Published online Feb 27, 2026. doi: 10.4240/wjgs.v18.i2.114483
Revised: November 24, 2025
Accepted: December 19, 2025
Published online: February 27, 2026
Processing time: 132 Days and 1.9 Hours
The global incidence of gastrointestinal tumors is continuously increasing. Sur
To construct a risk-prediction model for postoperative gastrointestinal dysfunc
Data from 176 patients who underwent gastrointestinal tumor surgery at the authors’ hospital between November 2022 and November 2024 were included. Patients were divided into groups according to Tilburg Frailty Scale scores on postoperative day 5. Risk factors were screened using univariate and multivariate logistic regression analyses to establish a model, and the effectiveness of pre
Seven factors including age, sex, body mass index, tumor stage, operative duration, and preoperative hemoglobin and albumin levels were identified as independent risk factors. The constructed model had an area under the receiver operating characteristic curve of 0.895. The incidence of postoperative gastrointestinal dysfunction in the intervention group was significantly lower than that in the control group using preventive management measures based on the model.
An effective risk-prediction model was constructed and independent risk factors were identified. Preventive management measures based on this model can reduce risk and provide a scientific basis for clinical practice.
Core Tip: The global incidence of gastrointestinal tumors is rising, and postoperative gastrointestinal dysfunction remains prevalent, severely impeding recovery. This study aimed to construct a risk prediction model for this complication and explore preventive management strategies. Using data from 176 patients, seven independent risk factors—such as age, tumor stage, and operative duration—were identified. The model demonstrated high predictive accuracy, with an area under the receiver operating characteristic curve of 0.895. Preventive measures based on the model significantly reduced dysfunction incidence, offering a scientific basis for clinical practice.
- Citation: Wang XH, Wang Y, Liu X, Ouyang XL, Xie F, Liao L. Construction of a risk prediction model for postoperative gastrointestinal dysfunction and prevention in patients with gastrointestinal tumors. World J Gastrointest Surg 2026; 18(2): 114483
- URL: https://www.wjgnet.com/1948-9366/full/v18/i2/114483.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i2.114483
In recent years, the global incidence of gastrointestinal tumors has continuously increased, emerging as a major disease that severely threatens human health[1]. According to global cancer data, released by the International Agency for Research on Cancer of the World Health Organization in 2020, gastric and colorectal cancers ranked fifth and third, respectively, in terms of the number of new cases worldwide, and fourth and second, respectively, in terms of the number of deaths. The burden of gastrointestinal tumors in China is equally severe[2]. Recent cancer statistics show that the incidence and mortality rates for gastric and colorectal tumors are the highest among all malignancies, imposing a heavy disease burden on society and families[3].
Surgical resection of tumor lesions is the primary treatment for gastrointestinal malignancies. However, postoperative gastrointestinal dysfunction remains a significant obstacle in the recovery process. Postoperative gastrointestinal dysfunction refers to a series of clinical symptoms characterized by abnormalities in gastrointestinal peristalsis, digestion, and absorption after surgery for gastrointestinal tumors. Studies have reported that approximately 30%-50% of patients who have undergone gastrointestinal tumor surgery develop varying degrees of postoperative gastrointestinal dys
Regarding treatment, although there are measures to promote the recovery of gastrointestinal function, such as early enteral nutrition, drug intervention, and traditional Chinese medicine physiotherapy, most of these methods are empirical. Precise risk prediction and personalized preventive management strategies are lacking[5,9]. Existing risk prediction studies are mostly limited to univariate analysis or small-sample research, and fail to comprehensively and systematically integrate multiple potential risk factors, resulting in insufficient accuracy and practicality of the prediction models[10]. Therefore, in-depth research aimed at constructing effective risk prediction models for postoperative gastrointestinal dysfunction in patients with gastrointestinal tumors and the development of targeted preventive management strategies is of great theoretical and practical significance for improving the quality of postoperative recovery and optimizing the allocation of medical resources.
The present study systematically analyzed clinical data from 176 patients who underwent surgery for gastrointestinal tumors at our hospital. Using advanced statistical methods, key risk factors for postoperative gastrointestinal dysfunction were screened, and an accurate risk prediction model was constructed[11]. Based on this model, scientifically based and effective preventive management measures will be developed, with the anticipation of providing reliable evidence for the early identification of high-risk patients and reducing the incidence of postoperative gastrointestinal dysfunction in clinical practice[12].
Data from 176 patients who underwent surgical treatment for gastrointestinal tumors at the Ganzhou People's Hospital between November 2022 and November 2024 were included. The inclusion criteria were as follows: (1) Pathologically confirmed gastrointestinal tumor(s); (2) First-time surgical treatment; and (3) The availability of complete clinical data. The exclusion criteria were as follows: (1) Coexisting severe dysfunction(s) of vital organs such as the heart, liver, and kidneys; (2) Preoperative gastrointestinal disorders; and (3) Withdrawal from the study.
On postoperative day 5, frailty was assessed according to the Tilburg Frailty Scale, and patients were subsequently divided into 2 groups: Dysfunction and non-dysfunction.
Sample size was estimated before patient enrollment based on the primary outcome (incidence of postoperative gastrointestinal dysfunction). Using a significance level (α) of 0.05 (two-sided), a power (1 - β) of 80%, an anticipated incidence rate of postoperative gastrointestinal dysfunction (P0) of 35% based on a literature review[4,5], and an expected clinically significant odds ratio (OR) for key risk factors (e.g., advanced tumor stage) of approximately 2.5, the required sample size was calculated. The following equation was applied to the case-control study design: n = [(Zα/2 + Zβ)2 × P
General patient information [e.g., age, sex, and body mass index (BMI)], disease-related data (e.g., tumor location and stage), surgery-related information (e.g., surgical approach and operative duration), and laboratory test indicators (e.g., preoperative hemoglobin and albumin levels) were collected. Univariate analysis was used to screen factors potentially associated with postoperative gastrointestinal dysfunction. Factors that were statistically significant in univariate analysis were further analyzed using multivariate logistic regression to identify independent risk factors, and a risk prediction model was subsequently constructed. Preventive management measures were developed based on the prediction model and their effectiveness was evaluated in patients who received preventive management.
All statistical analyses were performed using SPSS version 26.0 (IBM Corporation, Armonk, NY, United States). Measurement data are expressed as mean ± SD, and the t-test was used for intergroup comparisons. Count data are expressed as n (%), and the χ2 test was used for intergroup comparisons. Univariate analysis adopted the χ2 test or t-test, while multivariate analysis used logistic regression analysis. Differences with P < 0.05 were considered to be statistically significant.
Patients (n = 176) were divided into 2 postoperative groups: Gastrointestinal dysfunction (n = 52) and non-dysfunction (n = 124). Univariate analysis was performed for age, sex, BMI, tumor location, tumor stage, surgical approach, operative duration, and preoperative hemoglobin and albumin levels. The results are summarized in Table 1. Results of the analysis revealed statistically significant differences in age, sex, BMI, tumor stage, operative duration, and preoperative hemoglobin and albumin levels between the 2 groups (P < 0.05), with no statistically significant differences in tumor location or surgical approach (P > 0.05).
| Variable | Non-dysfunction group (n = 124) | Dysfunction group (n = 52) | t value | P value |
| Age (years) | 58.2 ± 8.5 | 65.3 ± 7.8 | 5.231 | < 0.001 |
| Gender (male/female) | 72/52 | 38/14 | 4.325 | 0.038 |
| BMI (kg/m2) | 23.5 ± 2.1 | 21.8 ± 2.3 | 4.123 | 0.043 |
| Tumor location (stomach/intestine) | 68/56 | 32/20 | 0.234 | 0.629 |
| Tumor stage (I-II/III-IV) | 86/38 | 24/28 | 5.678 | 0.017 |
| Surgical approach (open/laparoscopic) | 45/79 | 22/30 | 1.234 | 0.267 |
| Operation time (minute) | 180.5 ± 30.2 | 210.3 ± 35.6 | 4.876 | < 0.001 |
| Pre-operative hemoglobin (g/L) | 125.3 ± 15.6 | 108.2 ± 12.3 | 6.345 | < 0.001 |
| Pre-operative albumin (g/L) | 38.5 ± 3.2 | 34.2 ± 3.5 | 7.234 | < 0.001 |
Factors that were statistically significant in the univariate analysis—namely, age, sex, BMI, tumor stage, operative duration, and preoperative hemoglobin and albumin levels—were included in the multivariate logistic regression analysis. The results are summarized in Table 2. Results indicated that age, sex, BMI, tumor stage, operative duration, and preoperative hemoglobin and albumin levels were independent risk factors for postoperative gastrointestinal dysfunction in patients with gastrointestinal tumors (P < 0.05).
| Variable | B | SE | Wald | df | P value | OR | 95%CI |
| Age (year) | 0.085 | 0.032 | 7.123 | 1 | 0.008 | 1.089 | 1.023-1.160 |
| Gender (male vs female) | 0.852 | 0.345 | 6.123 | 1 | 0.013 | 2.346 | 1.182-4.658 |
| BMI (kg/m2) | -0.234 | 0.102 | 5.234 | 1 | 0.022 | 0.792 | 0.645-0.978 |
| Tumor stage (III-IV vs I-II) | 1.234 | 0.456 | 7.456 | 1 | 0.006 | 3.432 | 1.412-8.321 |
| Operation time (minute) | 0.012 | 0.004 | 8.234 | 1 | 0.004 | 1.012 | 1.004-1.020 |
| Pre-operative hemoglobin (g/L) | -0.023 | 0.008 | 8.123 | 1 | 0.004 | 0.977 | 0.962-0.993 |
| Pre-operative albumin (g/L) | -0.156 | 0.052 | 8.987 | 1 | 0.003 | 0.856 | 0.765-0.954 |
Based on results of multivariate logistic regression analysis, a risk prediction model for postoperative gastrointestinal dysfunction in patients with gastrointestinal tumors was constructed: Logit(P) = -8.234 + 0.085 × age + 0.852 × sex - 0.234 × BMI + 1.234 × tumor stage + 0.012 × operative duration - 0.023 × preoperative hemoglobin - 0.156 × preoperative albumin.
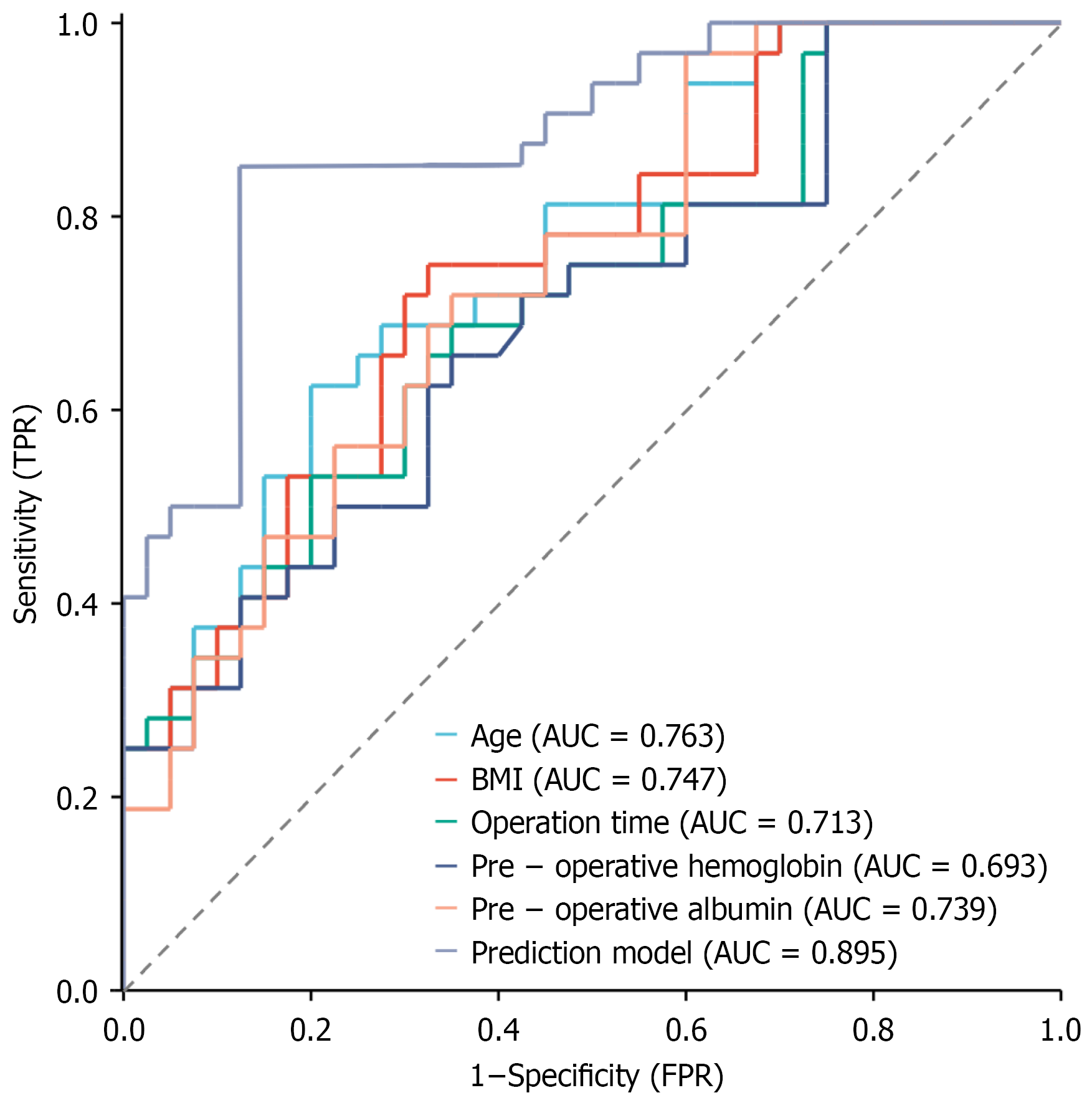
Receiver operating characteristic (ROC) curve analysis was performed using this model, and the results are summarized in Table 3 and Figure 1. The area under the ROC curve (AUC) for the prediction model was 0.895, indicating good predictive efficacy.
| Index | AUC | 95%CI | P value | Optimal cut-off value | Sensitivity (%) | Specificity (%) |
| Prediction model | 0.895 | 0.842-0.948 | 0.000 | 0.456 | 82.3 | 85.6 |
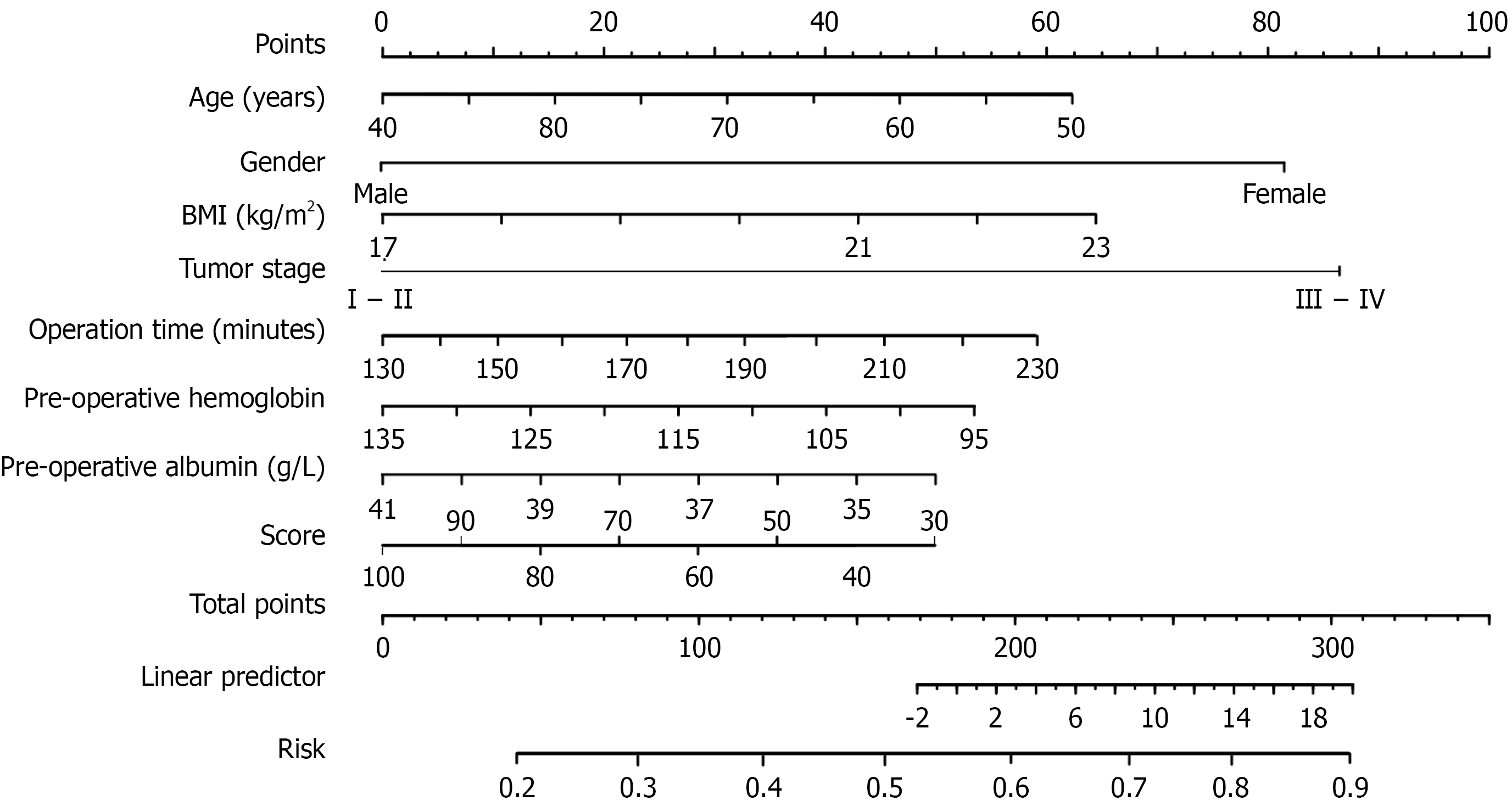
To further enhance the clinical practicality of the model and help healthcare providers to intuitively and efficiently assess the risk for postoperative gastrointestinal dysfunction in patients, this study constructed a predictive nomogram incorporating 7 independent risk factors (age, sex, BMI, tumor stage, operative duration, and preoperative hemoglobin and albumin levels) based on the independent risk factors and their regression coefficients from the aforementioned multivariate logistic regression model.
When using the nomogram, healthcare providers only need to locate the patient’s specific values on the coordinate axes corresponding to each risk factor, draw a vertical line upward from the values to intersect with the “points” axis to obtain the individual score for each factor, sum the individual scores of the 7 factors to obtain the total risk score, and then draw a vertical line downward from the total risk score to intersect with the “risk probability” axis.
This enables determination of individual risk probability of postoperative gastrointestinal dysfunction without the need for manual calculation of complex regression equations, significantly improving the clinical operability of the model presented in Figure 2.
According to the risk prediction model constructed, personalized preventive management measures, including nutritional support, early mobilization, and psychological intervention, were implemented for high-risk patients. Patients who received preventive management measures were included in the intervention group (n = 60) and those who did not receive such measures were included in the control group (n = 60). The incidence of postoperative gastrointestinal dysfunction was compared between the 2 groups, with the results reported in Table 4. Analysis revealed that the incidence of postoperative gastrointestinal dysfunction in the intervention group was significantly lower than that in the control group (P < 0.05), indicating that preventive management measures based on the risk prediction model could effectively reduce the risk for postoperative gastrointestinal dysfunction in patients who underwent surgery for gastrointestinal tumors.
| Group | n | Number of cases with postoperative gastrointestinal dysfunction | Incidence (%) | χ² | P value |
| Intervention group | 60 | 12 | 20.0 | 8.234 | 0.004 |
| Control group | 60 | 28 | 46.7 | 8.234 | 0.004 |
The present investigation systematically analyzed data from 176 patients who underwent surgery for gastrointestinal tumors, successfully constructed a risk-prediction model for postoperative gastrointestinal dysfunction, and verified the effectiveness of preventive management measures based on this model. The results revealed that age, sex, BMI, tumor stage, operative duration, and preoperative hemoglobin and albumin levels were independent risk factors for postope
Age plays a crucial role in the development of postoperative gastrointestinal dysfunction and is an important inde
Mechanisms underlying the influence of sex on postoperative gastrointestinal dysfunction are complex. The higher incidence of postoperative gastrointestinal dysfunction among males may be attributable to multiple factors. From a lifestyle perspective, the proportion of male smokers and drinkers was generally higher than that of female smokers and drinkers. These unhealthy habits can damage the gastrointestinal mucosa and affect gastrointestinal peristalsis and digestive function[16]. Androgens may affect movement and secretory functions of the gastrointestinal tract by regulating the expression of genes related to gastrointestinal motility. In addition, when faced with disease(s), adult males tend to ignore early symptoms, resulting in a later tumor stage at diagnosis, which indirectly increases the risk for postoperative gastrointestinal dysfunction.
A lower BMI reflects poorer nutritional status, and good nutritional reserves are the basis for the recovery of postoperative gastrointestinal function. Patients with a low BMI have insufficient nutrient reserves, such as proteins and fats, which cannot provide sufficient energy and raw materials for tissue repair and recovery of gastrointestinal function after surgery. Malnutrition weakens immune function in the body, increases the risk for postoperative infection(s), and negatively affects gastrointestinal function[17]. In this study, the risk for postoperative gastrointestinal dysfunction increased by 20.8% for every 1-unit decrease in BMI, highlighting the importance of nutritional status in postoperative recovery.
A later tumor stage indicates a wider invasion range of the tumor, and the corresponding surgical resection range and degree of trauma increase. Factors such as direct damage to the gastrointestinal tract during surgery, changes in local anatomical structures caused by lymph node dissection, and postoperative adhesions have serious impact(s) on gastrointestinal function. In addition, patients with advanced tumors often exhibit systemic inflammatory responses that exacerbate gastrointestinal dysfunction[18]. In this study, the risk for postoperative gastrointestinal dysfunction in patients with stage III-IV tumors was 3.432 times higher than that in those with stage I-II tumors, illustrating a close relationship between tumor stage and postoperative gastrointestinal dysfunction.
Prolonged operative duration increases intraoperative blood loss, leads to tissue ischemia and hypoxia, and causes intestinal mucosal damage and an imbalance in the intestinal flora. Simultaneously, prolonged exposure to anesthesia inhibits gastrointestinal peristalsis and interferes with neuroendocrine regulatory functions of the gastrointestinal tract[19]. Therefore, shortening the operative duration and optimizing the surgical process are of great significance for reducing the risk for postoperative gastrointestinal dysfunction.
Preoperative hemoglobin and albumin levels are important indicators of a patient’s nutritional status and body reserve capacity. Low hemoglobin and albumin levels not only indicate anemia and malnutrition but may also indicate impaired function of vital organs. This study found that for every 1 g/L decrease in preoperative hemoglobin level, the risk for postoperative gastrointestinal dysfunction increased by 2.3%; for every 1 g/L decrease in preoperative albumin level, the risk increased by 14.4%, again emphasizing the necessity of preoperative nutritional assessment and intervention[20,21].
The risk prediction model constructed in this study not only demonstrated good predictive efficacy (AUC = 0.895) but also exhibited significant advantages in clinical application value over those reported in similar studies. First, our model incorporates more comprehensive factors. Unlike the “I-FEED” score proposed by Alsharqawi et al[6], which only included postoperative indicators, such as eating status and bowel sounds, our model adds preoperative nutrition-related parameters (e.g., BMI, hemoglobin, and albumin), is better aligned with the clinical demand for “preoperative risk early warning”, and facilitates the timely identification and intervention of high-risk patients. Second, with an AUC of 0.895, it yields higher prediction accuracy, outperforming the model for postoperative intestinal dysfunction in rectal cancer developed by Qin et al[10] (AUC = 0.783); moreover, its sensitivity (82.3%) and specificity (85.6%) are both relatively high, ensuring a more reliable predictive performance. Third, it balances prevention and prediction. While most existing models focus solely on “risk identification”, this study provides personalized preventive measures (e.g., nutritional support, early ambulation, and psychological intervention) based on the model. Verification results show that the incidence rate in the intervention group (20.0%) was 40.5% lower than that in the control group (46.7%; Table 4), forming a “prediction-intervention” closed loop with greater clinical translation value.
The present study had some limitations that merit consideration. First, factors such as anesthesia methods (general vs epidural anesthesia), selection of analgesic drugs (opioids vs non-steroidal anti-inflammatory drugs), and intraoperative fluid volume were not included. Previous studies[22,23] have confirmed that opioids may increase the risk for dysfunction by inhibiting gastrointestinal motility; however, retrospective data for 18% of cases in our center lacked complete records of analgesic regimens; as such, to avoid data bias, these data were excluded from the model. Second, the aforementioned variables may have affected the results by interfering with the regulatory pathways involved in gastrointestinal motility. Future multicenter prospective studies are needed to systematically collect such data to further control for potentially confounding biases. In addition, the single-center design of the present study may have led to selection bias, and the follow-up was relatively short (long-term prognosis was not assessed). Subsequent studies should expand the sample size and extend the follow-up to optimize the generalizability of the model.
This study established a risk prediction model for postoperative gastrointestinal dysfunction in patients with gastroin
| 1. | Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76817] [Cited by in RCA: 69648] [Article Influence: 13929.6] [Reference Citation Analysis (45)] |
| 2. | Cao W, Chen HD, Yu YW, Li N, Chen WQ. Changing profiles of cancer burden worldwide and in China: a secondary analysis of the global cancer statistics 2020. Chin Med J (Engl). 2021;134:783-791. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2416] [Cited by in RCA: 2057] [Article Influence: 411.4] [Reference Citation Analysis (6)] |
| 3. | Xia C, Dong X, Li H, Cao M, Sun D, He S, Yang F, Yan X, Zhang S, Li N, Chen W. Cancer statistics in China and United States, 2022: profiles, trends, and determinants. Chin Med J (Engl). 2022;135:584-590. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3055] [Cited by in RCA: 2590] [Article Influence: 647.5] [Reference Citation Analysis (8)] |
| 4. | Mythen MG. Postoperative gastrointestinal tract dysfunction: an overview of causes and management strategies. Cleve Clin J Med. 2009;76 Suppl 4:S66-S71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 20] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 5. | Mythen MG. Postoperative gastrointestinal tract dysfunction. Anesth Analg. 2005;100:196-204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 80] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 6. | Alsharqawi N, Alhashemi M, Kaneva P, Baldini G, Fiore JF Jr, Feldman LS, Lee L. Validity of the I-FEED score for postoperative gastrointestinal function in patients undergoing colorectal surgery. Surg Endosc. 2020;34:2219-2226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 28] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 7. | Tan S, Wu G, Yu W, Li N. [Research advance in causes of postoperative gastrointestinal dysfunction]. Zhonghua Wei Chang Wai Ke Za Zhi. 2016;19:351-355. [PubMed] |
| 8. | Lam D, Jones O. Changes to gastrointestinal function after surgery for colorectal cancer. Best Pract Res Clin Gastroenterol. 2020;48-49:101705. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 9. | Osland E, Yunus RM, Khan S, Memon MA. Early versus traditional postoperative feeding in patients undergoing resectional gastrointestinal surgery: a meta-analysis. JPEN J Parenter Enteral Nutr. 2011;35:473-487. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 186] [Cited by in RCA: 171] [Article Influence: 11.4] [Reference Citation Analysis (3)] |
| 10. | Qin Q, Huang B, Wu A, Gao J, Liu X, Cao W, Ma T, Kuang Y, Guo J, Wu Q, Shao B, Guan Q, Yao H, Zhang X, Wang H; Chinese Radiation Intestinal Injury Research Group. Development and Validation of a Post-Radiotherapy Prediction Model for Bowel Dysfunction After Rectal Cancer Resection. Gastroenterology. 2023;165:1430-1442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 14] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 11. | Wang N, Zuo H, Xu Y, Zhou Y, Wei A, Li K. Relation of gut microbiota and postoperative gastrointestinal dysfunction in older patients with colon cancer undergoing elective colon resection: a protocol for a prospective, observational cohort study. BMJ Open. 2022;12:e057391. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 12. | Antonowicz S, Reddy S, Sgromo B. Gastrointestinal side effects of upper gastrointestinal cancer surgery. Best Pract Res Clin Gastroenterol. 2020;48-49:101706. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 13. | Tevis SE, Kennedy GD. Postoperative Complications: Looking Forward to a Safer Future. Clin Colon Rectal Surg. 2016;29:246-252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 126] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 14. | Gustafsson UO, Rockall TA, Wexner S, How KY, Emile S, Marchuk A, Fawcett WJ, Sioson M, Riedel B, Chahal R, Balfour A, Baldini G, de Groof EJ, Romagnoli S, Coca-Martinez M, Grass F, Brindle M, Hubner M. Guidelines for perioperative care in elective colorectal surgery: Enhanced Recovery After Surgery (ERAS) Society recommendations 2025. Surgery. 2025;184:109397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 64] [Article Influence: 64.0] [Reference Citation Analysis (0)] |
| 15. | Soenen S, Rayner CK, Jones KL, Horowitz M. The ageing gastrointestinal tract. Curr Opin Clin Nutr Metab Care. 2016;19:12-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 177] [Cited by in RCA: 165] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 16. | GBD 2019 Tobacco Collaborators. Spatial, temporal, and demographic patterns in prevalence of smoking tobacco use and attributable disease burden in 204 countries and territories, 1990-2019: a systematic analysis from the Global Burden of Disease Study 2019. Lancet. 2021;397:2337-2360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1322] [Cited by in RCA: 1139] [Article Influence: 227.8] [Reference Citation Analysis (0)] |
| 17. | Chen J, Ji X, Xing H. Risk factors and a nomogram model for postoperative delirium in elderly gastric cancer patients after laparoscopic gastrectomy. World J Surg Oncol. 2022;20:319. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 23] [Reference Citation Analysis (0)] |
| 18. | Nakamura M, Kido Y, Hosoya Y, Yano M, Nagai H, Monden M. Postoperative gastrointestinal dysfunction after 2-field versus 3-field lymph node dissection in patients with esophageal cancer. Surg Today. 2007;37:379-382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 23] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 19. | Braga M, Pecorelli N, Scatizzi M, Borghi F, Missana G, Radrizzani D; PeriOperative Italian Society. Enhanced Recovery Program in High-Risk Patients Undergoing Colorectal Surgery: Results from the PeriOperative Italian Society Registry. World J Surg. 2017;41:860-867. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 54] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 20. | Weimann A, Braga M, Carli F, Higashiguchi T, Hübner M, Klek S, Laviano A, Ljungqvist O, Lobo DN, Martindale RG, Waitzberg D, Bischoff SC, Singer P. ESPEN practical guideline: Clinical nutrition in surgery. Clin Nutr. 2021;40:4745-4761. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 659] [Cited by in RCA: 491] [Article Influence: 98.2] [Reference Citation Analysis (0)] |
| 21. | Zhang Y, Tan S, Wu G. ESPEN practical guideline: Clinical nutrition in surgery. Clin Nutr. 2021;40:5071. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 22. | Venara A, Neunlist M, Slim K, Barbieux J, Colas PA, Hamy A, Meurette G. Postoperative ileus: Pathophysiology, incidence, and prevention. J Visc Surg. 2016;153:439-446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 279] [Cited by in RCA: 222] [Article Influence: 22.2] [Reference Citation Analysis (4)] |
| 23. | Boden I. Appraisal of Clinical Practice Guideline: Guidelines for Perioperative Care in Elective Colorectal Surgery: Enhanced Recovery After Surgery (ERAS) Society Recommendations: 2018. J Physiother. 2024;70:71-72. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/