Copyright: ©Author(s) 2026.
World J Gastrointest Surg. Mar 27, 2026; 18(3): 115906
Published online Mar 27, 2026. doi: 10.4240/wjgs.v18.i3.115906
Published online Mar 27, 2026. doi: 10.4240/wjgs.v18.i3.115906
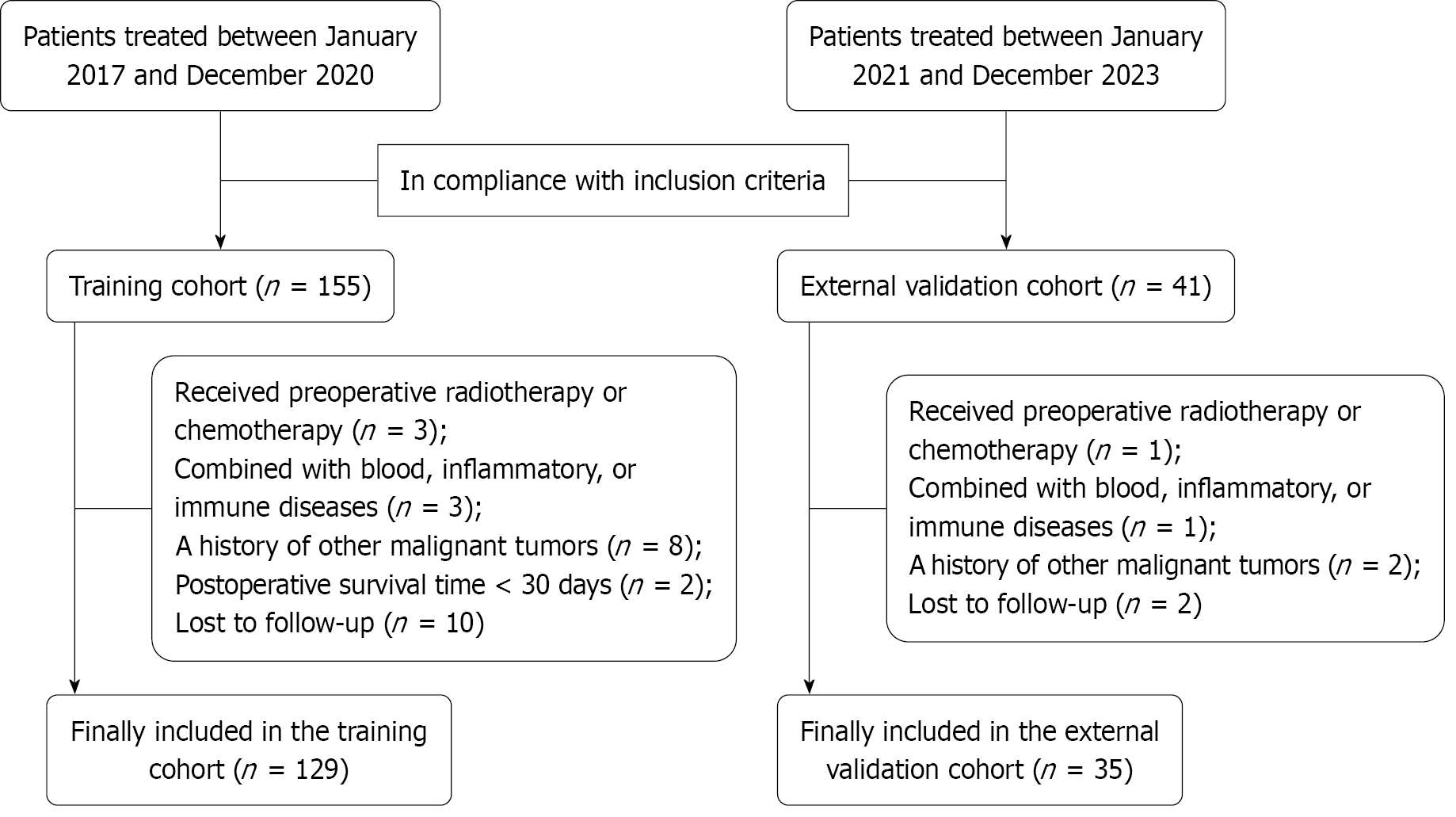
Figure 1
Flowchart for inclusion and exclusion of pancreatic ductal adenocarcinoma patients.
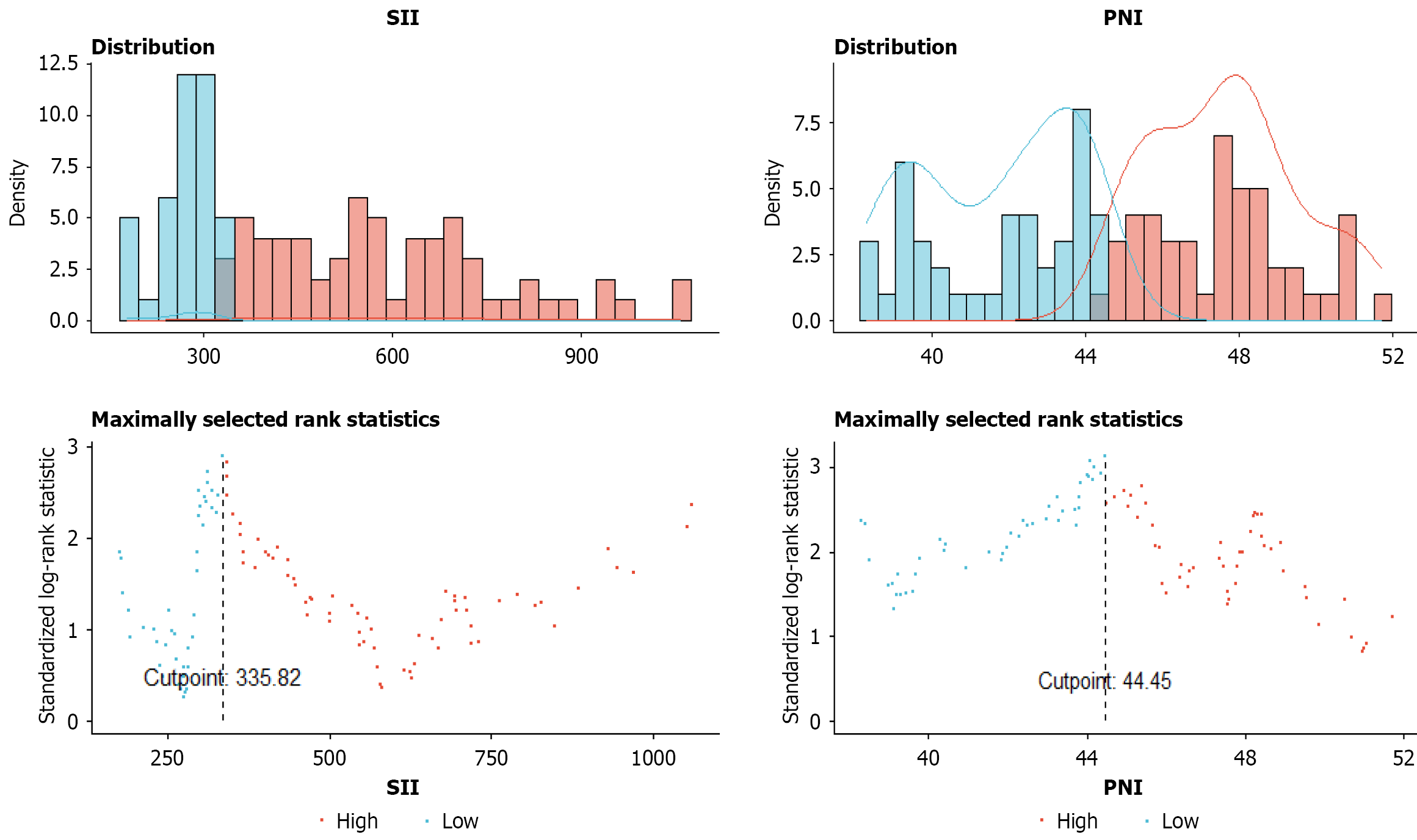
Figure 2 Calculation for the optimal systemic immune inflammation index and prognostic nutritional index cutoff values.
The basic principle of the surv-cutpoint function was based on patient overall survival, and the Kaplan-Meier curve was used to determine the best cutoff value. The survival differences between the two groups were tested by a log-rank test. The survival rates on both sides of a specific point had the greatest difference when the χ2 statistic of the log-rank test was the largest. The measured values corresponding to the maximum χ2 statistic were the optimal systemic immune inflammation index and prognostic nutritional index cutoff values. SII: Systemic immune inflammation index; PNI: Prognostic nutritional index; grps: Groups.
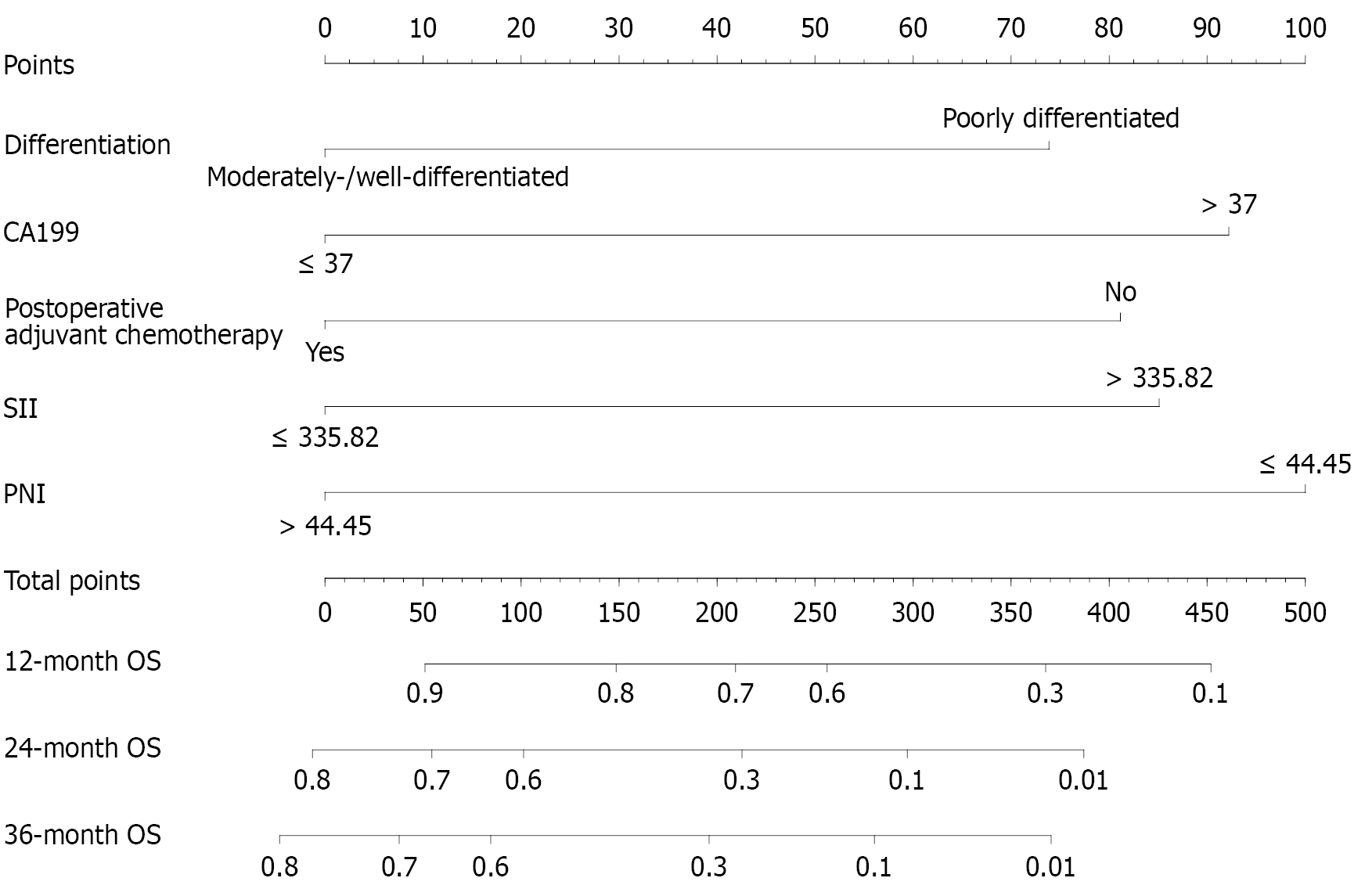
Figure 3 Nomogram model for predicting the 1-year, 2-year, and 3-year postoperative survival rates among patients with pancreatic ductal adenocarcinoma.
The assigned scores of relevant indicators were calculated through R language based on the regression coefficients of each variable. Usually the indicator with the largest absolute value of the regression coefficient was set at 100 points, and the scores of other indicators were converted based on the relative relationship between the regression coefficients and the benchmark indicator. Prognostic nutritional index ≤ 44.45 was 100 points; systemic immune inflammation index > 335.82 was 85 points; poorly differentiated tumor was 74 points; no postoperative adjuvant chemotherapy was 81 points; and the carbohydrate antigen 19-9 level > 37 U/mL was 92 points. The calculated total score was matched with the 1-year and 3-year survival rates. The higher the total score, the lower the postoperative survival rate. CA19-9: Carbohydrate antigen 19-9; SII: Systemic immune inflammation index; PNI: Prognostic nutritional index; OS: Overall survival.
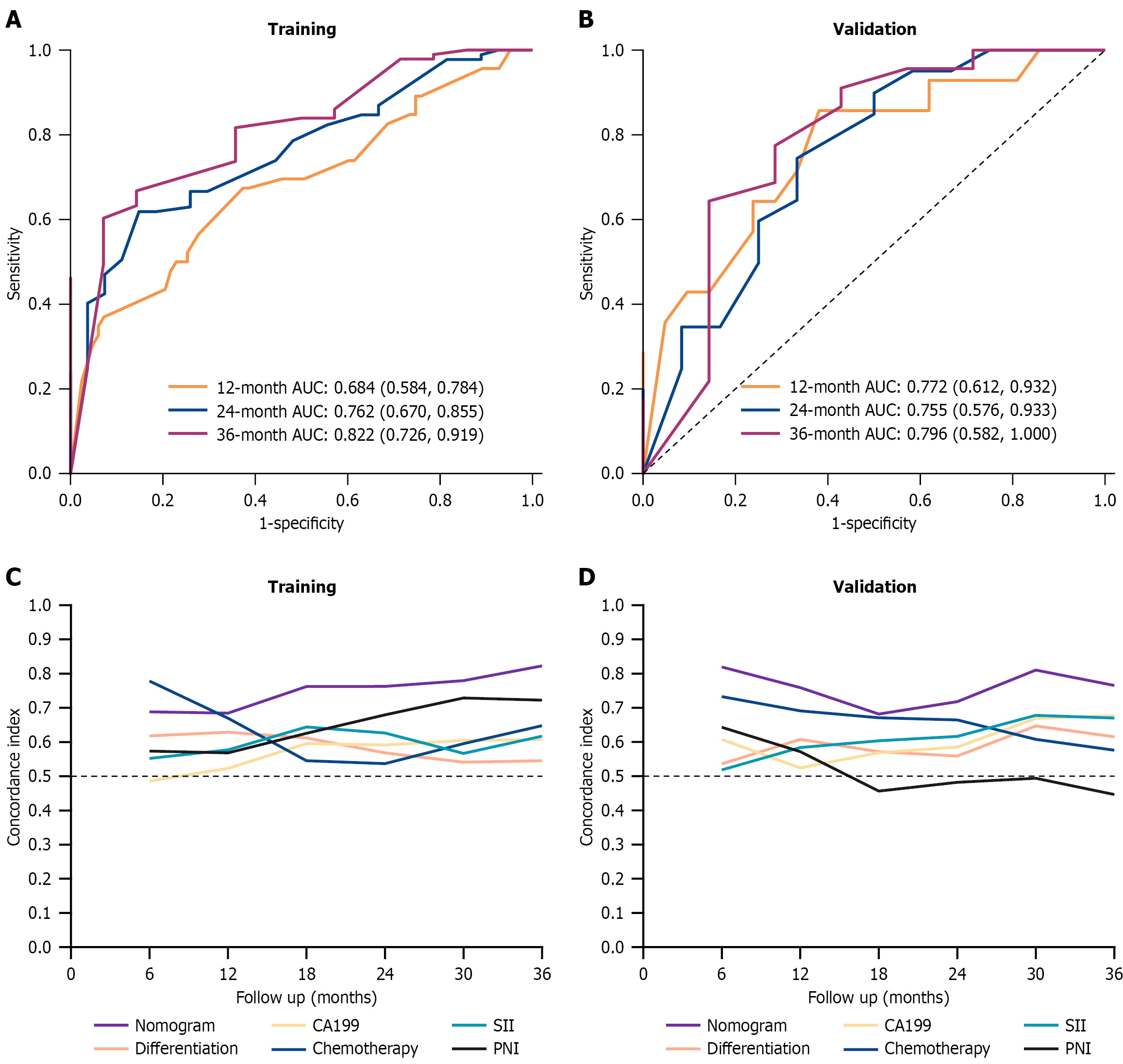
Figure 4 Discrimination evaluation and internal model validation.
A and B: Receiver operating characteristic curves of the nomogram prediction model in the training and external validation cohorts 1 year, 2 years, and 3 years after surgery; C and D: Time-dependent C-index analysis. AUC: Area under the receiver operating characteristic curve; CA19-9: Carbohydrate antigen 19-9; SII: Systemic immune inflammation index; PNI: Prognostic nutritional index.
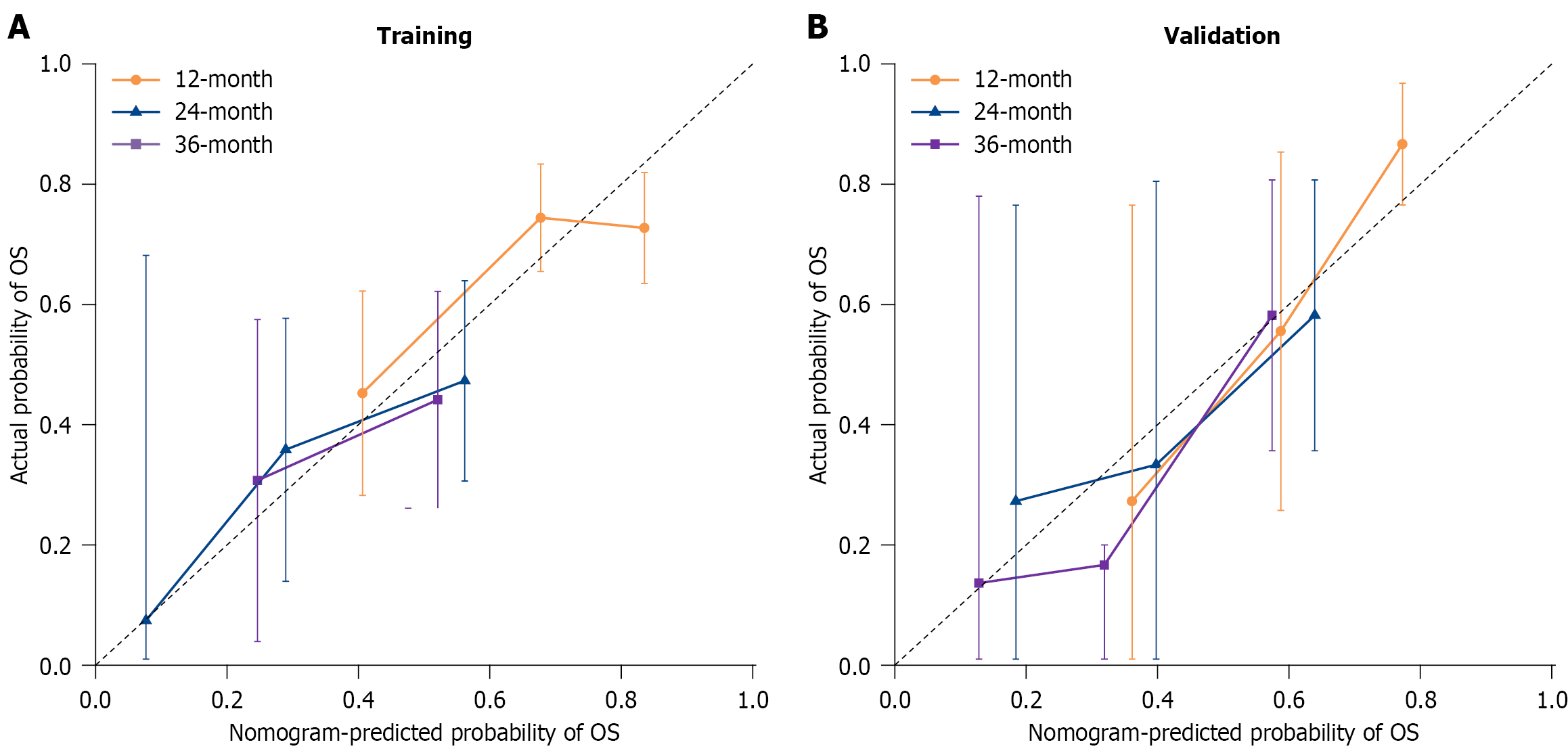
Figure 5 Calibration curves of the nomogram prediction model in the training and external validation cohorts for predicting overall survival 1 year, 2 years, and 3 years after surgery.
The horizontal axis represents the predicted probability of the model, and the vertical axis represents the actual probability. The higher the overlap between the predicted curve and the ideal curve, the better the consistency of the predicted model. A: The calibration curves of the nomogram prediction model in the training cohorts; B: The calibration curves of the nomogram prediction model in the external validation cohorts. OS: Overall survival.
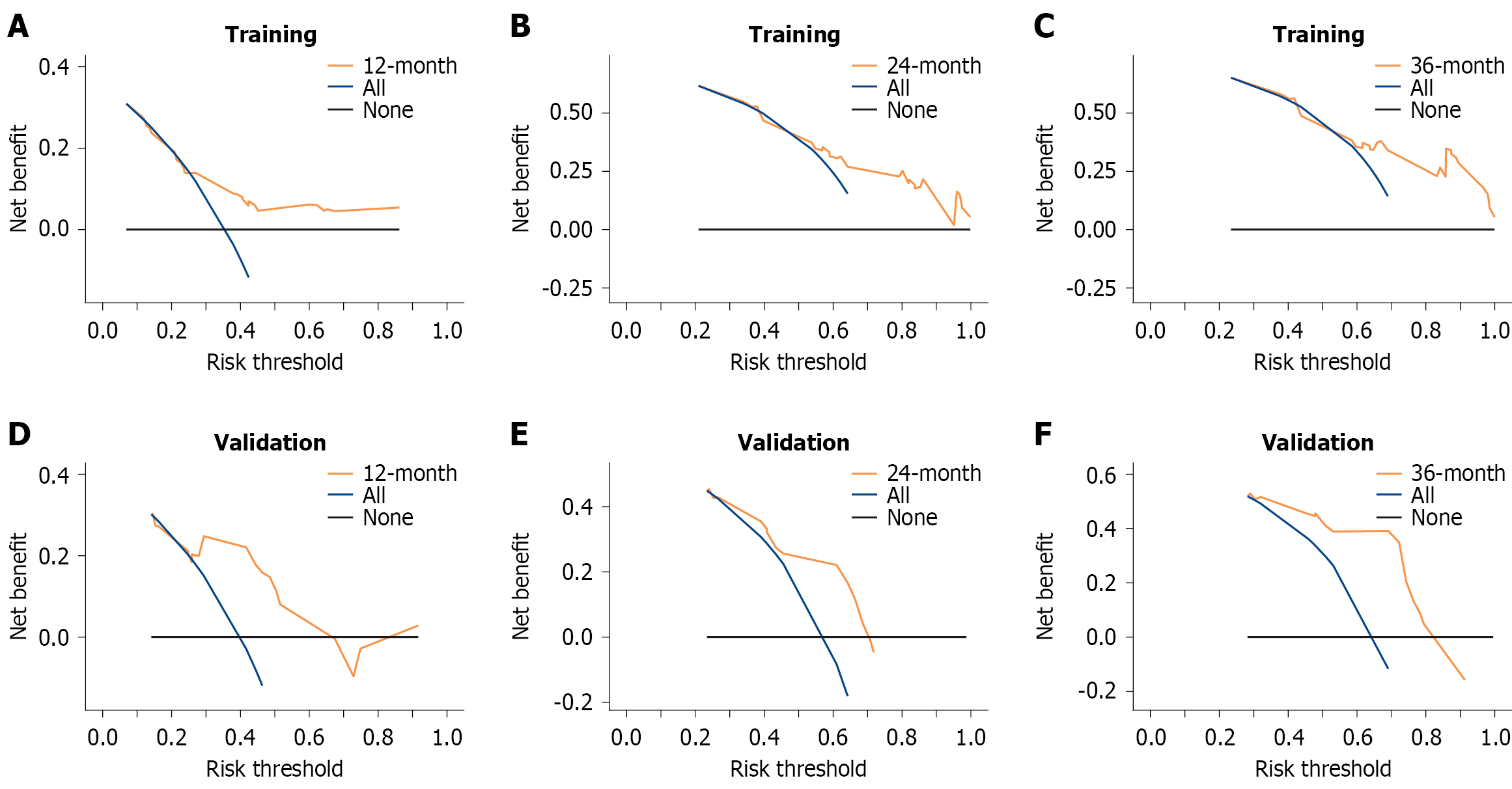
Figure 6 Decision curves of the nomogram prediction model in the training and external validation cohorts for predicting overall survival 1 year, 2 years, and 3 years after surgery.
The blue line represents the net benefit of the intervention strategy for all patients. The black line indicates the net benefit without any intervention strategy. The vertical axis represents the overall net benefit. A: The decision curves of the nomogram prediction model in the training cohorts for predicting overall survival 1 year after surgery; B: The decision curves of the nomogram prediction model in the training cohorts for predicting overall survival 2 years after surgery; C: The decision curves of the nomogram prediction model in the training cohorts for predicting overall survival 3 years after surgery; D: The decision curves of the nomogram prediction model in the external validation cohorts for predicting overall survival 1 year after surgery; E: The decision curves of the nomogram prediction model in the external validation cohorts for predicting overall survival 2 years after surgery; F: The decision curves of the nomogram prediction model in the external validation cohorts for predicting overall survival 3 years after surgery.
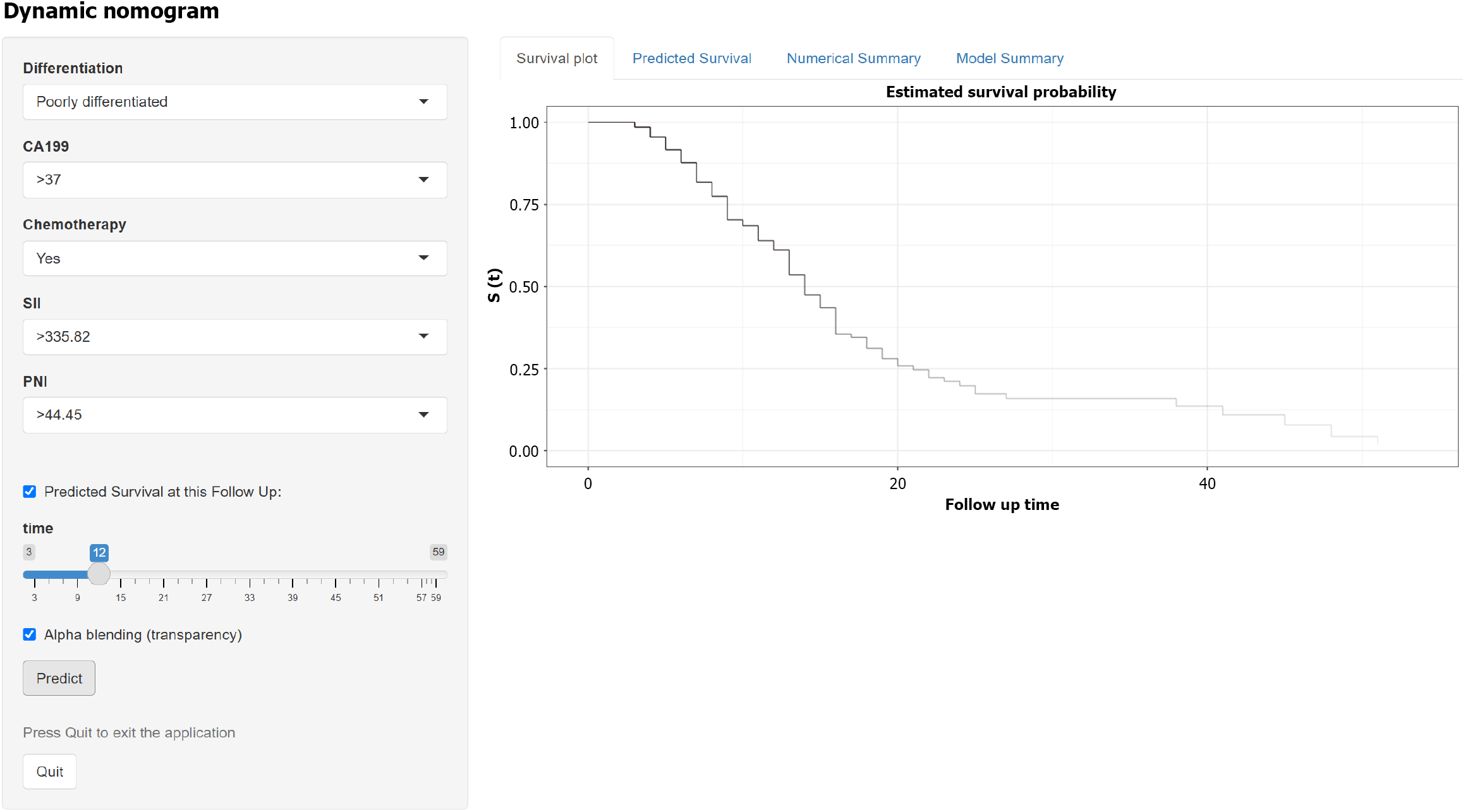
Figure 7
An example of using the online prediction website.
- Citation: Tang J, Ding SL, Wu Y, Wu YP, Yuan XB, Wu PF, Sha DS. Prognostic value of a nomogram model for pancreatic cancer incorporating the systemic immune-inflammation and prognostic nutritional indices. World J Gastrointest Surg 2026; 18(3): 115906
- URL: https://www.wjgnet.com/1948-9366/full/v18/i3/115906.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i3.115906