Published online Feb 27, 2026. doi: 10.4240/wjgs.v18.i2.115744
Revised: December 14, 2025
Accepted: December 25, 2025
Published online: February 27, 2026
Processing time: 125 Days and 20.4 Hours
Percutaneous cholecystostomy (PCT) is widely used for high-risk acute cholecy
To characterize timing, catheter management, survival, and follow-up outcomes after PCT in a high-risk cohort.
This single center retrospective cohort study included consecutive adult patients undergoing PCT placement for acute cholecystitis at a community hospital setting in New York between January 2012 and December 2024. The study population was grouped according to type of acute cholecystitis (calculous vs acalculous), according to the timing of PCT placement since diagnosis [early (≤ 4 days) vs late (> 4 days)], and according to level of care [intensive care unit (ICU) vs non-ICU patients]. Cox proportional hazards models were used to examine effects of PCT placement interval on mortality rates, after accounting for potential confounding factors (age, Charlson Comorbi
The population consisted of 174 patients who underwent PCT placement for acute cholecystitis between 2012 and 2024 at a community hospital in New York. Median time to PCT was 2 days (interquartile range 1-4). Overall, mortality was 21% (36/174) and was higher with delayed PCT (> 4 days) vs early PCT [35% (17/49) vs 15% (19/125), P = 0.001]. Catheter removal occurred in 13% (23/174), 55% (96/174) remained catheter-dependent, and 32% (56/174) had interval cholecystectomy. ICU admission was associated with prolonged catheter duration but was not associated with mortality. Kaplan-Meier analysis demonstrated a significantly higher survival rate in the early group compared with the late group (log-rank P = 0.006). In both unadjusted models and models adjusted for selected covariates, patients who had catheters placed late (> 4 days) had 2.5-fold higher risk of death than patients with early placement.
Early PCT was associated with higher survival in high-risk acute cholecystitis. High rates of catheter dependency highlight the need for standardized protocols and reassessment for definitive surgery.
Core Tip: Percutaneous cholecystostomy (PCT) is commonly used as a bridge or alternative to surgery in high-risk patients with acute cholecystitis, yet optimal timing and catheter management remain uncertain. In this retrospective study of 174 patients, early PCT (≤ 4 days from diagnosis) was associated with significantly higher survival compared to delayed intervention. Despite its clinical benefit, more than half of patients remained catheter-dependent, and only one-third underwent interval cholecystectomy. These findings highlight the need for standardized PCT management protocols and structured follow-up to optimize long-term outcomes in this fragile population.
- Citation: Hassanesfahani M, Giannis D, Marfo N, Kaur M, Quang CMPT, Miele A, Louis MA, Mandava NR. Defining endpoints in percutaneous cholecystostomy: Catheter management, patient survival, and long-term outcomes from a twelve-year retrospective study. World J Gastrointest Surg 2026; 18(2): 115744
- URL: https://www.wjgnet.com/1948-9366/full/v18/i2/115744.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i2.115744
Acute cholecystitis is an acute inflammatory condition of the gallbladder, typically precipitated by obstruction of the cystic duct, most often due to gallstones. However, acalculous forms are increasingly recognized in critically ill or postoperative patients. Acute cholecystitis accounts for approximately 3%-10% of hospital admissions for acute abdomi
Management options for acute cholecystitis include open cholecystectomy, laparoscopic cholecystectomy (LC), robotic-assisted cholecystectomy, and percutaneous cholecystostomy tube (PCT). LC has emerged as the gold standard treatment for most patients, considering its lower morbidity, shorter hospital length of stay, and faster recovery and return to daily activities compared to open surgery. In critically ill, poor surgical candidates, PCT placement is an alternative option[2,3].
PCT was first described in the early 1980s as a minimally invasive drainage technique for high-risk surgical candidates, as a temporizing measure to stabilize patients before interval LC or, in select cases, as definitive therapy-especially in elderly or critically ill patients with multiple comorbidities[4].
The Tokyo Guidelines, first published in 2007 and subsequently updated in 2013 (TG13) and 2018 (TG18), have provided an international framework for the diagnosis, severity grading, and treatment recommendations in acute cholecystitis. The TG18 version expanded the role of early PCT for grade II-III acute cholecystitis in high-risk surgical candidates, from a salvage rescue technique to a primary intervention in certain clinical scenarios[1]. The World Society of Emergency Surgery (WSES) 2020 guidelines further refined recommendations for PCT, emphasizing its use for immediate source control in patients with severe sepsis or septic shock who are unfit for surgery due to comorbidities/prohibitive operative risk. The WSES also highlighted the importance of defining patient selection criteria and standar
Despite the emerging evidence, important clinical questions remain unanswered, including the optimal timing for PCT placement, how long the PCT should remain in place, when and in which population definitive LC should follow PCT placement. Furthermore, real-world practices vary widely, especially in community and safety-net hospitals[5]. This study aims to address these questions through a review of a 12-year PCT placement experience in a community hospital, focusing on the management and outcomes in comparison to the current literature and established guidelines.
This single center retrospective cohort study included consecutive adult patients (18 years or older) undergoing PCT placement for acute cholecystitis at a community hospital setting in New York between January 2012 and December 2024. Calculous cholecystitis in this study refers to any diagnosis of acute cholecystitis in the presence of cholelithiasis (gallstones, sludge, or microlithiasis) on imaging, while acalculous cholecystitis is defined as acute cholecystitis in the absence of cholelithiasis. Institutional Review Board approval was obtained. Epic System Inc.'s Slicer Dicer was used to extract the records of eligible patients and variables of interest were automatically or manually extracted by three authors. The dataset was deidentified and patient consent was waived.
The study population was grouped according to type of acute cholecystitis (calculous vs acalculous), according to the timing of PCT placement since diagnosis [early (≤ 4 days) vs late (> 4 days)], and according to level of care [intensive care unit (ICU) vs non-ICU patients].
PCT irrigation followed a consistent institutional practice. Drains were irrigated twice daily while patients were inpatient. At discharge, patients and/or family members received standardized education regarding continued irrigation daily at home and as needed if output was decreasing. Additional irrigation was performed at scheduled outpatient follow-up visits.
Antibiotics for patients with acute cholecystitis, that are managed without surgery at index admission, are given for at least 4 days after source control (PCT placement) at our institution. However, antibiotic duration was individualized based on clinical response, microbiologic data when available, and treating team preference, rather than a fixed institutional protocol.
All patients were provided a scheduled follow-up at the outpatient surgery clinic within 2 weeks after discharge and were seen again at their schedule follow-up with Interventional radiology within 3-6 weeks after PCT placement for tract maturation cholangiography.
Tube removal was determined by a combination of clinical improvement, imaging findings, and cholangiographic confirmation of cystic duct patency prior to removal.
Variables of interest included patient demographics (age, sex, race/ethnicity), comorbidities (hypertension and diabetes mellitus), Charlson Comorbidity Index (CCI), type of cholecystitis (calculous, acalculous), level of care (ICU, non-ICU), type of antibiotics, culture data (blood culture, bile culture), important dates and timings (date of admission, date of PCT placement, date of discharge, date of surgery, date of outpatient follow-up, date of death), diagnostic imaging modalities [ultrasound (US), computerized tomography (CT), hepatobiliary iminodiacetic acid (HIDA)] scan, and main outcomes (overall mortality, catheter removal, interval LC, living as a catheter dependent).
Continuous variables were summarized as mean and SD or median and interquartile range (IQR), according to the shape of the variable distribution. Categorical variables as n (%). Univariate analysis between continuous variables were con
Several methods were used to examine the optimal cut-point for grouping patients into early vs late PCT placement. First, theoretically informed cut-points of > 2, > 3, > 4, and > 5 were examined. Cox proportional hazards models were then run and compared. Second, Cox proportional models were run using restricted cubic splines (RCS) and models with 3, 4 and 5 knots were compared.
Survival analysis was conducted using the Kaplan-Meier method and compared between groups using the log-rank test. Cox proportional hazards models were used to examine effects of PCT placement interval on mortality rates, after accounting for potential confounding factors. First, covariate analysis was used to assess potential confounding effects. Differences between patients who expired and those who were lost to follow-up (censored) were examined by age, CCI, cholecystitis type, ICU status, bile culture and blood culture. Significance was assessed via standardized mean differences (SMD) and associated 95% confidence intervals (CI). Covariate analysis was also conducted based on PCT placement status (early vs late). A Cox proportional hazards model was then used to examine the effect of PCT placement time on mortality, accounting for these covariates. Support for the model was based on visual interpretation of Schoenfeld residuals and formal hypothesis testing of the proportionality assumption, using the test outlined by Grambsch and Therneau[6]. Based on these diagnostics, the proportional hazards assumption was met and the model was correctly specified. Time-to-event was calculated from the date of PCT placement to the date of death or last follow-up. Censored observations represent patients who were alive at the end of their observation period. CI for survival estimates were computed using Greenwood’s formula. Statistical significance was set at P < 0.05. All statistical analyses were performed with the use of R language version 4.2.3 within the RStudio software version 2023.03.0+386.
The study population consisted of 174 patients, with a mean age of 71.7 years (SD: 15.8) and 53% (n = 92) were females (Table 1). Patients identified their race as Hispanic (29%, n = 50), White (28%, n = 49), Asian (28%, n = 49), Black/African-American (10%, n = 17); an additional 5% (n = 9) who did not self-identify in the aforementioned categories were grouped in the “other” category. Median CCI was 5 (IQR 4-7) and 29% (n = 50) were admitted to the ICU.
| Variables of interest | n (%), mean (SD) or median (IQR) |
| Demographics | |
| Age, mean (SD), years | 71.7 (15.8) |
| Gender | |
| Female | 92 (53) |
| Male | 82 (47) |
| Race | |
| Hispanic | 50 (29) |
| White | 49 (28) |
| Asian | 49 (28) |
| Black | 17 (10) |
| Other | 9 (5) |
| Comorbidities | |
| Hypertension | 124 (71) |
| Diabetes | 85 (49) |
| Clinical status | |
| Median CCI (IQR) | 5 (4–7) |
| ICU admission | 50 (29) |
| Antibiotics | |
| Piperacillin/tazobactam | 83 (48) |
| Cefoxitin | 25 (14) |
| Ceftriaxone | 10 (6) |
| Cefepime | 7 (4) |
| Ampicillin/sulbactam | 4 (2) |
| Vancomycin | 39 (22) |
| Ciprofloxacin | 13 (7) |
| Metronidazole | 32 (18) |
| Ertapenem | 2 (1) |
| Meropenem | 11 (6) |
| Aztreonam | 13 (7) |
| Doxycycline | 7 (4) |
Acute cholecystitis was diagnosed via US (n = 104; 60%), CT (n = 113; 65%), or HIDA (n = 4; 2%) and was calculous in 91% (n = 159) and acalculous in 9% (n = 15).
The main empiric antibiotics that were administered for the treatment of acute cholecystitis included piperacillin/tazobactam (48%), vancomycin (22%), metronidazole (18%), cefoxitin (14%), ciprofloxacin (7%), aztreonam (7%), meropenem (6%), ceftriaxone (6%), cefepime (4%), doxycycline (4%), ampicillin/sulbactam (2%), and ertapenem (1%).
The most frequently isolated microorganisms in blood and bile cultures were Escherichia coli (8% and 21%), Klebsiella pneumoniae (3% and 15%), and Enterococcus species (2% and 10%), followed by Enterobacter (1% and 5%), Clostridium perfringens (1% and 2%), and Acinetobacter (0% and 2%), respectively (Table 2). No growth was observed in 72% of blood and 35% of bile cultures.
| Microorganism | Blood culture | Bile culture |
| Escherichia coli | 14 (8) | 36 (21) |
| Enterococcus species | 3 (2) | 18 (10) |
| Klebsiella pneumoniae | 6 (3) | 26 (15) |
| Enterobacter | 1 (1) | 9 (5) |
| Acinetobacter | 0 (0) | 3 (2) |
| Clostridium perfrigens | 2 (1) | 3 (2) |
| No growth | 125 (72) | 61 (35) |
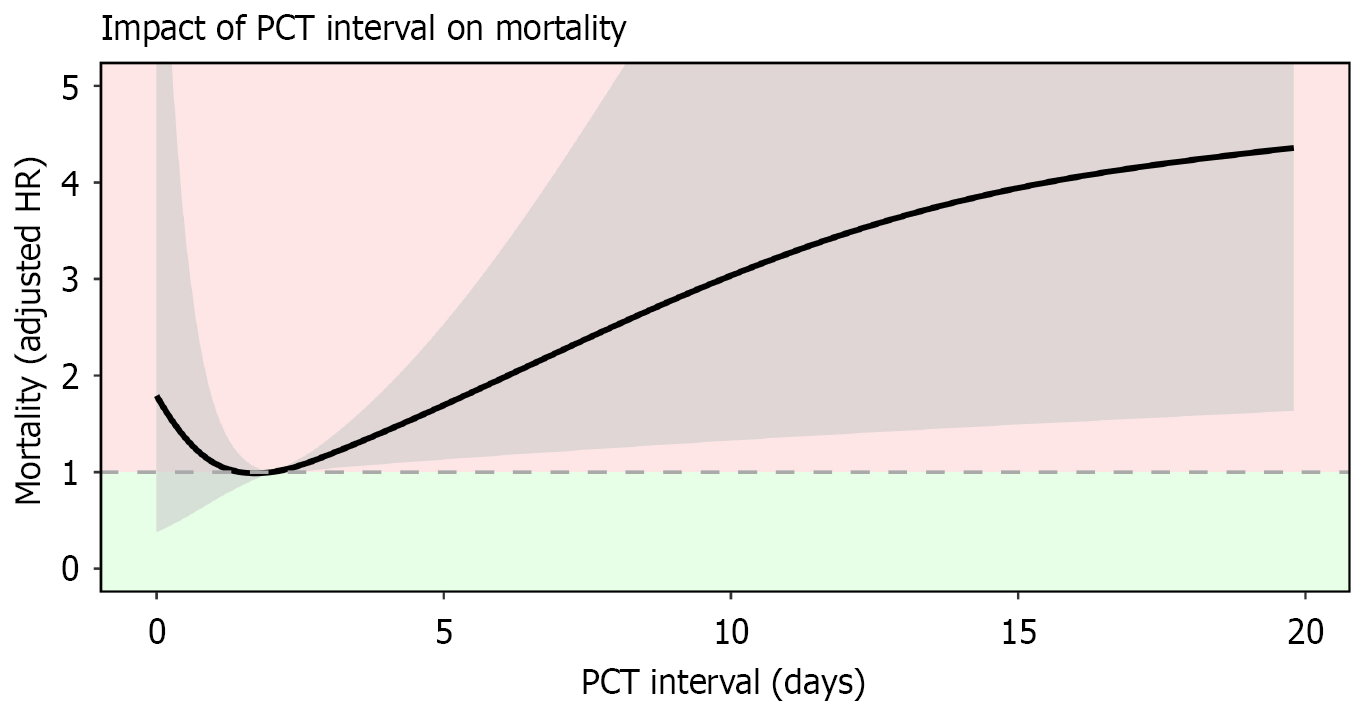
Median time from admission to PCT was 2 days (IQR 1-4). Median catheter duration was 45 days (IQR: 28-85). Cox proportional hazards models testing cut-points of late PCT placement defined at > 2, > 3, > 4, and > 5 days showed based on AIC that the model that best fit the data was the one which used a 4-day cut-off. RCS models with 3, 4 and 5 knots were also compared, with the 4-knot model demonstrating the best fit to the data based on logLik and AIC. Likelihood ratio tests indicated that the effect of longer PCT placement interval on increased risk for mortality was significant in both unadjusted (LR Test, P = 0.010) and adjusted (LR test, P = 0.008) RCS models. Visual inspection of adjusted HR of RCS models by PCT placement interval suggested some non-linearity, with an HR > 1 for patients with PCT interval < 2 days, and HR > 1 for patients with PCT interval > 3 days. As noted above, models using a cut-off of 3 did not outperform those using a cut-off of 4 days (Figure 1).
Covariate analysis conducted based on PCT placement status [early (n = 125) vs late (n = 49)] showed that patients with early PCT placement had significantly lower median CCI (SMD = -0.57, 95%CI: -0.92 to -0.23), and significantly higher rates of positive bile culture (SMD = 0.6, 95%CI: 0.25-0.95) and blood culture (SMD = 0.41, 95%CI: 0.05-0.76) (Table 3). There was no significant difference in PCT placement timing between acalculous & calculous patients (SMD = -0.40, 95%CI: -0.99 to 0.19). No differences by age or other covariates were observed between groups.
| Characteristic | Early (n = 125) | Late (n = 49) | SMD: Early vs late |
| Age | 75 (65, 85) | 74 (64, 81) | -0.13 (-0.47, 0.21) |
| CCI | 5.00 (3.00, 6.00) | 6.50 (4.00, 8.00) | -0.57 (-0.92, -0.23) |
| Cholecystitis type | 0.30 (-0.04, 0.64) | ||
| Acalculous | 14 (11) | 1 (2) | 0.30 (-0.04, 0.64) |
| Calculous | 111 (89) | 48 (98) | -0.30 (-0.64, 0.04) |
| ICU status | 0.08 (-0.26, 0.42) | ||
| ICU | 34 (27.0) | 16 (33) | -0.08 (-0.42, 0.26) |
| Non-ICU | 91 (73.0) | 33 (67) | 0.08 (-0.26, 0.42) |
| Bile culture | 0.60 (0.25, 0.95) | ||
| Negative | 35 (28) | 26 (53) | -0.60 (-0.95, -0.25) |
| Positive | 90 (72) | 23 (47) | 0.60 (0.25, 0.95) |
| Blood culture | 0.41 (0.05, 0.76) | ||
| Negative | 85 (68) | 40 (82) | -0.41 (-0.76, -0.05) |
| Positive | 40 (32) | 9 (18) | 0.41 (0.05, 0.76) |
Tract-maturation cholangiography was performed in 63% (109/174) and the number of cholangiographies varied, 37% (65/174) had one study, and 26% (44/174) underwent two or more studies (up to 4 studies). Among the 56 patients who ultimately underwent interval cholecystectomy, 64% (36/56) received at least one cholangiogram. Of the 118 patients who did not proceed to surgery, 73 were lost to follow-up within one year and 12 remained in follow-up beyond one year.
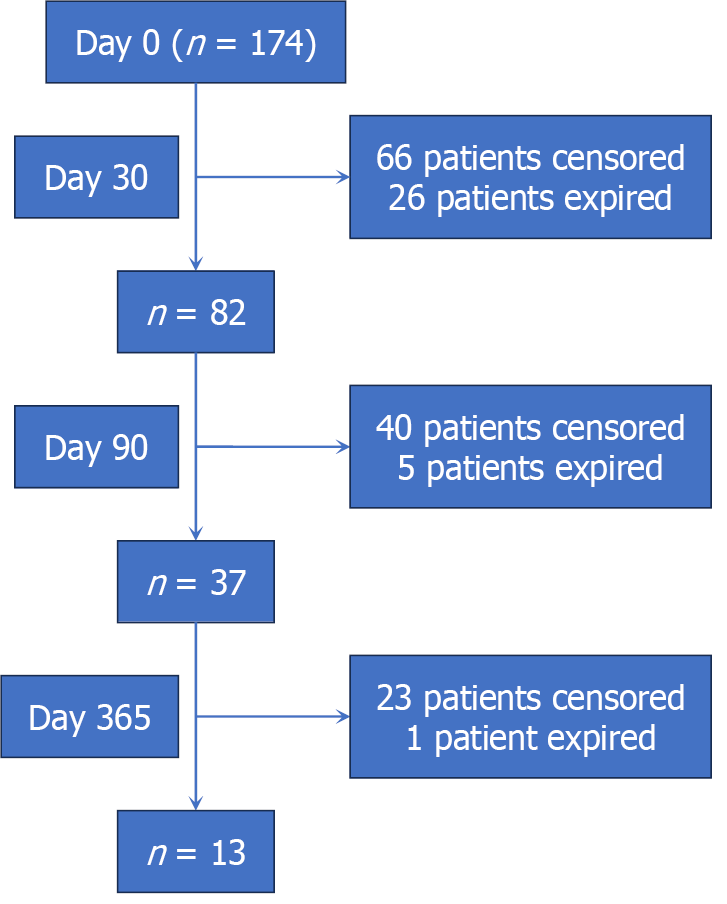
A detailed analysis of patient disposition at 30, 90, and 365 days following PCT placement was performed (Figure 2). Within 30 days, 26 patients died and 66 were censored. An additional 5 patients died and 40 were censored by 90 days, and 1 further death and 23 additional censored patients were recorded by 365 days. Overall, 82 patients had follow-up to at least 90 days, and 13 patients were followed beyond 1 year.
Catheter removal was documented in 13% (23/174); 32% (56/174) had interval cholecystectomy (Table 4). Mean time to surgery from PCT placement was 123 ± 281 days and 55% (96/174) remained catheter-dependent without having surgery (catheter remained in place at last follow-up or time of death). Catheter dependence was present in 79 non-expiring patients at their last documented follow-up, defined as a catheter dwell time greater than or equal to the duration of follow-up.
| Outcome | Early PCT (≤ 4 days) (n = 125) | Late PCT (> 4 days) (n = 49) |
| Catheter duration (days), median (IQR) | 40 (30-60) | 55 (45-80) |
| Overall mortality | 15% (19/125) | 35% (17/49) |
| Catheter removal | 14% (17/125) | 12% (6/49) |
| Interval LC performed | 38% (48/125) | 16% (8/49) |
ICU admission was associated with increased catheter duration (median 60 days vs 42 days) but was not significantly associated with mortality [ICU: 22% (11/50) vs non-ICU 20% (25/124), P > 0.05] (Table 5).
| Outcome | ICU patients (n = 50) | Non-ICU patients (n = 124) |
| Catheter duration (days), median (IQR) | 60 (40-85) | 42 (32-55) |
| Overall mortality | 22% (11/50) | 20% (25/124) |
| Catheter removal | 12% (6/50) | 14% (17/124) |
| Interval LC performed | 22% (11/50) | 36% (45/124) |
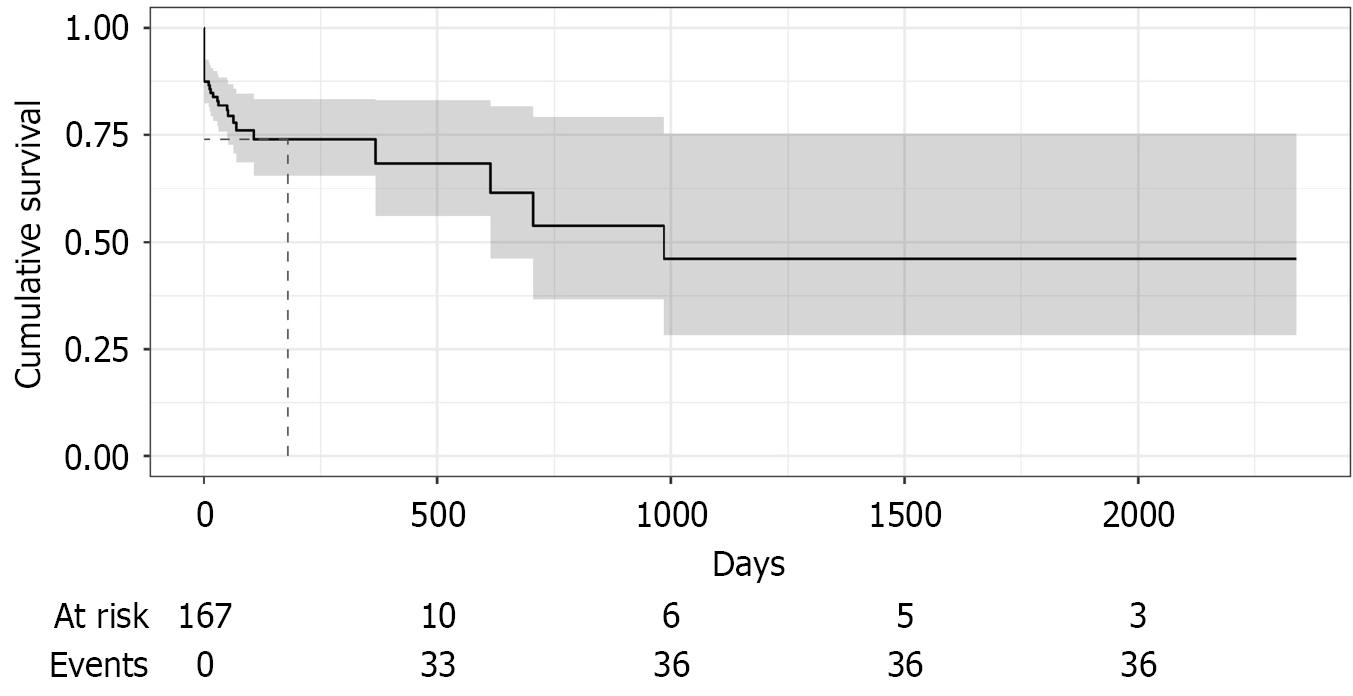
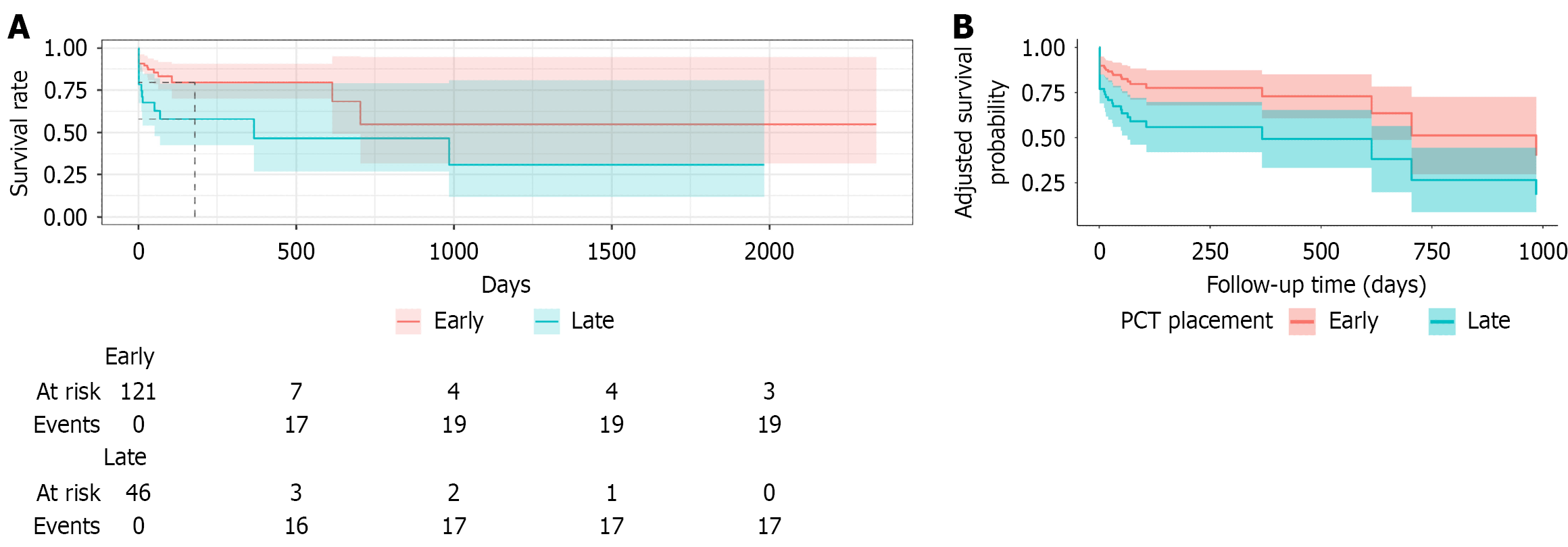
Overall, mortality was 21% (95%CI: 15.3-27.3; n = 36/174). Mortality was higher with delayed PCT (> 4 days) vs early PCT [35% (95%CI: 21.4-48.0; n = 17/49;) vs 15% (95%CI: 8.9%-21.5%; n = 19/125), P = 0.001] (Figures 3 and 4, Table 6).
| Outcome | Calculous (n = 159) | Acalculous (n = 15) |
| Catheter duration (days), median (IQR) | 49.5 (30-85) | 47 (9-94) |
| Overall mortality (%) | 22.6 (36/159) | 0 (0/15) |
| Catheter removal (%) | 13.2 (21/159) | 1.5 (2/15) |
| Interval LC performed (%) | 31 (49/159) | 47 (7/15) |
Covariate analysis was used to assess potential confounding effects and showed significant differences between those who expired and those who were censored or lost to follow up in age (SMD = -0.69, 95%CI: -1.1 to -0.32), with expired patients median age = 79 years and censored patients average age = 74 years, and by CCI (SMD = -0.81, 95%CI: -1.2 to
| Characteristic | Censored (n = 138) | Expired (n = 36) | SMD: Early vs late |
| Age | 74 (61, 83) | 79 (72, 88) | -0.69 (-1.07, -0.32) |
| CCI | 5.00 (3.00, 6.00) | 6.50 (5.50, 7.00) | -0.81 (-1.19, -0.43) |
| Cholecystitis type | 0.45 (0.08, 0.82) | ||
| Acalculous | 15 (11) | 0 (0) | 0.45 (0.08, 0.82) |
| Calculous | 123 (89) | 36 (100) | -0.45 (-0.82, -0.08) |
| ICU | 0.06 (-0.31, 0.43) | ||
| ICU | 39 (28) | 11 (31) | -0.06 (-0.43, 0.31) |
| Non-ICU | 99 (72) | 25 (69) | 0.06 (-0.31, 0.43) |
| Bile culture | 0.18 (-0.19, 0.55) | ||
| Negative | 50 (36) | 11 (31) | 0.18 (-0.19, 0.55) |
| Positive | 88 (64) | 25 (69) | -0.18 (-0.55, 0.19) |
| Blood culture | 0.01 (-0.37, 0.38) | ||
| Negative | 97 (70) | 28 (78) | 0.01 (-0.37, 0.38) |
| Positive | 41 (30) | 8 (22) | -0.01 (-0.38, 0.37) |
A Cox proportional hazards model was used to examine the effect of PCT placement time on mortality, accounting for age, CCI, cholecystitis type, ICU status, bile culture and blood culture data. In both unadjusted Cox models and models adjusted for selected covariates, patients who had catheters placed late (i.e., > 4 days) had 2.5 times greater hazard for mortality than patients who had placement early (Figure 4, Table 8). All deaths occurred in patients that never underwent surgery and mortality was zero in patients that had their catheter removed and in patients that underwent LC. Considering the observed mortality rate among acalculous cholecystitis patients (n = 0), a post-hoc sub-analysis that only included calculous patients and the primary analyses were repeated, with comparable results (Table 9).
| Characteristic | HR | 95%CI | P value |
| PCT placement interval | |||
| Early | |||
| Late | 2.58 | 1.25, 5.32 | 0.01 |
| Age | 1.05 | 1.01, 1.08 | 0.01 |
| Charlson Comorbidity Index | 1.14 | 0.98, 1.32 | 0.09 |
| ICU status | |||
| ICU | |||
| Non-ICU | 0.85 | 0.39, 1.82 | 0.7 |
| Blood culture | |||
| Negative | |||
| Positive | 0.67 | 0.28, 1.60 | 0.4 |
| Bile culture | |||
| Negative | |||
| Positive | 2.07 | 0.95, 4.49 | 0.07 |
| Characteristic | HR | 95%CI | P value |
| PCT placement interval | |||
| Early | |||
| Late | 2.51 | 1.21-5.18 | 0.01 |
| Age | 1.05 | 1.01-1.09 | 0.01 |
| Charlson Comorbidity Index | 1.11 | 0.96-1.29 | 0.2 |
| ICU status | |||
| ICU | |||
| Non-ICU | 0.93 | 0.44-2.00 | 0.9 |
| Blood culture | |||
| Negative | |||
| Positive | 0.67 | 0.28-1.60 | 0.4 |
| Bile culture | |||
| Negative | |||
| Positive | 2.21 | 1.02-4.79 | 0.04 |
Acute cholecystitis remains a common cause of surgical admissions and LC is the standard of care for the majority of patients. However, in elderly or critically ill individuals with significant comorbidities, the perioperative risk of LC is high[1]. In this context, PCT serves as a temporary and, in very high risk cases, a definitive long-term management strategy[4]. This study evaluated the timing of placement, catheter management, survival, and long-term outcomes in a community hospital serving a population with a high-comorbidity burden, aiming to contextualize our findings within current evidence and guidelines.
Although recently several studies and randomized controlled trials, like the CHOCOLATE trial suggest that early LC may provide superior long-term outcomes compared with PCT[7], the overall consensus remains aligned with the most recent international guidelines. The CHOCOLATE randomized trial demonstrated that LC is superior to PCT in high-risk acute cholecystitis, with lower rates of major complications, recurrent biliary events, and readmissions. In comparison, our findings reflect real-world practice in a safety-net population in which many patients were not operative candidates at presentation and PCT served as the only feasible source-control strategy. Within this context, the survival advantage associated with early PCT in our cohort reflects the timing of drainage among non-surgical candidates rather than a direct comparison between PCT and cholecystectomy. The Tokyo Guidelines 2018 recommend PCT as a safe and effective bridging therapy, particularly for patients with Grade II and III acute cholecystitis that are high-risk surgical candidates or require stabilization before definitive surgery. Of note, patient selection and disease severity play major roles in determining outcomes.
Acalculous cholecystitis is less common, but predominantly affects critically ill patients and is associated with increased morbidity and mortality, making timely drainage particularly important in this population[8].
Although many trials and studies are in consensus with the Tokyo Guidelines 2018, emphasizing PCT placement as a bridging intervention with scheduled reassessment for tube removal or interval LC, national data consistently demon
The approach to PCT, most commonly transhepatic or transperitoneal, has several implications in terms of complication rates, catheter stability, and subsequent management. Transhepatic access offers a more secure catheter tract and decreased rates of bile leakage, while transperitoneal access may be preferred due to operator choice or in patients with coagulopathy or variant hepatic anatomy. Current evidence suggests comparable overall success rates between the two approaches, but higher dislodgement rates with the transperitoneal route[12,13]. In this study, the majority of PCT place
Early source control is a central principle in the management of sepsis and severe infections, while delayed intervention is associated with increased mortality. In acute cholecystitis, timely PCT can halt the inflammatory process, prevent gallbladder gangrene or perforation, and reduce the risk of systemic complications such as septic shock or multi-organ failure[1].
Current evidence shows that delayed PCT (defined as PCT placement performed more than 3-4 days from hospital admission) is associated with significantly higher mortality[14]. Khasawneh et al[15] reported an approximately two-fold higher 30-day mortality when PCT was delayed beyond 4 days, even after adjusting for comorbidities. In this study, both calculous and acalculous cholecystitis cases were managed according to guidelines, with a median 2-day interval from admission to PCT.
Prolonged inflammation may result in a more technically challenging drainage procedure, longer catheter dwell time, higher rate of tube occlusion or dislodgement, and an increased risk of recurrent cholecystitis while awaiting further intervention[5,16].
In this study, delayed PCT (> 4 days from admission) was performed in almost one third of the population (49/174) and was associated with a significantly higher overall mortality compared with early PCT (35% vs 15%). Furthermore, delayed intervention was associated with longer catheter dwell times, suggesting that late drainage may also affect patients’ quality of life. The median time to PCT was 2 days, aligning with best-practice recommendations and potentially contributing to improved outcomes in early-treated patients. These findings reinforce the necessity of establishing institutional protocols that prioritize PCT placement within 72 hours of presentation for high-risk patients with acute cholecy
The optimal dwell time for PCT catheters remains a subject of ongoing debate[14]. Prolonged catheterization increases the risk of tube-related complications, including infection, bile leakage, occlusion, dislodgement, and local skin irritation, whereas premature removal is associated with recurrence of biliary stasis and inflammation due to cystic duct obstruction[17]. Tokyo Guidelines 2018 suggest maintaining the catheter for at least three weeks to facilitate tract maturation and resolution of inflammation[1]. However, the concept of “dual timing” has emerged, including the minimum safe dwell time to avoid complications related to premature removal and the maximum advisable dwell time beyond which risks of infection and patient discomfort increase substantially[18]. Lin et al[19] suggest the upper safe limit to be around 6–8 weeks, as extended dwell times have been linked to increased infection rates, granulation tissue formation, and difficult tube removal.
Catheter care is of utmost importance to prevent complications and maintain patency. Standard practice involves securing the tube without tension, maintaining a clean and dry exit site via local wound care, and performing daily insertion site inspections for signs of infection or bile leakage. Routine catheter irrigation, typically with 5-10 mL of sterile saline solution once or twice daily, is recommended to prevent occlusion and to maintain bile drainage[16]. Patients and caregivers should be educated on proper irrigation technique, dressing changes, local wound care, and early signs of catheter dysfunction, such as new onset pain, bile leakage, or decreased output. Detailed written instructions and follow-up calls may improve adherence and reduce emergency department visits for tube-related issues.
In this study, the median catheter dwell time is within the minimum and maximum advisable dwell times. However, this finding is the result of appropriate management of real-world challenges in a safety-net hospital, including patient and family education limitations, variable compliance with outpatient follow-up visits, especially for elderly patients, delays in scheduling cholangiography, and collaboration barriers between interventional radiology, medical, and surgical services.
ICU admission at the time of PCT placement is generally considered a marker of disease severity and has been associated with worse outcomes[20]. In our cohort, ICU admission was associated with longer dwell times, possibly due to delayed recovery/disease severity, though it was not associated with higher overall mortality.
Based on these study findings, a standardized removal protocol-including three-week to six weeks dwell time, routine catheter irrigation schedule, and timely cholangiographic confirmation of cystic duct patency prior to removal - may reduce complications and facilitate timely catheter discontinuation. However, cholangiography practices varied considerably (65% of patients underwent at least one tract-maturation study). Furthermore, inconsistent access to follow-up imaging and outpatient coordination likely contributed to ongoing catheter dependence in this population. These observations highlight the need for standardized, protocol-driven use of cholangiography to improve long-term outcomes after PCT.
Definitive cholecystectomy after stabilization with PCT is a critical step in the long-term management of acute cholecy
The need for interval LC after PCT varies depending on whether the underlying diagnosis is calculous or acalculous cholecystitis. In calculous cholecystitis, recurrence rates can be as high as 40% without surgery, as the gallbladder remains in place and is a persistent source of obstruction and inflammation. Definitive removal is generally recommended once the patient is stable. In contrast, acalculous cholecystitis, which often occurs in critically ill or postoperative patients, is a secondary phenomenon driven by ischemia, stasis, or systemic inflammation rather than mechanical obstruction. Once acalculous cholecystitis resolves, the gallbladder may return to normal function, and the risk of recurrence is substantially lower, making routine cholecystectomy less beneficial in the context of multiple comorbidities[16,24].
In this study, among patients with calculous cholecystitis, interval LC was performed in 31% (n = 49/159) of the population with acute calculous cholecystitis and 47% (n = 7/15) patients with acalculous cholecystitis. The most common barriers in patients that did not have surgery included persistent medical inoperability, patient refusal, and loss to follow-up. In a safety-net or community hospital setting, logistical and socioeconomic factors may delay or prevent appropriate follow-up and access to surgery. To address these barriers, structured reassessment pathways should be established. Such protocols might include scheduling LC at the time of discharge or PCT removal, implementing multidisciplinary review meetings to reassess surgical candidacy, and integrating patient navigation services to minimize loss to follow-up.
According to the most recent international guidelines, including the Tokyo Guidelines 2018[1] and the 2020 update from the WSES[2], there is no universally defined timeframe for interval cholecystectomy after PCT. These guidelines advocate for a bridge-to-surgery strategy, recommending that definitive cholecystectomy be performed once the patient’s overall condition has stabilized and any comorbidities have been appropriately optimized. This approach favors clinical judgment over defined intervals. Recent evidence, however, has sought to determine practical timing windows. Several studies have reported that operating too early - within the first two to four weeks after drainage - may increase operative difficulty and complication rates, while prolonged delays beyond eight weeks may predispose patients to recurrent biliary events. In summary, the current literature suggests that performing LC at approximately four to eight weeks after PCT provides a balanced and generally safe interval for most patients that are medically optimized for surgery[14,16].
Despite the aforementioned findings, it is worth noting that recent evidence points toward a gradual shift in the management of severe acute cholecystitis. While the latest guidelines recommend PCT as the preferred treatment for high-risk patients with acute cholecystitis[2,3], emerging evidence suggests that this paradigm may be changing. Based on advancements in laparoscopic techniques, improved perioperative optimization, next-generation antibiotic regimens, and enhanced intraoperative navigation systems, an increasing number of recently published studies have demonstrated the feasibility and safety of early LC in high-risk populations. In carefully selected patients, early surgical intervention may be considered as an alternative to percutaneous drainage. However, further experience, larger prospective trials, and long-term outcome data are still required before early LC can be considered as the new standard of care in high-risk surgical candidates[7,20,25].
Mortality after PCT reflects both the severity of acute cholecystitis and the underlying comorbidity burden of the patient population. Reported short-term mortality rates vary widely in the literature, ranging from 5% in relatively stable, selected patients to over 30% in critically ill or ICU populations[4,20]. The overall mortality rate of 21% in this study is consistent with prior reports and likely reflects a high-comorbidity, safety-net hospital cohort with a median CCI of 5. Higher CCI scores have been linked to decreased survival after PCT[26]. In the present study, patients with a CCI ≥ 6 had markedly higher mortality rates, underscoring the value of formal comorbidity scoring in risk stratification and evidence-based decision-making. Incorporating CCI into multidisciplinary discussions may help identify patients who would benefit the most from early intervention compared to those for whom PCT may be a bridge to palliative management.
In summary, our findings and prior evidence emphasize that mortality risk after PCT is a composite reflection of disease severity, burden of comorbidities, and timing of intervention. Standardizing early intervention protocols, integrating objective risk scores such as CCI, and prioritizing prompt drainage in deteriorating patients, regardless of ICU status, may improve survival in this high-risk population.
The retrospective nature of this study limits the generalizability of our findings and may be subject to selection bias and incomplete follow-up. Although a 12-year consecutive cohort was included, variations in practice patterns, documen
These findings highlight several important considerations in the management of high-risk acute cholecystitis with PCT. Prioritizing early PCT, ideally within 72 hours of presentation, is consistently associated with improved survival and should be incorporated into institutional protocols. Standardized catheter management pathways, such as maintaining a minimum dwell time of three weeks and confirming patency of the entire biliary tree (including the common bile duct) via cholangiography prior to removal, may reduce complications and improve patient outcomes. For patients stabilized after PCT performed for calculous cholecystitis, a definitive interval LC should be planned when clinically feasible; in contrast, routine LC after acalculous cholecystitis may not always be necessary and should be individualized. Reducing follow-up loss is critical, and while dedicated outpatient PCT clinics or structured navigation pathways may be feasible in high-volume centers, smaller or lower-volume practices may need to adapt their follow-up strategies to local resources. Optimal outcomes require an integrated multidisciplinary model involving surgery, interventional radiology, geriatrics, and primary care providers to ensure shared decision-making and continuity of care. Future research should focus on prospective, multicenter studies to define the optimal catheter dwell time, establish evidence-based criteria for early removal, and clarify the ideal timing and patient selection for interval LC vs lifelong catheter dependency. Further evidence is needed to evaluate strategies to reduce follow-up loss, assess the impact of structured outpatient pathways, and measure quality-of-life outcomes following PCT, particularly in diverse practice environments and safety-net popula
| 1. | Gallaher JR, Charles A. Acute Cholecystitis: A Review. JAMA. 2022;327:965-975. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 320] [Cited by in RCA: 243] [Article Influence: 60.8] [Reference Citation Analysis (1)] |
| 2. | Pisano M, Allievi N, Gurusamy K, Borzellino G, Cimbanassi S, Boerna D, Coccolini F, Tufo A, Di Martino M, Leung J, Sartelli M, Ceresoli M, Maier RV, Poiasina E, De Angelis N, Magnone S, Fugazzola P, Paolillo C, Coimbra R, Di Saverio S, De Simone B, Weber DG, Sakakushev BE, Lucianetti A, Kirkpatrick AW, Fraga GP, Wani I, Biffl WL, Chiara O, Abu-Zidan F, Moore EE, Leppäniemi A, Kluger Y, Catena F, Ansaloni L. 2020 World Society of Emergency Surgery updated guidelines for the diagnosis and treatment of acute calculus cholecystitis. World J Emerg Surg. 2020;15:61. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 435] [Cited by in RCA: 338] [Article Influence: 56.3] [Reference Citation Analysis (0)] |
| 3. | Overby DW, Apelgren KN, Richardson W, Fanelli R; Society of American Gastrointestinal and Endoscopic Surgeons. SAGES guidelines for the clinical application of laparoscopic biliary tract surgery. Surg Endosc. 2010;24:2368-2386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 242] [Cited by in RCA: 204] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 4. | Winbladh A, Gullstrand P, Svanvik J, Sandström P. Systematic review of cholecystostomy as a treatment option in acute cholecystitis. HPB (Oxford). 2009;11:183-193. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 293] [Cited by in RCA: 258] [Article Influence: 15.2] [Reference Citation Analysis (0)] |
| 5. | Colonna AL, Griffiths TM, Robison DC, Enniss TM, Young JB, McCrum ML, Nunez JM, Nirula R, Hardman RL. Cholecystostomy: Are we using it correctly? Am J Surg. 2019;217:1010-1015. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (1)] |
| 6. | Grambsch PM, Therneau TM. Proportional hazards tests and diagnostics based on weighted residuals. Biometrika. 1994;81:515-526. [DOI] [Full Text] |
| 7. | Loozen CS, van Santvoort HC, van Duijvendijk P, Besselink MG, Gouma DJ, Nieuwenhuijzen GA, Kelder JC, Donkervoort SC, van Geloven AA, Kruyt PM, Roos D, Kortram K, Kornmann VN, Pronk A, van der Peet DL, Crolla RM, van Ramshorst B, Bollen TL, Boerma D. Laparoscopic cholecystectomy versus percutaneous catheter drainage for acute cholecystitis in high risk patients (CHOCOLATE): multicentre randomised clinical trial. BMJ. 2018;363:k3965. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 242] [Cited by in RCA: 203] [Article Influence: 25.4] [Reference Citation Analysis (1)] |
| 8. | Barie PS, Eachempati SR. Acute acalculous cholecystitis. Gastroenterol Clin North Am. 2010;39:343-357, x. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 160] [Article Influence: 10.0] [Reference Citation Analysis (1)] |
| 9. | Melloul E, Denys A, Demartines N, Calmes JM, Schäfer M. Percutaneous drainage versus emergency cholecystectomy for the treatment of acute cholecystitis in critically ill patients: does it matter? World J Surg. 2011;35:826-833. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 84] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 10. | Wadhwa V, Trivedi PS, Makary MS, Strain DV, Ahmed O, Chick JFB, Charalel RA. Utilization and Outcomes of Cholecystostomy and Cholecystectomy in Patients Admitted With Acute Cholecystitis: A Nationwide Analysis. AJR Am J Roentgenol. 2021;216:1558-1565. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 11. | Podder S, Lung K, Ibrahim G, Koeneman S, Marks J, Cohen M, Kohli A. Barriers to interval cholecystectomy following percutaneous cholecystostomy in patients with acute calculous cholecystitis. Surg Endosc. 2025;39:8488-8497. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 12. | Jenkins PE, Maccormick A, Zhong J, Makris GC, Gafoor N, Chan D; on behalf of the MACAFI Collaborators. Transhepatic or transperitoneal technique for cholecystostomy: results of the multicentre retrospective audit of cholecystostomy and further interventions (MACAFI). Br J Radiol. 2023;96:20220279. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 13. | Bennett S, Shaida N, Godfrey E, Safranek P, O'Neill JR. A comparison of transhepatic versus transperitoneal cholecystostomy for acute calculous cholecystitis: a 5-year experience. J Surg Case Rep. 2021;2021:rjab410. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 14. | Woodward SG, Rios-Diaz AJ, Zheng R, McPartland C, Tholey R, Tatarian T, Palazzo F. Finding the Most Favorable Timing for Cholecystectomy after Percutaneous Cholecystostomy Tube Placement: An Analysis of Institutional and National Data. J Am Coll Surg. 2021;232:55-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (1)] |
| 15. | Khasawneh MA, Shamp A, Heller S, Zielinski MD, Jenkins DH, Osborn JB, Morris DS. Successful laparoscopic cholecystectomy after percutaneous cholecystostomy tube placement. J Trauma Acute Care Surg. 2015;78:100-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 2.1] [Reference Citation Analysis (2)] |
| 16. | Vaishnavi KB, Rice C, Khoo K, Rana M, Ahmad F, Higgins E, Williams TP, Chao C, Mileski WJ. Clinical outcomes of high-risk patients treated with percutaneous cholecystostomy tube drainage: application of comorbidity scores in a retrospective cohort study. Dig Med Res. 2021;4:66-66. [DOI] [Full Text] |
| 17. | Alvino DML, Fong ZV, McCarthy CJ, Velmahos G, Lillemoe KD, Mueller PR, Fagenholz PJ. Long-Term Outcomes Following Percutaneous Cholecystostomy Tube Placement for Treatment of Acute Calculous Cholecystitis. J Gastrointest Surg. 2017;21:761-769. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 60] [Article Influence: 6.7] [Reference Citation Analysis (1)] |
| 18. | Curry J, Chervu N, Cho NY, Hadaya J, Vadlakonda A, Kim S, Keeley J, Benharash P. Percutaneous cholecystostomy tube placement as a bridge to cholecystectomy for grade III acute cholecystitis: A national analysis. Surg Open Sci. 2024;18:6-10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 19. | Lin MH, Ni CF, Chiang HJ, Chen YT, Tsai CC, Chen YC, Huang SW, Chen YT, Wu JC, Cheng SJ, Tsai RJ, Chuang KI, Chen YC, Chiang Y, Hsieh KL. Optimal Timing of Percutaneous Cholecystostomy across Different Grades of Acute Cholecystitis: A Retrospective Cohort Study. J Vasc Interv Radiol. 2025;36:1105-1112.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 20. | Smith TJ, Manske JG, Mathiason MA, Kallies KJ, Kothari SN. Changing trends and outcomes in the use of percutaneous cholecystostomy tubes for acute cholecystitis. Ann Surg. 2013;257:1112-1115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 41] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 21. | Kaya C, Bozkurt E, Ömeroğlu S, Yazıcı P, İdiz UO, Tabakçı ÖN, Bostancı Ö, Mihmanlı M. Is Interval Cholecystectomy Necessary After Percutaneous Cholecystostomy in High-Risk Acute Cholecystitis Patients? Sisli Etfal Hastan Tip Bul. 2018;52:13-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 22. | Hung YL, Sung CM, Fu CY, Liao CH, Wang SY, Hsu JT, Yeh TS, Yeh CN, Jan YY. Management of Patients With Acute Cholecystitis After Percutaneous Cholecystostomy: From the Acute Stage to Definitive Surgical Treatment. Front Surg. 2021;8:616320. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 42] [Article Influence: 8.4] [Reference Citation Analysis (1)] |
| 23. | Kourounis G, Rooke ZC, McGuigan M, Georgiades F. Systematic review and meta-analysis of early vs late interval laparoscopic cholecystectomy following percutaneous cholecystostomy. HPB (Oxford). 2022;24:1405-1415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 24. | Fanciulli G, Favara G, Maugeri A, Barchitta M, Agodi A, Basile G. Comparing percutaneous treatment and cholecystectomy outcomes in acute cholecystitis patients: a systematic review and meta-analysis. World J Emerg Surg. 2025;20:50. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 10] [Article Influence: 10.0] [Reference Citation Analysis (1)] |
| 25. | Huang H, Zhang H, Yang D, Wang W, Zhang X. Percutaneous cholecystostomy versus emergency cholecystectomy for the treatment of acute calculous cholecystitis in high-risk surgical patients: a meta-analysis and systematic review. Updates Surg. 2022;74:55-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 26. | Flexer SM, Peter MB, Durham-Hall AC, Ausobsky JR. Patient outcomes after treatment with percutaneous cholecystostomy for biliary sepsis. Ann R Coll Surg Engl. 2014;96:229-233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/