Published online Feb 27, 2026. doi: 10.4240/wjgs.v18.i2.114286
Revised: December 3, 2025
Accepted: January 6, 2026
Published online: February 27, 2026
Processing time: 121 Days and 1.6 Hours
Colorectal cancer (CRC) is among the most common malignant tumors. Che
To evaluate the feasibility of using PTCs to guide personalized chemotherapy for patients with CRC.
Fresh tumor tissues from 54 patients with resectable CRC were used to establish PTCs. Culture duration, diameter, and morphology were monitored. Hemato
PTCs exhibited inter- and intrapatient variability in growth rate, diameter, and morphology. Comparable propor
Accounting for patient’s pathological and clinical characteristics, PTC-based drug-sensitivity testing can help accurately identify effective agents for CRC in vitro. This novel preclinical platform may reduce unnecessary chemotherapy and support precision oncology.
Core Tip: Colorectal cancer (CRC) is one of the most common malignant tumors. Currently, FOLFOX serves as the standard postoperative adjuvant chemotherapy for resectable CRC, while FOLFIRI and FOLFOXIRI are widely used for metastatic or recurrent disease, typically in combination with targeted therapies. The microtumor patient-derived tumor-like cell clusters (PTC) drug sensitivity testing technology involves establishing an in vitro tumor model highly similar to the patient's actual tumor using their own tumor cells. This model is then utilized for drug sensitivity testing, enabling the screening and recommendation of effective treatment regimens while filtering out ineffective options based on therapeutic efficacy. This study primarily evaluates the feasibility of using PTC to guide individualized chemotherapy for CRC patients.
- Citation: Lin FL, Zhou LM, Ye YJ, Luo XL, Ji XL, Chen SQ, Xie JG, Jin BF, Liu ZD. Individual chemotherapy for patients colorectal cancer based on patient-derived tumor-like cell clusters. World J Gastrointest Surg 2026; 18(2): 114286
- URL: https://www.wjgnet.com/1948-9366/full/v18/i2/114286.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i2.114286
Colorectal cancer (CRC) is among the most common malignancy worldwide[1], with more than 1 million new cases annually. By 2030, global incidence is projected to reach 2.2 million cases and 1.1 million deaths[2,3]. Approximately 83% of patients with CRC are at an advanced stage at initial diagnosis, and may already exhibit distant metastases with poor prognosis. Currently, FOLFOX is the standard postoperative adjuvant chemotherapy for resectable CRC, while FOLFIRI and FOLFOXIRI are widely applied in metastatic or recurrent phases, often in combination with targeted agents. Studies have shown that approximately 25% of stage II and 50%-60% of stage III patients develop recurrence or metastasis within five years of radical resection, although FOLFOX adjuvant therapy improves survival[4]. The response rate of FOLFIRI in advanced rectal cancer exceeds 40%[5]. In the GERCOR study, FOLFOX and FOLFIRI were alternated as first- and second-line therapies, and multiple clinical trials demonstrated that FOLFOXIRI is more effective than FOLFIRI for metastatic CRC, although with markedly increased toxicity[6-9]. Additional evidence shows that survival in advanced CRC is associated with receiving all three agents during the disease course, as patients treated with all three drugs achieve the best outcomes[10,11]. CRC is highly heterogeneous[12], and patients with similar pathological features can show substantial differences in treatment response and survival[11,13]. Current therapy approaches therefore remain insufficient, underscoring the need for predictive models capable of optimizing individualized chemotherapy.
In vitro tumor models offer a promising platform for personalized medicine. Patient-derived organoids (PDOs), a representative tumor model, have been widely used to explore physiological and pathological mechanisms and to predict drug sensitivity[14]. However, several factors limit their clinical application. PDOs rely primarily on stem-cell proliferation and differentiation, resulting in relatively uniform cell composition and lack of stromal components. In addition, the use of Matrigel as an extracellular matrix substitute hinders standardization. Patient-derived tumor-like cell clusters (PTCs) are self-assembled tumor microspheres composed of primary epithelial cells, fibroblasts, and immune cells. PTCs closely mirror the morphology, biomarkers, genetic variation, and clinical drug sensitivity of the original tumor tissue[15], combining advantages of both PDOs and spheres to better simulate the tumor microenvironment. A PTC-based drug assay demonstrated 96.6% clinical consistency in advanced gastrointestinal cancer[15]. Moreover, the assay dynamically guided chemotherapy selection for a patients diagnosed with mucinous adenocarcinoma after conventional treatment, indicating its predictive value for drug efficacy in CRC and its potential to support precision, individualized treatment.
In this study, PTC models were established from 54 patients with CRC. We analyzed similarities and differences in PTC growth rate, diameter, and morphology, and then evaluated patient sensitivity to FOLFOX, FOLFIRI, and FOLFOXIRI using PTC drug assays. The feasibility of applying PTC models to guide individualized precision medicine was assessed at both the individual and cohort levels.
All patients enrolled in this study were newly diagnosed with resectable CRC. Human tissue samples were obtained after surgery at the 903rd Hospital of PLA Joint Logistic Support Force. Fresh tissue was immediately placed in specialized preservation solution and sent to the laboratory for processing. Ethical approval was obtained from the ethical com
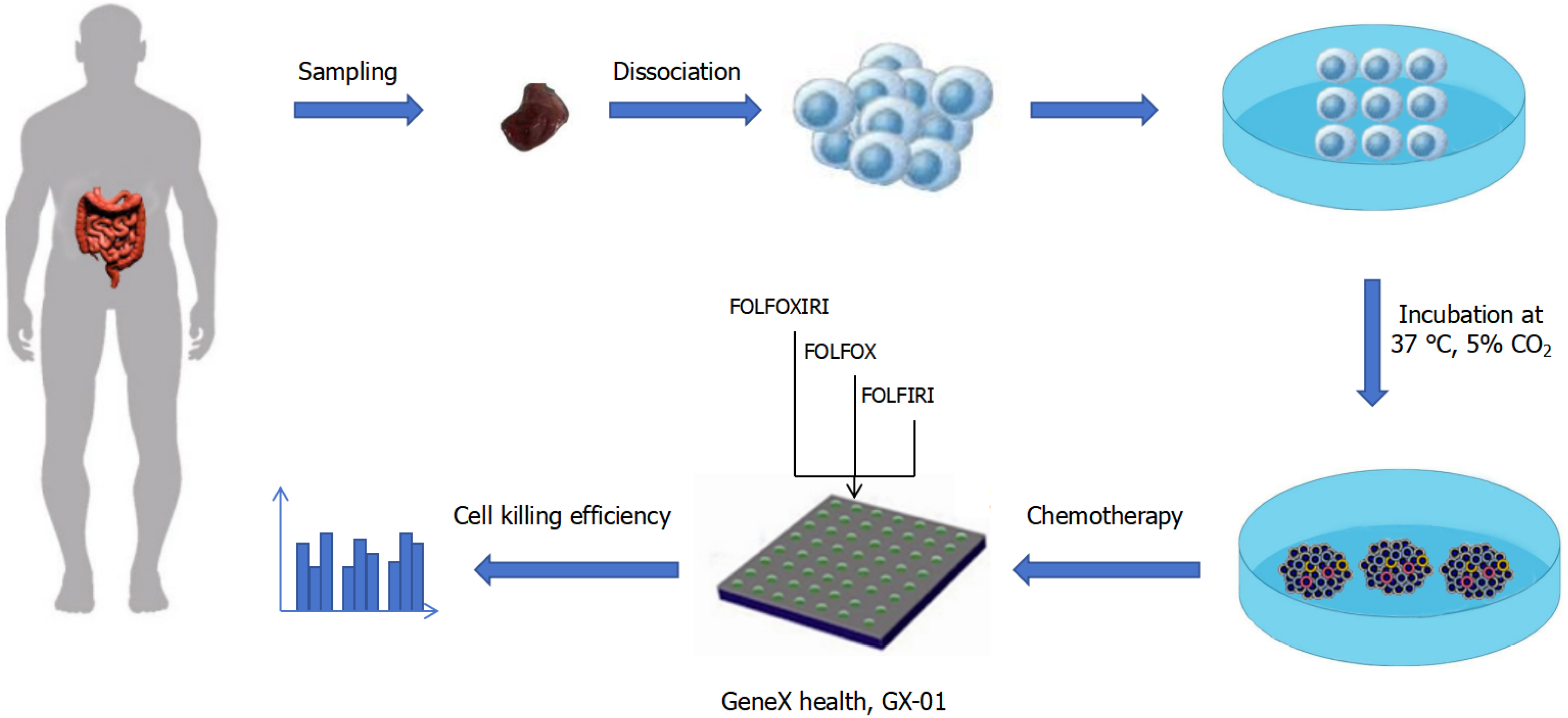
PTC culture followed established protocols[15]. Fresh tumor tissue (≥ 20 mg) without necrotic or fatty areas was collected, and multi-point sampling was used to reduce the effects of intratumoral heterogeneity. Tissues were washed 2-3 times with phosphate-buffered saline (PBS) containing 100 U/mL penicillin-streptomycin (Thermo Fisher Scientific), minced, and digested with collagenase I, II, and IV (200 U/mL each; Thermo Fisher Scientific) at 37 °C for approximately 1 hour, with pipetting every 20 minutes to facilitate digestion. Digestion was terminated with DMEM containing FBS, cells were filtered through 40-100 μm filters, centrifuged for 5 minutes (300 × g, 4 °C), resuspended in PTC growth medium, and seeded in low-attachment dishes at 105 cells/cm2. Cultures were maintained at 37 °C with 5% CO2, with medium changes every 2-3 days. Daily imaging was performed to monitor PTC size and morphology, and culture duration and diameter were recorded once PTCs reached the stationary phase.
The PTC suspension was centrifuged at 300 × g for 2 minutes and fixed in 4% paraformaldehyde overnight. PTCs were mounted unto slides, permeabilized with 1% TritonX-100 solution for 10 minutes, and nuclei were stained with hematoxylin for 10 seconds. After differentiation with 1% hydrochloric acid for 4 seconds, eosin was applied dropwise to stain the cytoplasm. Cells were dehydrated through an ascending ethanol series, cleared in xylene, and sealed with neutral balsam. Samples were examined under a Nikon Ti2-U microscope.
Paraffin sections were dewaxed and rehydrated through a graded ethanol series, then placed in citric acid buffer (pH 6.0) and heated to boiling twice for 5 minutes. Sections were blocked with 5% goat serum for 1 hour at room temperature. The primary antibody CK8/18 (Abcam) was diluted 1:100 and incubated overnight at 4 °C. After incubation with the secondary antibody for 30 minutes, DAB (Proteintech) was applied to achieve appropriate staining intensity. Imaging was performed using a Nikon Ti2-U microscope.
Drug testing followed published procedures[15]. PTCs > 40 μm in diameter were collected using 40-μm filters (BD Falcon), centrifuged at 300 × g for 10 minutes, washed with PBS, and resuspended in 100 μL PTC growth medium. A suspension containing 30-50 PTCs was seeded into Teflon-modified chips (GeneX Health, GX-01). The drug combinations FOLFOXIRI (fluorouracil + oxaliplatin + irinotecan), FOLFOX (fluorouracil + oxaliplatin), and FOLFIRI (fluorouracil + irinotecan) were added to the wells. Drug concentrations for fluorouracil, oxaliplatin, and irinotecan were 2, 4, and 1 uM, respectively, consistent with optimized efficacy concentrations reported previously[15]. Each treatment was performed in triplicate. Wells were imaged using a Nikon Ti-U microscope system and incubated at 37 °C with 5% CO2.
After seven days of treatment, wells were imaged again to evaluate drug effects by measuring changes in microsphere area. PTC drug sensitivity was determined using PA value, defined as the ratio of PTC area after treatment to that before treatment. Cell viability following exposure to drug A was calculated as: PAi = SAi,t1/SAi,t0, PA = (1/n)Σ(n, i = 1)PAi., where S is the PTC area per well, n is the number of replicates, and t0 and t1 denote measurement time points[16]. A PA cutoff of 0.7 was used to classify drug sensitivity.
Cell viability was assessed using the Celltiter-Glo reagent (Promega) according to the manufacturer’s instructions, and luminescence was recorded using a Synergy plate reader (BioTek).
Results from the drug-sensitivity assays are expressed as mean ± SD for three replicates per group. t-test and ANOVA were used for between-group comparisons; P < 0.05 was considered statistically significant.
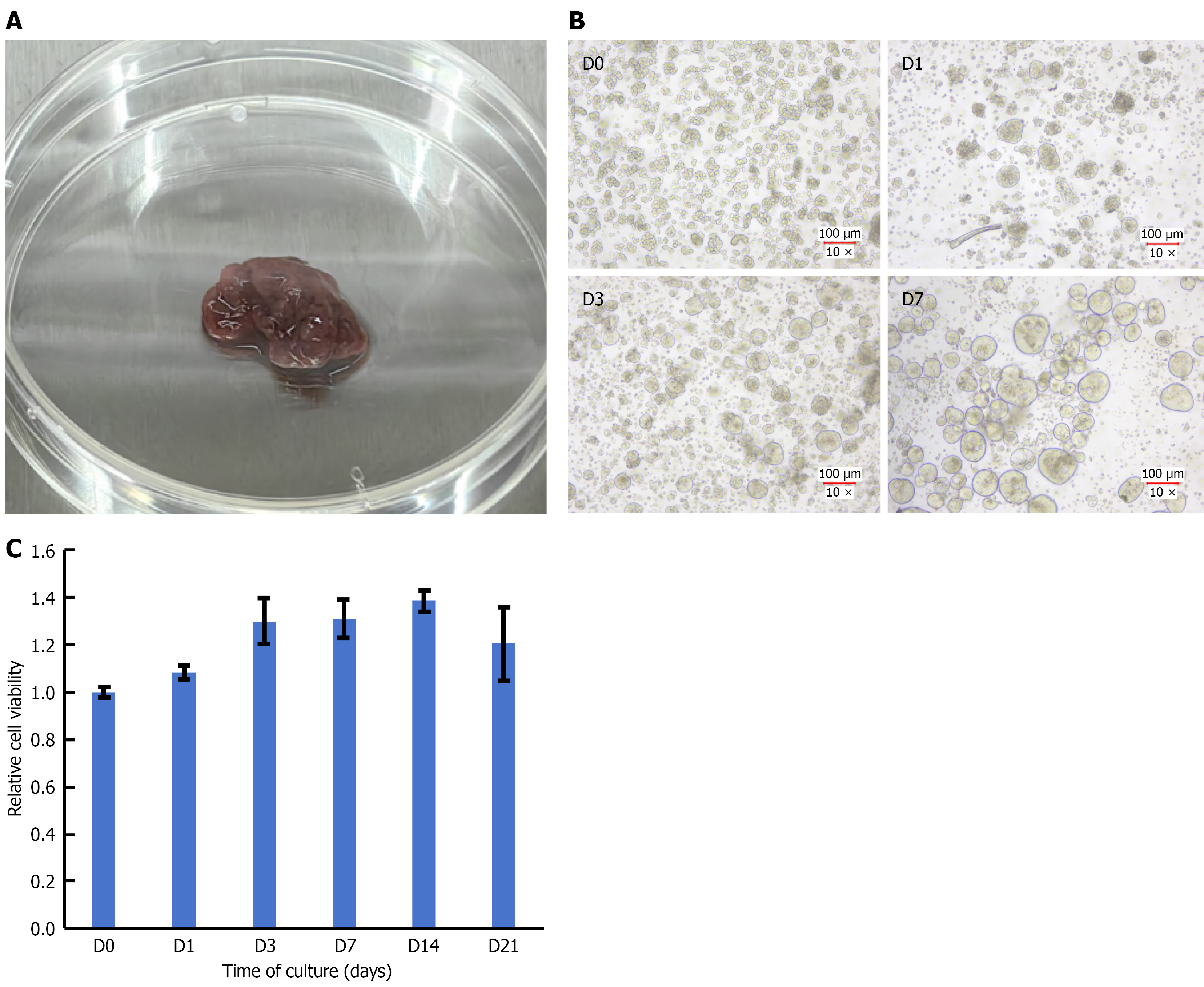
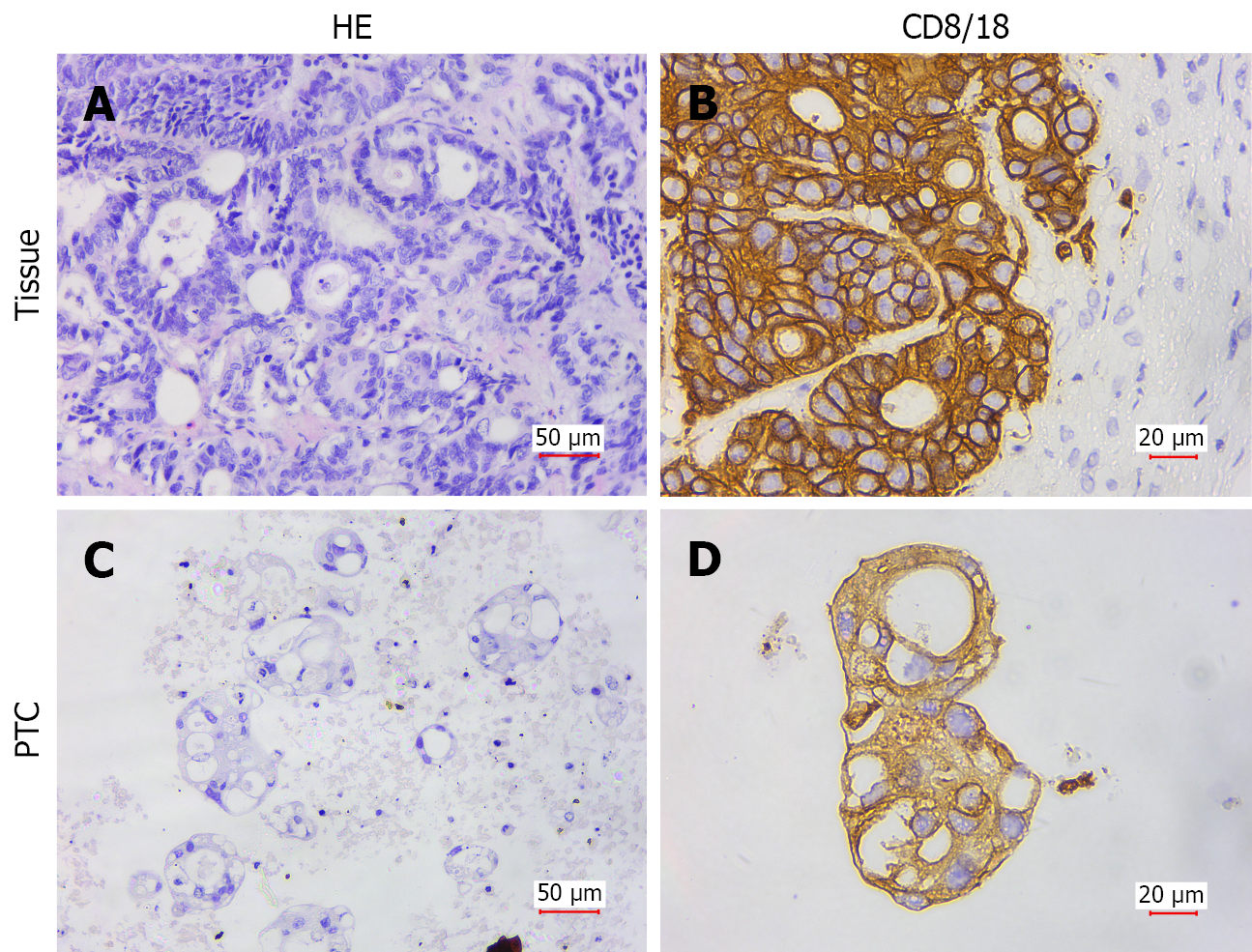
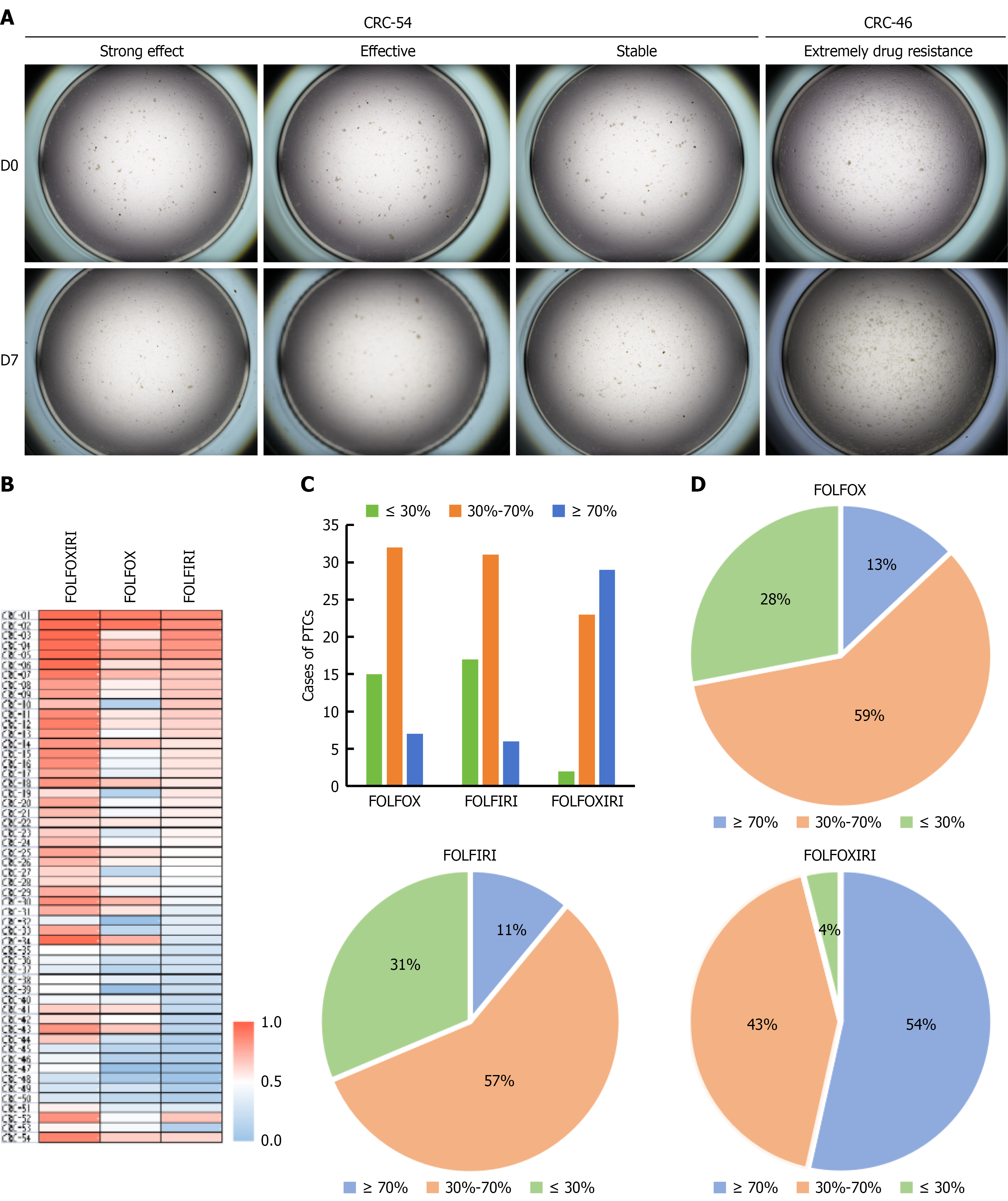
In this study, 54 PTC models were successfully established from resectable CRC patients (29 male and 25 female patients; median age, 62 years), as outlined in Figure 1. Baseline clinical characteristics were comparable across patients, with most tumors classified as medium-low or moderately differentiated adenocarcinomas (Supplementary Table 1). Primary tumor tissues were mechanically minced, digested with collagenase, filtered, centrifuged, and seeded into low-attachment dishes at appropriate densities (Figure 2A and B, D0). Within 2-7 days, dissociated epithelial, fibroblast, and immune cells migrated and self-assembled into PTC clones measuring 40-300 μm in diameter in response to growth and inhibitory factors in PTC medium, including epidermal growth factor, fibroblast growth factor, and a transforming growth factor-β inhibitor (Figure 2B, D1, D3, and D7). AS a newly developed three-dimensional in vitro culture system, PTC formed rapidly, with distinct microsphere structures observable as early as 24 hours (Figure 2B, D1). Cell viability assays showed that PTC microspheres remained stable for approximately three weeks (Figure 2C). To confirm that cultured PTCs retained properties of their parental tumors-an essential criterion for in vitro tumor modeling-we compared histological and immunohistochemical features between PTCs and matched primary tissues. The cultured microspheres preserved clear adenocarcinoma morphology and epithelial lineage (Figure 3).
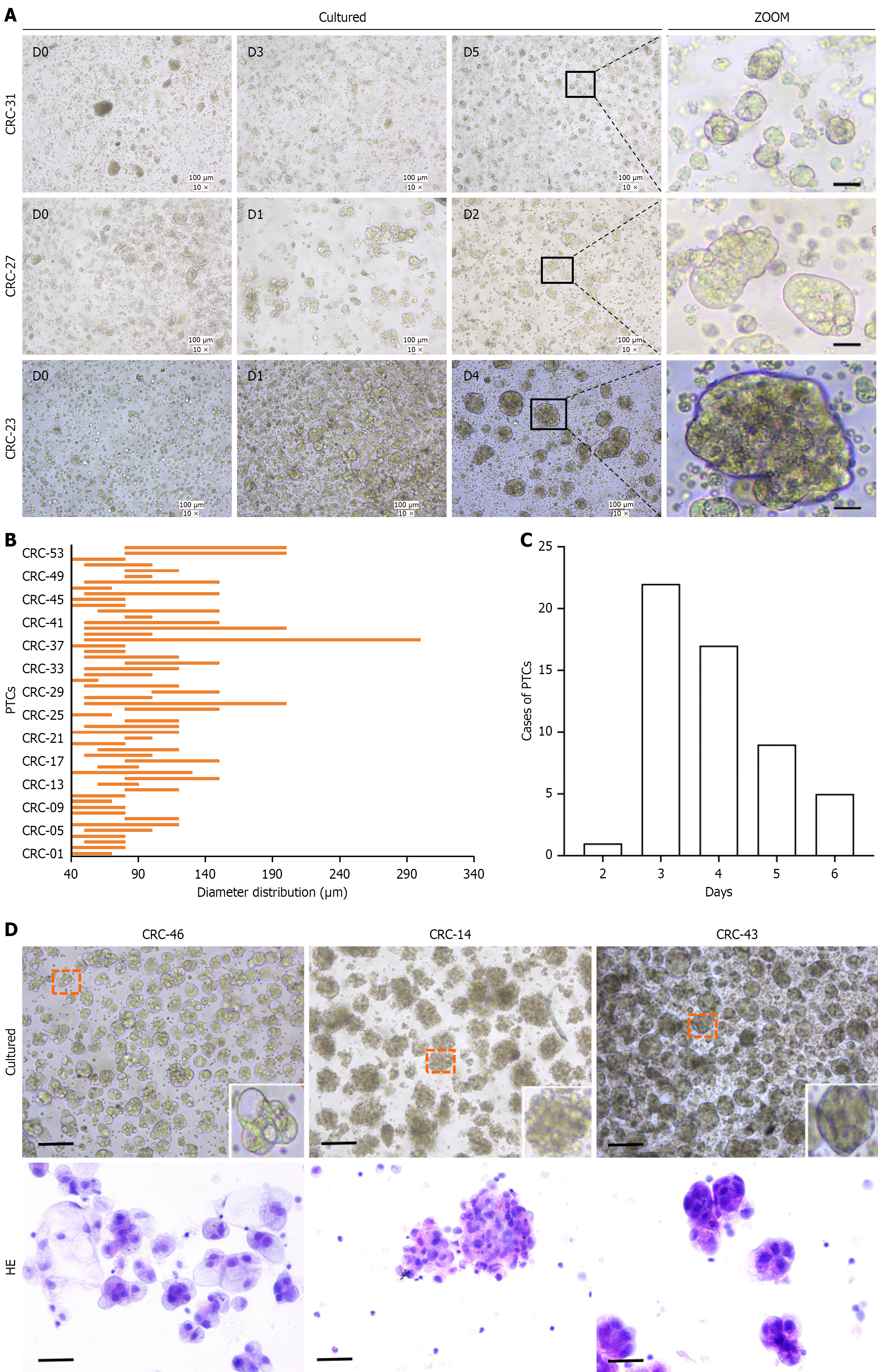
PTCs demonstrated marked heterogeneity across patients in formation rate, diameter, and density. CRC-27 and CRC-23 generated typical PTCs within 24 hours; CRC-27 clusters grew steadily in diameter and volume during the first 24 hours and reached a plateau by day 2, whereas CRC-23 clusters grew to 50-120 μm in diameter within five days following migration and self-assembly of dissociated primary cells. In contrast, CRC-31 clusters aggregated more slowly, forming spheres only by day 3 and remaining relatively uniform in diameter at 40-60 μm (Figure 4A). Overall, PTC diameters varied widely, typically at 50-150 μm, with some reaching 200 μm (CRC-40, CRC-27, CRC-53, and CRC-54) or 300 μm (CRC-38). Intrapatient variability was also evident: Some patients produced relatively uniform clusters-CRC-42 (80-100 μm) and CRC-01 (40-70 μm-whereas CRC-38 exhibited internal heterogeneity, spanning 50-300 μm. CRC-27 PTC also showed broad size distribution (50-200 μm) (Figure 4B). The mechanisms underlying this diversity require further investigation. Notably, PTCs from 48 patients reached a growth plateau within 3-5 days of culturing, reaching the criteria for drug-sensitivity testing once proliferation had largely ceased (Figure 4C).
Hematoxylin and eosin (HE) staining was then performed to assess PTC morphology. Histological analysis demonstrated nuclear and cellular atypia, including nuclear enlargement, malformation, and disproportionate nucleoplasm (Figure 4D). These findings indicate that PTC models in vitro reflect tumor-associated morphological features. Distinct structural patterns were also observed: Some PTCs contained variably sized acini (CRC-46), others showed a non-dense phenotype with irregular boundaries (CRC-14), whereas CRC-43 exhibited a dense, compact microsphere structure with clearly defined margins (Figure 4D).
All 54 patient-derived PTC models were evaluated for drug sensitivity once they reached a stable state. Drug concentration and exposure time are critical variables in in vitro susceptibility assessments; therefore, we used the optimized efficacy concentrations reported in the original literature[15]. FOLFOX, an oxaliplatin-based regimen, is a standard clinical chemotherapy for CRC. As shown in Figure 5A, both FOLFOX and FOLFIRI exerted cytotoxic effects on PTCs. Overall, 39 PTC models (72%) were sensitive to FOLFOX and 37 (68%) to FOLFIRI (Figure 5B), suggesting broadly comparable proportions of responsive patients-consistent with multiple clinical reports. FOLFOX produced strong cytotoxicity in 7 cases (18%), with killing efficiencies exceeding 70%, and reached a maximum efficacy of 92%, characterized by complete microsphere disintegration and extensive apoptosis. In total, 32 cases (82%) demonstrated effective FOLFOX-mediated cytotoxicity. FOLFIRI yielded similar patterns, with 6 cases (16%) showing strong cytotoxicity and 31 cases (84%) showing effective cytotoxicity. No significant differences were observed in the distributions of sensitive populations between the two regimens (Figure 5C and D). Sensitivity and resistance to FOLFOX, FOLFIRI, and FOLFOXIRI varied markedly across PTC models, reflecting inherent tumor heterogenicity (Figure 5A and C). Thus, PTCs capture clinically relevant diversity in therapeutic response and can model solid tumor behavior in vitro.
Drug-sensitivity testing with FOLFOXIRI revealed that 97% of PTCs were responsive: 29 cases (54%) showed strong cytotoxicity, and 23 cases (43%) showed effective cytotoxicity, whereas only 2 cases (4%) were stable or resistant (Figure 5B). These results indicate apotent antitumor activity of FOLFOXIRI in CRC, suggesting that certain patient subgroups may derive substantial benefit from it. Nonetheless, factors such as tumor stage, genetic variant, functional status, economic situation, and regimen-specific toxicities should inform clinical decision-making, and patient tolerance must be carefully evaluated.
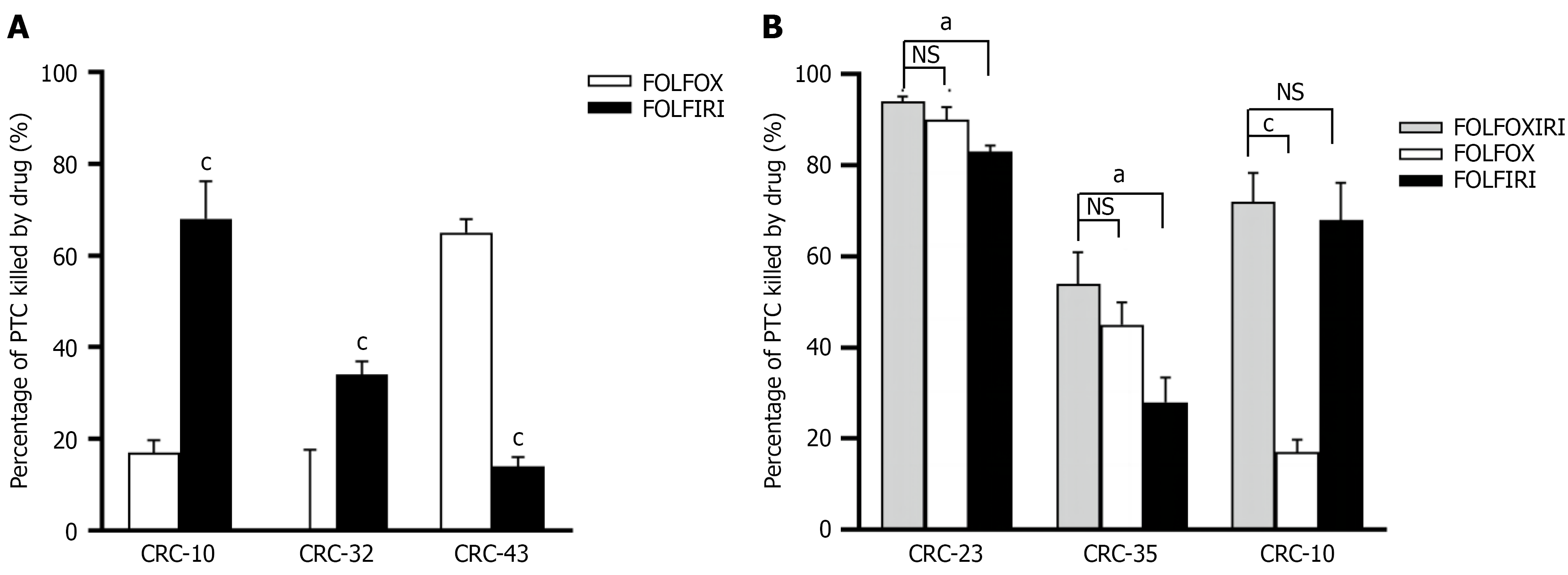
The findings above showed that FOLFOX and FOLFIRI were broadly similar in both drug-sensitive population proportions and killing efficiencies, consistent with their mutual use as first- and second-line regimens in clinical practice. However, individual patients exhibited substantial differences in sensitivity to the two regimens. For example, malignant tumor cells from CRC-10 responded effectively to FOLFIRI (KE = 68%) but remained largely stable under FOLFOX, with only 17% killing efficiency. Likewise, CRC-32 was strongly resistant to FOLFOX while demonstrating moderate sensitivity to FOLFIRI (KE = 34%). In contrast, CRC-43, 65showed marked responsiveness to FOLFOX, with 65% of clusters effectively killed, whereas cluster size changed only slightly following FOLFIRI treatment (Figure 6A). These differences likely reflect intrinsic cellular and molecular characteristics of individual tumors as well as prior treatment history. Overall, although population-level analysis revealed no major differences between FOLFOX and FOLFIRI, the regimens are not interchangeable for many patients. Using a 10% killing-ratio threshold, approximately 60% of patients could identify a more effective chemotherapy regimen through personalized PTC-based testing. Patients who did not respond to standard first-line therapy may therefore receive timelier and more appropriate alternative treatments. By directly visualizing drug-induced cytotoxicity in a patient’s own tumor cells, PTC drug-testing helps evaluate therapeutic sensitivity and reduce the likelihood of ineffective treatment, thereby advancing precision oncology.
Additionally, in vitro PTC drug-sensitivity testing may help reduce toxicity associated with overtreatment. Although FOLFOXIRI exhibited killing effects in 97% of PTC models from 54 patients with CRC, triple-drug (FOLFOXIRI) and doublet regimens (FOLFOX or FOLFIRI) yielded comparable cytotoxicity in 26 patients (48%). For instance, cell viability values for FOLFOXIRI and FOLFOX were similarly high in CRC-2 (0.94 and 0.90), and comparable viability was observed in CRC-35 (0.54 and 0.45). Partial disintegration of CRC-10 PTC clusters following FOLFOXIRI or FOLFIRI exposure also showed similar cytotoxic efficiencies (0.72 and 0.68) (Figure 6B). These findings suggest that, for some patients, doublet regimens may offer antitumor effects similar to FOLFOXIRI. To balance efficacy with tolerability, therapeutic decisions should incorporate drug toxicity profiles, safety considerations, and patient-specific tolerance.
CRC is a highly heterogeneous malignancy, and inter- and intrapatient heterogeneity substantially influence tumor growth, invasiveness, drug sensitivity, and prognosis. In this study, PTC models derived from primary CRC tissue exhibited distinct growth rates, microsphere diameters, and histomorphologies. Most PTCs, ranging from 40-300 μm in diameter, met drug-testing criteria after in vitro culture for 3-5 days. Considerable size diversity was observed both across and within patients; for instance, CRC-38 clusters ranged from 50-300 μm. HE staining revealed three morphological patterns-acinus, non-dense, and dense types. Similar morphological and developmental variation has been reported for organoid systems. For example, Kopper et al[17] demonstrated wide morphological diversity among ovary carcinoma organoids using scanning electron microscopy, ranging from cystic to dense, with high-grade serous organoids showing differences in circularity and cellular cohesiveness. Epithelial ovarian cancer organoids also vary in morphology and generally form within 2-4 weeks, exhibiting dense and cystic types[18]. Likewise, lung adenocarcinoma organoids vary by histological subtype, with acinar carcinoma organoids adopting adenomatous and cystic morphologies and solid carcinoma organoids appearing dense and compact[19]. Heterogeneity across and within tumors likely reflects combined environmental and genetic influences. In 2017, Jaxon Laboratory of the United States, the Genome Institute of Singapore, and the National Cancer Research Center of Singapore identified two distinct subtypes of cancer-associated fibroblasts using a novel algorithm for reference component analysis, advancing the classification of CRC cells and improving prognostic assessment[20]. In 2018, single-cell sequencing revealed marked genetic uniqueness within CRC tissue, with individual organoids displaying extensive diversity in mutational burden and epigenetic features[21]. Giving the pronounced inter- and intra-PTC heterogeneity observed in this study, further analyses integrating clinicopathological and genetic characteristics are warranted.
Currently, FOLFOX is the standard postoperative adjuvant chemotherapy regimen for resectable CRC, whereas FOLFIRI and FOLFOXIRI are widely used for metastatic or recurrent phase, often in combination with targeted therapy. Clinical trials have shown comparable efficacy between FOLFOX and FOLFIRI, with no significant differences[22-24]. In GERCOR’s study, FOLFOX and FOLFIRI regimens served as first- and second-line options interchangeably[25]. Despite this evidence, a subset of patients consistently show poor response to standard treatments. Precision medicine in contrast to evidence-based population approaches, emphasizes individualized benefit. The PTC model used in this study supports individualized prediction of treatment response through systematic modeling and clinical comparison[15]. Among the 54 PTCs evaluated, 72% and 68% were sensitive to FOLFOX and FOLFIRI, respectively, with most showing effective cytotoxicity. Notably, 97% of patients showed sensitivity to FOLFOXIRI, indicating broad potential benefit. Multiple clinical studies have similarly shown that FOLFOXIRI outperforms FOLFIRI in metastatic CRC, prolonging progression-free survival by 2.9 months and overall survival by 5.9 months[6-9]. However, FOLFOXIRI is associated with higher rates of peripheral neurotoxicity, leukopenia, myelosuppression, and diarrhea[26]. Its wider use in Europe and the United States reflects both differences in UGT1A1 polymorphisms and distinct drug tolerability profiles between Chinese and Western populations[27,28]. Consequently, several studies have explored modified dosing and infusion strategies to optimize long-term patient outcomes.
Although FOLFOX and FOLFIRI-used interchangeably as first- and second-line regimens-show no significant differences in efficacy, 20%-50% of patients never receive second-line therapy after completing the first. This highlights the importance of selecting the optimal initial regimen. In this study, PTC-derived drug responses were not uniform: CRC-10 was more sensitive to FOLFIRI than FOLFOX, whereas CRC-43 showed the opposite pattern. Although FOLFOXIRI demonstrated stronger activity and benefited a broader patient population, 48% of PTCs exhibited similar inhibitory responses to FOLFOXIRI or FOLFIRI as to FOLFOXIRI. In such cases, double therapy may be more appropriate when considering treatment cost, toxicity, and overall benefit. Clinical selection between FOLFOX and FOLFIRI typically depends on disease stage, tumor location, comorbidities, and patient performance status. FOLFIRI is more frequently administered in patients with stage II disease, whereas FOLFOX is more common in stage IV disease[29], possibly reflecting greater tolerability of FOLFIRI in earlier stages and the perceived superior efficacy of FOLFOX in advanced diseases[30]. Recent trials suggest that FOLFIRI may be more effective in patients with microsatellite instability-high tumors[31], whereas FOLFOX may be preferred for RAS wild-type disease[32]. The expanding integration of targeted therapy and immunotherapy also shapes regimen selection, as these agents may be combined with-or replace-traditional chemotherapy[33]. Consequently, additional clinical validation is required to determine whether PTC-guided testing can reliably inform adjuvant chemotherapy decisions for CRC. Moreover, intratumoral heterogeneity remains a major challenge for in vitro drug-sensitivity testing; multi-site sampling and systematic comparison with clinical outcomes may help reduce its impact. In clinical practice, treatment decisions must carefully balance antitumor efficacy and toxicity to achieve precise, individualized therapy.
We successfully established a series of in vitro tumor models that capture both intra- and interpatient heterogeneity and show potential for predicting chemotherapy responsiveness and prognosis in CRC. By integrating statistical analyses of in vitro drug testing with prior clinical evidence, we demonstrated the preliminary feasibility of individualized precision chemotherapy. Future studies will focus on validating the clinical utility of the PTC model and elucidating mechanisms of drug resistance at genomic, molecular, cellular, and proteomic levels to strengthen its application in personalized chemotherapy.
The authors are thankful for the patients who agreed to participate in the study. Zhejiang GeneX Precision Medicine Co., Ltd provided significant support and assistance for model construction and drug testing in vitro.
| 1. | Xi Y, Xu P. Global colorectal cancer burden in 2020 and projections to 2040. Transl Oncol. 2021;14:101174. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2033] [Cited by in RCA: 1639] [Article Influence: 327.8] [Reference Citation Analysis (7)] |
| 2. | Testa U, Pelosi E, Castelli G. Colorectal cancer: genetic abnormalities, tumor progression, tumor heterogeneity, clonal evolution and tumor-initiating cells. Med Sci (Basel). 2018;6:31. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 184] [Cited by in RCA: 195] [Article Influence: 24.4] [Reference Citation Analysis (2)] |
| 3. | Łukaszewicz-Zając M, Mroczko B. Circulating Biomarkers of Colorectal Cancer (CRC)-Their Utility in Diagnosis and Prognosis. J Clin Med. 2021;10:2391. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (1)] |
| 4. | André T, Boni C, Mounedji-Boudiaf L, Navarro M, Tabernero J, Hickish T, Topham C, Zaninelli M, Clingan P, Bridgewater J, Tabah-Fisch I, de Gramont A; Multicenter International Study of Oxaliplatin/5-Fluorouracil/Leucovorin in the Adjuvant Treatment of Colon Cancer (MOSAIC) Investigators. Oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment for colon cancer. N Engl J Med. 2004;350:2343-2351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3029] [Cited by in RCA: 2696] [Article Influence: 122.5] [Reference Citation Analysis (3)] |
| 5. | Kim DY, Paek TY, Oh SY, Kim YB, Lee JH, Lee MY, Choi ZS, Suh KW. Pretreatment selection of regimen according to genetic analysis improves the efficacy of chemotherapy in the first line treatment of metastatic colorectal cancer. J Surg Oncol. 2014;109:250-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 6. | Souglakos J, Androulakis N, Syrigos K, Polyzos A, Ziras N, Athanasiadis A, Kakolyris S, Tsousis S, Kouroussis Ch, Vamvakas L, Kalykaki A, Samonis G, Mavroudis D, Georgoulias V. FOLFOXIRI (folinic acid, 5-fluorouracil, oxaliplatin and irinotecan) vs FOLFIRI (folinic acid, 5-fluorouracil and irinotecan) as first-line treatment in metastatic colorectal cancer (MCC): a multicentre randomised phase III trial from the Hellenic Oncology Research Group (HORG). Br J Cancer. 2006;94:798-805. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 310] [Cited by in RCA: 275] [Article Influence: 13.8] [Reference Citation Analysis (1)] |
| 7. | Marques RP, Duarte GS, Sterrantino C, Pais HL, Quintela A, Martins AP, Costa J. Triplet (FOLFOXIRI) versus doublet (FOLFOX or FOLFIRI) backbone chemotherapy as first-line treatment of metastatic colorectal cancer: A systematic review and meta-analysis. Crit Rev Oncol Hematol. 2017;118:54-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 63] [Article Influence: 7.0] [Reference Citation Analysis (4)] |
| 8. | Cremolini C, Loupakis F, Antoniotti C, Lupi C, Sensi E, Lonardi S, Mezi S, Tomasello G, Ronzoni M, Zaniboni A, Tonini G, Carlomagno C, Allegrini G, Chiara S, D'Amico M, Granetto C, Cazzaniga M, Boni L, Fontanini G, Falcone A. FOLFOXIRI plus bevacizumab versus FOLFIRI plus bevacizumab as first-line treatment of patients with metastatic colorectal cancer: updated overall survival and molecular subgroup analyses of the open-label, phase 3 TRIBE study. Lancet Oncol. 2015;16:1306-1315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 921] [Cited by in RCA: 836] [Article Influence: 76.0] [Reference Citation Analysis (2)] |
| 9. | Cremolini C, Antoniotti C, Rossini D, Lonardi S, Loupakis F, Pietrantonio F, Bordonaro R, Latiano TP, Tamburini E, Santini D, Passardi A, Marmorino F, Grande R, Aprile G, Zaniboni A, Murgioni S, Granetto C, Buonadonna A, Moretto R, Corallo S, Cordio S, Antonuzzo L, Tomasello G, Masi G, Ronzoni M, Di Donato S, Carlomagno C, Clavarezza M, Ritorto G, Mambrini A, Roselli M, Cupini S, Mammoliti S, Fenocchio E, Corgna E, Zagonel V, Fontanini G, Ugolini C, Boni L, Falcone A; GONO Foundation Investigators. Upfront FOLFOXIRI plus bevacizumab and reintroduction after progression versus mFOLFOX6 plus bevacizumab followed by FOLFIRI plus bevacizumab in the treatment of patients with metastatic colorectal cancer (TRIBE2): a multicentre, open-label, phase 3, randomised, controlled trial. Lancet Oncol. 2020;21:497-507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 271] [Cited by in RCA: 252] [Article Influence: 42.0] [Reference Citation Analysis (0)] |
| 10. | Grothey A, Sargent D, Goldberg RM, Schmoll HJ. Survival of patients with advanced colorectal cancer improves with the availability of fluorouracil-leucovorin, irinotecan, and oxaliplatin in the course of treatment. J Clin Oncol. 2004;22:1209-1214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 857] [Cited by in RCA: 765] [Article Influence: 34.8] [Reference Citation Analysis (0)] |
| 11. | Buyse M, Thirion P, Carlson RW, Burzykowski T, Molenberghs G, Piedbois P. Relation between tumour response to first-line chemotherapy and survival in advanced colorectal cancer: a meta-analysis. Meta-Analysis Group in Cancer. Lancet. 2000;356:373-378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 317] [Cited by in RCA: 280] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 12. | Chan DKH, Buczacki SJA. Tumour heterogeneity and evolutionary dynamics in colorectal cancer. Oncogenesis. 2021;10:53. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 13. | Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53448] [Cited by in RCA: 56088] [Article Influence: 7011.0] [Reference Citation Analysis (10)] |
| 14. | Clevers H. Modeling Development and Disease with Organoids. Cell. 2016;165:1586-1597. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2646] [Cited by in RCA: 2270] [Article Influence: 227.0] [Reference Citation Analysis (0)] |
| 15. | Yin S, Xi R, Wu A, Wang S, Li Y, Wang C, Tang L, Xia Y, Yang D, Li J, Ye B, Yu Y, Wang J, Zhang H, Ren F, Zhang Y, Shen D, Wang L, Ying X, Li Z, Bu Z, Ji X, Gao X, Jia Y, Jia Z, Li N, Li Z, Ji JF, Xi JJ. Patient-derived tumor-like cell clusters for drug testing in cancer therapy. Sci Transl Med. 2020;12:eaaz1723. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 68] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 16. | Tsuchida Y, Therasse P. Response evaluation criteria in solid tumors (RECIST): new guidelines. Med Pediatr Oncol. 2001;37:1-3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 156] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 17. | Kopper O, de Witte CJ, Lõhmussaar K, Valle-Inclan JE, Hami N, Kester L, Balgobind AV, Korving J, Proost N, Begthel H, van Wijk LM, Revilla SA, Theeuwsen R, van de Ven M, van Roosmalen MJ, Ponsioen B, Ho VWH, Neel BG, Bosse T, Gaarenstroom KN, Vrieling H, Vreeswijk MPG, van Diest PJ, Witteveen PO, Jonges T, Bos JL, van Oudenaarden A, Zweemer RP, Snippert HJG, Kloosterman WP, Clevers H. An organoid platform for ovarian cancer captures intra- and interpatient heterogeneity. Nat Med. 2019;25:838-849. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 304] [Cited by in RCA: 629] [Article Influence: 89.9] [Reference Citation Analysis (0)] |
| 18. | Maenhoudt N, Defraye C, Boretto M, Jan Z, Heremans R, Boeckx B, Hermans F, Arijs I, Cox B, Van Nieuwenhuysen E, Vergote I, Van Rompuy AS, Lambrechts D, Timmerman D, Vankelecom H. Developing Organoids from Ovarian Cancer as Experimental and Preclinical Models. Stem Cell Reports. 2020;14:717-729. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 148] [Article Influence: 24.7] [Reference Citation Analysis (0)] |
| 19. | Li Z, Qian Y, Li W, Liu L, Yu L, Liu X, Wu G, Wang Y, Luo W, Fang F, Liu Y, Song F, Cai Z, Chen W, Huang W. Human Lung Adenocarcinoma-Derived Organoid Models for Drug Screening. iScience. 2020;23:101411. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 106] [Article Influence: 17.7] [Reference Citation Analysis (0)] |
| 20. | Li H, Courtois ET, Sengupta D, Tan Y, Chen KH, Goh JJL, Kong SL, Chua C, Hon LK, Tan WS, Wong M, Choi PJ, Wee LJK, Hillmer AM, Tan IB, Robson P, Prabhakar S. Reference component analysis of single-cell transcriptomes elucidates cellular heterogeneity in human colorectal tumors. Nat Genet. 2017;49:708-718. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 988] [Cited by in RCA: 811] [Article Influence: 90.1] [Reference Citation Analysis (1)] |
| 21. | Roerink SF, Sasaki N, Lee-Six H, Young MD, Alexandrov LB, Behjati S, Mitchell TJ, Grossmann S, Lightfoot H, Egan DA, Pronk A, Smakman N, van Gorp J, Anderson E, Gamble SJ, Alder C, van de Wetering M, Campbell PJ, Stratton MR, Clevers H. Intra-tumour diversification in colorectal cancer at the single-cell level. Nature. 2018;556:457-462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 497] [Cited by in RCA: 415] [Article Influence: 51.9] [Reference Citation Analysis (2)] |
| 22. | Falcone A, Ricci S, Brunetti I, Pfanner E, Allegrini G, Barbara C, Crinò L, Benedetti G, Evangelista W, Fanchini L, Cortesi E, Picone V, Vitello S, Chiara S, Granetto C, Porcile G, Fioretto L, Orlandini C, Andreuccetti M, Masi G; Gruppo Oncologico Nord Ovest. Phase III trial of infusional fluorouracil, leucovorin, oxaliplatin, and irinotecan (FOLFOXIRI) compared with infusional fluorouracil, leucovorin, and irinotecan (FOLFIRI) as first-line treatment for metastatic colorectal cancer: the Gruppo Oncologico Nord Ovest. J Clin Oncol. 2007;25:1670-1676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 987] [Cited by in RCA: 876] [Article Influence: 46.1] [Reference Citation Analysis (2)] |
| 23. | Saltz LB, Clarke S, Díaz-Rubio E, Scheithauer W, Figer A, Wong R, Koski S, Lichinitser M, Yang TS, Rivera F, Couture F, Sirzén F, Cassidy J. Bevacizumab in combination with oxaliplatin-based chemotherapy as first-line therapy in metastatic colorectal cancer: a randomized phase III study. J Clin Oncol. 2008;26:2013-2019. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2410] [Cited by in RCA: 2207] [Article Influence: 122.6] [Reference Citation Analysis (6)] |
| 24. | Hochster HS, Hart LL, Ramanathan RK, Childs BH, Hainsworth JD, Cohn AL, Wong L, Fehrenbacher L, Abubakr Y, Saif MW, Schwartzberg L, Hedrick E. Safety and efficacy of oxaliplatin and fluoropyrimidine regimens with or without bevacizumab as first-line treatment of metastatic colorectal cancer: results of the TREE Study. J Clin Oncol. 2008;26:3523-3529. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 447] [Cited by in RCA: 436] [Article Influence: 24.2] [Reference Citation Analysis (1)] |
| 25. | Tournigand C, André T, Achille E, Lledo G, Flesh M, Mery-Mignard D, Quinaux E, Couteau C, Buyse M, Ganem G, Landi B, Colin P, Louvet C, de Gramont A. FOLFIRI followed by FOLFOX6 or the reverse sequence in advanced colorectal cancer: a randomized GERCOR study. J Clin Oncol. 2004;22:229-237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2364] [Cited by in RCA: 2109] [Article Influence: 95.9] [Reference Citation Analysis (8)] |
| 26. | Innocenti F, Undevia SD, Iyer L, Chen PX, Das S, Kocherginsky M, Karrison T, Janisch L, Ramírez J, Rudin CM, Vokes EE, Ratain MJ. Genetic variants in the UDP-glucuronosyltransferase 1A1 gene predict the risk of severe neutropenia of irinotecan. J Clin Oncol. 2004;22:1382-1388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 821] [Cited by in RCA: 696] [Article Influence: 31.6] [Reference Citation Analysis (4)] |
| 27. | Su YY, Chiang NJ, Chang JS, Wang YW, Shen BN, Li YJ, Hwang DY, Shan YS, Chen LT. The association between UGT1A1 polymorphisms and treatment toxicities of liposomal irinotecan. ESMO Open. 2023;8:100746. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 28. | Ma X, Han S, Liu Y, Liu JT, Fang J, Zhang YH. Pharmacogenetic impact of UGT1A1 polymorphisms on pulmonary neuroendocrine tumours treated with metronomic irinotecan-based chemotherapy in Chinese populations. J Pharm Pharmacol. 2020;72:1528-1535. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 7] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 29. | Mulya PAS, Satriyasa BK, Dewi NWS, Ernawati DK. FOLFOX vs. FOLFIRI in Colorectal Adenocarcinoma: A Retrospective Study of Treatment Patterns, Side Effects, and Treatment Response. Biosci Med Biomed Transl Res. 2025;9:6667-6680. [DOI] [Full Text] |
| 30. | Mohelnikova-Duchonova B, Melichar B, Soucek P. FOLFOX/FOLFIRI pharmacogenetics: the call for a personalized approach in colorectal cancer therapy. World J Gastroenterol. 2014;20:10316-10330. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 46] [Cited by in RCA: 67] [Article Influence: 5.6] [Reference Citation Analysis (1)] |
| 31. | Sun BL. Current Microsatellite Instability Testing in Management of Colorectal Cancer. Clin Colorectal Cancer. 2021;20:e12-e20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 43] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 32. | Raimondi A, Nichetti F, Stahler A, Wasan HS, Aranda E, Randon G, Kurreck A, Meade AM, Díaz-Rubio E, Niger M, Stintzing S, Palermo F, Trarbach T, Prisciandaro M, Sommerhäuser G, Fisher D, Morano F, Pietrantonio F, Modest DP. Optimal maintenance strategy following FOLFOX plus anti-EGFR induction therapy in patients with RAS wild type metastatic colorectal cancer: An individual patient data pooled analysis of randomised clinical trials. Eur J Cancer. 2023;190:112945. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 33. | Lin ZP, Hu XL, Chen D, Huang DB, Zou XG, Zhong H, Xu SX, Chen Y, Li XQ, Zhang J. Efficacy and safety of targeted therapy plus immunotherapy combined with hepatic artery infusion chemotherapy (FOLFOX) for unresectable hepatocarcinoma. World J Gastroenterol. 2024;30:2321-2331. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 13] [Reference Citation Analysis (5)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/