Published online Feb 27, 2026. doi: 10.4240/wjgs.v18.i2.114378
Revised: December 21, 2025
Accepted: January 7, 2026
Published online: February 27, 2026
Processing time: 114 Days and 2.1 Hours
Despite therapeutic advances, outcomes in hepatocellular carcinoma (HCC) re
To compare efficacy and prognosis in HCC patients treated with synchronous vs sequential transcatheter arterial chemoembolization (TACE) and microwave ablation (MWA).
A total of 106 patients with HCC admitted between March 2022 and March 2024 were included. Patients receiving concurrent TACE and MWA constituted the synchronous group (n = 56), while those treated with TACE followed by MWA formed the sequential group (n = 50). Intergroup comparisons encompassed cura
No significant intergroup differences were observed in curative efficacy, ablation power, prognosis, or overall complication rates (P > 0.05). However, the synchronous group required fewer ablation needles and shorter ablation durations than the sequential group (P < 0.05), AFP, AFP-L3, TBIL, ALT, and AST levels significantly decreased after treatment in both groups (P < 0.05), with no significant differences between groups (P > 0.05).
Synchronous TACE combined with MWA is non-inferior to the sequential approach regarding therapeutic efficacy, survival outcomes, safety, and tumor control in HCC. Notably, the synchronous strategy offers procedural advan
Core Tip: This study comparatively evaluated synchronous versus sequential combinations of transcatheter arterial chemoembolization and microwave ablation for hepatocellular carcinoma. Both strategies effectively controlled tumor lesions, inhibited tumor markers, and improved liver function, with no significant differences in patient outcomes. Importantly, the synchronous approach significantly reduced ablation attempts and procedure duration, indicating a modest procedural advantage over the sequential regimen.
- Citation: Yu Y, Yuan W, Lei J, Zhao CB, Tao CG, Liu SH, Wang BL. Synchronous vs sequential combination of transarterial chemoembolization and microwave ablation for hepatocellular carcinoma: Efficacy and prognosis. World J Gastrointest Surg 2026; 18(2): 114378
- URL: https://www.wjgnet.com/1948-9366/full/v18/i2/114378.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i2.114378
Hepatocellular carcinoma (HCC) is a highly heterogeneous malignancy characterized by high mortality and global prevalence. It most commonly arises in the context of chronic liver disease, including long-term hepatitis B virus or hepatitis C virus infection, metabolic dysfunction-associated steatotic liver disease, and alcohol-related liver injury[1,2]. Global data from 2020 reported nearly one million new HCC cases and more than 830000 deaths[3], with projections indicating a 55% increase in incidence over the next 15 years. Prognosis varies substantially by disease stage: Over 70% of patients diagnosed at an early stage survive beyond 5 years compared with approximately 20% of those with advanced disease[4,5]. Management options for HCC include surgical resection as well as radiotherapy, systemic chemotherapy, molecularly targeted therapies, transcatheter arterial chemoembolization (TACE), and microwave ablation (MWA). Although these modalities continue to evolve, improving therapeutic efficacy and prognosis remains a clinical priority[6,7], largely due to the often asymptomatic early onset of HCC and the emergence of treatment resistance[8]. Consequently, continued exploration of optimized therapeutic strategies is warranted.
TACE is a widely used locoregional therapy for unresectable HCC, aiming to alleviate symptoms and enhance quality of life[9]. By embolizing tumor-feeding vessels, TACE induces localized ischemia, while the concomitant delivery of chemotherapeutic agents enhances tumor necrosis. This approach has been shown to improve survival outcomes, reduce tumor burden, and support postoperative liver function[10]. MWA, another locoregional modality, achieves higher intratumoral temperatures and larger ablation zones in a shorter time than single-needle radiofrequency ablation (RFA), providing greater resistance to the heat-sink effect near major vessels and improve local tumor control[11]. Combining TACE with MWA can yield synergistic effects, as arterial occlusion enhances the efficacy of subsequent ablation. Se
However, evidence comparing synchronous vs sequential application of TACE and MWA remains limited. This study therefore aimed to evaluate and compare the therapeutic efficacy and prognostic outcomes of these two strategies, with the goal of providing clinical evidence to inform optimized HCC management.
This study evaluated 106 patients with HCC treated between March 2022 and March 2024. Patients were allocated to a synchronous group (n = 56), in which TACE was performed concurrently with MWA, or a sequential group (n = 56), in which TACE was followed by MWA. Baseline demographic and clinical characteristics were comparable between groups, with no statistically significant differences observed (P > 0.05), supporting intergroup comparability.
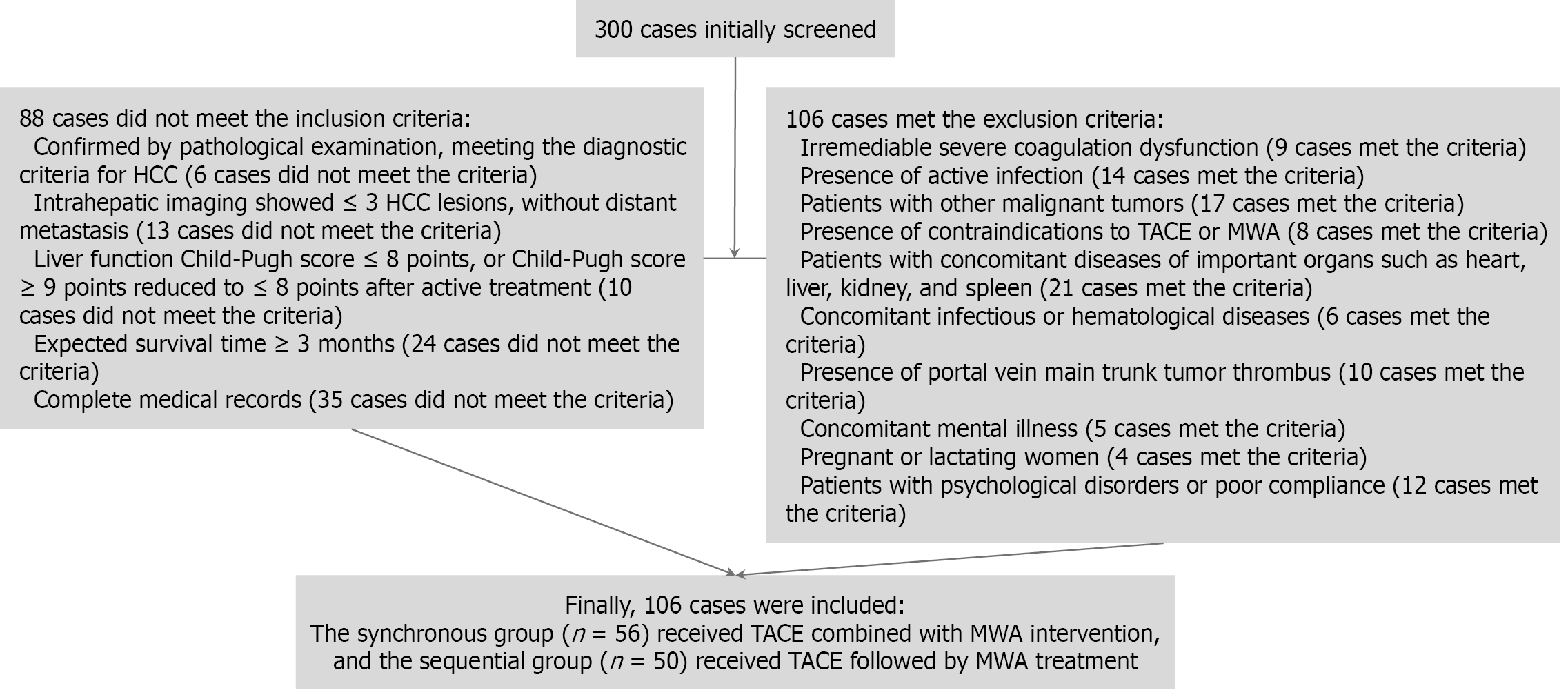
Inclusion criteria were as follows: (1) Pathological confirmation of HCC[13]; (2) Imaging evidence of no more than three intrahepatic lesions and no extrahepatic metastasis; (3) Child-Pugh score ≤ 8, or initially ≥ 9 but reduced to ≤ 8 after treatment[14]; (4) Expected survival ≥ 3 months; and (5) Complete clinical records.
Exclusion criteria were as follows: (1) Severe uncorrectable coagulation disorders; (2) Active infection; (3) Concomitant malignancies; (4) Contraindications to TACE or MWA; (5) Severe cardiac, hepatic, or renal dysfunction; infectious or hematological disease; (6) Main portal vein tumor thrombosis; psychiatric disorders; (7) Pregnancy or lactation; (8) Cognitive impairment; or (9) Poor treatment compliance.
The screening process for patient inclusion and exclusion is shown in Figure 1.
Patients in the synchronous group underwent combined TACE-MWA during the same treatment session. After routine disinfection, draping, and local anesthesia, femoral artery access was obtained using a modified Seldinger technique. A 5F hepatic arteriography catheter was inserted to delineate tumor location, number, size, and feeding arteries. Target vessels were selectively catheterized under microscopic guidance, followed by slow injection of 6-12 mL cisplatin and 10-20 mL lipiodol emulsion. Embolization was completed with gelatin sponge particles until tumor-feeding arteries were largely occluded.
In the sequential group, TACE was followed by MWA performed 1-4 weeks later, using single-needle ablation. Patient position (supine or lateral) was determined based on preoperative imaging. Under computed tomography (CT) guidance, the puncture site and depth were defined. The MWA needle was advanced through the tumor and extended approximately 0.5 cm beyond the distal margin. Ablation was performed at 50-80 W for 5-10 minutes per site. The needle was then repositioned to ensure overlapping ablation zones. After completing the planned ablations, power was reduced to 30 W, and the needle was withdrawn gradually with intermittent activation to ablate the needle tract and mitigate the risk of needle track metastasis. A repeat hepatic CT scan was performed to evaluate immediate efficacy and identify complications such as bleeding or organ injury.
Efficacy: Treatment response was assessed using modified Response Evaluation Criteria in Solid Tumors criteria and categorized as complete remission (CR; all target lesion disappearance sustained for at least four weeks), partial remission (PR; lesion size decrease > 50%, though not meeting CR criteria for over four weeks), progressive disease (lesion growth of more than 25% or the development of new lesions), or stable disease (SD; lesion size reduction < 50% or increase ≤ 25%). The objective response rate (ORR) comprised CR + PR, and the disease control rate (DCR) comprised CR + PR + SD[15].
Ablation-related parameters: The number of ablation needles, ablation duration, and power settings were recorded.
Prognostic outcomes: Progression-free survival (PFS) and overall survival (OS) were evaluated at 3 months, 6 months, and 12 months after treatment.
Tumor biomarkers: Fasting venous blood (5 mL) was collected before and after treatment. Serum alpha-fetoprotein (AFP) and AFP-L3 levels were measured using enzyme-linked immunosorbent assay.
Liver function indices: Total bilirubin (TBIL) level was measured using the diazo method, while alanine aminotransferase (ALT) and aspartate aminotransferase (AST) activities were determined using the IFCC rate method.
Complications: Adverse events, including pyrexia, abdominal pain, and gastrointestinal reactions, were recorded, and overall complication incidence was calculated.
Continuous variables are presented as mean ± SEM. Between-group comparisons were performed using independent-samples t-tests, and within-group pre- and post-treatment comparisons used paired t-tests. Categorical variables are expressed as frequencies or percentages and were compared using the χ2 test. All statistical analyses were conducted with SPSS version 23.0, with statistical significance defined as P < 0.05.
Baseline characteristics are summarized in Table 1. The synchronous and sequential groups were comparable with no statistically significant differences in sex, age, Child-Pugh grade, tumor diameter, clinical stage, or lesion count (P > 0.05).
| Data | Synchronous group (n = 56) | Sequential group (n = 50) | χ2/t | P value |
| Sex (male/female) | 32/24 | 28/22 | 0.014 | 0.906 |
| Age (years) | 60.12 ± 5.94 | 59.82 ± 6.94 | 0.240 | 0.811 |
| Child-Pugh classification (A/B) | 30/26 | 30/20 | 0.444 | 0.505 |
| Tumor diameter (cm) | 4.48 ± 0.80 | 4.40 ± 0.91 | 0.482 | 0.631 |
| Clinical staging (II/III/IV) | 18/26/12 | 13/27/10 | 0.670 | 0.716 |
| Number of lesions (single/multiple) | 19/37 | 20/30 | 0.419 | 0.518 |
Response evaluation (Table 2) showed no significant statistical differences between groups (P > 0.05). The synchronous group achieved an ORR of 78.57% and a DCR of 89.29%, compared with an ORR of 72.00% and a DCR of 88.00% in the sequential group.
| Response | Synchronous group (n = 56) | Sequential group (n = 50) | χ2 | P value |
| CR | 23 (41.07) | 17 (34.00) | ||
| PR | 21 (37.50) | 19 (38.00) | ||
| SD | 6 (10.71) | 8 (16.00) | ||
| PD | 6 (10.71) | 6 (12.00) | ||
| ORR | 44 (78.57) | 36 (72.00) | 0.616 | 0.433 |
| DCR | 50 (89.29) | 44 (88.00) | 0.043 | 0.835 |
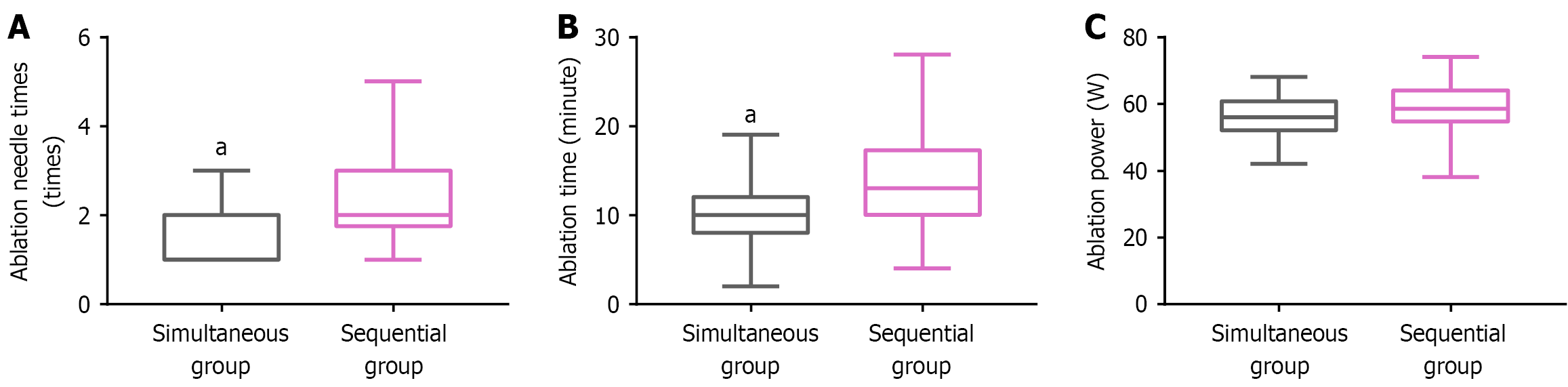
As shown in Figure 2, the synchronous approach required fewer ablation needle insertions and a shorter procedure duration than the sequential approach (P < 0.05), while ablation power did not differ significantly between intergroup differences (P > 0.05).
Prognostic outcomes are summarized in Table 3. PFS was similar between groups (P > 0.05). OS outcomes at 3 months, 6 months, and 12 months also showed no statistically significant intergroup differences (P > 0.05).
| Prognosis | Synchronous group (n = 56) | Sequential group (n = 50) | χ2/t | P value |
| PFS (months) | 7.09 ± 2.60 | 7.58 ± 2.27 | 1.028 | 0.306 |
| OS | ||||
| 3 months after treatment | 56 (100.00) | 49 (98.00) | 1.131 | 0.288 |
| 6 months after treatment | 56 (100.00) | 48 (96.00) | 2.283 | 0.131 |
| 12 months after treatment | 51 (91.07) | 45 (90.00) | 0.035 | 0.851 |
As illustrated in Figure 3, AFP and AFP-L3 levels decreased significantly after treatment within each group (P < 0.05). However, no significant differences were observed between groups at baseline or after intervention (P > 0.05).
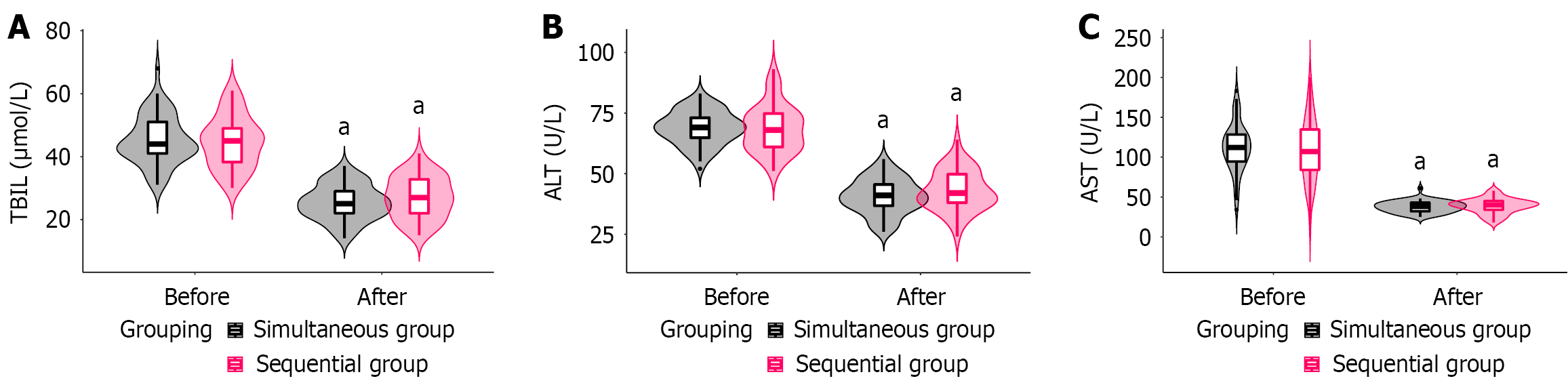
Baseline TBIL, ALT, and AST levels were comparable between groups (P > 0.05; Figure 4). Post-treatment, all indices declined significantly in both cohorts (P < 0.01), with no significant intergroup differences (P > 0.05).
As shown in Table 4, the overall complications—including pyrexia, abdominal pain, and gastrointestinal responses—was similar between the synchronous (14.29%) and sequential (18.00%) groups (P > 0.05).
| Complications | Synchronous group (n = 56) | Sequential group (n = 50) | χ2 | P value |
| Pyrexia | 2 (3.57) | 3 (6.00) | ||
| Abdominal pain | 3 (5.36) | 2 (4.00) | ||
| Gastrointestinal responses | 3 (5.36) | 4 (8.00) | ||
| Total | 8 (14.29) | 9 (18.00) | 0.271 | 0.603 |
Previous studies have consistently demonstrated the clinical benefits of combining TACE with MWA for HCC. Adwan et al[16] reported that, within the Milan Criteria, combined TACE-MWA regimen outperformed MWA alone in efficacy and safety and was associated with improved OS. Keshavarz and Raman[17] further showed that TACE-MWA achieved more favorable outcomes than TACE combined with RFA or cryoablation, particularly in patients younger than 60 years with tumors ≤ 3 cm. In addition to tumor size and number, Liu et al[18] identified the interval between TACE and RFA as a critical determinant of OS and PFS in patients with medium-to-large or recurrent multifocal HCC, suggesting that treatment sequencing may influence outcomes.
The present study compared concurrent TACE-MWA with sequential TACE followed by MWA in terms of efficacy and prognosis. Both strategies demonstrated comparable efficacy, as reflected by similar ORR and DCR values, indicating that tumor control did not differ significantly between concurrent and sequential delivery. This finding aligns with results reported by Wang et al[19] using propensity score matching. Shi et al[20] observed that TACE-MWA in patients with primary and metastatic HCC altered systemic inflammatory markers [neutrophil-to-lymphocyte ratio and liver function indicators (bilirubin)], suggesting that the combined approach may affect the tumor microenvironment and systemic inflammatory status. The specific mechanisms underlying these effects warrant further investigation using tumor tissue-based analyses. Analysis of ablation-related parameters revealed that, although ablation power used was comparable, concurrent TACE-MWA approach required fewer needle insertions and shorter ablation duration. This likely reflects reduced heat loss after TACE-induced vascular embolization, as well as enhanced heat distribution asso
Tumor biomarker testing demonstrated significant post-intervention reductions of similar magnitude in AFP and AFP-L3 levels across both groups. As established biomarkers for HCC, AFP and its variant AFP-L3—which results from glycolylation—are clinically significant; AFP-L3 exhibits higher specificity than AFP, and both are useful for estimating the risk of HCC recurrence[23,24]. Comparable patterns were observed for liver function indices: Concurrent and sequential TACE-MWA strategies produced comparable reductions in TBIL, ALT, and AST levels in patients with HCC. From a safety perspective, a non-significant trend toward a lower complication rate was noted with concurrent vs sequential TACE-MWA, indicating comparable clinical safety for both approaches. These findings align with those of Özen et al[25], who reported that concurrent TACE with percutaneous thermal ablation (using either MWA or RFA) for solitary HCC is well tolerated and effective for improving local control without lethal complications.
This study has several limitations. First, clinical indicators such as hospital stay, recovery course, intraoperative trauma, and medical costs were not assessed and should be incorporated to better define the potential advantages of synchronous TACE + MWA. Second, the absence of health-economic measures, including direct and indirect medical costs, limits cost-effectiveness evaluation; future analyses should address this to inform resource optimization. Third, to refine ablation strategy guidance, differences in tumor perfusion, actual ablation volume, and necrosis extent between groups warrant analysis. Fourth, treatment effects stratified by tumor size were not evaluated; future studies should assess outcomes by tumor size to identify optimal beneficiary subgroups. Finally, data on 2-3-year follow-up, recurrence sites, and subsequent treatments were unavailable; comprehensive long-term follow-up is needed to better characterize prognosis.
In summary, combining TACE with MWA—whether administered concurrently or sequentially—effectively controls tumor growth in HCC, yielding comparable survival outcomes, reducing AFP and AFP-L3 levels, and promoting liver function recovery. Notably, the synchronous approach requires fewer ablations and shorter procedure times, conferring a clear clinical advantage.
| 1. | Yu B, Ma W. Biomarker discovery in hepatocellular carcinoma (HCC) for personalized treatment and enhanced prognosis. Cytokine Growth Factor Rev. 2024;79:29-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 51] [Article Influence: 25.5] [Reference Citation Analysis (0)] |
| 2. | Hwang SY, Danpanichkul P, Agopian V, Mehta N, Parikh ND, Abou-Alfa GK, Singal AG, Yang JD. Hepatocellular carcinoma: updates on epidemiology, surveillance, diagnosis and treatment. Clin Mol Hepatol. 2025;31:S228-S254. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 169] [Cited by in RCA: 152] [Article Influence: 152.0] [Reference Citation Analysis (0)] |
| 3. | Burciu C, Sirli R, Bende R, Vuletici D, Miutescu B, Moga T, Bende F, Popescu A, Sporea I, Koppandi O, Miutescu E, Iovanescu D, Danila M. Paraneoplastic Syndromes in Hepatocellular Carcinoma, Epidemiology, and Survival: A Retrospective Seven Years Study. Medicina (Kaunas). 2024;60:552. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 4. | Xin X, Cheng X, Zeng F, Xu Q, Hou L. The Role of TGF-β/SMAD Signaling in Hepatocellular Carcinoma: from Mechanism to Therapy and Prognosis. Int J Biol Sci. 2024;20:1436-1451. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 74] [Cited by in RCA: 66] [Article Influence: 33.0] [Reference Citation Analysis (0)] |
| 5. | Calderon-Martinez E, Landazuri-Navas S, Vilchez E, Cantu-Hernandez R, Mosquera-Moscoso J, Encalada S, Al Lami Z, Zevallos-Delgado C, Cinicola J. Prognostic Scores and Survival Rates by Etiology of Hepatocellular Carcinoma: A Review. J Clin Med Res. 2023;15:200-207. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 71] [Cited by in RCA: 63] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 6. | Singal AG, Kanwal F, Llovet JM. Global trends in hepatocellular carcinoma epidemiology: implications for screening, prevention and therapy. Nat Rev Clin Oncol. 2023;20:864-884. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 713] [Cited by in RCA: 603] [Article Influence: 201.0] [Reference Citation Analysis (2)] |
| 7. | Tan BB, Fu Y, Shao MH, Chen HL, Liu P, Fan C, Zhang H. Combined transarterial chemoembolization and tislelizumab for patients with unresectable hepatocellular carcinoma. World J Gastrointest Surg. 2024;16:2829-2841. [PubMed] [DOI] [Full Text] |
| 8. | Ladd AD, Duarte S, Sahin I, Zarrinpar A. Mechanisms of drug resistance in HCC. Hepatology. 2024;79:926-940. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 232] [Cited by in RCA: 193] [Article Influence: 96.5] [Reference Citation Analysis (1)] |
| 9. | Duan R, Gong F, Wang Y, Huang C, Wu J, Hu L, Liu M, Qiu S, Lu L, Lin Y. Transarterial chemoembolization (TACE) plus tyrosine kinase inhibitors versus TACE in patients with hepatocellular carcinoma: a systematic review and meta-analysis. World J Surg Oncol. 2023;21:120. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 24] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 10. | Fite EL, Makary MS. Transarterial Chemoembolization Treatment Paradigms for Hepatocellular Carcinoma. Cancers (Basel). 2024;16:2430. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 11.5] [Reference Citation Analysis (2)] |
| 11. | Sugimoto K, Imajo K, Kuroda H, Murohisa G, Shiozawa K, Sakamaki K, Wada T, Takeuchi H, Endo K, Abe T, Matsui T, Murakami T, Yoneda M, Nakajima A, Kokubu S, Itoi T. Microwave ablation vs. single-needle radiofrequency ablation for the treatment of HCC up to 4 cm: A randomized-controlled trial. JHEP Rep. 2025;7:101269. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 12. | Zhang C, Qin Y, Song Y, Liu Y, Zhu X. Transarterial Chemoembolization Combined with Microwave Ablation in Elderly Patients with Recurrent Medium or Large Hepatocellular Carcinoma. J Hepatocell Carcinoma. 2024;11:2005-2017. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 13. | Xie DY, Zhu K, Ren ZG, Zhou J, Fan J, Gao Q. A review of 2022 Chinese clinical guidelines on the management of hepatocellular carcinoma: updates and insights. Hepatobiliary Surg Nutr. 2023;12:216-228. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 134] [Cited by in RCA: 125] [Article Influence: 41.7] [Reference Citation Analysis (0)] |
| 14. | Aly A, Fulcher N, Seal B, Pham T, Wang Y, Paulson S, He AR. Clinical outcomes by Child-Pugh Class in patients with advanced hepatocellular carcinoma in a community oncology setting. Hepat Oncol. 2023;10:HEP47. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 15. | Karagiannakis DS. Systemic Treatment in Intermediate Stage (Barcelona Clinic Liver Cancer-B) Hepatocellular Carcinoma. Cancers (Basel). 2023;16:51. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 16. | Adwan H, Adwan M, Vogl TJ. Combination Therapy of Bland Transarterial Embolization and Microwave Ablation for Hepatocellular Carcinoma within the Milan Criteria Leads to Significantly Higher Overall Survival. Cancers (Basel). 2023;15:5076. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 17. | Keshavarz P, Raman SS. Comparison of combined transarterial chemoembolization and ablations in patients with hepatocellular carcinoma: a systematic review and meta-analysis. Abdom Radiol (NY). 2022;47:1009-1023. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 18. | Liu D, Liu M, Su L, Wang Y, Zhang X, Long H, Kuang M, Xie X, Lin M. Transarterial Chemoembolization Followed by Radiofrequency Ablation for Hepatocellular Carcinoma: Impact of the Time Interval between the Two Treatments on Outcome. J Vasc Interv Radiol. 2019;30:1879-1886. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 19. | Wang HY, Zhang GX, Fan WZ, Li JW, Hao SF, Ouyang YS, Li JP, Liu WD. Simultaneous versus sequential transcatheter arterial chemoembolization combined with microwave ablation for hepatocellular carcinoma: A retrospective propensity score-matched analysis. Hepatobiliary Pancreat Dis Int. 2025;24:286-293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 20. | Shi Q, Zhang Z, Zhang W, Ma J, Yang M, Luo J, Liu L, Yan Z. Microwave ablation combined with transarterial chemoembolization containing doxorubicin hydrochloride liposome for treating primary and metastatic liver cancers. J Interv Med. 2023;6:121-125. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 21. | Li W, Ni CF. Current status of the combination therapy of transarterial chemoembolization and local ablation for hepatocellular carcinoma. Abdom Radiol (NY). 2019;44:2268-2275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 39] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 22. | Tang L, Hou Y, Huang Z, Huang J. Transarterial Chemoembolization Combined with Lenvatinib Plus Sequential Microwave Ablation for Large Hepatocellular Carcinoma Beyond Up-to-Seven Criteria: A Retrospective Cohort Study. Acad Radiol. 2024;31:2795-2806. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 23. | Ren T, Hou X, Zhang X, Chen D, Li J, Zhu Y, Liu Z, Yang D. Validation of combined AFP, AFP-L3, and PIVKA II for diagnosis and monitoring of hepatocellular carcinoma in Chinese patients. Heliyon. 2023;9:e21906. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 24. | Norman JS, Li PJ, Kotwani P, Shui AM, Yao F, Mehta N. AFP-L3 and DCP strongly predict early hepatocellular carcinoma recurrence after liver transplantation. J Hepatol. 2023;79:1469-1477. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 124] [Cited by in RCA: 110] [Article Influence: 36.7] [Reference Citation Analysis (0)] |
| 25. | Özen Ö, Boyvat F, Zeydanlı T, Kesim Ç, Karakaya E, Haberal M. Transarterial Chemoembolization Combined with Simultaneous Thermal Ablation for Solitary Hepatocellular Carcinomas in Regions with a High Risk of Recurrence. Exp Clin Transplant. 2023;21:512-519. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/