Published online Feb 27, 2026. doi: 10.4240/wjgs.v18.i2.115978
Revised: November 27, 2025
Accepted: January 7, 2026
Published online: February 27, 2026
Processing time: 119 Days and 5.2 Hours
The optimal anastomotic technique for total laparoscopic right hemicolectomy remains a subject of debate. Novel intracorporeal approaches may help overcome current challenges.
To compare the efficacy of improved π anastomosis and overlap anastomosis.
A retrospective analysis was conducted on 110 colon cancer patients who underwent total laparoscopic radical right hemicolectomy at the Department of Gastrointestinal Surgery, Suzhou Ninth People’s Hospital, from January 2022 to May 2024. All patients underwent intracorporeal reconstruction, with 42 patients receiving improved π anastomosis and 68 patients undergoing overlap anasto
No significant differences were observed between the two groups in terms of operative time, number of lymph nodes dissected, or postoperative hospital stay (P > 0.05). However, the anastomosis time was significantly shorter in the im
The improved π anastomosis in total laparoscopic radical right hemicolectomy ensures high safety and favorable short-term outcomes. It offers a shorter learning curve and quicker proficiency compared to overlap anastomosis, reducing operative times and technical demands on surgeons.
Core Tip: A retrospective analysis was conducted on 110 colon cancer patients who underwent total laparoscopic radical right hemicolectomy. This study compares the efficacy of improved π anastomosis and overlap anastomosis. No significant differences were observed between the two groups in terms of operative time, number of lymph nodes dissected, or postoperative hospital stay, however, the anastomosis time was significantly shorter in the improved π anastomosis group. The learning curve analysis shows that improved π anastomosis achieves proficiency earlier than overlap anastomosis.
- Citation: Wu JZ, Liu PF, Gu ZG, Zhang Z, Zhou ZY, Shen T. Application of improved π anastomosis in totally laparoscopic radical right hemicolectomy. World J Gastrointest Surg 2026; 18(2): 115978
- URL: https://www.wjgnet.com/1948-9366/full/v18/i2/115978.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i2.115978
Right-sided colon cancer constitutes approximately 40% of all cases of colon cancer[1]. The complexity of its anatomy, particularly variations in “Henle’s trunk”, increases the surgical challenges of laparoscopic right hemicolectomy. Unlike sigmoid colectomy or anterior rectal resection, right hemicolectomy requires varied approaches for access, lymphadenectomy and anastomosis, with no standardized protocol. While routinely performed in high-volume centers, laparoscopic right hemicolectomy often necessitates an auxiliary incision for extraluminal ileocolic anastomosis. In obese patients, thickened abdominal walls and shortened mesentery further complicate the procedure, increasing the risk of venous bleeding or mesenteric injury[2].
Studies have shown that intracorporeal anastomosis offers more advantages compared to extracorporeal anastomosis, including faster recovery of bowel function, reduced postoperative pain, and a lower incidence of abdominal incisional hernias[3,4]. The two main intracorporeal anastomotic methods in laparoscopic right hemicolectomy are isoperistaltic (overlap) anastomosis and anti-peristaltic functional end-to-end (π) anastomosis[5-7]. Leveraging extensive experience in laparoscopic-assisted surgery, we proposed and refined the improved π anastomosis technique to optimize intracorporeal ileocolic anastomosis in radical laparoscopic right hemicolectomy.
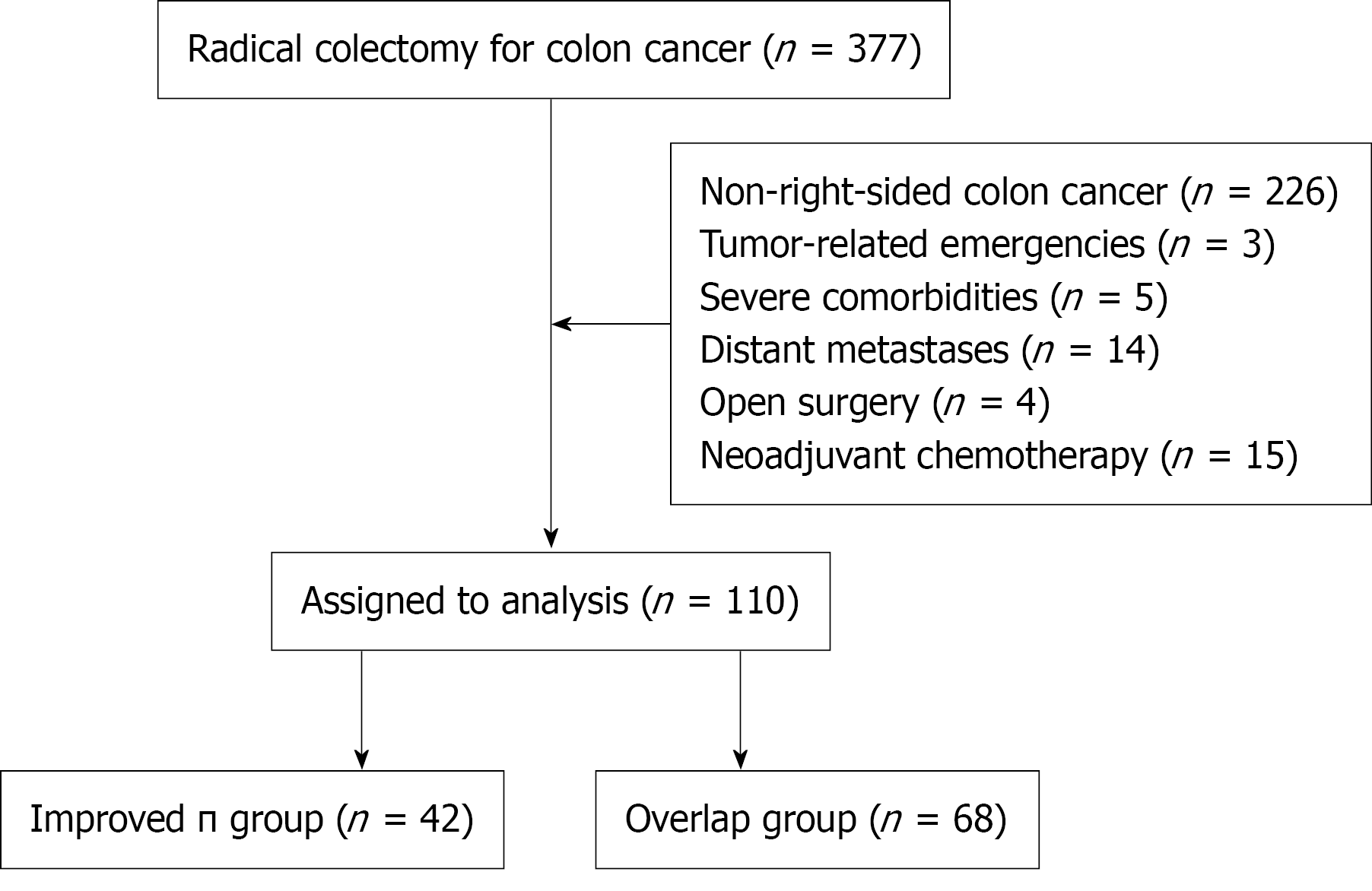
A retrospective analysis was performed on the clinical data of 377 colon cancer patients who underwent radical colectomy at the Department of Gastrointestinal Surgery, Suzhou Ninth People’s Hospital, from January 2022 to May 2024. The inclusion criteria were as follows: (1) Preoperative colonoscopy and biopsy confirmed colonic adenocarcinoma, with tumors located in the ileocecal region, ascending colon, or transverse colon near the hepatic flexure; (2) No tumor-related emergencies (e.g., obstruction, bleeding, or perforation) and completion of preoperative bowel preparation; and (3) No severe underlying diseases. The exclusion criteria were as follows: (1) Evidence of distant metastases in the liver, lungs, brain, bones, or other sites; (2) Open surgery; and (3) Preoperative neoadjuvant chemotherapy. A total of 110 patients were included, all of whom underwent intracorporeal reconstruction by the same surgeon. Among them, 42 patients received improved π anastomosis, and 68 patients underwent overlap anastomosis (Figure 1). The study protocol conformed to the Declaration of Helsinki and was approved by the Ethics Committee of the Suzhou Ninth People’s Hospital.
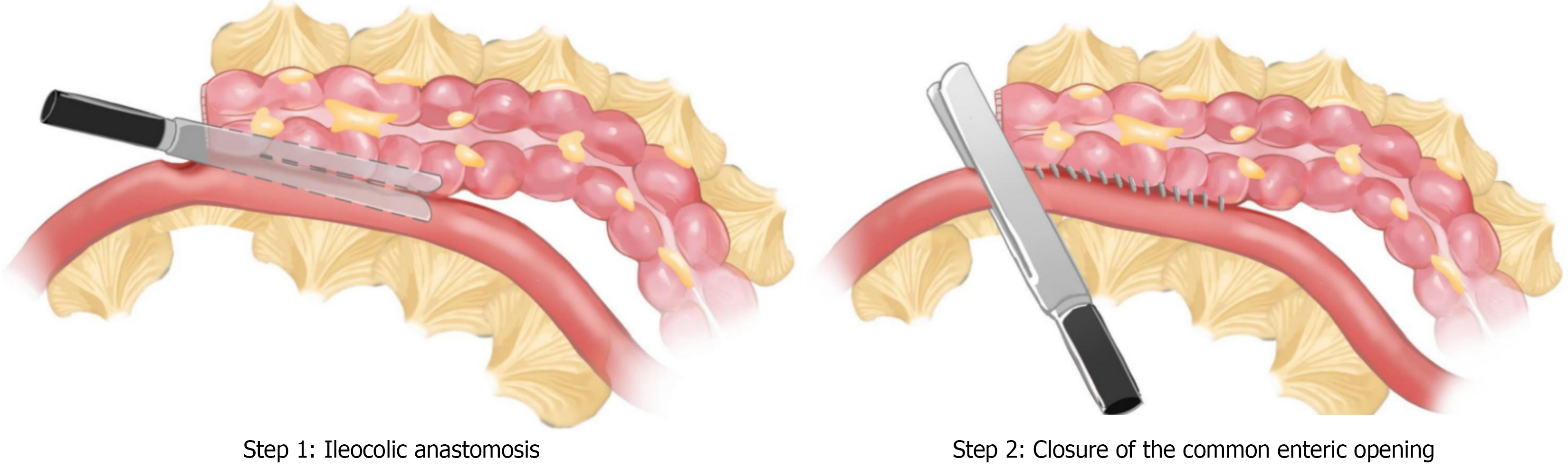
After anesthesia was successfully administered, the patient was positioned in the supine split-leg position, and pneumoperitoneum was established via a needle inserted through the umbilicus, maintaining a pressure of 14 mmHg. A 10 mm trocar was inserted approximately 4 cm below the umbilicus as the observation hole. A 12 mm trocar was placed 4 cm below the left midclavicular line as the primary operating port, while 5 mm trocars were positioned 2 cm below the umbilicus on the left and right midaxillary lines and 3 cm below the right midaxillary line at the rib margin. The procedures for improved π anastomosis are detailed below and illustrated schematically in Figure 2.
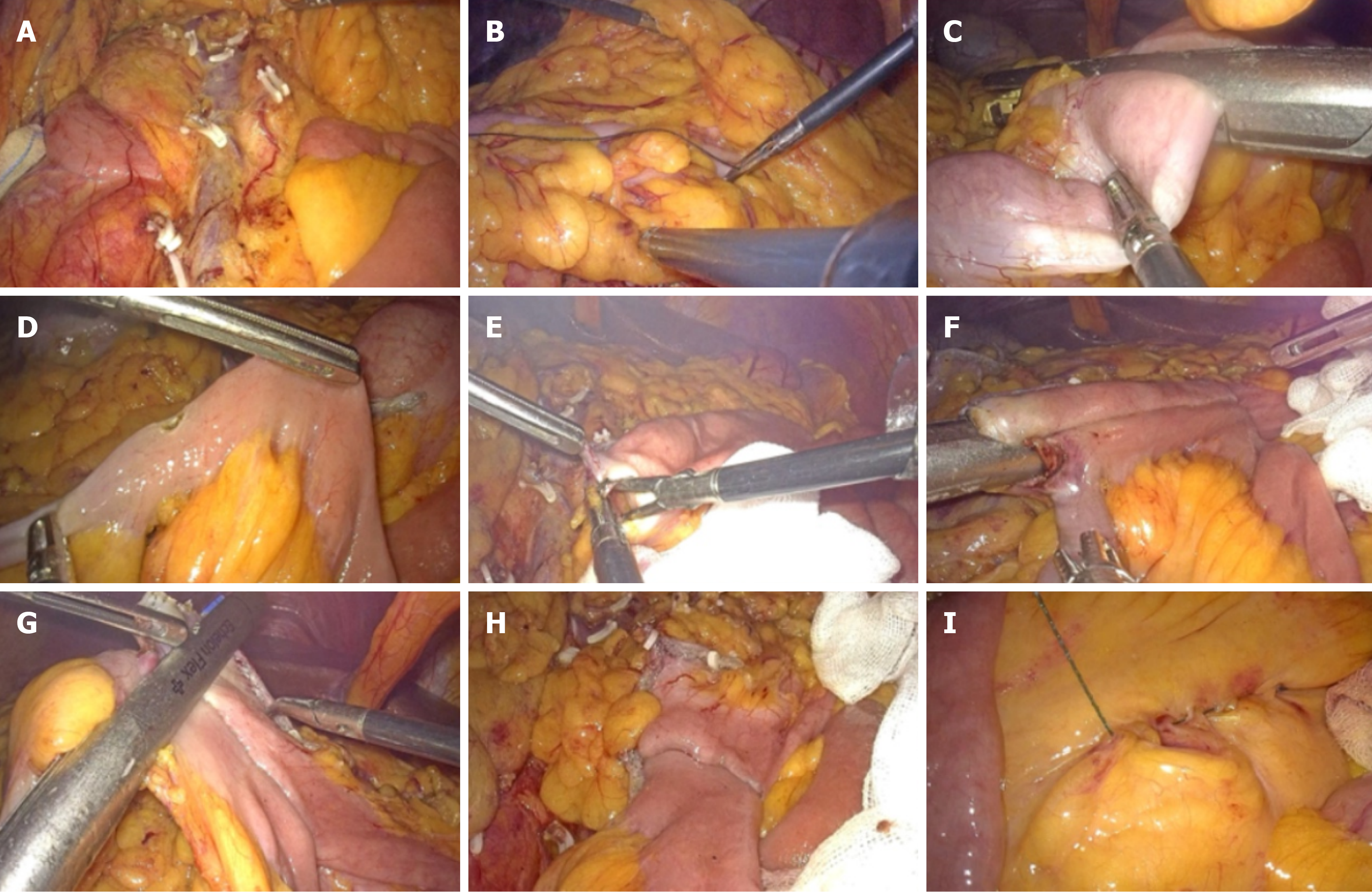
For the improved π group, the abdominal cavity was routinely explored (Figure 3), and the caudal-to-intermediate approach was employed to dissect regional lymph nodes and adipose tissue, completing the dissociation of the right colon (Figure 3A). The ileal mesentery, 15 cm proximal to the ileocecal region, and the mesentery at the predetermined transverse colon resection site were incised. The anastomosis between the ileum and transverse colon was performed using a three-step method: Step 1: The transverse colon was transected using a Johnson linear cutting and stapling device (Figure 3C). Step 2: The ileum and transverse colon were aligned against the direction of peristalsis. A 0.5 cm incision was made on the anti-mesenteric side of the transverse colon stump, while a 0.5 cm incision was made at the anti-mesenteric edge of the predetermined ileal transection line using electrocoagulation (Figure 3D and E). The jaws of the linear cutter were positioned within the ileum and transverse colon, respectively, to create a side-to-side ileocolonic anastomosis (Figure 3F). Step 3: The common opening of the ileocolonic anastomosis was closed with a linear cutting stapler, which simultaneously transected the ileum, completing the anastomosis (Figure 3G and H). The anastomosis was inspected for bleeding and stenosis, and the mesenteric defect was closed (Figure 3I).
For the overlap group, the abdominal cavity was routinely explored, and the caudal-to-intermediate approach was used to dissect regional lymph nodes and adipose tissue, completing the mobilization of the right colon. The ileal mesentery, 15 cm proximal to the ileocecal region, and the mesentery at the predetermined transverse colon resection site were incised. Step 1: The transverse colon and ileum were transected using a Johnson linear cutting and stapling device. Step 2: The ileum and transverse colon were aligned along the direction of peristalsis. A 0.5 cm incision was made on the anti-mesenteric side, 6 cm from the transverse colon stump, and another 0.5 cm incision was made along the anti-mesenteric edge of the ileal transection line. The jaws of the linear cutter were positioned within the ileum and transverse colon to complete the side-to-side ileocolonic anastomosis. Step 3: The common opening of the ileocolonic anastomosis was closed using barbed sutures. The mesenteric defect was subsequently closed.
Finally, the abdominal cavity was inspected for bleeding and a drainage tube was placed. The specimen was extracted through the umbilical incision, completing the procedure. (The following are the manufacturers and specifications of key intraoperative instruments: Stapler: Johnson and Johnson Echelon Flex™ powered stapler; shaft length: 340 mm; suture line length: 60 mm. Staple Cartridge: Ethicon Endo-Surgery, LLC; model: GST60W/B/D; length: 60 mm; staple height: 2.6/3.6/3.8 mm. Barbed Suture: COVIDEN V-Loc™ 180 non-absorbable suture; size: 3-0; length: 15 cm; radius: 26 mm; 2 Metric.)
Patients’ baseline data included age, gender, body mass index (BMI), American Society of Anesthesiologists (ASA) score, tumor diameter, tumor location, and clinical stage. Surgical parameters recorded were operative time (minute), anastomotic time (minute), blood loss (mL), time to first flatus (hour), time to first oral intake (hour), hospital stay (days), postoperative hospital stay (days), anastomotic complications (leakage, stenosis, bleeding), abdominal abscess, bowel obstruction, and other complications (pneumonia, wound infection, urinary tract infection). Additional records included Clavien-Dindo classification of complications and bowel frequency (times/day). Postoperative pathological data encompassed lymph nodes dissected, distal resection margin (cm), positive resection margins, and pathological tumor, node, and metastasis stage.
BMI was calculated as weight (kg) divided by height (m) squared. ASA scores were assessed using the American Society of Anesthesiologists grading system, and tumor location was classified per the World Health Organization tumor classification system[8]. Clinical and pathological staging followed the 8th edition of the American Joint Committee on Cancer Staging Manual. Complications were graded using the Clavien-Dindo criteria[9]. Bowel frequency was assessed through outpatient or telephone follow-up, calculated as the average number of daily bowel movements within the first three months postoperatively. All patients underwent standardized postoperative follow-up, scheduled every 3-6 months during the first two years after surgery, with a minimum follow-up period of 6 months.
In the comparison of baseline characteristics, postoperative recovery, complications, and pathological information between the improved π group and the overlap group, normally distributed continuous variables were expressed as the mean ± SD and were analyzed using the independent samples t-test or one-way analysis of variance for between-group comparisons; non-normally distributed continuous variables were expressed as the median (interquartile range) and analyzed using the Mann-Whitney U test. Categorical and hierarchical variables were expressed as n (%) and analyzed using the χ2 test; when cell frequencies were < 5, the Fisher exact test was used. Two-sided P values were reported for all analyses, and P < 0.05 was considered statistically significant. Linear regression was used to analyze trends in operative times and to calculate R2 values, and cumulative sum (CUSUM) analysis identified proficiency inflection points. All statistical analyses were completed using SPSS software (version 17.0, IBM Corp., Armonk, NY, United States) and R software (version 4.4.1).
Table 1 shows the preoperative demographic characteristics of the patients. The median age was 59 years in the improved π group (n = 42) and 56 years in the overlap group (n = 68). Both groups had a higher proportion of male patients (59.5% vs 54.4%), and the most common tumor location was the ascending colon (61.9% vs 66.2%). The majority of patients in both groups were classified as clinical tumor stage II (73.8% vs 66.2%). No statistically significant differences (P > 0.05) were observed between the two groups in terms of gender, age, BMI (kg/m2), ASA score, tumor diameter, tumor location, or clinical tumor, node, and metastasis stage. Therefore, perioperative data comparisons can be performed directly without requiring propensity score matching.
| Characteristic | Improved π group (n = 42) | Overlap group (n = 68) | P value |
| Age (years) | 59 (49-66) | 56 (43-70) | 0.137 |
| Sex | 0.599 | ||
| Male | 25 (59.5) | 37 (54.4) | |
| Female | 17 (40.5) | 31 (45.6) | |
| BMI (kg/m2), mean ± SD | 24.74 ± 7.03 | 25.22 ± 6.88 | 0.665 |
| ASA score | 0.514 | ||
| I | 2 (4.8) | 7 (10.3) | |
| II | 29 (69.0) | 47 (69.1) | |
| III | 11 (26.2) | 14 (20.6) | |
| Tumor size (cm), mean ± SD | 3.17 ± 1.24 | 3.09 ± 2.85 | 0.300 |
| Tumor location | 0.726 | ||
| Cecum | 12 (28.6) | 15 (22.0) | |
| Ascending colon | 26 (61.9) | 45 (66.2) | |
| Transverse colon | 4 (9.5) | 8 (11.8) | |
| cTNM stage | 0.589 | ||
| I | 4 (9.5) | 6 (8.8) | |
| II | 31 (73.8) | 45 (66.2) | |
| III | 7 (16.7) | 17 (25.0) | |
Table 2 summarizes the intraoperative details, postoperative bowel recovery, and complications. While there was no significant difference in operative time or blood loss between the two groups, the anastomosis time in the improved π anastomosis group was significantly shorter than that in the overlap anastomosis group (13.54 ± 4.47 minutes vs 21.69 ± 8.51 minutes, P < 0.001). Although the time to the first postoperative anal flatus was longer in the improved π anastomosis group compared to the overlap anastomosis group (68.64 ± 9.31 hours vs 59.07 ± 9.67 hours, P < 0.05), there was no statistically significant difference between the two groups in terms of postoperative hospital stay (9.00 ± 2.04 days vs 8.69 ± 1.92 days, P > 0.05). Neither group experienced anastomotic leakage or stenosis; however, one patient in the overlap group developed anastomotic bleeding, which was successfully treated with endoscopic hemostasis. The overall complication rates and non-gastrointestinal complications showed no significant differences between the two groups. At the 3-month postoperative follow-up, the mean daily bowel movement frequency was significantly lower in the improved π group compared to the overlap group (3.11 times/day vs 4.43 times/day, P = 0.017).
| Characteristic | Improved π group (n = 42) | Overlap group (n = 68) | P value |
| Operative time (minute), mean ± SD | 173.44 ± 60.01 | 181.72 ± 65.40 | 0.177 |
| Anastomotic time (minute), mean ± SD | 13.54 ± 4.47 | 21.69 ± 8.51 | < 0.001 |
| Blood loss (mL), mean ± SD | 30.47 ± 6.12 | 28.18 ± 9.07 | 0.133 |
| First time to flatus (hour), mean ± SD | 68.64 ± 9.31 | 59.07 ± 9.67 | 0.049 |
| Time to first oral intake (hour), mean ± SD | 112.62 ± 23.01 | 108.18 ± 26.58 | 0.442 |
| Hospital stay (day), mean ± SD | 13.03 ± 4.44 | 12.59 ± 3.91 | 0.425 |
| Postoperative hospital stay (day), mean ± SD | 9.00 ± 2.04 | 8.69 ± 1.92 | 0.169 |
| Anastomotic complications | 0 | 1 (1.5) | 1.000 |
| Leakage | 0 | 0 | - |
| Stenosis | 0 | 0 | - |
| Bleeding | 0 | 1 | - |
| Abdominal abscess | 0 | 0 | - |
| Bowel obstruction | 0 | 0 | - |
| Other complications | 5 (11.9) | 8 (11.8) | 0.982 |
| Pneumonia | 2 (40.0) | 2 (25.0) | - |
| Wound infection | 2 (40.0) | 2 (25.0) | - |
| Urinary infection | 1 (20.0) | 4 (50.0) | - |
| Clavien-Dindo classification | 3 (7.1) | 8(11.8) | 0.647 |
| I | 0 | 4 (50.0) | - |
| II | 3 (100) | 3 (37.5) | - |
| III | 0 | 1 (12.5) | - |
| Bowel frequency (times/day) | 3.11 ± 0.56 | 4.43 ± 1.71 | 0.017 |
Table 3 presents the pathological information of the two groups. There were no statistically significant differences in the number of lymph nodes dissected, distal margin distance, rate of positive margins, or pathological staging between the groups (P > 0.05).
| Characteristic | Improved π group (n = 42) | Overlap group (n = 68) | P value |
| Lymph nodes dissected | 25.83 ± 5.65 | 26.02 ± 4.51 | 0.750 |
| Distal resection margin (cm) | 8.18 (6.01-14.28) | 8.93 (6.77-15.30) | 0.524 |
| Positive resection margin cases | 0 | 0 | - |
| pTNM stage, n (%) | 0.790 | ||
| I | 4 (9.5) | 5 (7.4) | |
| II | 29 (69.0) | 51 (75.0) | |
| III | 9 (21.4) | 12 (17.6) | |
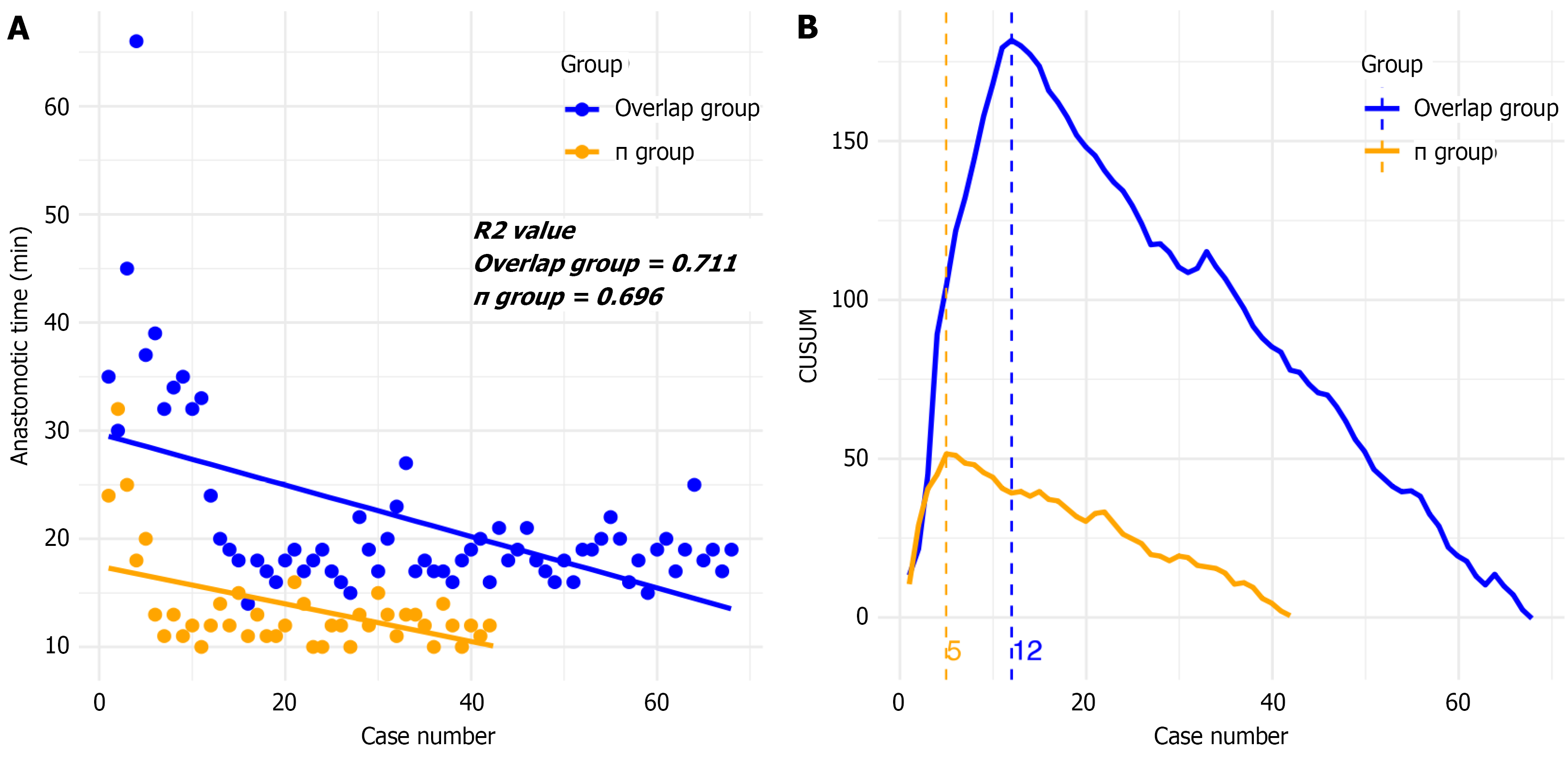
The learning curve analysis is shown in Figure 4. The scatter plot demonstrates that operative times for both improved π anastomosis and overlap anastomosis decreased with increasing case numbers, reflecting the learning effect. The linear trend showed good fit for both procedures, with R2 values of 0.696 for the improved π group and 0.711 for the overlap group. Operative times for improved π anastomosis were overall shorter than those for overlap anastomosis throughout the learning curve. The CUSUM plot identified inflection points at the 5th case for improved π anastomosis and the 12th case for overlap anastomosis, indicating that proficiency is achieved earlier with improved π anastomosis. These findings suggest that improved π anastomosis has a shorter learning curve, reducing the technical demands on surgeons and facilitating quicker mastery of the procedure.
Recent advances in surgical instruments, particularly linear staplers and barbed sutures, have significantly accelerated the development of intracorporeal anastomosis in laparoscopic surgery. Studies have highlighted its advantages in laparoscopic right hemicolectomy, demonstrating favorable short-term outcomes. Compared to extracorporeal anastomosis, intracorporeal anastomosis reduces intestinal and mesenteric traction while minimizing air exposure, promoting faster postoperative recovery[10]. It eliminates the need for bowel externalization, lowering the risk of venous bleeding and mesenteric traction injuries, thereby reducing intraoperative blood loss and the incidence of mesenteric venous and portal vein thrombosis[11]. Additionally, it allows flexible specimen extraction through various sites, such as a lower abdominal transverse incision, rectum, or vagina[12].
A comparative study[2,13] found that the distal resection margin was significantly longer in the intracorporeal anastomosis group than in the extracorporeal group. In the latter, an additional abdominal incision was required to externalize the intestine for resection and anastomosis. Difficulty in externalization, especially in patients with thicker abdominal walls or shorter mesenteries, may result in a shorter distal margin. In contrast, intracorporeal anastomosis is unaffected by body habitus, BMI, or mesenteric length, ensuring greater procedural consistency. Additionally, a 10 cm silk suture can be used during intracorporeal anastomosis to precisely measure the colon, ensuring an accurate distal margin while avoiding unnecessary mesenteric dissection and bowel resection (Figure 3B).
Studies suggest that intracorporeal anastomosis may elevate white blood cell counts and C-reactive protein levels, indicating a heightened inflammatory response, possibly due to bacterial contamination from intestinal content leakage into the abdominal cavity[13,14]. Unlike extracorporeal anastomosis, intracorporeal anastomosis poses a risk of spillage, especially in patients with inadequate bowel preparation or obstructive tumors, potentially leading to postoperative abscess formation or even tumor implantation and metastasis. Some studies have found no significant difference in postoperative abdominal implantation and metastasis rates between intracorporeal and extracorporeal anastomosis; however, these studies were limited by small sample sizes and short follow-up periods[2,13]. Preventing intra-abdominal contamination is crucial. No patients in either group developed postoperative infections, likely due to the following precautions: (1) Thorough preoperative bowel preparation; (2) Opting for extra-luminal anastomosis in cases of partial obstruction; (3) Placing gauze around the anastomotic site before opening the lumen (Figure 3E); and (4) Minimizing the time needed to close the common opening. If contamination occurred, the abdominal cavity was irrigated with at least 1000 mL of saline until the effluent was clear.
Manual end-to-end anastomosis for total laparoscopic reconstruction in colon cancer has been reported but remains technically demanding, limiting its adoption[15]. Overlap and π anastomosis are the main intracorporeal reconstruction methods, with linear staplers significantly reducing operative time and simplifying the procedure compared to manual anastomosis[16]. However, both techniques involve two stumps and a common opening, increasing wound size and risks of anastomotic leakage and bleeding. Closing the common opening also requires additional staplers or barbed sutures, raising costs. Jian-Cheng et al[17] reported that improved overlap anastomosis is straightforward, reducing the ileal blind end effect, though it may increase the risk of anastomotic stenosis and requires careful technique. π anastomosis necessitates extra mesenteric mobilization and division to reduce tension, with proper ileocolic alignment before anastomosis. Compared to overlap anastomosis, it involves more intestinal resection, prolonging operative time. Du et al[18] introduced U-tie functional end-to-end anastomosis, reducing stapler use but adding technical complexity, however, it was not suitable for patients with a BMI greater than 28.
We refined π anastomosis by using a linear stapler to close the common opening of the ileocolonic anastomosis while simultaneously transecting the ileum. A total of 42 patients underwent functional end-to-end anastomosis with three stapler cartridges, eliminating the need for manual closure (e.g., with barbed sutures). This modification significantly reduced intracorporeal reconstruction time to approximately 13 minutes, notably shorter than in the overlap group (13.54 ± 4.47 vs 21.69 ± 8.51, P < 0.001). The linear trend of the learning curve demonstrated good model fit, with R2 values of 0.696 for the improved π group and 0.711 for the overlap group (Figure 4A). The CUSUM plot (Figure 4B) identified inflection points at the 5th case for improved π anastomosis and the 12th case for overlap anastomosis, indicating that proficiency is achieved earlier with improved π anastomosis. These findings suggest that improved π anastomosis has a shorter learning curve, reducing the technical demands on surgeons and facilitating quicker mastery of the procedure. The improved π anastomosis offers several advantages: Elevating the preserved distal ileum in a reverse peristaltic alignment with the transverse colon improves control over the anastomotic angle and reduces the risk of torsion; full exposure of the common opening ensures smooth closure and minimizes stenosis; the three-step technique, using a linear stapler for ileocolic side-to-side anastomosis, simultaneous common opening closure, and ileum transection, limits unnecessary bowel resection and reduces operative time; compared to overlap anastomosis, it also lowers the need for additional stapler cartridges or barbed sutures. Previous studies[19,20] reported earlier bowel function recovery with overlap anastomosis, possibly due to peristaltic alignment. In our study, the improved π anastomosis group had a longer postoperative anal exhaust time than the overlap group (68.64 ± 9.31 hours vs 59.07 ± 9.67 hours, P < 0.05), but postoperative hospital stay was similar (9.00 ± 2.04 days vs 8.69 ± 1.92 days, P > 0.05). The absence of the ileocecal valve after right hemicolectomy increases bowel movements; however, reverse peristaltic reconstruction in the improved π anastomosis group resulted in fewer bowel movements in the first three postoperative months compared to the overlap group (3.11 ± 0.56 times/day vs 4.43 ± 1.71 times/day, P < 0.05), potentially improving postoperative quality of life.
Nevertheless, this study has several limitations: The intrinsic selection bias associated with its retrospective design; a limited sample size, which constrains the capacity to perform additional subgroup analyses for postoperative observational metrics; and insufficient follow-up duration, which obstructs intergroup comparisons across varying time points and impedes the capacity to draw more authoritative conclusions.
In conclusion, the improved π anastomosis articulated in this study streamlines the surgical procedure, shortens the duration of anastomosis, minimizes dependence on the operator’s technical expertise, and presents advantages such as reliable outcomes and cost efficiency.
| 1. | Patel M, McSorley ST, Park JH, Roxburgh CSD, Edwards J, Horgan PG, McMillan DC. The relationship between right-sided tumour location, tumour microenvironment, systemic inflammation, adjuvant therapy and survival in patients undergoing surgery for colon and rectal cancer. Br J Cancer. 2018;118:705-712. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 55] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 2. | Sakurai T, Yamaguchi T, Noguchi T, Sakamoto T, Mukai T, Hiyoshi Y, Nagasaki T, Akiyoshi T, Fukunaga Y. Short-term outcomes of intracorporeal versus extracorporeal anastomosis in laparoscopic surgery for right-sided colon cancer: A propensity score-matched study. Asian J Endosc Surg. 2023;16:14-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 3. | Ozawa H, Sakamoto J, Nakanishi H, Fujita S. Short-term outcomes of intracorporeal versus extracorporeal anastomosis after laparoscopic colectomy: a propensity score-matched cohort study from a single institution. Surg Today. 2022;52:616-623. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 4. | Zhang M, Lu Z, Zheng Z, Cheng P, Zhou H, Wang X. Comparison of short-term outcomes between totally laparoscopic right colectomy and laparoscopic-assisted right colectomy: a retrospective study in a single institution on 300 consecutive patients. Surg Endosc. 2022;36:176-184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 5. | Portale G, Cipollari C, Pedon S, Benacchio L, Fiscon V. A novel and simple technique to close the enterotomy after intracorporeal anastomosis in laparoscopic right hemicolectomy. Updates Surg. 2021;73:1435-1442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 6. | Tajima JY, Nagayama S, Hiyoshi Y, Mukai T, Nagasaki T, Yamaguchi T, Akiyoshi T, Konishi T, Fukunaga Y. Colonic delta-shaped anastomosis using linear staplers in laparoscopic colectomy. Tech Coloproctol. 2021;25:473-474. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 7. | Hanna MH, Hwang GS, Phelan MJ, Bui TL, Carmichael JC, Mills SD, Stamos MJ, Pigazzi A. Laparoscopic right hemicolectomy: short- and long-term outcomes of intracorporeal versus extracorporeal anastomosis. Surg Endosc. 2016;30:3933-3942. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 77] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 8. | Nagtegaal ID, Odze RD, Klimstra D, Paradis V, Rugge M, Schirmacher P, Washington KM, Carneiro F, Cree IA; WHO Classification of Tumours Editorial Board. The 2019 WHO classification of tumours of the digestive system. Histopathology. 2020;76:182-188. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3203] [Cited by in RCA: 2836] [Article Influence: 472.7] [Reference Citation Analysis (0)] |
| 9. | Clavien PA, Barkun J, de Oliveira ML, Vauthey JN, Dindo D, Schulick RD, de Santibañes E, Pekolj J, Slankamenac K, Bassi C, Graf R, Vonlanthen R, Padbury R, Cameron JL, Makuuchi M. The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg. 2009;250:187-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9980] [Cited by in RCA: 9257] [Article Influence: 544.5] [Reference Citation Analysis (0)] |
| 10. | Krouchev R, Tamana S, Nguyen N, Yorke E, Harris D, Sampath S. Total intracorporeal anastomosis for right hemicolectomy: Experience from a Canadian center. Am J Surg. 2018;215:905-908. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 11. | Anania G, Santini M, Scagliarini L, Marzetti A, Vedana L, Marino S, Gregorio C, Resta G, Cavallesco G. A totally mini-invasive approach for colorectal laparoscopic surgery. World J Gastroenterol. 2012;18:3869-3874. [PubMed] [DOI] [Full Text] |
| 12. | Li XW, Wang CY, Zhang JJ, Ge Z, Lin XH, Hu JH. Short-term efficacy of transvaginal specimen extraction for right colon cancer based on propensity score matching: A retrospective cohort study. Int J Surg. 2019;72:102-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 30] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 13. | Liao CK, Chern YJ, Lin YC, Hsu YJ, Chiang JM, Tsai WS, Hsieh PS, Hung HY, Yeh CY, You JF. Short- and medium-term outcomes of intracorporeal versus extracorporeal anastomosis in laparoscopic right colectomy: a propensity score-matched study. World J Surg Oncol. 2021;19:6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 14. | Mari GM, Crippa J, Costanzi ATM, Pellegrino R, Siracusa C, Berardi V, Maggioni D. Intracorporeal Anastomosis Reduces Surgical Stress Response in Laparoscopic Right Hemicolectomy: A Prospective Randomized Trial. Surg Laparosc Endosc Percutan Tech. 2018;28:77-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 54] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 15. | Milone M, Elmore U, Allaix ME, Bianchi PP, Biondi A, Boni L, Bracale U, Cassinotti E, Ceccarelli G, Corcione F, Cuccurullo D, Degiuli M, De Manzini N, D'Ugo D, Formisano G, Manigrasso M, Morino M, Palmisano S, Persiani R, Reddavid R, Rondelli F, Velotti N, Rosati R, De Palma GD. Fashioning enterotomy closure after totally laparoscopic ileocolic anastomosis for right colon cancer: a multicenter experience. Surg Endosc. 2020;34:557-563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 33] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 16. | Zhou HT, Wang P, Liang JW, Su H, Zhou ZX. Short-term outcomes of overlapped delta-shaped anastomosis, an innovative intracorporeal anastomosis technique, in totally laparoscopic colectomy for colon cancer. World J Gastroenterol. 2017;23:6726-6732. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 8] [Cited by in RCA: 13] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 17. | Jian-Cheng T, Shu-Sheng W, Bo Z, Jian F, Liang Z. Total laparoscopic right hemicolectomy with 3-step stapled intracorporeal isoperistaltic ileocolic anastomosis for colon cancer: An evaluation of short-term outcomes. Medicine (Baltimore). 2016;95:e5538. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 29] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 18. | Du Q, Zheng Z, Ma Q, Zhong J, Wang Y, Yang L, Zhou Z. A novel U-tied semi-manual anastomosis in totally laparoscopic colectomy. Asian J Surg. 2024;47:990-992. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 19. | Matsuda A, Miyashita M, Matsumoto S, Sakurazawa N, Takahashi G, Yamada M, Uchida E. Isoperistaltic versus antiperistaltic stapled side-to-side anastomosis for colon cancer surgery: a randomized controlled trial. J Surg Res. 2015;196:107-112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 28] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 20. | Ibáñez N, Abrisqueta J, Luján J, Hernández Q, Rufete MD, Parrilla P. Isoperistaltic versus antiperistaltic ileocolic anastomosis. Does it really matter? Results from a randomised clinical trial (ISOVANTI). Surg Endosc. 2019;33:2850-2857. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 27] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/