North America, Asia, and Latin America underscores the global relevance of this paradigm and emphasizes the need for context-specific adaptations of the proposed algorithms. Ultimately, overcoming the limitations of the outdated binary paradigm, namely recurrent symptoms and functional failure, depends on aligning therapeutic strategies with each patient’s pathophysiology, with true surgical success defined as the restoration of comfort, complete evacuation, and a sustained quality of life.
| 1. | Altomare DF, Picciariello A, Memeo R, Fanelli M, Digennaro R, Chetta N, De Fazio M. Pelvic floor function following ventral rectopexy versus STARR in the treatment of obstructed defecation. Tech Coloproctol. 2018;22:289-294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 15] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 2. | Bhandarkar S, Kalikar V, Supe A, Patankar SR. Comparison of Surgical Outcomes of Laparoscopic Ventral Mesh Rectopexy and Stapled Transanal Rectal Resection for Managing Obstructed Defecation Syndrome. World J Colorectal Surg. 2024;13:77-82. [DOI] [Full Text] |
| 3. | Bunni J, Laugharne MJ. Pathophysiological basis, clinical assessment, investigation and management of patients with obstruction defecation syndrome. Langenbecks Arch Surg. 2023;408:75. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 4. | Valiyeva S, Tiso D, Cerri P, Pisciaroli A, Pietroletti R. Results of medical treatment with psyllium, lactobacillus, and tryptophan (Plurilac® Trio) in obstructive defecation syndrome. Front Surg. 2024;11:1361049. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 5. | Schiano di Visconte M. Comparative clinical outcomes of ventral rectopexy versus STARR in the management of obstructed defecation syndrome: a clinical review. Updates Surg. 2025;77:1645-1655. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 6. | Schiano di Visconte M, Nicolì F, Pasquali A, Bellio G. Clinical outcomes of stapled transanal rectal resection for obstructed defaecation syndrome at 10-year follow-up. Colorectal Dis. 2018;20:614-622. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 7. | Madbouly KM, Mohii AD. Laparoscopic Ventral Rectopexy Versus Stapled Transanal Rectal Resection for Treatment of Obstructed Defecation in the Elderly: Long-term Results of a Prospective Randomized Study. Dis Colon Rectum. 2019;62:47-55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 22] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 8. | Consten EC, van Iersel JJ, Verheijen PM, Broeders IA, Wolthuis AM, D'Hoore A. Long-term Outcome After Laparoscopic Ventral Mesh Rectopexy: An Observational Study of 919 Consecutive Patients. Ann Surg. 2015;262:742-7; discussion 747. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 136] [Article Influence: 12.4] [Reference Citation Analysis (1)] |
| 9. | Picciariello A, Rinaldi M, Grossi U, Trompetto M, Graziano G, Altomare DF; SICCR ODS Study Group, Gallo G. Time trend in the surgical management of obstructed defecation syndrome: a multicenter experience on behalf of the Italian Society of Colorectal Surgery (SICCR). Tech Coloproctol. 2022;26:963-971. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 10. | Borie F, Bigourdan JM, Pissas MH, Guillon F. Laparoscopic ventral rectopexy for the treatment of outlet obstruction associated with recto-anal intussusception and rectocele: a valid alternative to STARR procedure in patients with anal sphincter weakness. Clin Res Hepatol Gastroenterol. 2014;38:528-534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 20] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 11. | Podzemny V, Pescatori LC, Pescatori M. Management of obstructed defecation. World J Gastroenterol. 2015;21:1053-1060. [PubMed] [DOI] [Full Text] |
| 12. | Ratuapli SK, Bharucha AE, Noelting J, Harvey DM, Zinsmeister AR. Phenotypic identification and classification of functional defecatory disorders using high-resolution anorectal manometry. Gastroenterology. 2013;144:314-322.e2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 164] [Cited by in RCA: 140] [Article Influence: 10.8] [Reference Citation Analysis (1)] |
| 13. | Patcharatrakul T, Rao SSC. Update on the Pathophysiology and Management of Anorectal Disorders. Gut Liver. 2018;12:375-384. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 52] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 14. | Tabatabaei SM, Mousavie SH, Vaseghi H, Sedaghat HK, Alemrajabi M. The effect of total colectomy with posterior suture rectopexy in patients with internal prolapse and colonic inertia; A case series on 9 women. Int J Surg Case Rep. 2025;131:111327. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 15. | Alavi K, Thorsen AJ, Fang SH, Burgess PL, Trevisani G, Lightner AL, Feingold DL, Paquette IM; Clinical Practice Guidelines Committee of the American Society of Colon and Rectal Surgeons. The American Society of Colon and Rectal Surgeons Clinical Practice Guidelines for the Evaluation and Management of Chronic Constipation. Dis Colon Rectum. 2024;67:1244-1257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 15] [Article Influence: 7.5] [Reference Citation Analysis (1)] |
| 16. | Kaplan JA, Simianu VV. Pelvic Floor Nonrelaxation: Approach to Evaluation and Treatment. Clin Colon Rectal Surg. 2021;34:49-55. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 17. | Zhang B, Ding JH, Yin SH, Zhang M, Zhao K. Stapled transanal rectal resection for obstructed defecation syndrome associated with rectocele and rectal intussusception. World J Gastroenterol. 2010;16:2542-2548. [PubMed] [DOI] [Full Text] |
| 18. | Ripamonti L, Guttadauro A, Lo Bianco G, Rennis M, Maternini M, Cioffi G, Chiarelli M, De Simone M, Cioffi U, Gabrielli F. Stapled Transanal Rectal Resection (Starr) in the Treatment of Obstructed Defecation: A Systematic Review. Front Surg. 2022;9:790287. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 19. | Renzi A, Marano L, Talento P, Brusciano L, Pezzolla A, Izzo D, Antropoli C, D'Aniello F, Di Sarno G, Minieri G, Cantore G, Terracciano G, Barbato D, Docimo L, Antropoli M, Palumbo A, Lanza M, Caputi EM, Brillantino A. Transverse perineal support improves long-term outcomes in patients undergoing stapled transanal rectal resection for obstructed defecation syndrome: a multicenter observational case-control study. Ann Coloproctol. 2025;41:330-337. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 20. | Longo A, Boller B, Gloeckler M, Crafa F, Gianfreda V, Giannantoni C, del Re L, Ceriani C. Multiorgan Pelvic Prolapses. Indian J Colo-Rectal Surg. 2021;4:1-11. [DOI] [Full Text] |
| 21. | Ceci F, Spaziani E, Corelli S, Casciaro G, Martellucci A, Costantino A, Napoleoni A, Cipriani B, Nicodemi S, Di Grazia C, Avallone M, Orsini S, Tudisco A, Aiuti F, Stagnitti F. Technique and outcomes about a new laparoscopic procedure: the Pelvic Organ Prolapse Suspension (POPS). G Chir. 2013;34:141-144. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 22. | Sah S, Mehta R, Bushi G, Balaraman AK, Pandey S, Brar M, Simiyu BW, Verma A. Laparoscopic pelvic organ prolapse suspension vs. ventral mesh rectopexy in rectal prolapse. Int J Surg Open. 2024;62:838-839. [DOI] [Full Text] |
| 23. | Lukacz ES, Sridhar A, Chermansky CJ, Rahn DD, Harvie HS, Gantz MG, Varner RE, Korbly NB, Mazloomdoost D; Eunice Kennedy Shriver National Institute of Child Health and Human Development Pelvic Floor Disorders Network (PFDN). Sexual Activity and Dyspareunia 1 Year After Surgical Repair of Pelvic Organ Prolapse. Obstet Gynecol. 2020;136:492-500. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 24. | Altomare DF, Binda G, Ganio E, De Nardi P, Giamundo P, Pescatori M; Rectal Prolapse Study Group. Long-term outcome of Altemeier's procedure for rectal prolapse. Dis Colon Rectum. 2009;52:698-703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 59] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 25. | Sipahi M, Arslan E, Börekçi H, Aytekin FÖ, Külah B, Banlı O. Perineal rectosigmoidectomy for incarcerated rectal prolapse (Altemeier's procedure). Ulus Cerrahi Derg. 2016;32:217-220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 26. | Lee S, Kye BH, Kim HJ, Cho HM, Kim JG. Delorme's Procedure for Complete Rectal Prolapse: Does It Still Have It's Own Role? J Korean Soc Coloproctol. 2012;28:13-18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 27. | Park SY, Cho SB, Park CH, Joo JK, Joo YE, Kim HS, Choi SK, Rew JS. Surgical correction is ineffective for improvement of dyssynergic defecation in patients with rectal prolapse. J Neurogastroenterol Motil. 2013;19:85-89. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 28. | Mellgren A, Zafar MH. Obstructed defecation syndrome. Ann Laparosc Endosc Surg. 2022;7:18-18. [DOI] [Full Text] |
| 29. | Burns JB, Haraki AE, Crawford J, Parker-Autry CY. Fecal Incontinence Outcomes Following Transvaginal Posterior Vaginal Wall Repair. Int Urogynecol J. 2025;36:1061-1066. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 30. | De Robles MS, Young CJ. Transperineal rectocele repair is ideal for patients presenting with fecal incontinence. Ann Coloproctol. 2022;38:376-379. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 31. | Martellucci J, Annicchiarico A, Scheiterle M, Trompetto M, Prosperi P. Sacral Neuromodulation for defecation disorders after non oncologic pelvic surgery. Int J Colorectal Dis. 2023;39:2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 32. | Renzi A, Brillantino A, Di Sarno G, d'Aniello F, Bianco P, Iacobellis F, Reginelli A, Grassi R. Transverse Perineal Support: A Novel Surgical Treatment for Perineal Descent in Patients With Obstructed Defecation Syndrome. Dis Colon Rectum. 2016;59:557-564. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 21] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 33. | Gervasi MC, Brancato G, Crepaz L, Tfaily A, Di Leo A. Laparoscopic ventral mesh rectopexy with and without transverse perineal support using biological mesh for rectal prolapse and perineal descent: postoperative course and functional outcomes. Ann Coloproctol. 2025;41:453-461. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 34. | Piloni V, Tosi P, Vernelli M. MR-defecography in obstructed defecation syndrome (ODS): technique, diagnostic criteria and grading. Tech Coloproctol. 2013;17:501-510. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 30] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 35. | Belilos EA, Post Z, Anderson S, DeMeo M. The Clinical Utility of Anorectal Manometry: A Review of Current Practices. Gastro Hep Adv. 2025;4:100562. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 36. | Ortengren AR, Ramkissoon RA, Chey WD, Baker JR, Staller K, Iturrino J, Shah ED. Anorectal manometry to diagnose dyssynergic defecation: Systematic review and meta-analysis of diagnostic test accuracy. Neurogastroenterol Motil. 2021;33:e14137. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 37. | Perek A, Ergün S. Obstructed Defecation Syndrome. Turk J Colorectal Dis. 2021;32:88-98. [DOI] [Full Text] |
| 38. | Laitakari KE, Mäkelä-Kaikkonen JK, Kössi J, Kairaluoma M, Koivurova S, Pollari L, Ohtonen P, Rautio TT. Mid-term functional and quality of life outcomes of robotic and laparoscopic ventral mesh rectopexy: multicenter comparative matched-pair analyses. Tech Coloproctol. 2022;26:253-260. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 39. | Faried M, El Nakeeb A, Youssef M, Omar W, El Monem HA. Comparative study between surgical and non-surgical treatment of anismus in patients with symptoms of obstructed defecation: a prospective randomized study. J Gastrointest Surg. 2010;14:1235-1243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 31] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 40. | Ashoush F, Abdelrahim A, Ali O, Kamali D, Harrison S, Reddy A, Elshazly W, Sultan M, Saafan T, Abounozha S, Ahmed M. A Randomized Controlled Trial Comparing the Efficacy of Bilateral Percutaneous Tibial Nerve Stimulation Versus Biofeedback Pelvic Floor Muscle Training in the Management of Obstructed Defecation Syndrome. Cureus. 2025;17:e80885. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 41. | Hidaka J, Elfeki H, Duelund-Jakobsen J, Laurberg S, Lundby L. Functional Outcome after Laparoscopic Posterior Sutured Rectopexy Versus Ventral Mesh Rectopexy for Rectal Prolapse: Six-year Follow-up of a Double-blind, Randomized Single-center Study. EClinicalMedicine. 2019;16:18-22. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 37] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 42. | Solari S, Martellucci J, Ascanelli S, Sturiale A, Annicchiarico A, Fabiani B, Prosperi P, Carcoforo P, Naldini G. Predictive factors for functional failure of ventral mesh rectopexy in the treatment of rectal prolapse and obstructed defecation. Tech Coloproctol. 2022;26:973-979. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 43. | Mandovra P, Kalikar V, Patankar R. Clinical outcomes of stapled transanal rectal resection (STARR) for obstructed defecation syndrome: Is STARR really a star on long-term follow-up? World J Colorectal Surg. 2020;9:45. [DOI] [Full Text] |
| 44. | Barra M, Trilling B, Mastronicola G, Sage PY, Roudier A, Foote A, Tidadini F, Fournier J, Faucheron JL. Long-term outcome of laparoscopic ventral rectopexy for full-thickness rectal prolapse: the PEXITY study. Tech Coloproctol. 2025;29:68. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 45. | Mäkelä-Kaikkonen J, Rautio T, Kairaluoma M, Carpelan-Holmström M, Kössi J, Rautio A, Ohtonen P, Mäkelä J. Does Ventral Rectopexy Improve Pelvic Floor Function in the Long Term? Dis Colon Rectum. 2018;61:230-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 24] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 46. | Gagliardi G, Pescatori M, Altomare DF, Binda GA, Bottini C, Dodi G, Filingeri V, Milito G, Rinaldi M, Romano G, Spazzafumo L, Trompetto M; Italian Society of Colo-Rectal Surgery (SICCR). Results, outcome predictors, and complications after stapled transanal rectal resection for obstructed defecation. Dis Colon Rectum. 2008;51:186-95; discussion 195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 127] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 47. | Hess GF, Nocera F, Taha-Mehlitz S, Christen S, von Strauss Und Torney M, Steinemann DC. Mesh-associated complications in minimally invasive ventral mesh rectopexy: a systematic review. Surg Endosc. 2024;38:7073-7082. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 48. | Mandovra P, Kalikar V, Patankar RV. Laparoscopic ventral mesh rectopexy for obstructive defecation syndrome: Follow-up in the Indian population. J Minim Access Surg. 2021;17:305-310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/