Published online Feb 27, 2026. doi: 10.4240/wjgs.v18.i2.115830
Revised: November 27, 2025
Accepted: January 7, 2026
Published online: February 27, 2026
Processing time: 123 Days and 1 Hours
Despite the enhanced detection capability of capsule endoscopy for small intes
To investigate the risk factors for small intestinal mucosal injury (SIMI) in non
We conducted a retrospective analysis of clinical data from patients undergoing capsule endoscopy at our institution and Shanxi Bethune Hospital from August 2012 to January 2025. Multivariate analysis was performed to identify indepen
In the primary cohort, SIMI was identified in 114 out of 181 NSAID users. Multivariate analysis revealed that advanced age, smoking, proton pump inhibitor use, elevated body mass index, high triglyceride levels, and increased low-density lipoprotein were independent risk factors for SIMI. The research team developed a risk prediction model for estimating the risk of mucosal injury, which achieved an area under the curve of 0.775 (95% confidence interval: 0.700-0.849) in the derivation set. The model exhibited an area under the curve of 0.797 (95% confidence interval: 0.643-0.952) in the validation cohort.
We identified risk factors for SIMI in NSAID users and established a predictive model, which may facilitate early identification of high-risk populations and guide clinical interventions.
Core Tip: This study identified advanced age, smoking, proton pump inhibitor use, elevated body mass index, high triglyceride, and low-density lipoprotein levels as independent risk factors for small intestinal mucosal injury in nonsteroidal anti-inflammatory drug users. A predictive nomogram was developed based on these factors. The model demonstrated good predictive accuracy, with an area under the curve of 0.775 in the derivation cohort and 0.797 in the validation cohort. This tool can facilitate early identification of high-risk individuals and guide clinical interventions to prevent small intestinal mucosal injury.
- Citation: Zhao NH, Zhao LR, Yao J, Niu LY, Yang LJ, Lin YJ, Liu M. Development and validation of a risk prediction model for small intestinal mucosal injury in nonsteroidal anti-inflammatory drug users. World J Gastrointest Surg 2026; 18(2): 115830
- URL: https://www.wjgnet.com/1948-9366/full/v18/i2/115830.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i2.115830
With the aging population and increasing incidence of cardiovascular and cerebrovascular diseases, nonsteroidal anti-inflammatory drugs (NSAIDs), including aspirin, have become one of the most widely used medications worldwide. NSAIDs are extensively employed for analgesia and anti-inflammatory purposes, while low-dose aspirin is commonly used for the prevention and treatment of cardiovascular events and stroke[1]. It is well-established that NSAID use is associated with an elevated risk of upper gastrointestinal mucosal injury, manifesting as peptic ulcers and/or bleeding[2,3]. Studies indicate that such NSAID-related gastrointestinal complications are not confined to the upper tract - NSAIDs are also recognized as a leading cause of obscure gastrointestinal bleeding, with lesions frequently localized in the small intestine[4].
Unlike upper gastrointestinal injury, which can be promptly assessed via conventional endoscopy, small intestinal mucosal damage remains diagnostically challenging due to its anatomical inaccessibility. Capsule endoscopy is a highly useful and noninvasive method for evaluating small bowel diseases and is considered one of the best modalities for assessing small intestinal mucosal lesions[5-8]. With advancements in capsule endoscopy, small bowel pathology has garnered increasing attention among clinicians.
NSAID-induced small intestinal injury is a clinically significant yet underrecognized condition[9]. The risk factors for NSAID-associated small intestinal mucosal injury (SIMI) remain unclear, and its endoscopic and clinical features are often difficult to delineate accurately due to overlapping pathologies such as inflammatory bowel disease or intestinal tuberculosis. Of particular concern is that NSAID-related small intestinal injury frequently progresses asymptomatically until severe complications arise, including occult bleeding, protein-losing enteropathy, or iron-deficiency anemia[10]. Therefore, early identification of high-risk individuals and precise characterization of injury patterns are crucial for reducing morbidity. Accordingly, this study aimed to develop and validate a risk prediction model for SIMI in NSAID users by identifying key risk factors and characterizing endoscopic features based on capsule endoscopy findings.
This retrospective study reviewed medical records of patients who underwent capsule endoscopy at Tongji Hospital and Shanxi Bethune Hospital between August 2012 and January 2025. Exclusion criteria included: (1) Age < 18 years; (2) Diagnosed inflammatory bowel disease; (3) Intestinal tuberculosis; (4) Henoch-Schönlein purpura; and (5) Incomplete clinical data. We collected comprehensive clinical data (including demographic characteristics, medical history, medication use, and laboratory tests) and capsule endoscopy findings. NSAID users were defined as patients who had received continuous oral NSAID treatment for at least 2 weeks prior to the capsule endoscopy procedure. The Human Investigation Committee of Tongji Medical College, Huazhong University of Science and Technology approved this study, approval No. 2024-S079. Details that might disclose the identity of the patients had been omitted.
The examination was performed using OMOM, PillCam SB 3, or PillCam SB 2 capsule endoscopy systems. Although these systems differ in technical specifications such as image resolution and field of view, they are all clinically validated for the detection of significant mucosal injury. Patients fasted overnight prior to the procedure and underwent bowel preparation with oral polyethylene glycol electrolyte powder. After capsule ingestion, clear liquids were permitted at 2 hours and light meals at 4 hours.
All previous capsule endoscopy records were retrospectively evaluated, with reinterpretation of the original images. Two expert endoscopists, each with experience in over 300 capsule endoscopy interpretations, independently reviewed all recordings. A third senior endoscopist with over 20 years of experience and more than 500 interpretations served as the adjudicator for any discrepancies. All identified lesions were systematically documented, and corresponding images were archived for further analysis.
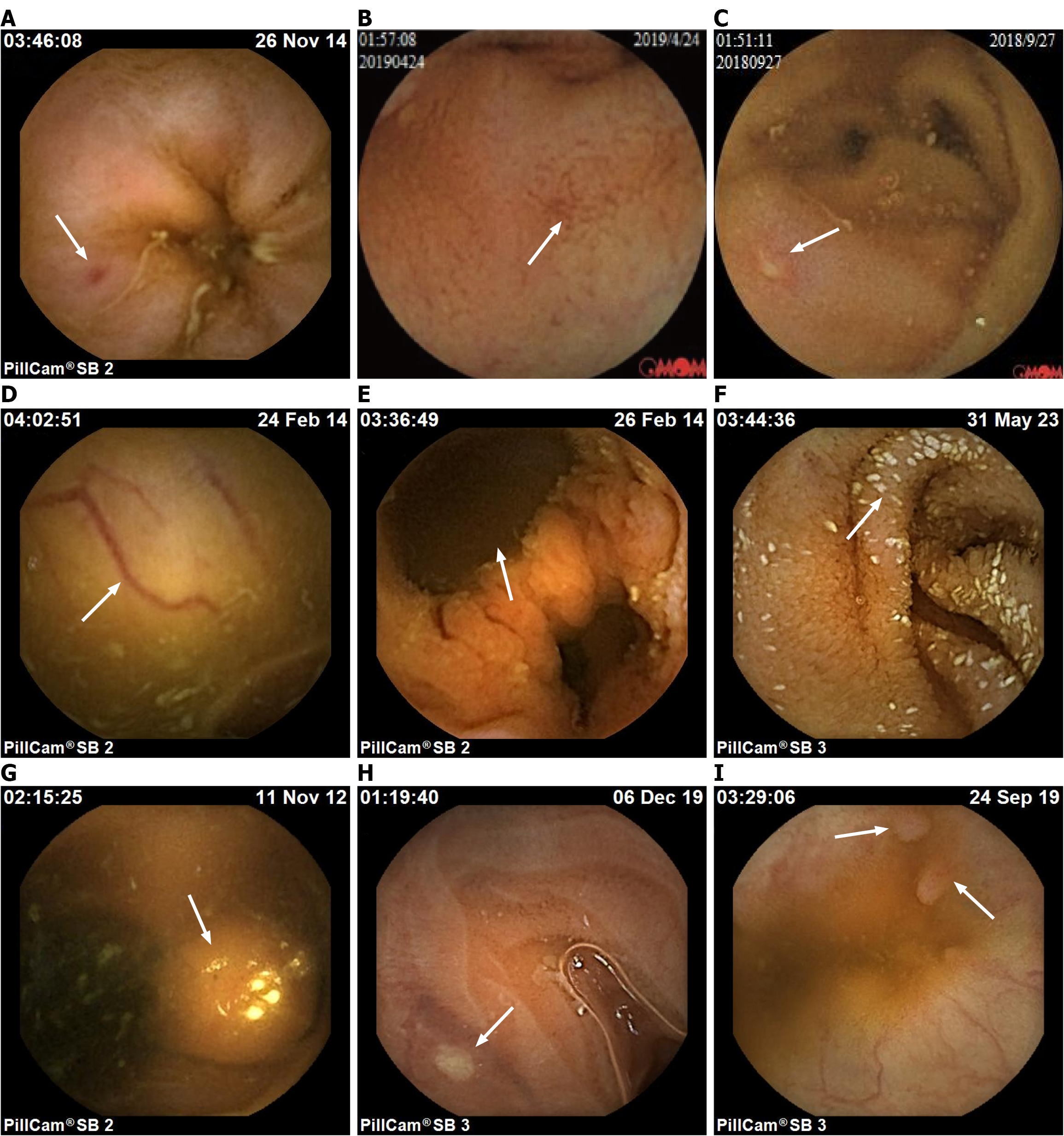
Small intestinal lesions were categorized into seven distinct phenotypes: (1) Erythema: Defined as focal red spots or crimson mucosal areas with preserved villous architecture[11]; (2) Erosions: Discrete lesions featuring central pallor surrounded by congested mucosa with villous loss[12]; (3) Ulcers: Circumscribed mucosal breaks with exudate-covered bases and typically elevated margins[13]; (4) Protrusions: Including inflammatory hyperplasia, lymphoid follicles, stromal tumors, polyps, extrinsic compressions, or indeterminate masses; (5) Vascular malformations; (6) Diverticula: Outpouchings from the small bowel wall; and (7) Lymphangiectasia[7]. Representative images are presented in Figure 1.
Following image review, subjects presenting with erythema, erosions, or ulcers in the small intestine were classified into the SIMI group, while all remaining subjects were assigned to the non mucosal injury group[7].
Categorical variables were expressed as n (%) and compared using the χ² test or Fisher’s exact test, as appropriate. Continuous variables were assessed for normality. Normally distributed data are presented as mean ± SD and compared using the independent samples t test, while non-normally distributed data are expressed as median (interquartile range) and compared using the Mann-Whitney U test. Variables with a statistically significant association in the univariate analysis (P < 0.05) were identified as candidates and were all simultaneously entered into the multivariate logistic regression model. Independent influencing factors were identified through multivariate regression, and a nomogram drawn was constructed primarily based on these results. The predictive performance of the nomogram model was evaluated by the area under the curve (AUC), calibration curve, and decision curve analysis. Additionally, internal validation was carried out. The nomogram drawn was generated using R software (version 4.3.3), while all other sta
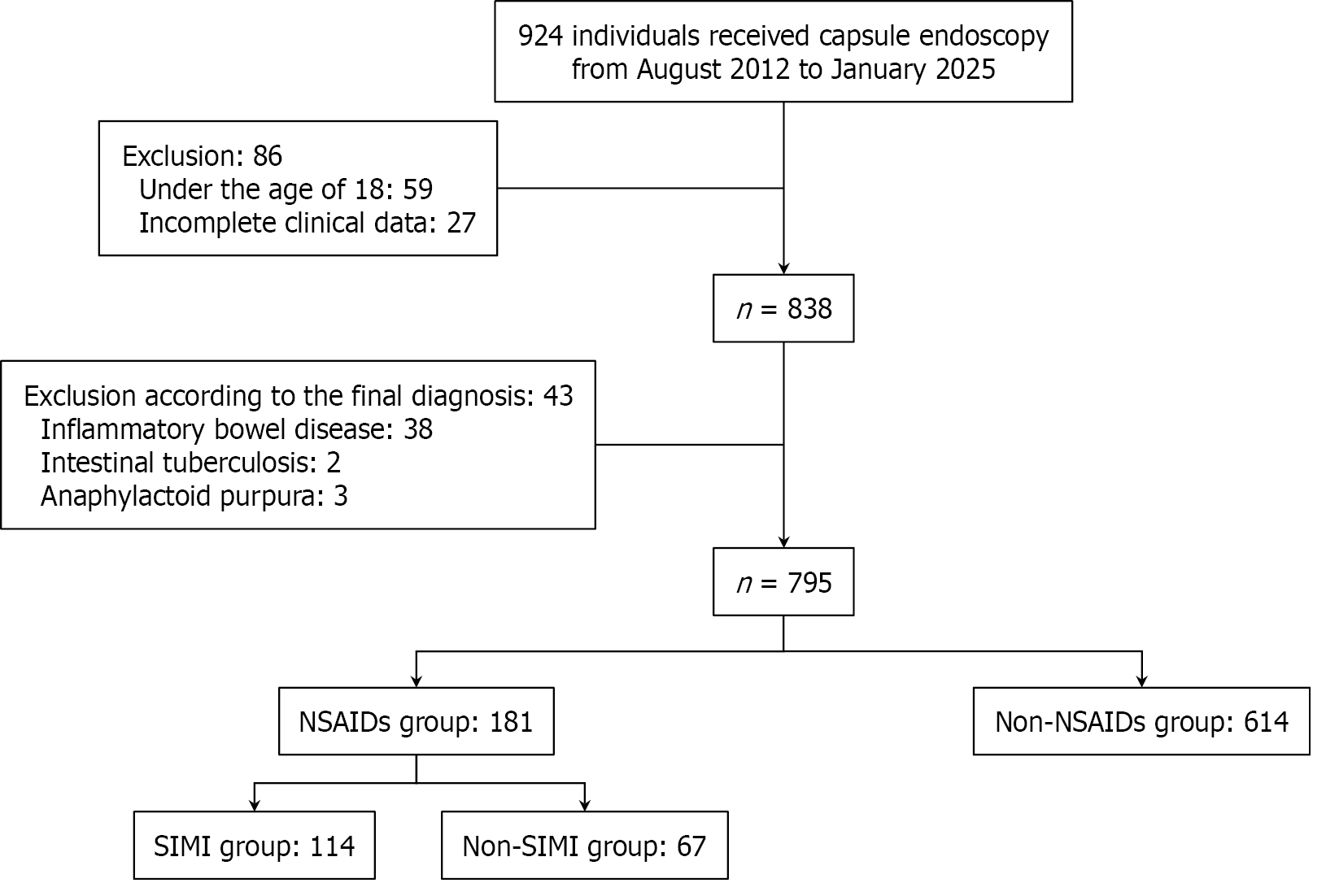
Between August 2012 and January 2025, 924 patients underwent capsule endoscopy at two participating hospitals. After excluding 59 patients aged < 18 years, 27 with incomplete clinical data, 38 with confirmed inflammatory bowel disease, 2 with intestinal tuberculosis, and 3 with anaphylactoid purpura, 795 participants were ultimately included in this study (Figure 2).
The cohort had a median age of 55 years, with 459 males (57.74%) and a mean body mass index (BMI) of 22.87 ± 3.39 kg/m2. Current smokers accounted for 43.65% of participants, while 28.05% reported alcohol consumption. Among the 795 participants, 747 (93.96%) presented with gastrointestinal symptoms - predominantly gastrointestinal bleeding (the most common manifestation), followed by abdominal pain, diarrhea, and bloating. The remaining 48 participants were asymptomatic individuals undergoing routine screening (Table 1). Table 1 also summarizes the endoscopic characteristics of small intestinal lesions. Significant small bowel pathology was detected in 546 patients (68.68%), with erosions and protrusions being the most prevalent findings. The majority of lesions involved both jejunal and ileal segments.
| Variables | n = 795 | NSAID group (n = 181) | Non-NSAID group (n = 614) | P value |
| Sex, male | 459 (57.74) | 108 (59.67) | 351 (57.17) | 0.549 |
| Age (years) | 55.00 (43.00, 64.50) | 60.00 (52.00, 66.00) | 52.50 (39.00, 62.25) | < 0.001 |
| BMI (kg/m2) | 22.87 ± 3.39 | 23.45±2.73 | 22.39 ± 3.80 | 0.003 |
| Smoking habit | 347 (43.65) | 83 (45.86) | 264 (43.00) | 0.495 |
| Alcohol consumption | 223 (28.05) | 55 (30.39) | 168 (27.36) | 0.426 |
| Comorbidity | ||||
| Hypertension | 305 (38.36) | 92 (50.83) | 213 (34.69) | < 0.001 |
| Diabetes mellitus | 111 (13.96) | 29 (16.02) | 82 (13.36) | 0.363 |
| Cardiovascular disease | 153 (19.25) | 106 (58.56) | 47 (7.65) | < 0.001 |
| Stroke | 59 (7.42) | 46 (25.41) | 13 (2.12) | < 0.001 |
| Liver cirrhosis | 15 (1.89) | 3 (1.66) | 12 (1.95) | 0.796 |
| Renal disease | 14 (1.76) | 5 (2.76) | 9 (1.47) | 0.224 |
| Rheumatic/autoimmune disorders | 23 (2.89) | 19 (10.50) | 4 (1.09) | < 0.001 |
| Presenting symptoms | < 0.001 | |||
| Abdominal pain | 191 (24.02) | 28 (15.47) | 163 (26.55) | |
| Diarrhea | 53 (6.67) | 4 (2.21) | 49 (7.98) | |
| Gastrointestinal bleeding | 483 (60.75) | 140 (77.35) | 343 (55.86) | |
| Abdominal distension | 20 (2.52) | 0 (0.00) | 20 (3.26) | |
| Asymptomatic (health check-up) | 48 (6.04) | 9 (4.97) | 39 (6.35) | |
| Hb (g/L) | 97.92 ± 31.31 | 91.05 ± 26.53 | 99.20 ± 32.00 | 0.058 |
| WBC (× 109/L) | 6.04 ± 4.70 | 5.85 ± 1.97 | 6.07 ± 4.98 | 0.788 |
| Alb (g/L) | 39.26 ± 5.46 | 38.70 ± 5.54 | 39.37 ± 5.45 | 0.440 |
| TG (mmol/L) | 1.46 ± 0.52 | 1.50 ± 0.59 | 1.41 ± 0.43 | 0.103 |
| LDL (mmol/L) | 2.74 (1.69, 3.29) | 2.86 (1.88, 3.42) | 2.49 (1.59, 3.22) | 0.190 |
| HDL (mmol/L) | 1.27 (1.07, 1.43) | 1.25 (1.07, 1.42) | 1.29 (1.16, 1.45) | 0.335 |
| Presence of significant small intestinal lesions | 546 (68.68) | 153 (84.53) | 393 (64.01) | < 0.001 |
| Location of lesions | < 0.001 | |||
| Predominantly jejunal | 177 (22.26) | 85 (46.96) | 92 (14.98) | |
| Predominantly ileal | 173 (21.76) | 31 (17.13) | 142 (23.13) | |
| Both jejunal and ileal | 196 (24.65) | 37 (20.44) | 159 (25.90) | |
| Endoscopic manifestations | ||||
| Erythema | 59 (7.42) | 34 (18.78) | 25 (4.07) | < 0.001 |
| Erosions | 209 (26.29) | 87 (48.07) | 122 (19.87) | < 0.001 |
| Ulcers | 90(11.32) | 32 (17.68) | 58 (9.45) | 0.002 |
| Protrusions | 131 (16.48) | 17 (9.39) | 114 (18.57) | 0.003 |
| Vascular malformations | 115 (14.47) | 17 (9.39) | 98 (15.96) | 0.027 |
| Diverticula | 42 (5.28) | 9 (4.97) | 33 (5.37) | 0.832 |
| Lymphangiectasia | 21(2.64) | 7 (3.87) | 14 (2.28) | 0.242 |
For further characterization of NSAID-related effects, patients were stratified by NSAID use into the NSAID group (n = 181) and non-NSAID group (n = 614). Compared with non-NSAID users, NSAID users were significantly older (P < 0.001) and had higher BMI values (P = 0.003). Gastrointestinal bleeding was more prevalent in the NSAID group, whereas abdominal pain, diarrhea, bloating, and asymptomatic cases were more common in the non-NSAID group (P < 0.001). The NSAID group also showed higher rates of hypertension, cardiovascular disease, cerebral infarction, and rheumatic/autoimmune disorders (all P < 0.001). No significant intergroup differences were observed in gender distribution, smoking/alcohol habits, diabetes, liver cirrhosis, renal diseases, white blood cell count, hemoglobin or lipid levels.
The NSAID group demonstrated a significantly higher incidence of small intestinal lesions compared to the non-NSAID group (P < 0.001), with predominant involvement of the jejunum. Erythema, erosions, and ulcers are the primary manifestations of NSAID-associated small intestinal injury. In contrast, the non-NSAID group showed significantly higher rates of protrusions and vascular malformations (Table 1).
The 181 NSAID-exposed patients were stratified into two subgroups based on the presence of SIMI: The NSAID SIMI group (n = 114) and NSAID non mucosal injury group (n = 67). Compared to the non mucosal injury subgroup, patients with SIMI were significantly older (P = 0.009), had higher smoking rates (P = 0.017), elevated BMI (P = 0.011), higher triglyceride (TG) levels (P = 0.007), higher low-density lipoprotein (LDL) levels (P = 0.004), and a greater rate of proton pump inhibitor (PPI) use (P = 0.025). No significant differences were observed between groups regarding gender, alcohol consumption, clinical symptoms, indications for NSAID use, NSAID types or duration of use, leukocyte count, hemo
| Variables | SIMI group (n = 114) | Non-SIMI group (n = 67) | P value |
| Sex, male | 70 (61.40) | 38 (56.72) | 0.535 |
| Age (years) | 60.64 ± 10.22 | 56.29 ± 9.48 | 0.009 |
| Smoking habit | 60 (52.63) | 23 (34.33) | 0.017 |
| Alcohol consumption | 36 (31.58) | 19 (28.36) | 0.649 |
| BMI (kg/m2) | 23.87 ± 2.67 | 22.73 ± 2.72 | 0.011 |
| Presenting symptoms | 0.627 | ||
| Abdominal pain | 15 (13.16) | 13 (19.40) | |
| Diarrhea | 2 (1.75) | 2 (2.99) | |
| Gastrointestinal bleeding | 91 (79.82) | 49 (73.13) | |
| Asymptomatic (health check-up) | 6 (5.26) | 3 (4.48) | |
| Indications for NSAID use | 0.644 | ||
| Cardiovascular disease | 77 (67.54) | 43 (64.18) | |
| Other | 37 (32.46) | 24 (35.82) | |
| NSAID types | 0.404 | ||
| Aspirin | 88 (77.19) | 48 (71.64) | |
| Other NSAIDs | 26 (22.81) | 19 (28.36) | |
| Prolonged use (≥ 3 months) | 96 (84.21) | 54 (80.60) | 0.533 |
| PPI | 69 (60.52) | 29 (43.28) | 0.025 |
| WBC (× 109/L) | 5.83 ± 2.07 | 5.95 ± 1.55 | 0.887 |
| Hb (g/L) | 89.69 ± 24.28 | 98.19 ± 37.49 | 0.412 |
| Alb (g/L) | 38.36 ± 5.50 | 39.73 ± 5.77 | 0.461 |
| TG (mmol/L) | 1.60 ± 0.62 | 1.34 ± 0.49 | 0.007 |
| LDL (mmol/L) | 3.03 (1.95, 3.51) | 2.36 (1.58, 2.97) | 0.004 |
| HDL (mmol/L) | 1.25 (1.07, 1.42) | 1.25 (1.05, 1.44) | 0.802 |
| Characteristic | Univariate analysis | Multivariate analysis | ||
| OR (95%CI) | P value | OR (95%CI) | P value | |
| Age | 1.045 (1.010-1.082) | 0.011 | 1.043 (1.003-1.084) | 0.033 |
| Smoking | 2.126 (1.139-3.968) | 0.018 | 2.352 (1.088-5.086) | 0.030 |
| BMI | 1.174 (1.035-1.331) | 0.013 | 1.176 (1.019-1.358) | 0.026 |
| PPI | 2.009 (1.089-3.705) | 0.025 | 2.099 (1.003-4.390) | 0.049 |
| TG | 2.279 (1.232-4.215) | 0.009 | 2.427 (1.196-4.924) | 0.014 |
| LDL | 1.557 (1.107-2.188) | 0.011 | 1.606 (1.089-2.367) | 0.017 |
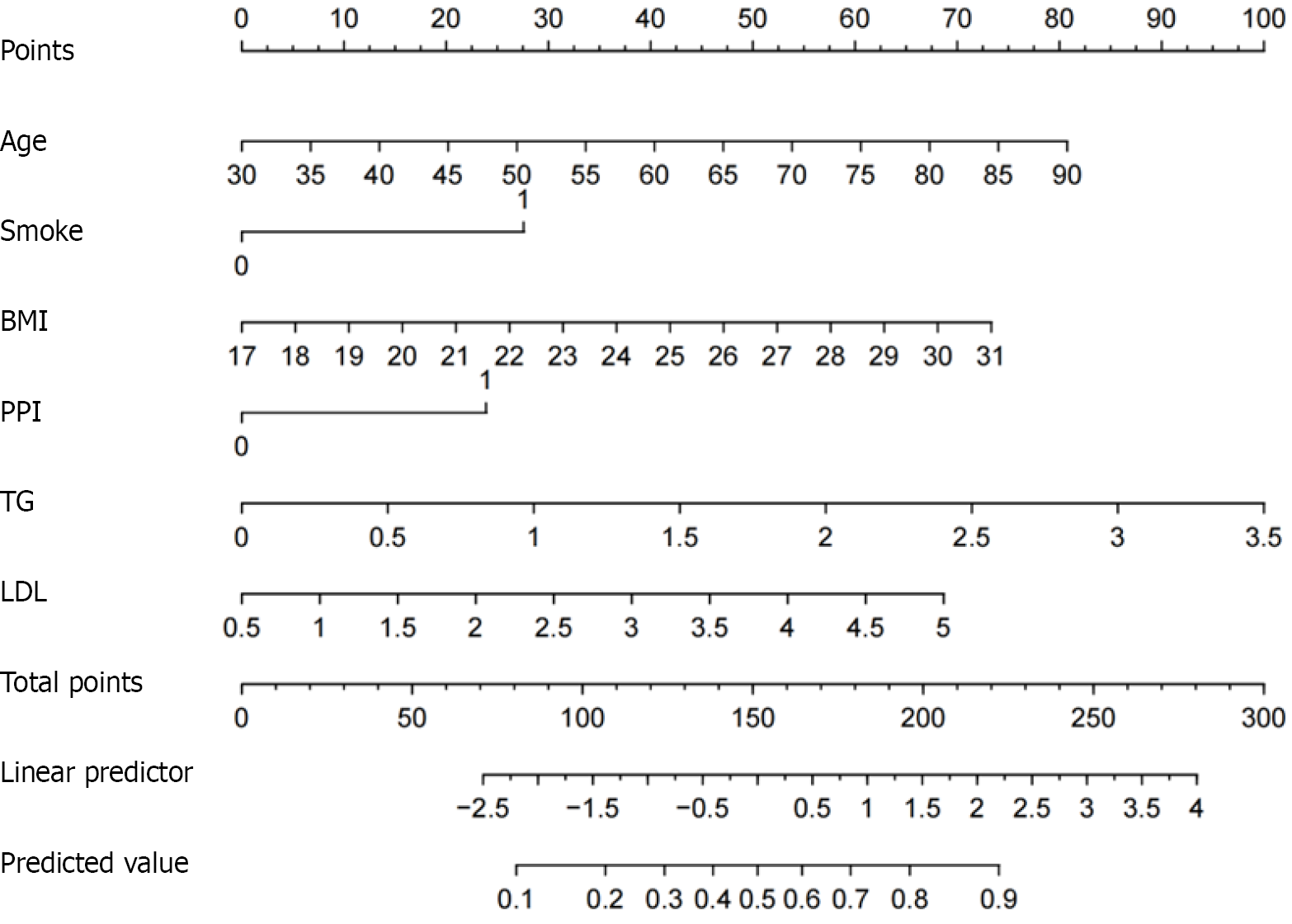
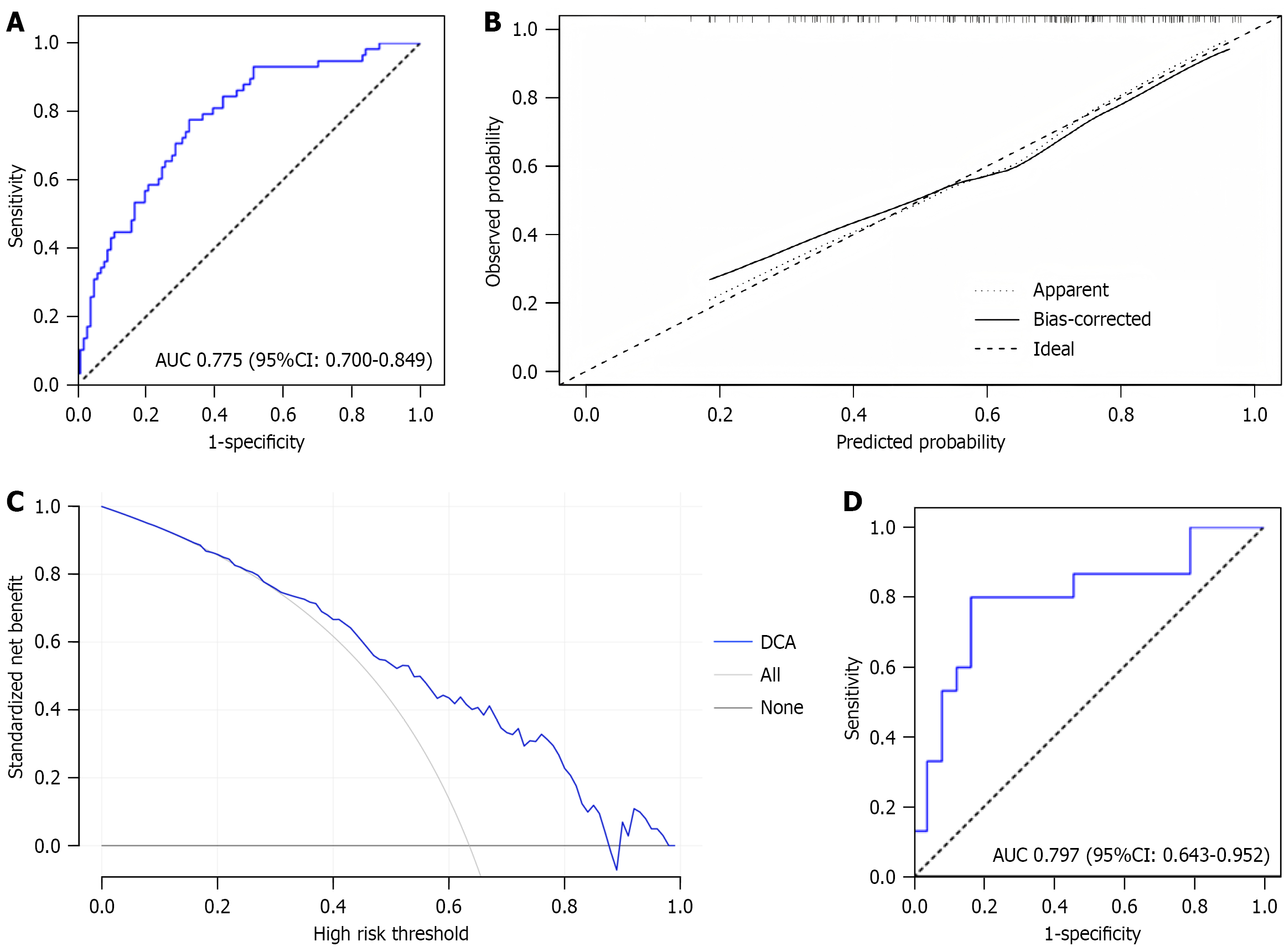
The logistic regression model demonstrated good fit using the Hosmer and Lemeshow test (χ2 = 2.759, P = 0.949). Based on the results of the multivariate logistic regression analysis, a risk prediction nomogram model for SIMI in NSAID users was constructed (Figure 3). A receiver operating characteristic curve was drawn to estimate the predictive accuracy of the nomogram, and the AUC was 0.775 (Figure 4A). The calibration curve demonstrated a strong agreement between the observed and predicted probabilities (Figure 4B). Decision curve analysis showed that the predictive model provided substantial net clinical benefit across a wide range of threshold probabilities (approximately 0.2 to 0.88) (Figure 4C). For validation, we utilized a cohort of 37 patients who underwent capsule endoscopy at two hospitals between February and July 2025. Among them, 23 patients were diagnosed with SIMI, while 14 showed no evidence of injury. The results demonstrated that the risk scoring system exhibited good predictive accuracy, achieving an AUC of 0.797 (Figure 4D).
SIMI represents a clinically significant pathological condition with multifactorial etiology and diverse manifestations, particularly with the growing recognition of NSAID-induced enteropathy. Extensive prior research has established the association between NSAID use and small intestinal mucosal damage[7,14,15]. This study systematically analyzed the risk factors and clinical endoscopic characteristics of mucosal injury in NSAID users. By rigorously excluding patients with confounding factors - such as inflammatory bowel disease and tuberculosis - we identified key risk factors asso
Consistent with previous clinical trials indicating that 55% to 75% of chronic NSAID users exhibit mucosal injury[16,17], our study also found a higher incidence of small intestinal lesions among NSAID-exposed patients. Notably, in agreement with the findings reported by Ehrhard et al[18], small intestinal lesions in NSAID users predominantly presented as erythema, erosions, and ulcers, and occurred more frequently in the jejunum. This pattern may be attributed to the richer villous architecture, larger absorptive surface area, and higher local concentrations of drugs and bile acids in the jejunal mucosa.
Multivariate analysis identified six independent risk factors: Advanced age, smoking, PPI use, elevated BMI, higher TG levels, and increased LDL. Previous studies have indicated that advanced age is associated with diminished mucosal repair capacity and increased susceptibility to NSAIDs[19,20].
Cigarette-derived reactive oxygen species may directly damage gastrointestinal mucosa, compromising small intestinal barrier integrity, while smoking-induced gut microbiota alterations could indirectly impair small intestinal epithelium[21,22]. The association between PPI use and SIMI is particularly noteworthy. Although PPIs effectively reduce upper gastrointestinal damage, they may alter gut microbiota and increase intestinal permeability, thereby potentially exacerbating NSAID-related lower gastrointestinal injury[23]. However, the potential for confounding by indication must be considered. Specifically, patients with a higher baseline risk for gastrointestinal complications (e.g., those with a history of ulcers) are more likely to be co-prescribed NSAIDs and PPIs. While our multivariate model adjusted for PPI use as a covariate, the inherent channeling of PPI to higher-risk patients may introduce residual confounding. The relationship between PPI co-therapy and SIMI risk warrants further investigation in prospective studies designed to better control for this confounding.
Elevated BMI demonstrated significant association with SIMI in our study, aligning with Sun et al’s findings[21] identifying obesity as a SIMI risk factor. Pathophysiologically, obesity may indirectly contribute to SIMI through gut microbiota alterations, characterized by increased Firmicutes/Prevotella ratios and decreased beneficial bacteria (Bacteroides, Bifidobacterium, Lactobacillus), which reduce short-chain fatty acid production critical for epithelial barrier integrity[24]. Additionally, adipose-derived proinflammatory mediators (leptin, tumor necrosis factor-α) may exacerbate NSAID-induced mucosal damage[25,26]. Obese patients may also develop intestinal barrier dysfunction through downregulation of tight junction proteins[27], which may exacerbate NSAID-induced epithelial apoptosis and bacterial translocation. Hyperlipidemia is associated with intestinal barrier dysfunction and gut microbiota dysbiosis[28]. In rat models, hyperlipidemia has been shown to induce intestinal barrier injury, accompanied by downregulated expression of tight junction proteins ZO-1 and occludin, as well as reduced villus height and thinning of the muscular layer in the small intestine[29]. Furthermore, studies have demonstrated significant alterations in the gut microbiota of hyperlipidemic mice, which subsequently compromise the intestinal mucosal barrier[30]. Additionally, elevated levels of deoxycholic acid resulting from a high-fat diet can impair intestinal stem cells by reducing interleukin-22 expression in the ileum[31]. Moreover, studies have indicated that the lipid-lowering agent fenofibrate can ameliorate intestinal injury and promote mucosal recovery in rat models[32]. Similarly, atorvastatin has been shown to alleviate radiation-induced intestinal damage through activation of autophagy and exertion of antioxidant effects[33]. These findings underscore the necessity of weight and lipid management in high-risk populations.
The nomogram developed in this study demonstrated good discriminatory ability, with an AUC of 0.775 in the derivation cohort and 0.797 in the validation cohort. These results indicate that the model exhibits satisfactory predictive accuracy and generalizability. Furthermore, bootstrap validation revealed excellent calibration performance, further supporting its potential for clinical application.
A key strength of this model lies in its reliance on readily available clinical parameters - including demographic characteristics, lifestyle habits, routine laboratory tests, and medication history. This facilitates the early identification of high-risk patients prior to initiating long-term NSAID therapy. For such individuals, clinicians may consider implementing preventive strategies.
In recent years, machine learning-based artificial intelligence technologies for medical image recognition have advanced significantly[34]. Building on the clinical risk factors and practical model identified in our study, artificial intelligence presents a transformative avenue for future research. For instance, deep learning algorithms could be applied to automatically analyze entire capsule endoscopy videos, capturing subtle, sub-visual mucosal features imperceptible to the human eye. Furthermore, multi-modal data fusion can integrate these imaging phenotypes with clinical and microbiome data to construct next-generation, personalized prediction tools, promising a deeper understanding of NSAID-induced enteropathy and enabling precise risk stratification.
This study has inherent limitations due to its retrospective design, including potential selection bias and unmeasured confounders such as dietary habits. Furthermore, the relatively small sample size of NSAID users, particularly in the validation cohort, may affect the model's stability and the reliability of the validation results, which also precluded detailed comparisons of different NSAID subtypes’ enterotoxic effects and limited statistical power to detect rare outcomes (e.g., strictures or perforations). Moreover, the model’s performance lacks external validation, and its generalizability to other populations requires further confirmation in future independent studies. The absence of biomarker data (e.g., fecal calprotectin) also restricted our ability to correlate endoscopic findings with subclinical inflammation or barrier dysfunction. Future prospective studies should incorporate multi-omics approaches (metagenomics, metabolomics) to investigate the mechanistic interplay between gut microbiota and NSAIDs, thereby identifying novel therapeutic targets.
In summary, we identified key risk factors and constructed a reliable predictive model for SIMI in NSAID users. This tool could assist clinicians in identifying high-risk individuals who may benefit from closer monitoring, alternative therapies, or prophylactic measures.
| 1. | Antithrombotic Trialists' Collaboration. Collaborative meta-analysis of randomised trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients. BMJ. 2002;324:71-86. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5315] [Cited by in RCA: 4407] [Article Influence: 183.6] [Reference Citation Analysis (4)] |
| 2. | Weil J, Colin-Jones D, Langman M, Lawson D, Logan R, Murphy M, Rawlins M, Vessey M, Wainwright P. Prophylactic aspirin and risk of peptic ulcer bleeding. BMJ. 1995;310:827-830. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 420] [Cited by in RCA: 374] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 3. | Sakamoto C, Sugano K, Ota S, Sakaki N, Takahashi S, Yoshida Y, Tsukui T, Osawa H, Sakurai Y, Yoshino J, Mizokami Y, Mine T, Arakawa T, Kuwayama H, Saigenji K, Yakabi K, Chiba T, Shimosegawa T, Sheehan JE, Perez-Gutthann S, Yamaguchi T, Kaufman DW, Sato T, Kubota K, Terano A. Case-control study on the association of upper gastrointestinal bleeding and nonsteroidal anti-inflammatory drugs in Japan. Eur J Clin Pharmacol. 2006;62:765-772. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 94] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 4. | Fujimori S, Seo T, Gudis K, Ehara A, Kobayashi T, Mitsui K, Yonezawa M, Tanaka S, Tatsuguchi A, Sakamoto C. Prevention of nonsteroidal anti-inflammatory drug-induced small-intestinal injury by prostaglandin: a pilot randomized controlled trial evaluated by capsule endoscopy. Gastrointest Endosc. 2009;69:1339-1346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 97] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 5. | Min YW, Chang DK. The Role of Capsule Endoscopy in Patients with Obscure Gastrointestinal Bleeding. Clin Endosc. 2016;49:16-20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 1.4] [Reference Citation Analysis (4)] |
| 6. | Cheung DY, Kim JS, Shim KN, Choi MG; Korean Gut Image Study Group. The Usefulness of Capsule Endoscopy for Small Bowel Tumors. Clin Endosc. 2016;49:21-25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 34] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 7. | Lee SP, Lee J, Kae SH, Jang HJ, Jung ES. Effect of Nonsteroidal Anti-inflammatory Agents on Small Intestinal Injuries as Evaluated by Capsule Endoscopy. Dig Dis Sci. 2021;66:2724-2731. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 8. | Saurin JC, Beneche N, Chambon C, Pioche M. Challenges and Future of Wireless Capsule Endoscopy. Clin Endosc. 2016;49:26-29. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 15] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 9. | Sample JW, Solanki MH, Thiels C, Bingener J. Nonsteroidal anti-inflammatory drug-induced small bowel strictures (diaphragm disease) - an under-recognized cause of small bowel obstruction. J Gastrointest Surg. 2024;28:1430-1435. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 10. | Shin SJ, Noh CK, Lim SG, Lee KM, Lee KJ. Non-steroidal anti-inflammatory drug-induced enteropathy. Intest Res. 2017;15:446-455. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 70] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 11. | Iwamoto J, Mizokami Y, Saito Y, Shimokobe K, Honda A, Ikegami T, Matsuzaki Y. Small-bowel mucosal injuries in low-dose aspirin users with obscure gastrointestinal bleeding. World J Gastroenterol. 2014;20:13133-13138. [PubMed] [DOI] [Full Text] |
| 12. | Niikura R, Yamada A, Maki K, Nakamura M, Watabe H, Fujishiro M, Oka S, Esaki M, Fujimori S, Nakajima A, Ohmiya N, Matsumoto T, Tanaka S, Koike K, Sakamoto C. Associations between drugs and small-bowel mucosal bleeding: Multicenter capsule-endoscopy study. Dig Endosc. 2018;30:79-89. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 20] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 13. | Endo H, Sakai E, Taniguchi L, Kessoku T, Komiya Y, Ezuka A, Kawamura H, Taguri M, Higurashi T, Ohkubo H, Yamada E, Takahashi H, Inamori M, Maeda S, Sakaguchi T, Hata Y, Nagase H, Nakajima A. Risk factors for small-bowel mucosal breaks in chronic low-dose aspirin users: data from a prospective multicenter capsule endoscopy registry. Gastrointest Endosc. 2014;80:826-834. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 60] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 14. | Maiden L, Thjodleifsson B, Theodors A, Gonzalez J, Bjarnason I. A quantitative analysis of NSAID-induced small bowel pathology by capsule enteroscopy. Gastroenterology. 2005;128:1172-1178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 411] [Cited by in RCA: 358] [Article Influence: 17.0] [Reference Citation Analysis (4)] |
| 15. | Washio E, Esaki M, Maehata Y, Miyazaki M, Kobayashi H, Ishikawa H, Kitazono T, Matsumoto T. Proton Pump Inhibitors Increase Incidence of Nonsteroidal Anti-Inflammatory Drug-Induced Small Bowel Injury: A Randomized, Placebo-Controlled Trial. Clin Gastroenterol Hepatol. 2016;14:809-815.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 99] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 16. | Allison MC, Howatson AG, Torrance CJ, Lee FD, Russell RI. Gastrointestinal damage associated with the use of nonsteroidal antiinflammatory drugs. N Engl J Med. 1992;327:749-754. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 792] [Cited by in RCA: 660] [Article Influence: 19.4] [Reference Citation Analysis (0)] |
| 17. | Ishihara M, Ohmiya N, Nakamura M, Funasaka K, Miyahara R, Ohno E, Kawashima H, Itoh A, Hirooka Y, Watanabe O, Ando T, Goto H. Risk factors of symptomatic NSAID-induced small intestinal injury and diaphragm disease. Aliment Pharmacol Ther. 2014;40:538-547. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 35] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 18. | Ehrhard F, Nazeyrollas P, Brixi H, Heurgue-Berlot A, Thiéfin G. Proximal predominance of small bowel injury associated with uncoated low-dose aspirin therapy: a video capsule study in chronic users. Eur J Gastroenterol Hepatol. 2013;25:1265-1272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 19. | Connell E, Le Gall G, Pontifex MG, Sami S, Cryan JF, Clarke G, Müller M, Vauzour D. Microbial-derived metabolites as a risk factor of age-related cognitive decline and dementia. Mol Neurodegener. 2022;17:43. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 153] [Article Influence: 38.3] [Reference Citation Analysis (0)] |
| 20. | Ribeiro H, Rodrigues I, Napoleão L, Lira L, Marques D, Veríssimo M, Andrade JP, Dourado M. Non-steroidal anti-inflammatory drugs (NSAIDs), pain and aging: Adjusting prescription to patient features. Biomed Pharmacother. 2022;150:112958. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 67] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 21. | Sun X, Wang F, Liu J, Wu L, Wang Z, Chen X, Wang M, Zeng Q. Risk factors for small-intestinal mucosal breaks beyond aspirin. J Gastroenterol Hepatol. 2022;37:1596-1602. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 22. | Yoshihara T, Oikawa Y, Kato T, Kessoku T, Kobayashi T, Kato S, Misawa N, Ashikari K, Fuyuki A, Ohkubo H, Higurashi T, Tateishi Y, Tanaka Y, Nakajima S, Ohno H, Wada K, Nakajima A. The protective effect of Bifidobacterium bifidum G9-1 against mucus degradation by Akkermansia muciniphila following small intestine injury caused by a proton pump inhibitor and aspirin. Gut Microbes. 2020;11:1385-1404. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 53] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 23. | Nadatani Y, Watanabe T, Suda W, Nakata A, Matsumoto Y, Kosaka S, Higashimori A, Otani K, Hosomi S, Tanaka F, Nagami Y, Kamata N, Taira K, Yamagami H, Tanigawa T, Hattori M, Fujiwara Y. Gastric acid inhibitor aggravates indomethacin-induced small intestinal injury via reducing Lactobacillus johnsonii. Sci Rep. 2019;9:17490. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 40] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 24. | Amabebe E, Robert FO, Agbalalah T, Orubu ESF. Microbial dysbiosis-induced obesity: role of gut microbiota in homoeostasis of energy metabolism. Br J Nutr. 2020;123:1127-1137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 290] [Article Influence: 48.3] [Reference Citation Analysis (0)] |
| 25. | Ahmed B, Sultana R, Greene MW. Adipose tissue and insulin resistance in obese. Biomed Pharmacother. 2021;137:111315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 839] [Cited by in RCA: 656] [Article Influence: 131.2] [Reference Citation Analysis (1)] |
| 26. | Hagberg CE, Spalding KL. White adipocyte dysfunction and obesity-associated pathologies in humans. Nat Rev Mol Cell Biol. 2024;25:270-289. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 135] [Article Influence: 67.5] [Reference Citation Analysis (0)] |
| 27. | Fernández-García P, Taxerås SD, Reyes-Farias M, González L, Soria-Gondek A, Pellitero S, Tarascó J, Moreno P, Sumoy L, Stephens JM, Yoo LG, Galán M, Izquierdo A, Medina-Gómez G, Herrero L, Corrales P, Villarroya F, Cereijo R, Sánchez-Infantes D. Claudin-1 as a novel target gene induced in obesity and associated to inflammation, fibrosis, and cell differentiation. Eur J Endocrinol. 2024;190:201-210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 28. | Liu Z, Tang R, Liu J, Zhang Z, Li Y, Zhao R. Epicatechin and β-glucan from whole highland barley grain ameliorates hyperlipidemia associated with attenuating intestinal barrier dysfunction and modulating gut microbiota in high-fat-diet-fed mice. Int J Biol Macromol. 2024;278:134917. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 29. | Zuo Z, Liu S, Pang W, Lu B, Sun W, Zhang N, Zhou X, Zhang D, Wang Y. Beneficial Effect of Kidney Bean Resistant Starch on Hyperlipidemia-Induced Acute Pancreatitis and Related Intestinal Barrier Damage in Rats. Molecules. 2022;27:2783. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 30. | Hamilton MK, Boudry G, Lemay DG, Raybould HE. Changes in intestinal barrier function and gut microbiota in high-fat diet-fed rats are dynamic and region dependent. Am J Physiol Gastrointest Liver Physiol. 2015;308:G840-G851. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 203] [Cited by in RCA: 245] [Article Influence: 22.3] [Reference Citation Analysis (0)] |
| 31. | Xu J, Huang D, Xu X, Wu X, Liu L, Niu W, Lu L, Zhou H. An elevated deoxycholic acid level induced by high-fat feeding damages intestinal stem cells by reducing the ileal IL-22. Biochem Biophys Res Commun. 2021;579:153-160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 32. | Sukhotnik I, Nissimov N, Ben Shahar Y, Moati D, Bitterman N, Pollak Y, Berkowitz D, Coran AG, Bitterman A. Fenofibrate reduces intestinal damage and improves intestinal recovery following intestinal ischemia-reperfusion injury in a rat. Pediatr Surg Int. 2016;32:1193-1200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 33. | Wei MF, Cheng CH, Wen SY, Lin JC, Chen YH, Wang CW, Lee YH, Kuo SH. Atorvastatin Attenuates Radiotherapy-Induced Intestinal Damage through Activation of Autophagy and Antioxidant Effects. Oxid Med Cell Longev. 2022;2022:7957255. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 34. | Huang TY, Zhan SQ, Chen PJ, Yang CW, Lu HH. Accurate diagnosis of endoscopic mucosal healing in ulcerative colitis using deep learning and machine learning. J Chin Med Assoc. 2021;84:678-681. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (1)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/