Published online Feb 27, 2026. doi: 10.4240/wjgs.v18.i2.114607
Revised: December 4, 2025
Accepted: December 24, 2025
Published online: February 27, 2026
Processing time: 121 Days and 1.9 Hours
Malnutrition is highly prevalent in gastric cancer patients receiving adjuvant chemotherapy and may compromise treatment completion and survival out
To investigate the impact of nutritional status on treatment completion and prognosis during adjuvant chemotherapy following gastric cancer surgery, providing scientific evidence for clinical nutritional intervention strategies.
A retrospective analysis was conducted on clinical data of 80 patients who received adjuvant chemotherapy after gastric cancer surgery from January 2020 to June 2024. Nutritional status was assessed using Nutritional Risk Screening 2002 (NRS2002), Controlling Nutritional Status, Prognostic Nutritional Index (PNI), and Glasgow Prognostic Score before and after chemotherapy. Patients were divided into nutritional risk group (NRS2002 ≥ 3 points, n = 37) and non-nutritional risk group (< 3 points, n = 43) based on NRS2002 scores. Treatment completion, adverse reactions, and survival prognosis were evaluated. Logistic regression analysis was used to identify risk factors affecting treatment comple
Thirty-seven patients (46.2%) had nutritional risk before chemotherapy. The nutritional risk group had significantly lower treatment completion rate compared to the non-nutritional risk group (75.7% vs 95.3%, P = 0.009), insufficient relative dose intensity (78.6% ± 14.2% vs 92.1% ± 8.7%, P < 0.001), and significantly higher incidence rates of ≥ grade 3 hematologic and non-hematologic toxicities (P < 0.05). With a median follow-up of 28 months, the nutritional risk group had significantly lower 3-year disease-free survival (DFS) and overall survival (OS) rates compared to the non-nutritional risk group (62.1% vs 83.7%, P = 0.018; 72.4% vs 90.7%, P = 0.023). Multivariate analysis showed that NRS2002 ≥ 3 points was an independent risk factor for treatment completion [odds ratio = 4.829, 95% confidence interval (CI): 1.542-15.114, P = 0.007], DFS [hazard ratio (HR) = 2.847, 95%CI: 1.124-7.214, P = 0.027], and OS (HR = 3.524, 95%CI: 1.089-11.404, P = 0.036).
Nutritional status significantly affects treatment completion and prognosis in gastric cancer patients receiving postoperative adjuvant chemotherapy. Both NRS2002 and PNI demonstrate important predictive value, with NRS2002 showing the most consistent performance in predicting treatment completion and survival outcomes. Clinical practice should emphasize nutritional risk assessment and dynamic monitoring, and develop individualized nutritional intervention strategies to improve chemotherapy completion rates and patient outcomes.
Core Tip: This study investigated the impact of nutritional status on treatment completion and prognosis in gastric cancer patients undergoing adjuvant chemotherapy after surgery. Nutritional Risk Screening 2002 ≥ 3 was identified as an independent risk factor for reduced chemotherapy completion and poor survival. Patients with nutritional risk experienced more severe toxicities, lower relative dose intensity, and worse 3-year disease-free and overall survival. Routine nutritional risk assessment and timely intervention may improve chemotherapy adherence and outcomes in gastric cancer patients.
- Citation: Zhou K, Tu RF, Lu LH, Zhang H. Impact of nutritional status on treatment completion and prognosis during adjuvant chemotherapy following gastric cancer surgery. World J Gastrointest Surg 2026; 18(2): 114607
- URL: https://www.wjgnet.com/1948-9366/full/v18/i2/114607.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i2.114607
Gastric cancer is one of the most common gastrointestinal malignancies worldwide, ranking 5th in incidence and 4th in mortality among malignant tumors, posing a serious threat to human health[1]. China ranks among the highest globally in both gastric cancer incidence and mortality, with approximately 400000 new cases and 300000 deaths annually, creating a substantial disease burden[2]. Surgical resection remains the primary means of curative treatment for gastric cancer; however, even after radical resection, a considerable proportion of patients still experience recurrence and metastasis. Postoperative adjuvant chemotherapy has been proven to significantly improve disease-free survival (DFS) and overall survival (OS) in gastric cancer patients, making it an important component of comprehensive gastric cancer treatment[3].
Malnutrition is prevalent in malignant tumor patients, with an incidence rate of 40%-80%. Gastric cancer patients have an even higher incidence of malnutrition due to factors such as tumor consumption, surgical trauma, and postoperative digestive function changes[4]. Nutritional status not only affects patients' quality of life but is also closely related to treatment tolerance, complication rates, treatment completion, and long-term prognosis. Studies have shown that malnourished tumor patients have increased chemotherapy adverse reaction rates and decreased treatment compliance, leading to dose reductions or treatment interruptions that ultimately affect treatment efficacy[5].
Currently, there are numerous tools for assessing nutritional status in tumor patients clinically, including Nutritional Risk Screening 2002 (NRS2002), Controlling Nutritional Status (CONUT) score, Prognostic Nutritional Index (PNI), and Glasgow Prognostic Score (GPS). These assessment tools reflect patients' nutritional status and inflammatory response levels from different perspectives and have important value in predicting tumor patient prognosis[6]. However, systematic studies on the dynamic changes in nutritional status during postoperative adjuvant chemotherapy for gastric cancer and their impact on treatment completion and prognosis are relatively scarce, and the value of different nutritional assessment tools in predicting treatment completion during adjuvant chemotherapy in gastric cancer patients needs further validation[7].
Therefore, this study retrospectively analyzed clinical data from gastric cancer patients receiving postoperative adjuvant chemotherapy, used multiple nutritional assessment tools to evaluate changes in patients' nutritional status before and after chemotherapy, and explored the impact of nutritional status on chemotherapy completion, adverse reaction occurrence, and long-term prognosis. This provides scientific evidence for developing individualized nutritional intervention strategies in clinical practice to improve gastric cancer patients' adjuvant chemotherapy completion rates and treatment efficacy.
This study was a single-center retrospective study that explored the impact of nutritional status on treatment completion and prognosis by analyzing clinical data from gastric cancer patients receiving postoperative adjuvant chemotherapy.
Eighty gastric cancer patients who received postoperative adjuvant chemotherapy at our hospital from January 2020 to June 2024 were selected as study subjects.
Inclusion criteria: (1) Pathologically confirmed gastric adenocarcinoma; (2) Received radical gastric cancer resection (R0 resection); (3) Postoperative pathological staging of Ib-III (AJCC 8th edition TNM staging); (4) Started standardized adjuvant chemotherapy within 4-8 weeks postoperatively; (5) Age 18-80 years; (6) Eastern Cooperative Oncology Group performance status score 0-2; (7) Basically normal major organ function; and (8) Complete clinical data and comprehensive follow-up data.
Exclusion criteria: (1) Received preoperative neoadjuvant chemotherapy or radiotherapy; (2) Concurrent or previous other malignant tumors; (3) Severe heart, lung, liver, kidney, or other major organ dysfunction; (4) Severe infection, immunodeficiency diseases, or autoimmune diseases; (5) Pregnant or lactating women; (6) Mental illness preventing treatment cooperation; and (7) Lost to follow-up or follow-up time less than 12 months.
The following patient data were retrospectively collected through the hospital electronic medical record system.
General clinical data: Gender, age, height, weight, body mass index (BMI), smoking history (years of smoking and daily cigarette consumption), drinking history (years of drinking and daily alcohol intake), comorbidities (hypertension, diabetes, cardiovascular and cerebrovascular diseases, etc.).
Tumor-related data: Tumor location (gastric fundus, body, antrum, total stomach), gross type (ulcerative, fungating, infiltrative, stenotic), histological type (high, moderate, low differentiated adenocarcinoma, signet ring cell carcinoma, mucinous adenocarcinoma, etc.), degree of differentiation, depth of invasion (T stage), lymph node metastasis (N stage), TNM staging, vascular invasion, neural invasion, preoperative tumor marker levels [carcinoembryonic antigen (CEA), carbohydrate antigen 19-9, carbohydrate antigen 72-4], etc.
Nutritional status assessment was performed before chemotherapy initiation (baseline) and after chemotherapy completion (endpoint), using standardized evaluation during the same time period (morning fasting state).
NRS2002: The NRS2002 scale assesses two dimensions: Degree of nutritional status impairment and disease severity: (1) Nutritional status impairment score (0-3 points): 0 points for normal nutritional status; 1 point for weight loss > 5% in recent 3 months, or food intake 50%-75% of normal requirements in recent week; 2 points for weight loss > 5% in recent 2 months, or food intake 25%-50% of normal requirements in recent week, or BMI 18.5-20.5 kg/m2 with impaired general condition; 3 points for weight loss > 5% in recent month (or > 15% in recent 3 months), or food intake 0%-25% of normal requirements in recent week, or BMI < 18.5 kg/m²; (2) Disease severity score (0-3 points): 0 points for normal nutritional needs; 1 point for hip fracture, chronic disease with acute complications, cirrhosis, chronic obstructive pulmonary disease, hemodialysis, diabetes; 2 points for major surgery, stroke, severe pneumonia, hematologic malignancy, solid malignancy; 3 points for head trauma, bone marrow transplant, critically ill patients (intensive care unit); and (3) Age adjustment: Add 1 point for patients ≥ 70 years. Total score = nutritional status impairment score + disease severity score + age adjustment score, with ≥ 3 points indicating nutritional risk.
CONUT score: CONUT score assesses nutritional status based on three indicators: Serum albumin (ALB), total cholesterol, and total lymphocyte count: (1) Serum ALB (g/L) scoring: ≥ 35 scores 0 points, 30-34.9 scores 2 points, 25-29.9 scores 4 points, < 25 scores 6 points; (2) Total cholesterol (mmol/L) scoring: ≥ 4.66 scores 0 points, 3.63-4.65 scores 1 point, 2.60-3.62 scores 2 points, < 2.60 scores 3 points; and (3) Total lymphocyte count (× 109/L) scoring: ≥ 1.60 scores 0 points, 1.20-1.59 scores 1 point, 0.80-1.19 scores 2 points, < 0.80 scores 3 points. Total score is the sum of three indicator scores (0-12 points), where 0-1 points indicates normal nutrition, 2-4 points indicates mild malnutrition, 5-8 points indicates moderate malnutrition, and 9-12 points indicates severe malnutrition.
PNI: PNI calculation formula: PNI = serum ALB (g/L) + 5 × total lymphocyte count (× 109/L). This index comprehensively reflects patients' protein nutritional status and immune function level, with PNI ≥ 45 indicating normal nutrition and PNI < 45 indicating malnutrition. Studies have shown that PNI is closely related to tumor patient prognosis and is an effective tool for assessing nutritional status and predicting prognosis.
GPS: GPS assesses patients' systemic inflammatory nutritional status based on C-reactive protein (CRP) and serum ALB levels: (1) CRP ≤ 10 mg/L and ALB ≥ 35 g/L scores 0 points (low risk); (2) CRP > 10 mg/L or ALB < 35 g/L (only one abnormal) scores 1 point (moderate risk); and (3) CRP > 10 mg/L and ALB < 35 g/L (both abnormal) scores 2 points (high risk). GPS combines inflammatory response with nutritional status, better reflecting the overall health status of tumor patients and is an important indicator for predicting prognosis.
Weight loss percentage calculation: Weight loss percentage (%) = (preoperative weight - current weight)/preoperative weight × 100%.
Treatment completion assessment used multidimensional indicators including relative dose intensity (RDI), cycle completion rate, and treatment compliance for comprehensive evaluation: (1) RDI calculation: RDI (%) = (actual dose/standard dose) × (actual treatment time/standard treatment time) × 100%, with RDI ≥ 85% defined as adequate dose intensity and RDI < 85% defined as inadequate dose intensity; (2) Cycle completion rate assessment: Statistics of the proportion of actual completed chemotherapy cycles to planned cycles, with completion rate ≥ 80% defined as high completion, 60%-79% as moderate completion, and < 60% as low completion; and (3) Dose adjustment assessment: Recording dose reductions (reduction ≥ 20%), treatment delays (delay time ≥ 7 days), and early termination due to adverse reactions or poor patient tolerance.
Safety indicators: Chemotherapy-related adverse reactions were assessed for severity (grades 1-5) according to the National Cancer Institute Common Terminology Criteria for Adverse Events version 5.0, recording incidence rates of each grade level, with particular focus on ≥ grade 3 severe adverse reactions. Specific categories include: (1) Hematologic toxicity: Leukopenia, neutropenia, thrombocytopenia, anemia; and (2) Non-hematologic toxicity: Nausea and vomiting, diarrhea, mucositis, hand-foot syndrome, peripheral neuropathy, hepatic dysfunction, renal dysfunction, etc.
Efficacy indicators: (1) DFS: Time interval from chemotherapy start date to first detection of tumor recurrence, distant metastasis, or death from any cause; (2) OS: Time interval from chemotherapy start date to death from any cause or last follow-up; and (3) Chemotherapy completion rate: Proportion of patients who actually completed planned chemotherapy cycles.
Follow-up was conducted through outpatient visits combined with telephone follow-up. After chemotherapy completion, follow-up was conducted at the following time points: Every 3 months for the first 2 years, every 6 months for years 3-5, and annually after 5 years.
Data analysis was performed using SPSS 27.0 statistical software. Quantitative data were tested for normality using the Shapiro-Wilk test. Those conforming to normal distribution were expressed as mean ± SD and compared between groups using independent samples t-test; those with non-normal distribution were expressed as median (interquartile range) [M (P25, P75)] and analyzed using Mann-Whitney U test. Categorical data were expressed as n (%), with group comparisons using χ² test or Fisher's exact test.
Univariate and multivariate logistic regression analyses were used to analyze risk factors affecting chemotherapy completion, with results expressed as odds ratio (OR) and 95% confidence interval (CI).
Survival analysis used the Kaplan-Meier method to estimate survival curves, with log-rank test comparing survival differences between groups. Cox proportional hazards regression model was used to analyze independent prognostic factors, with Schoenfeld residual test validating proportional hazards assumptions. Results were expressed as hazard ratio (HR) and 95%CI. For survival analysis, patients lost to follow-up were treated as censored data, with censoring time as the last follow-up time.
Multivariate analysis used stepwise forward method, including variables with P < 0.10 in univariate analysis, with exclusion criterion of P > 0.10. Specifically, candidate variables included age (≥ 65 years), BMI (< 22 kg/m²), NRS2002 score (≥ 3 points), CONUT score (≥ 5 points), PNI (< 45), GPS score (≥ 1 point), TNM stage (III), and preoperative CEA (≥ 5 ng/mL). Variables that did not reach statistical significance in the final model were excluded during the stepwise selection process, including age, BMI, CONUT score, GPS score, and preoperative CEA levels. All statistical tests were two-sided, with P < 0.05 considered statistically significant.
This research protocol was approved by our hospital's ethics committee and complies with the ethical requirements of the Declaration of Helsinki. Due to the retrospective nature of the study, the ethics committee agreed to waive patient informed consent after review, but strictly protected patient privacy information. All data underwent de-identification processing for use only in this research and were not disclosed to third parties. The research process strictly followed medical research ethical principles to ensure patient information security.
This study included 80 gastric cancer patients receiving postoperative adjuvant chemotherapy, including 49 males (61.2%) and 31 females (38.8%). Patient ages ranged from 39-78 years, with a median age of 62 years. Based on NRS2002 scores before chemotherapy initiation, 37 patients (46.2%) were in the nutritional risk group and 43 patients (53.8%) were in the non-nutritional risk group. No statistically significant differences were found between the two groups in general data such as gender, age, smoking history, and drinking history (P > 0.05). The nutritional risk group had significantly lower BMI compared to the non-nutritional risk group, with statistically significant difference (P = 0.007). Regarding tumor-related data, there was a trend toward difference in TNM staging distribution (P = 0.054), with a higher proportion of stage III patients in the nutritional risk group (Table 1).
| Item | Total (n = 80) | Nutritional risk group (n = 37) | Non-nutritional risk group (n = 43) | χ²/t | P value |
| General clinical data | |||||
| Gender | 0.089 | 0.765 | |||
| Male | 49 (61.2) | 23 (62.2) | 26 (60.5) | ||
| Female | 31 (38.8) | 14 (37.8) | 17 (39.5) | ||
| Age (years) | 62.4 ± 11.2 | 63.8 ± 10.9 | 61.2 ± 11.4 | 1.084 | 0.282 |
| BMI (kg/m2) | 22.3 ± 3.8 | 21.1 ± 3.4 | 23.3 ± 3.9 | -2.765 | 0.007 |
| Smoking history | 34 (42.5) | 17 (45.9) | 17 (39.5) | 0.331 | 0.565 |
| Drinking history | 28 (35.0) | 14 (37.8) | 14 (32.6) | 0.237 | 0.626 |
| Comorbidities | |||||
| Hypertension | 31 (38.8) | 16 (43.2) | 15 (34.9) | 0.579 | 0.447 |
| Diabetes | 18 (22.5) | 9 (24.3) | 9 (20.9) | 0.125 | 0.724 |
| Cardiovascular disease | 12 (15.0) | 7 (18.9) | 5 (11.6) | 0.835 | 0.361 |
| Tumor-related data | |||||
| Tumor location | 2.341 | 0.505 | |||
| Fundus | 8 (10.0) | 5 (13.5) | 3 (7.0) | ||
| Body | 22 (27.5) | 11 (29.7) | 11 (25.6) | ||
| Antrum | 42 (52.5) | 18 (48.6) | 24 (55.8) | ||
| Total stomach | 8 (10.0) | 3 (8.1) | 5 (11.6) | ||
| Gross type | 3.247 | 0.355 | |||
| Ulcerative | 45 (56.2) | 22 (59.5) | 23 (53.5) | ||
| Fungating | 18 (22.5) | 8 (21.6) | 10 (23.3) | ||
| Infiltrative | 14 (17.5) | 5 (13.5) | 9 (20.9) | ||
| Stenotic | 3 (3.8) | 2 (5.4) | 1 (2.3) | ||
| Histological type | 4.892 | 0.180 | |||
| Well-differentiated adenocarcinoma | 15 (18.8) | 5 (13.5) | 10 (23.3) | ||
| Moderately-differentiated adenocarcinoma | 28 (35.0) | 12 (32.4) | 16 (37.2) | ||
| Poorly-differentiated adenocarcinoma | 31 (38.7) | 17 (45.9) | 14 (32.6) | ||
| Signet ring cell carcinoma | 6 (7.5) | 3 (8.1) | 3 (7.0) | ||
| T stage | 3.892 | 0.273 | |||
| T1 | 12 (15.0) | 4 (10.8) | 8 (18.6) | ||
| T2 | 23 (28.8) | 9 (24.3) | 14 (32.6) | ||
| T3 | 31 (38.7) | 16 (43.2) | 15 (34.9) | ||
| T4 | 14 (17.5) | 8 (21.6) | 6 (14.0) | ||
| N stage | 4.537 | 0.103 | |||
| N0 | 22 (27.5) | 8 (21.6) | 14 (32.6) | ||
| N1 | 26 (32.5) | 10 (27.0) | 16 (37.2) | ||
| N2 | 19 (23.8) | 11 (29.7) | 8 (18.6) | ||
| N3 | 13 (16.2) | 8 (21.6) | 5 (11.6) | ||
| TNM stage | 5.847 | 0.054 | |||
| Ib | 12 (15.0) | 3 (8.1) | 9 (20.9) | ||
| II | 35 (43.8) | 14 (37.8) | 21 (48.8) | ||
| III | 33 (41.2) | 20 (54.1) | 13 (30.2) | ||
| Vascular invasion | 38 (47.5) | 21 (56.8) | 17 (39.5) | 2.379 | 0.123 |
| Neural invasion | 29 (36.2) | 16 (43.2) | 13 (30.2) | 1.436 | 0.231 |
| Preoperative CEA (ng/mL) | 8.2 ± 15.3 | 10.1 ± 18.2 | 6.7 ± 12.1 | 1.015 | 0.313 |
| Preoperative CA19-9 (U/mL) | 42.8 ± 86.4 | 51.7 ± 102.3 | 35.2 ± 71.8 | 0.852 | 0.397 |
| Preoperative CA72-4 (U/mL) | 15.6 ± 32.4 | 19.8 ± 38.7 | 12.1 ± 26.2 | 1.038 | 0.302 |
Before chemotherapy initiation, 37 of 80 patients (46.2%) had nutritional risk (NRS2002 ≥ 3 points). Results from various nutritional assessment tools showed that patients in the nutritional risk group had significantly higher NRS2002 scores, CONUT scores, and weight loss percentages compared to the non-nutritional risk group, while PNI scores were significantly lower, with statistically significant differences (P < 0.001). GPS score distribution showed statistically significant differences between groups (P < 0.001). After chemotherapy completion, both groups showed varying degrees of nutritional status decline, with more pronounced decline in the nutritional risk group (Table 2).
| Nutritional assessment indicator | Nutritional risk group (n = 37) | Non-nutritional risk group (n = 43) | t/Z | P value |
| Baseline nutritional status | ||||
| NRS2002 score (points, mean ± SD) | 4.2 ± 1.1 | 2.1 ± 0.8 | 10.174 | < 0.001 |
| CONUT score [M (P25, P75)] | 4 (3, 6) | 2 (1, 3) | -4.832 | < 0.001 |
| PNI (mean ± SD) | 42.1 ± 6.8 | 48.9 ± 5.2 | -4.952 | < 0.001 |
| GPS score, n (%) | ||||
| 0 points | 8 (21.6) | 32 (74.4) | 22.356 | < 0.001 |
| 1 point | 18 (48.6) | 9 (20.9) | ||
| 2 points | 11 (29.7) | 2 (4.7) | ||
| Weight loss percentage (%) | 8.7 ± 4.2 | 3.1 ± 2.8 | 7.089 | < 0.001 |
| Post-chemotherapy nutritional status | ||||
| NRS2002 score (points, mean ± SD) | 4.8 ± 1.3 | 2.4 ± 0.9 | 9.635 | < 0.001 |
| CONUT score [M (P25, P75)] | 5 (4, 7) | 2 (1, 4) | -5.247 | < 0.001 |
| PNI (mean ± SD) | 39.8 ± 7.1 | 47.6 ± 5.8 | -5.401 | < 0.001 |
| Weight loss percentage (%, mean ± SD) | 12.4 ± 5.1 | 4.8 ± 3.2 | 7.852 | < 0.001 |
Among the 80 patients, 69 (86.2%) completed all planned chemotherapy cycles. The nutritional risk group had a treatment completion rate of 75.7% (28/37), significantly lower than the non-nutritional risk group's 95.3% (41/43), with statistically significant difference (P = 0.009). The nutritional risk group had significantly lower RDI compared to the non-nutritional risk group (P < 0.001), with significantly higher rates of dose reduction, treatment delay, and early termination compared to the non-nutritional risk group (P < 0.05) (Table 3).
| Item | Nutritional risk group (n = 37) | Non-nutritional risk group (n = 43) | χ²/t | P value |
| Completed all planned cycles | 28 (75.7) | 41 (95.3) | 6.859 | 0.009 |
| RDI (%) | 78.6 ± 14.2 | 92.1 ± 8.7 | -5.142 | < 0.001 |
| RDI grouping | ||||
| ≥ 85% | 18 (48.6) | 38 (88.4) | 15.671 | < 0.001 |
| < 85% | 19 (51.4) | 5 (11.6) | ||
| Actual completion cycle proportion | ||||
| 100% | 28 (75.7) | 41 (95.3) | 8.762 | 0.013 |
| 80%-99% | 6 (16.2) | 2 (4.7) | ||
| < 80% | 3 (8.1) | 0 (0) | ||
| Treatment adjustment | ||||
| Dose reduction | 15 (40.5) | 7 (16.3) | 6.025 | 0.014 |
| Treatment delay | 18 (48.6) | 9 (20.9) | 6.896 | 0.009 |
| Early termination | 9 (24.3) | 2 (4.7) | 6.859 | 0.009 |
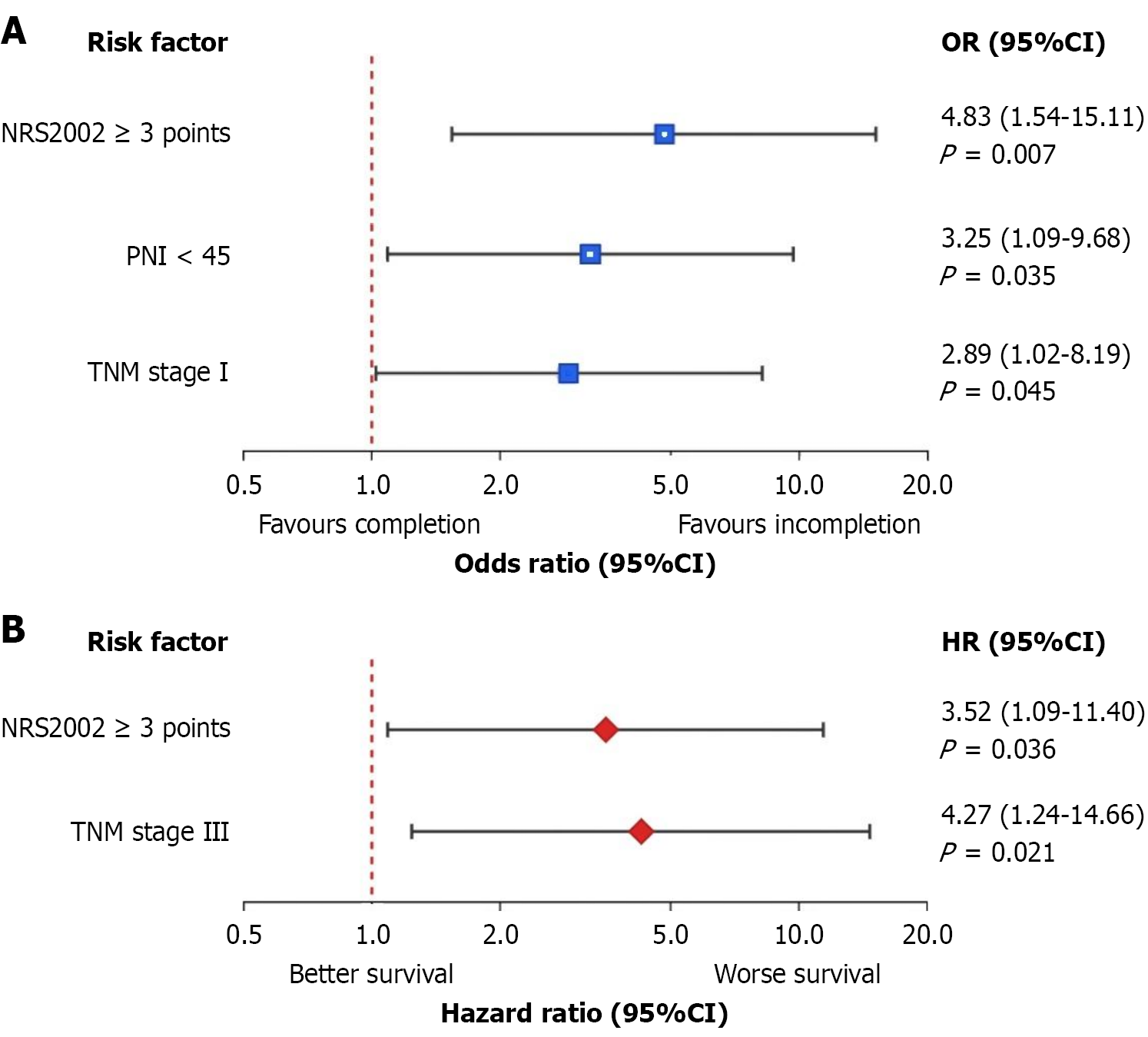
Univariate logistic regression analysis showed that age, BMI, NRS2002 score, CONUT score, PNI, GPS score, TNM stage, and preoperative CEA levels were associated with treatment completion (P < 0.10). Multivariate logistic regression analysis results showed that NRS2002 score ≥ 3 points (OR = 4.829, 95%CI: 1.542-15.114, P = 0.007), PNI < 45 (OR = 3.247, 95%CI: 1.089-9.683, P = 0.035), and TNM stage III (OR = 2.894, 95%CI: 1.023-8.187, P = 0.045) were independent risk factors affecting treatment completion (Table 4 and Figure 1A).
| Factor | Univariate analysis | Multivariate analysis | ||
| OR (95%CI) | P value | OR (95%CI) | P value | |
| Age (≥ 65 years) | 2.341 (0.896-6.118) | 0.082 | ||
| BMI (< 22 kg/m2) | 2.156 (0.824-5.642) | 0.118 | ||
| NRS2002 ≥ 3 points | 5.892 (1.842-18.849) | 0.003 | 4.829 (1.542-15.114) | 0.007 |
| CONUT score ≥ 5 points | 3.784 (1.369-10.464) | 0.010 | ||
| PNI < 45 | 4.125 (1.476-11.524) | 0.007 | 3.247 (1.089-9.683) | 0.035 |
| GPS score ≥ 1 point | 2.678 (0.989-7.250) | 0.053 | ||
| TNM stage III | 3.542 (1.289-9.735) | 0.014 | 2.894 (1.023-8.187) | 0.045 |
| Preoperative CEA (≥ 5 ng/mL) | 2.134 (0.792-5.745) | 0.133 | ||
During chemotherapy, all 80 patients experienced varying degrees of adverse reactions. The nutritional risk group had significantly higher rates of ≥ grade 3 hematologic and non-hematologic toxicities compared to the non-nutritional risk group (P < 0.05). The rates of ≥ grade 3 toxicity for leukopenia, neutropenia, nausea and vomiting, and diarrhea in the nutritional risk group were 43.2%, 40.5%, 32.4%, and 27.0%, respectively, significantly higher than the non-nutritional risk group's 18.6%, 16.3%, 14.0%, and 9.3% (P < 0.05) (Table 5).
| Adverse reaction | Nutritional risk group (n = 37) | Non-nutritional risk group (n = 43) | P value | ||
| Any grade | ≥ grade 3 | Any grade | ≥ grade 3 | ||
| Hematologic toxicity | |||||
| Leukopenia | 32 (86.5) | 16 (43.2) | 31 (72.1) | 8 (18.6) | 0.015 |
| Neutropenia | 30 (81.1) | 15 (40.5) | 29 (67.4) | 7 (16.3) | 0.012 |
| Thrombocytopenia | 24 (64.9) | 8 (21.6) | 22 (51.2) | 4 (9.3) | 0.127 |
| Anemia | 28 (75.7) | 6 (16.2) | 26 (60.5) | 3 (7.0) | 0.201 |
| Non-hematologic toxicity | |||||
| Nausea and vomiting | 34 (91.9) | 12 (32.4) | 36 (83.7) | 6 (14.0) | 0.042 |
| Diarrhea | 22 (59.5) | 10 (27.0) | 19 (44.2) | 4 (9.3) | 0.039 |
| Mucositis | 18 (48.6) | 5 (13.5) | 15 (34.9) | 2 (4.7) | 0.159 |
| Hand-foot syndrome | 25 (67.6) | 7 (18.9) | 24 (55.8) | 4 (9.3) | 0.221 |
| Peripheral neuropathy | 21 (56.8) | 4 (10.8) | 19 (44.2) | 2 (4.7) | 0.298 |
| Hepatic dysfunction | 16 (43.2) | 3 (8.1) | 13 (30.2) | 1 (2.3) | 0.242 |
| Renal dysfunction | 12 (32.4) | 2 (5.4) | 8 (18.6) | 1 (2.3) | 0.398 |
Follow-up ended on July 30, 2025, with a median follow-up time of 28 months (range 13-55 months). During the study period, 24 disease progression events occurred (including 9 local recurrences and 15 distant metastases), with 14 patient deaths. The nutritional risk group had 17 disease progressions (45.9%), significantly higher than the non-nutritional risk group's 7 cases (16.3%) (P = 0.003); the nutritional risk group had 10 deaths (27.0%), higher than the non-nutritional risk group's 4 cases (9.3%) (P = 0.032). The nutritional risk group's 3-year DFS was 62.1%, significantly lower than the non-nutritional risk group's 83.7% (Log-rank P = 0.018); 3-year OS was 72.4%, significantly lower than the non-nutritional risk group's 90.7% (Log-rank P = 0.023) (Table 6).
| Survival indicator | Nutritional risk group (n = 37) | Non-nutritional risk group (n = 43) | χ² | P value |
| Disease progression | 17 (45.9) | 7 (16.3) | 8.973 | 0.003 |
| Local recurrence | 6 (16.2) | 3 (7.0) | ||
| Distant metastasis | 11 (29.7) | 4 (9.3) | ||
| Death | 10 (27.0) | 4 (9.3) | 4.589 | 0.032 |
| Median DFS (months, 95%CI) | Not reached | Not reached | 5.573 | 0.018 |
| 3-year DFS rate (%) | 62.1 | 83.7 | ||
| Median OS (months, 95%CI) | Not reached | Not reached | 5.234 | 0.023 |
| 3-year OS rate (%) | 72.4 | 90.7 |
DFS factor analysis: Univariate Cox regression analysis showed that NRS2002 ≥ 3 points, CONUT ≥ 5 points, PNI < 45, GPS ≥ 1 point, TNM stage III, vascular invasion, and RDI < 85% were associated with DFS (P < 0.10). Multivariate Cox regression analysis results showed that NRS2002 ≥ 3 points (HR = 2.847, 95%CI: 1.124-7.214, P = 0.027), TNM stage III (HR = 3.162, 95%CI: 1.287-7.768, P = 0.012), and RDI < 85% (HR = 2.536, 95%CI: 1.089-5.903, P = 0.031) were independent risk factors for DFS (Table 7).
| Factor | Univariate analysis | Multivariate analysis | ||
| HR (95%CI) | P value | HR (95%CI) | P value | |
| NRS2002 ≥ 3 points | 3.241 (1.315-7.985) | 0.011 | 2.847 (1.124-7.214) | 0.027 |
| CONUT ≥ 5 points | 2.687 (1.142-6.319) | 0.024 | ||
| PNI < 45 | 2.834 (1.189-6.754) | 0.019 | ||
| GPS ≥ 1 point | 2.156 (0.886-5.245) | 0.090 | ||
| TNM stage III | 4.125 (1.754-9.702) | 0.001 | 3.162 (1.287-7.768) | 0.012 |
| Vascular invasion | 2.341 (0.987-5.551) | 0.053 | ||
| RDI < 85% | 3.156 (1.387-7.182) | 0.006 | 2.536 (1.089-5.903) | 0.031 |
OS factor analysis: Univariate Cox regression analysis showed that age ≥ 65 years, NRS2002 ≥ 3 points, PNI < 45, GPS ≥ 1 point, TNM stage III, neural invasion, and RDI < 85% were associated with OS (P < 0.10). Multivariate Cox regression analysis results showed that NRS2002 ≥ 3 points (HR = 3.524, 95%CI: 1.089-11.404, P = 0.036) and TNM stage III (HR = 4.267, 95%CI: 1.241-14.658, P = 0.021) were independent risk factors for OS (Table 8 and Figure 1B).
| Factor | Univariate analysis | Multivariate analysis | ||
| HR (95%CI) | P value | HR (95%CI) | P value | |
| Age ≥ 65 years | 2.341 (0.756-7.254) | 0.140 | ||
| NRS2002 ≥ 3 points | 4.267 (1.274-14.289) | 0.018 | 3.524 (1.089-11.404) | 0.036 |
| PNI < 45 | 3.156 (1.024-9.724) | 0.045 | ||
| GPS ≥ 1 point | 2.789 (0.821-9.480) | 0.100 | ||
| TNM stage III | 5.234 (1.578-17.357) | 0.007 | 4.267 (1.241-14.658) | 0.021 |
| Neural invasion | 2.456 (0.798-7.564) | 0.118 | ||
| RDI < 85% | 2.987 (0.975-9.156) | 0.055 | ||
During chemotherapy, both groups showed varying degrees of nutritional status decline. The nutritional risk group had significantly greater increases in NRS2002 scores, CONUT scores, and weight loss percentages compared to the non-nutritional risk group before and after chemotherapy (P < 0.05), while the PNI decline showed no statistically significant difference between groups (P = 0.102). Eight patients (21.6%) in the nutritional risk group progressed from mild-moderate malnutrition to severe malnutrition after chemotherapy, while only 2 patients (4.7%) in the non-nutritional risk group experienced this progression (P = 0.025) (Table 9).
| Nutritional indicator change | Nutritional risk group (n = 37) | Non-nutritional risk group (n = 43) | t/χ² | P value |
| NRS2002 score change (points, mean ± SD) | 0.6 ± 0.8 | 0.3 ± 0.5 | 2.074 | 0.042 |
| CONUT score change [M (P25, P75)] | 1 (0, 2) | 0 (0, 1) | -2.891 | 0.004 |
| PNI change (mean ± SD) | -2.3 ± 3.1 | -1.3 ± 2.4 | -1.653 | 0.102 |
| Weight loss percentage change (%, mean ± SD) | 3.7 ± 2.8 | 1.7 ± 1.9 | 3.742 | < 0.001 |
| Nutritional status progression, n (%) | 8 (21.6) | 2 (4.7) | 5.025 | 0.025 |
To explore whether the predictive value of nutritional indicators differed by sex and disease severity, we performed subgroup analyses. In male patients (n = 49), NRS2002 ≥ 3 points was significantly associated with lower treatment completion rate (OR = 4.215, 95%CI: 1.089-16.314, P = 0.037), while in female patients (n = 31), the association showed a similar trend but did not reach statistical significance (OR = 5.624, 95%CI: 0.892-35.462, P = 0.066), likely due to the smaller sample size. When stratified by TNM stage, the predictive value of NRS2002 was more pronounced in stage III patients (OR = 6.127, 95%CI: 1.456-25.783, P = 0.013) compared to stage II patients (OR = 3.542, 95%CI: 0.687-18.264, P = 0.131). Similarly, the impact of nutritional risk on 3-year DFS was greater in stage III patients (HR = 3.524, 95%CI: 1.124-11.047, P = 0.031) than in stage II patients (HR = 2.156, 95%CI: 0.542-8.573, P = 0.276). These findings suggest that nutritional status may have a more significant impact on treatment outcomes in patients with more advanced disease, although these subgroup results should be interpreted with caution given the limited sample sizes and require validation in larger studies (Table 10).
| Subgroup | n | OR/HR (95%CI) | P value | Outcome |
| Stratified by sex (treatment completion) | ||||
| Male | 49 | 4.215 (1.089-16.314) | 0.037 | OR |
| Female | 31 | 5.624 (0.892-35.462) | 0.066 | OR |
| Stratified by TNM stage (treatment completion) | ||||
| Stage II | 47 | 3.542 (0.687-18.264) | 0.131 | OR |
| Stage III | 33 | 6.127 (1.456-25.783) | 0.013 | OR |
| Stratified by TNM stage (3-year DFS) | ||||
| Stage II | 47 | 2.156 (0.542-8.573) | 0.276 | HR |
| Stage III | 33 | 3.524 (1.124-11.047) | 0.031 | HR |
This study found that 46.2% of gastric cancer patients had nutritional risk before chemotherapy initiation, consistent with international reports of 40%-60%[8]. The nutritional risk group had significantly lower treatment completion rates compared to the non-nutritional risk group (75.7% vs 95.3%), markedly insufficient RDI, and significantly higher rates of dose reduction, treatment delay, and early termination. These results are highly consistent with previous studies[9,10].
The mechanisms by which malnutrition affects treatment completion are multifaceted. First, malnourished patients have compromised immune function and poor tolerance to chemotherapy drugs, making them more susceptible to severe adverse reactions, leading to dose adjustments or treatment interruptions[11]. This study showed significantly higher rates of ≥ grade 3 hematologic and non-hematologic toxicities in the nutritional risk group, confirming this mechanism. Second, malnutrition affects drug metabolism and clearance, potentially increasing drug toxicity[12]. Additionally, patients with poor nutritional status often have declining performance status, affecting treatment compliance[13].
The mechanisms by which malnutrition affects drug metabolism warrant further elaboration. Protein-energy malnutrition may reduce hepatic cytochrome P450 enzyme system activity, impairing the biotransformation of chemotherapeutic agents. Additionally, hypoalbuminemia, commonly observed in malnourished patients, alters plasma protein binding of drugs, resulting in increased free drug concentrations and potentially enhanced toxicity. Furthermore, impaired hepatic synthetic function and altered body fluid distribution in malnourished patients affect drug volume of distribution and clearance rates, leading to prolonged drug exposure and increased adverse effects.
Multivariate analysis showed that NRS2002 ≥ 3 points was an independent risk factor affecting treatment completion (OR = 4.829), suggesting that NRS2002 as a comprehensive nutritional assessment tool has important value in predicting chemotherapy tolerance. NRS2002 not only assesses the degree of nutritional status impairment but also considers disease severity and age factors, providing a more comprehensive reflection of patients' overall condition[14]. PNI < 45 was also an independent risk factor, consistent with other study results[15]. PNI combines serum ALB and lymphocyte count, reflecting both protein nutritional status and immune function levels, thus having unique advantages in predicting chemotherapy tolerance[16].
A direct comparison of the predictive performance of different nutritional assessment tools in this study reveals notable differences. NRS2002 demonstrated the most consistent predictive value across all endpoints, remaining significant in multivariate analyses for both treatment completion (OR = 4.829) and survival outcomes (DFS: HR = 2.847; OS: HR = 3.524). PNI showed independent predictive value for treatment completion (OR = 3.247) but did not retain significance in survival analyses after adjustment for other factors. CONUT and GPS scores, while significantly elevated in the nutritional risk group in univariate analyses, did not enter the final multivariate models, suggesting their predictive efficacy may be less robust than NRS2002 and PNI. The superior performance of NRS2002 may be attributed to its multidimensional assessment approach, which integrates nutritional status impairment, disease severity, and age adjustment, thereby providing a more comprehensive evaluation of patients’ overall condition and treatment tolerance capacity. These findings support NRS2002 as the recommended primary screening tool for gastric cancer patients undergoing adjuvant chemotherapy, with PNI serving as a valuable complementary indicator.
This study employed multiple nutritional assessment tools for comprehensive evaluation, showing that different tools each have characteristics. NRS2002, as the standardized nutritional risk screening tool recommended by the European Society for Clinical Nutrition and Metabolism, is widely used in hospitalized patients, and this study confirmed its good predictive value in gastric cancer patients[17]. CONUT score, based on objective biochemical indicators, is simple and easy to implement, showing significant elevation in the nutritional risk group, but did not enter the final model in multivariate analysis, possibly because it mainly reflects biochemical nutritional status rather than comprehensive risk[18].
PNI as a PNI not only performed excellently in predicting treatment completion but has also been proven closely related to prognosis in various tumor patients in previous studies[19]. GPS score combines inflammatory response and nutritional status, embodying the concept of the inflammation-nutrition axis and has important significance in malignant tumor patient prognosis assessment[20]. In this study, GPS scores were significantly elevated in the nutritional risk group but were marginally significant in multivariate analysis, suggesting its predictive value may not be as stable as NRS2002 and PNI.
Weight loss as a traditional nutritional assessment indicator showed that the nutritional risk group had significantly higher weight loss percentages compared to the control group, closely related to factors such as postoperative digestive function changes and reduced food intake in gastric cancer patients[21]. Weight loss reflects the degree of patient nutritional reserve depletion and is an important indicator for assessing nutritional status.
Survival analysis results showed that the nutritional risk group had significantly lower 3-year DFS and OS compared to the non-nutritional risk group, consistent with multiple domestic and international study results[22,23]. The mechanisms by which nutritional status affects prognosis are complex and include several aspects.
First, malnutrition directly affects treatment completion, leading to decreased treatment efficacy. This study's Cox regression analysis showed that RDI < 85% was an independent risk factor for DFS (HR = 2.536), confirming the close relationship between chemotherapy dose intensity and prognosis[24]. Second, malnourished patients have compromised immune function and weakened anti-tumor immunity, making them more susceptible to tumor recurrence and metastasis[25]. Additionally, malnutrition is associated with adverse outcomes such as surgical complications and prolonged hospitalization, indirectly affecting patient prognosis[26].
NRS2002 ≥ 3 points was an independent risk factor for DFS (HR = 2.847) and OS (HR = 3.524) in multivariate analysis, further confirming the important value of nutritional risk screening in prognosis assessment. This result suggests that nutritional status assessment before chemotherapy initiation is not only meaningful for guiding treatment decisions but is also an important basis for prognostic stratification[27].
This study observed dynamic changes in nutritional status during chemotherapy, finding that both groups showed varying degrees of nutritional status decline, but the nutritional risk group showed more pronounced decline. This phenomenon reflects the toxic effects of chemotherapy drugs and differences in patient tolerance to treatment[28]. In the nutritional risk group, 21.6% of patients experienced further nutritional status deterioration after chemotherapy, significantly higher than the control group, suggesting that malnutrition has self-reinforcing characteristics, where patients with worse nutritional status are more prone to further nutritional deterioration.
This finding has important clinical significance, suggesting that patients with nutritional risk need close nutritional monitoring and active nutritional intervention throughout the entire chemotherapy course. Nutritional assessment only before chemotherapy is insufficient; establishing a dynamic assessment and intervention system is necessary.
The results of this study have important guiding significance for clinical practice. First, nutritional risk screening should be incorporated into routine assessment procedures for postoperative adjuvant chemotherapy in gastric cancer, with validated assessment tools such as NRS2002 and PNI recommended. Second, for patients with nutritional risk, active nutritional intervention should be implemented before chemotherapy initiation, including nutritional counseling, nutritional supplementation, and enteral or parenteral nutritional support when necessary. Third, nutritional monitoring should be strengthened during chemotherapy, with intervention strategies adjusted timely based on nutritional status changes.
Additionally, this study suggests that clinicians should fully consider patients' nutritional status when formulating chemotherapy regimens. For patients with high nutritional risk, relatively gentle chemotherapy regimens or prolonged chemotherapy intervals may be necessary to improve treatment completion rates and safety.
This study has certain limitations. First, this is a single-center retrospective study with a relatively small sample size, potentially subject to selection bias. Second, nutritional intervention measures were not standardized, which may affect result accuracy; importantly, because nutritional interventions were not controlled or standardized in this retrospective study, the observed outcomes reflect “real-world” clinical practice patterns rather than the potential benefits of structured nutritional support programs. This distinction is crucial, as the current findings demonstrate the natural course of nutritional status during chemotherapy without systematic intervention, underscoring the need for prospective randomized controlled trials with standardized nutritional intervention protocols to definitively establish the clinical benefits of targeted nutritional support.
Based on this study's results, future research should focus on the following aspects: First, conducting multicenter prospective studies to expand sample size and validate the applicability of nutritional assessment tools in different populations. Second, exploring the effectiveness of individualized nutritional intervention strategies and establishing precision intervention models based on nutritional risk stratification. Third, studying the correlation between nutritional status and chemotherapy drug metabolism and immune function to deeply elucidate the molecular mechanisms by which nutrition affects prognosis. Fourth, developing comprehensive prognostic prediction models integrating multidimensional information to provide more precise tools for clinical decision-making.
In conclusion, this study confirmed the important impact of nutritional status on treatment completion and prognosis in gastric cancer patients receiving postoperative adjuvant chemotherapy, providing scientific evidence for optimizing nutritional management and treatment strategies for gastric cancer patients. Clinical practice should emphasize nutritional risk screening and intervention to improve patients' treatment outcomes and quality of life.
| 1. | Morgan E, Arnold M, Camargo MC, Gini A, Kunzmann AT, Matsuda T, Meheus F, Verhoeven RHA, Vignat J, Laversanne M, Ferlay J, Soerjomataram I. The current and future incidence and mortality of gastric cancer in 185 countries, 2020-40: A population-based modelling study. EClinicalMedicine. 2022;47:101404. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 604] [Cited by in RCA: 533] [Article Influence: 133.3] [Reference Citation Analysis (4)] |
| 2. | Yan X, Lei L, Li H, Cao M, Yang F, He S, Zhang S, Teng Y, Li Q, Xia C, Chen W. Stomach cancer burden in China: Epidemiology and prevention. Chin J Cancer Res. 2023;35:81-91. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 46] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 3. | Takayama T, Tsuji Y. Updated Adjuvant Chemotherapy for Gastric Cancer. J Clin Med. 2023;12:6727. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 4. | Mullin GE, Fan L, Sulo S, Partridge J. The Association between Oral Nutritional Supplements and 30-Day Hospital Readmissions of Malnourished Patients at a US Academic Medical Center. J Acad Nutr Diet. 2019;119:1168-1175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 32] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 5. | Wu L, Zhong Y, Wu D, Xu P, Ruan X, Yan J, Liu J, Li X. Immunomodulatory Factor TIM3 of Cytolytic Active Genes Affected the Survival and Prognosis of Lung Adenocarcinoma Patients by Multi-Omics Analysis. Biomedicines. 2022;10:2248. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 90] [Cited by in RCA: 85] [Article Influence: 21.3] [Reference Citation Analysis (1)] |
| 6. | Yu M, Li X, Chen M, Liu L, Yao T, Li J, Su W. Prognostic potential of nutritional risk screening and assessment tools in predicting survival of patients with pancreatic neoplasms: a systematic review. Nutr J. 2024;23:17. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 18] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 7. | Cho JW, Youn J, Kim EM, Choi MG, Lee JE. Associations of patient-generated subjective global assessment (PG-SGA) and NUTRISCORE with survival in gastric cancer patients: timing matters, a retrospective cohort study. BMC Gastroenterol. 2022;22:468. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 8. | Wu L, Liu Q, Ruan X, Luan X, Zhong Y, Liu J, Yan J, Li X. Multiple Omics Analysis of the Role of RBM10 Gene Instability in Immune Regulation and Drug Sensitivity in Patients with Lung Adenocarcinoma (LUAD). Biomedicines. 2023;11:1861. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 91] [Cited by in RCA: 78] [Article Influence: 26.0] [Reference Citation Analysis (1)] |
| 9. | Yun JH, Song GJ, Son MW, Lee MS. Global Leadership Initiative on Malnutrition Criteria and Immunonutritional Status Predict Chemoadherence and Survival in Stage II/III Gastric Cancer Treated with XELOX Chemotherapy. Nutrients. 2024;16:3468. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 10. | Lee S, Kang DH, Ahn TS, Kim SS, Yun JH, Kim HJ, Seo SH, Kim TW, Kong HJ, Baek MJ. The Impact of Pre-Chemotherapy Body Composition and Immunonutritional Markers on Chemotherapy Adherence in Stage III Colorectal Cancer Patients. J Clin Med. 2023;12:1423. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 11. | Chan WL, Yun HB, Cheung EE, Liu M, Hou LY, Lam KO, Wong IY, Chiu WK, Law S, Kwong D. Association of sarcopenia with severe chemotherapy toxicities and survival in patients with advanced gastric cancer. Oncologist. 2024;29:e1272-e1279. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 12. | Arends J. Malnutrition in cancer patients: Causes, consequences and treatment options. Eur J Surg Oncol. 2024;50:107074. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 123] [Article Influence: 61.5] [Reference Citation Analysis (0)] |
| 13. | Gándara-Mireles JA, Lares-Asseff I, Reyes Espinoza EA, Córdova Hurtado LP, Payan Gándara H, Botello Ortiz M, Loera Castañeda V, Patrón Romero L, Almanza Reyes H. Nutritional Status as a Risk Factor for Doxorubicin Cardiotoxicity in Mexican Children with Acute Lymphoblastic Leukemia. Nutr Cancer. 2024;76:952-962. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 14. | Tu Y, Chen F, Yu Q, Song L, Chen M. Application of NRS2002 and PG-SGA in nutritional assessment for perioperative patients with head and neck squamous cell carcinoma: An observational study. Medicine (Baltimore). 2024;103:e40025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 15. | Wu L, Zheng Y, Ruan X, Wu D, Xu P, Liu J, Wu D, Li X. Long-chain noncoding ribonucleic acids affect the survival and prognosis of patients with esophageal adenocarcinoma through the autophagy pathway: construction of a prognostic model. Anticancer Drugs. . 2022;33:e590-e603. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 91] [Article Influence: 22.8] [Reference Citation Analysis (4)] |
| 16. | Peng P, Chen L, Shen Q, Xu Z, Ding X. Prognostic Nutritional Index (PNI) and Controlling Nutritional Status (CONUT) score for predicting outcomes of breast cancer: A systematic review and meta-analysis. Pak J Med Sci. 2023;39:1535-1541. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 36] [Reference Citation Analysis (0)] |
| 17. | Trollebø MA, Skeie E, Revheim I, Stangeland H, Erstein MH, Grønning MK, Tangvik RJ, Morken MH, Nygård O, Eagan TML, Rosendahl-Riise H, Dierkes J. Comparison of nutritional risk screening with NRS2002 and the GLIM diagnostic criteria for malnutrition in hospitalized patients. Sci Rep. 2022;12:19743. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 21] [Reference Citation Analysis (0)] |
| 18. | Matysiak K, Hojdis A, Szewczuk M. Controlling Nutritional Status (CONUT) Score as Prognostic Indicator in Stage IV Gastric Cancer with Chronic Intestinal Failure. Nutrients. 2024;16:4052. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 19. | Wu L, Zhong Y, Yu X, Wu D, Xu P, Lv L, Ruan X, Liu Q, Feng Y, Liu J, Li X. Selective poly adenylation predicts the efficacy of immunotherapy in patients with lung adenocarcinoma by multiple omics research. Anticancer Drugs. 2022;33:943-959. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 86] [Article Influence: 21.5] [Reference Citation Analysis (1)] |
| 20. | Li KJ, Zhang ZY, Wang K, Sulayman S, Zeng XY, Liu J, Chen Y, Zhao ZL. Prognostic scoring system using inflammation- and nutrition-related biomarkers to predict prognosis in stage I-III colorectal cancer patients. World J Gastroenterol. 2025;31:104588. [PubMed] [DOI] [Full Text] |
| 21. | Mękal D, Sobocki J, Badowska-Kozakiewicz A, Sygit K, Cipora E, Bandurska E, Czerw A, Deptała A. Evaluation of Nutritional Status and the Impact of Nutritional Treatment in Patients with Pancreatic Cancer. Cancers (Basel). 2023;15:3816. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 26] [Reference Citation Analysis (0)] |
| 22. | Wu L, Li X, Qian X, Wang S, Liu J, Yan J. Lipid Nanoparticle (LNP) Delivery Carrier-Assisted Targeted Controlled Release mRNA Vaccines in Tumor Immunity. Vaccines (Basel). 2024;12:186. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 92] [Cited by in RCA: 85] [Article Influence: 42.5] [Reference Citation Analysis (0)] |
| 23. | Zhang J, Li M, Zhang L, Kuang T, Yu J, Wang W. Prognostic value of controlling nutritional status on clinical and survival outcomes in cancer patients treated with immunotherapy. Sci Rep. 2023;13:17715. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 24. | Wonders KY, Schmitz K, Harness J. Dose Delays, Dose Reductions, and Relative Total Dose Intensity in Patients With Advanced Cancer Who Exercised During Neoadjuvant Chemotherapy Treatment. Integr Cancer Ther. 2023;22:15347354231168368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 25. | Liang X, Cheng Z, Chen X, Li J. Prognosis analysis of necroptosis-related genes in colorectal cancer based on bioinformatic analysis. Front Genet. 2022;13:955424. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 20] [Reference Citation Analysis (0)] |
| 26. | Wu L, Yang L, Qian X, Hu W, Wang S, Yan J. Mannan-Decorated Lipid Calcium Phosphate Nanoparticle Vaccine Increased the Antitumor Immune Response by Modulating the Tumor Microenvironment. J Funct Biomater. 2024;15:229. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 20] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 27. | Wang P, Jiang L, Soh KL, Ying Y, Liu Y, Huang X, Tan Y, Soh KG. Mini Nutritional Assessment for Adult Cancer Patients: A Systematic Review and Meta-Analysis. Nutr Cancer. 2023;75:61-72. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 28. | Alonso Domínguez T, Civera Andrés M, Santiago Crespo JA, García Malpartida K, Botella Romero F. Digestive toxicity in cancer treatments. Bibliographic review. Influence on nutritional status. Endocrinol Diabetes Nutr (Engl Ed). 2023;70:136-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/