©Author(s) (or their employer(s)) 2026.
World J Gastrointest Surg. Feb 27, 2026; 18(2): 115830
Published online Feb 27, 2026. doi: 10.4240/wjgs.v18.i2.115830
Published online Feb 27, 2026. doi: 10.4240/wjgs.v18.i2.115830
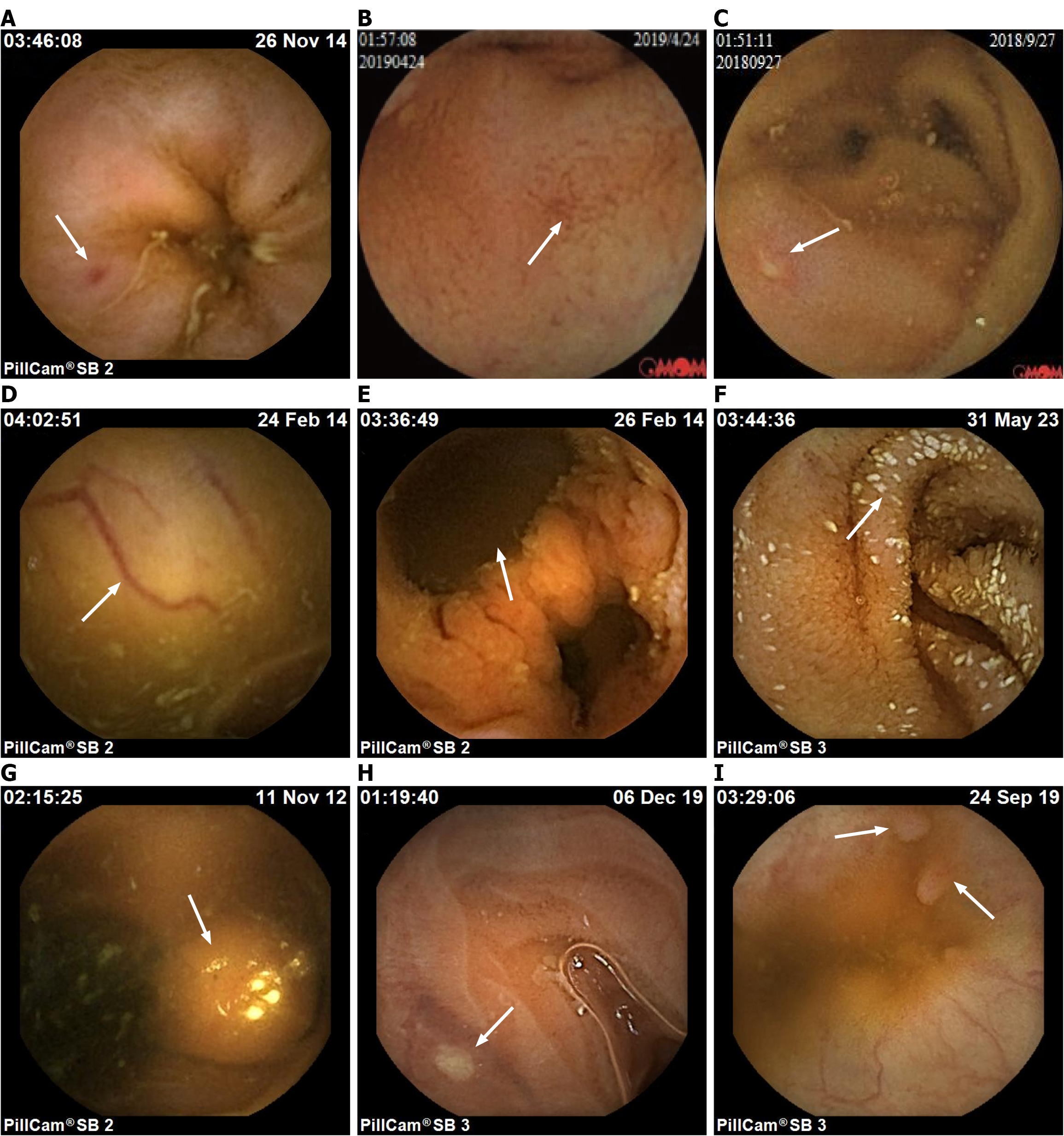
Figure 1 Representative image of small intestine lesions.
A: Erythema; B: Erosion; C: Ulcer; D: Vascular malformation; E: Diverticulum; F: Lymphan
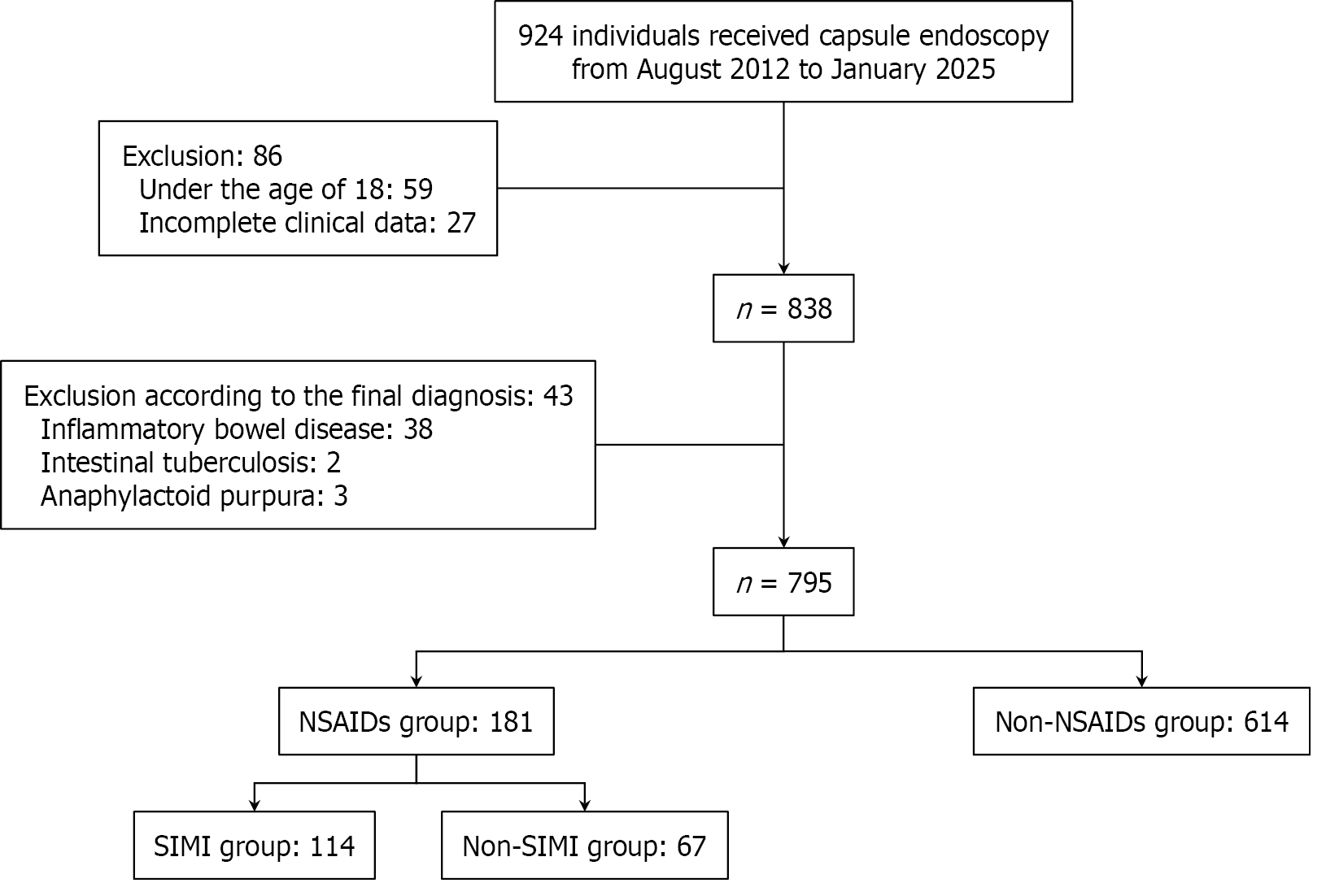
Figure 2 Flowchart of patient enrollment and exclusion criteria in the current study.
NSAIDs: Nonsteroidal anti-inflammatory drugs; SIMI: Small intestinal mucosal injury.
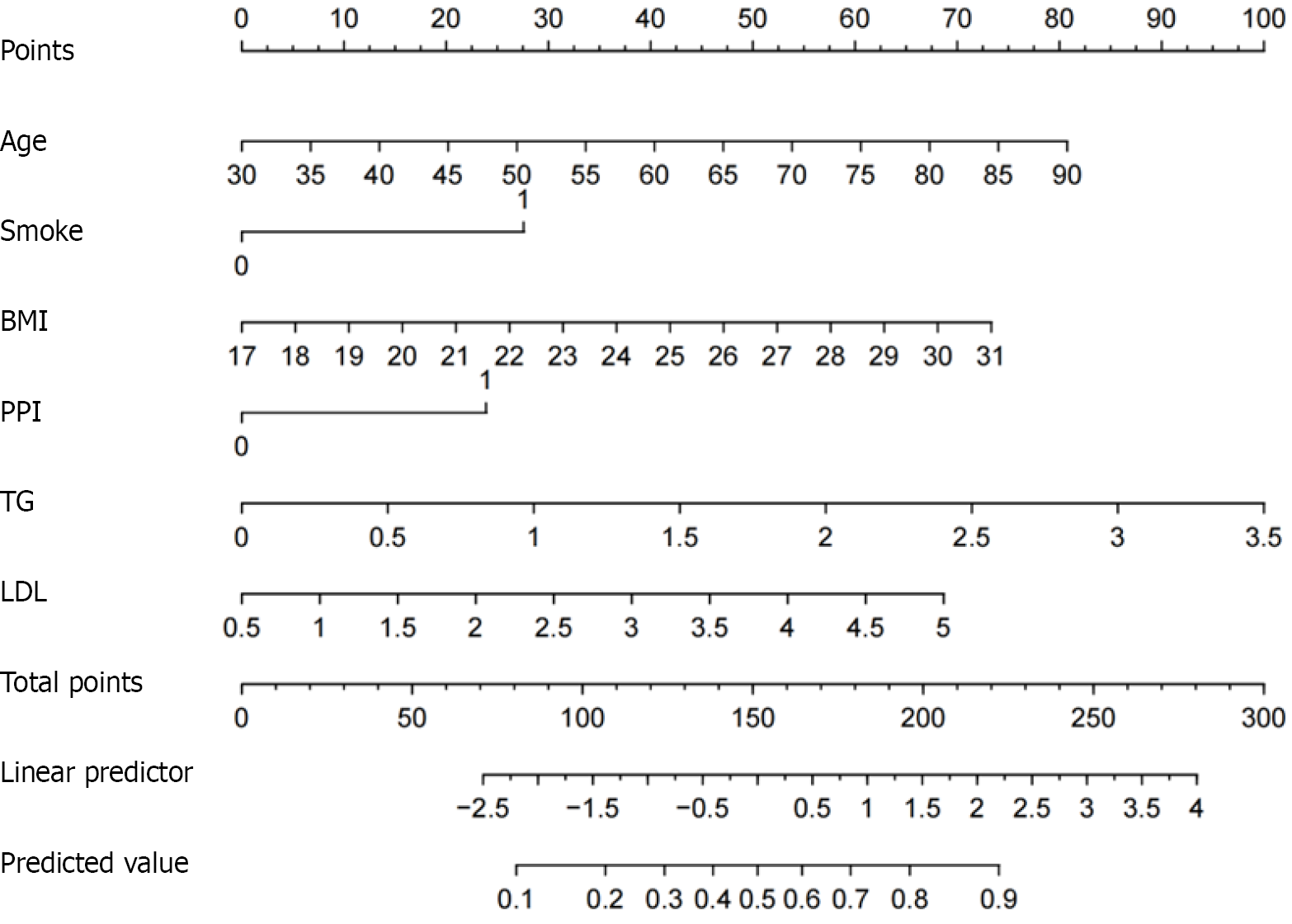
Figure 3 A nomogram for assessing the risk of small intestinal mucosal injury in nonsteroidal anti-inflammatory drug users.
BMI: Body mass index; PPI: Proton pump inhibitor; TG: Triglyceride LDL: Low density lipoprotein.
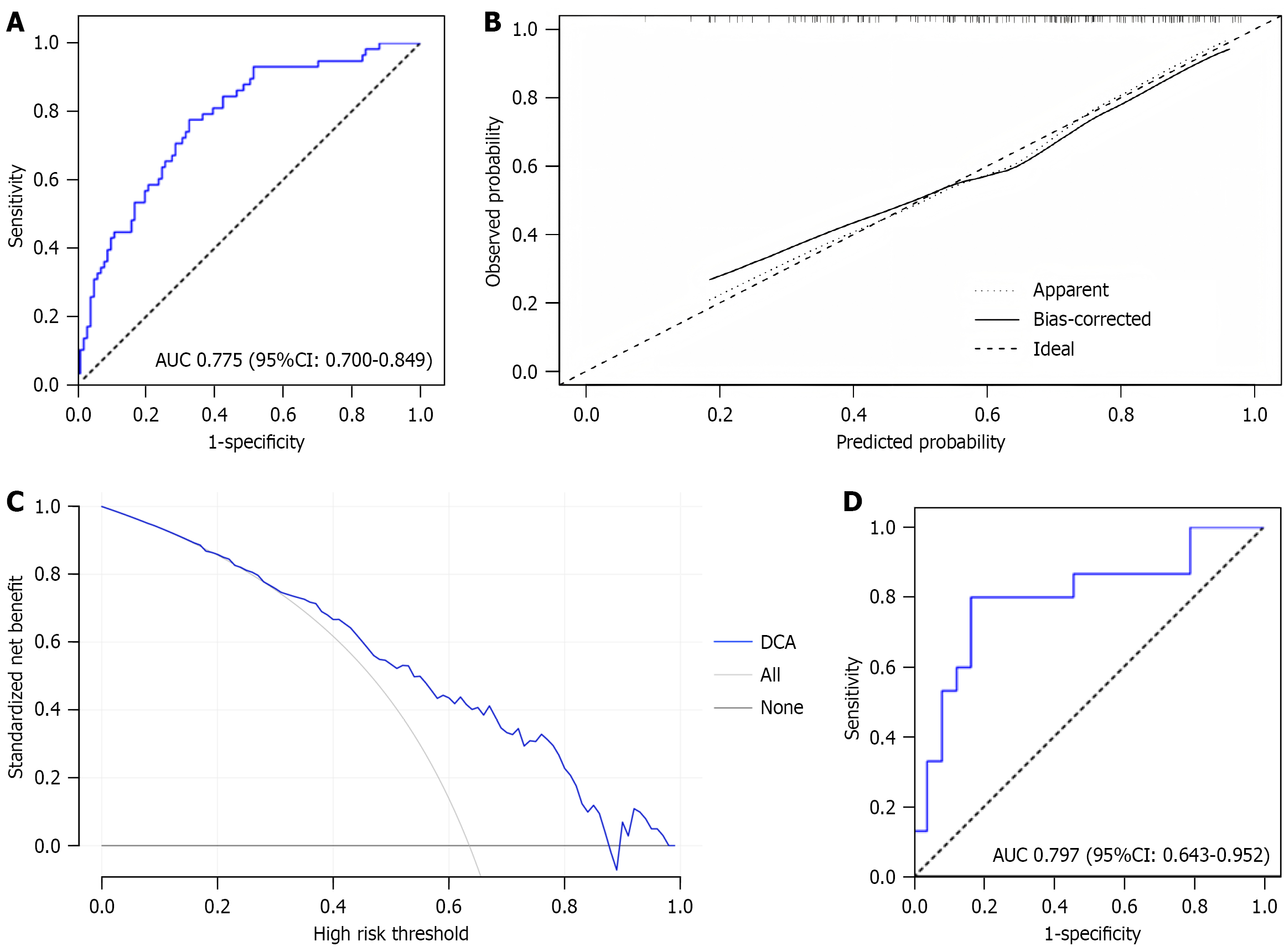
Figure 4 Evaluation of the risk prediction model for small intestinal mucosal injury in both the development and validation sets.
A: Receiver operating characteristic curve of the development set, with an area under the curve value of 0.775; B: Calibration curve of this nomogram model; C: Decision curve analysis of this nomogram model; D: Receiver operating characteristic curve of the validation set, with an area under the curve value of 0.797. AUC: Area under the curve; CI: Confidence interval; DCA: Decision curve analysis.
- Citation: Zhao NH, Zhao LR, Yao J, Niu LY, Yang LJ, Lin YJ, Liu M. Development and validation of a risk prediction model for small intestinal mucosal injury in nonsteroidal anti-inflammatory drug users. World J Gastrointest Surg 2026; 18(2): 115830
- URL: https://www.wjgnet.com/1948-9366/full/v18/i2/115830.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i2.115830