Published online May 15, 2026. doi: 10.4239/wjd.v17.i5.118141
Revised: February 24, 2026
Accepted: April 3, 2026
Published online: May 15, 2026
Processing time: 137 Days and 21.6 Hours
Diabetic retinopathy (DR) is a prevalent and vision-threatening microvascular complication of type 2 diabetes mellitus (T2DM). Although traditional risk factors for DR are well established, the roles of vitamin D (VD) and genetic variations, particularly the VD receptor (VDR) FokI polymorphism (rs2228570), remain not fully elucidated in the pathogenesis of DR and are under active investigation. VD exerts anti-inflammatory, anti-oxidative, and anti-angiogenic effects that are crucial for retinal microvascular homeostasis. We hypothesized that serum VD levels and the VDR FokI polymorphism are associated with susceptibility to DR.
To investigate associations of serum VD levels and the VDR FokI polymorphism with the risk of DR in T2DM patients in Kunming, China.
This case-control study was conducted at the First Affiliated Hospital of Kunming Medical University. Participants were recruited and categorized into three groups: (1) 115 patients with DR; (2) 130 T2DM patients without retinopathy; and (3) 58 healthy controls. Serum 25-hydroxyvitamin D levels were measured by chemiluminescence immunoassay. VDR FokI (rs2228570) genotyping was performed using Sanger sequencing.
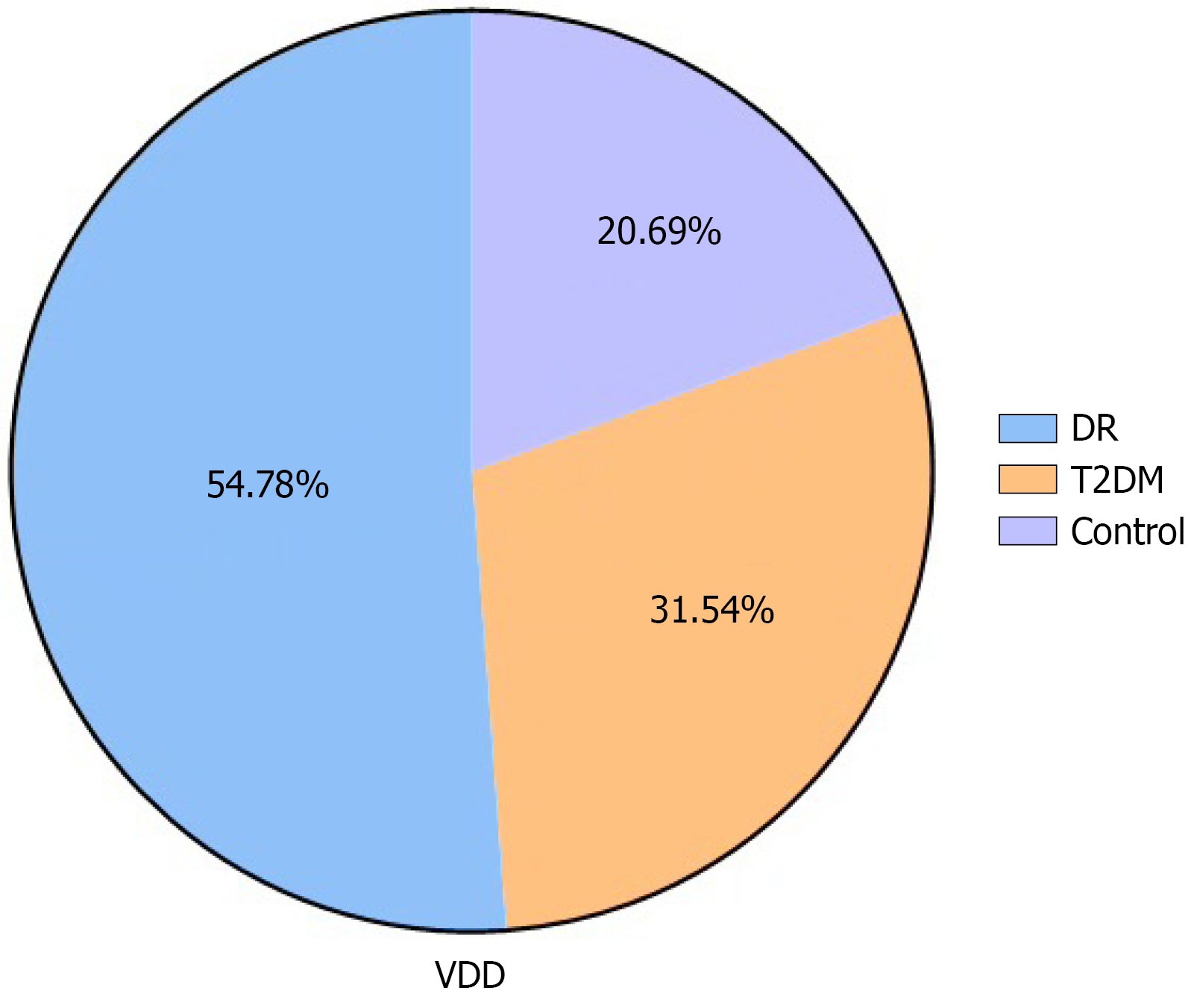
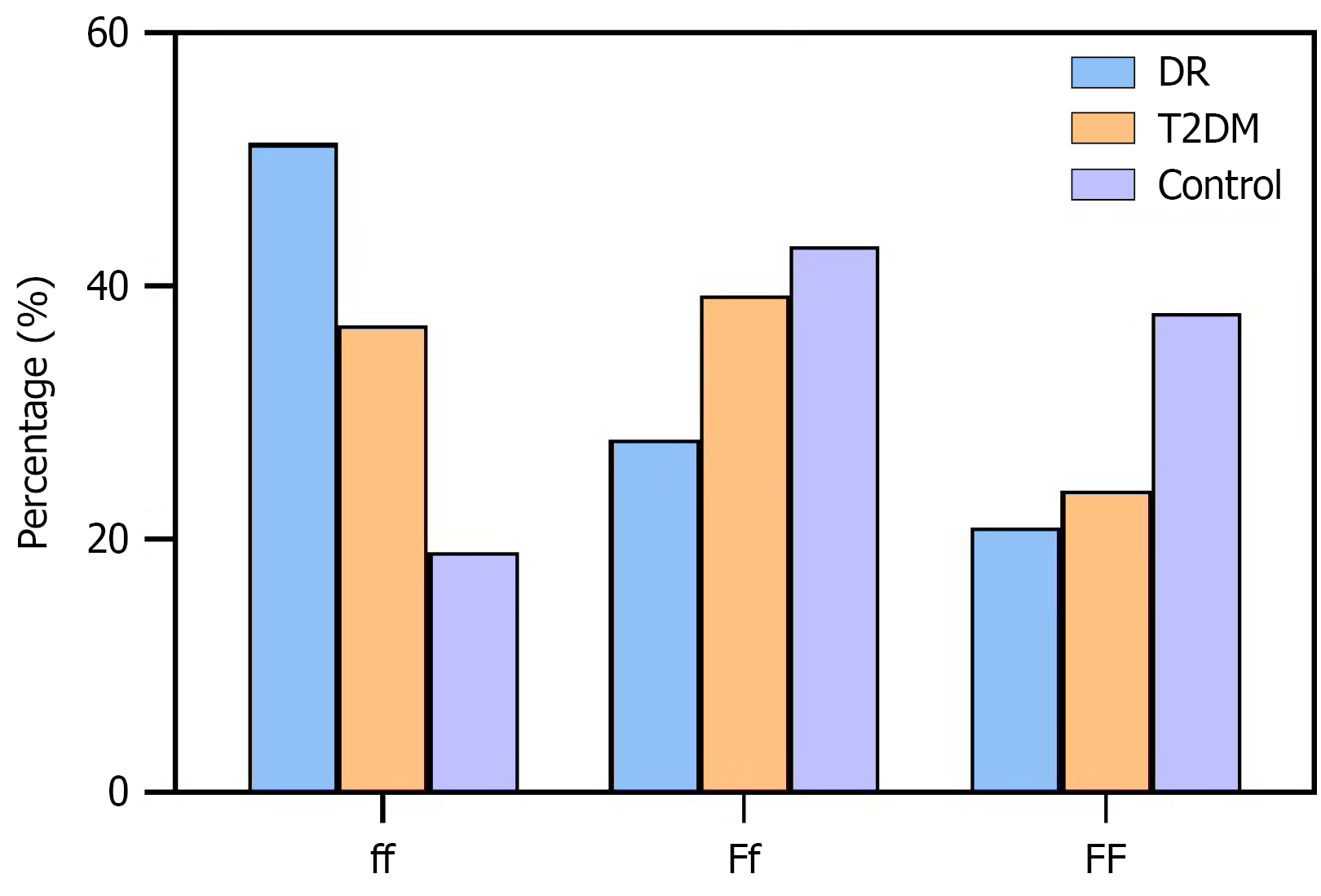
The prevalence of VD deficiency (VDD) was significantly higher in the DR group (54.78%) compared with the T2DM (31.54%) and control groups (20.69%). VDD was significantly associated with an increased risk of DR relative to T2DM patients and healthy controls [odds ratio (OR) = 3.09, 95%CI: 1.90-5.01, P < 0.001]. Genetic analysis revealed that both the FokI ff genotype (OR = 2.36; 95%CI: 1.27-4.39; P = 0.007) and f allele (OR = 1.67; 95%CI: 1.18-2.36; P = 0.004) were substantially more prevalent in DR patients than in non-retinopathy T2DM patients and controls, suggesting that these genetic variants are associated with the development of DR.
VDD and the VDR FokI ff genotype are independent risk factors for DR in T2DM patients in Kunming, China. Their combined assessment may aid in DR risk stratification and early intervention.
Core Tip: This study identified vitamin D (VD) deficiency and VD receptor FokI ff genotype as independent risk factors for diabetic retinopathy in type 2 diabetes mellitus patients. Assessment of VD deficiency status and VD receptor FokI genotyping may facilitate diabetic retinopathy risk stratification and early intervention.
- Citation: Liu ZH, Ran ZQ, Yang R, Chen LX, Wang ZZ, Li YY, Han R. Vitamin D, vitamin D receptor FokI polymorphism, and diabetic retinopathy: A case-control study in Kunming, China. World J Diabetes 2026; 17(5): 118141
- URL: https://www.wjgnet.com/1948-9358/full/v17/i5/118141.htm
- DOI: https://dx.doi.org/10.4239/wjd.v17.i5.118141
Type 2 diabetes mellitus (T2DM) has become a global pandemic, with its microvascular complications imposing a substantial burden on healthcare systems and diminishing patients’ quality of life[1]. Among these complications, diabetic retinopathy (DR) remains a leading cause of acquired blindness in the working-age population worldwide[2]. The pathogenesis of DR is multifactorial, involving chronic hyperglycemia, oxidative stress, inflammation, and genetic predisposition, leading to the breakdown of the blood-retinal barrier[3]. Despite established risk factors such as prolonged diabetes duration, poor glycemic control, and hypertension, substantial individual variability in DR susceptibility remains unexplained[4]. This underscores the critical need to identify novel and modifiable risk factors to improve risk stratification and enable preemptive interventions.
In recent years, the non-skeletal roles of vitamin D (VD) have gained prominence, particularly its potential invol
The biological efficacy of VD is mediated by the VDR, whose functional integrity is largely determined by its genetic sequence. Notably, polymorphisms within the VDR gene can influence the receptor’s transcriptional activity, thereby modulating the entire VD signaling cascade[8]. The FokI single nucleotide polymorphism (rs2228570) within the VDR gene is of particular significance. This polymorphism involves a T>C transition in the start codon of exon 2, which disrupts the primary translation initiation site. This forces translation to begin from a downstream AUG codon, producing a truncated 424-amino acid protein instead of the full-length 427-amino acid variant[9]. Functional analyses indicate that this truncated protein, associated with the C allele (often denoted as the F allele), exhibits higher transcriptional activity and enhanced responsiveness to 1,25-dihydroxyvitamin D3 compared with the full-length protein. This functional alteration may modulate VDR signaling in target cells and tissues[10]. Although associations between the FokI polymorphism and various diseases are well-documented, its precise role in the development and progression of DR remains unclear, representing a significant gap in our current understanding.
The association between the VDR FokI polymorphism and DR has been inconsistent across previous studies involving different ethnic populations. A meta-analysis by Jiao et al[11], comprising 636 DR patients and 1035 controls, identified a significant association in Asian populations, in which the FokI FF genotype appeared to confer a reduced risk of DR; however, this association was not observed in Caucasian populations. Similarly, a case-control study conducted in Brazil by Neves et al[12] reported no significant relationship between the FokI polymorphism and DR susceptibility. These ethnic discrepancies may reflect complex gene-environment interactions, especially differences in population-level VD status[13]. This study aimed to elucidate their potential roles in DR pathogenesis and evaluate their clinical relevance for risk assessment.
This case-control study enrolled a total of 303 unrelated participants of Han Chinese ethnicity. The cohort consisted of 115 patients with DR, 130 diabetic patients without DR, and 58 healthy controls, all of whom were long-term residents (≥ 10 years) of Kunming, China. Patients were recruited from the clinical departments of the First Affiliated Hospital of Kunming Medical University, while healthy controls were consecutively recruited from individuals undergoing routine health examinations at the same institution’s Health Examination Center between January and December 2024. The study protocol was approved by the Institutional Review Board of the hospital, and written informed consent was obtained from all participants prior to enrollment.
The diagnosis of T2DM was confirmed in accordance with the 2023 American Diabetes Association diagnostic criteria[14], which defined T2DM as a fasting plasma glucose ≥ 7.0 mmol/L, a 2-hour plasma glucose ≥ 11.1 mmol/L during an oral glucose tolerance test, or a glycated hemoglobin level ≥ 6.5%. DR was diagnosed based on the presence of characteristic funduscopic changes, such as microaneurysms, hemorrhages, hard exudates, and/or signs of neovascularization. All participants underwent a comprehensive ocular examination, where fundus evaluation was performed using slit-lamp biomicroscopy through pharmacologically dilated pupils. All assessments were conducted by an experienced ophthalmologist. Exclusion criteria included: (1) A history of hyperparathyroidism, chronic liver disease, or heart failure; (2) Other significant retinal pathologies; (3) Recent VD supplementation; and (4) Chronic systemic infections. Individuals unable to provide reliable responses or unwilling to provide written informed consent were also excluded.
The sample size for this case-control study was calculated a priori for the primary genetic objective of detecting an association between the VDR FokI polymorphism and DR risk. The calculation relied on comparing genotype frequency between the DR group and the T2DM control group.
Sample size estimation was informed by a previous case-control study conducted in a Northwestern Chinese (Xinjiang Uygur Autonomous Region) population, which successfully identified a significant association between the VDR polymorphisms and DR using 151 participants in the DR group and 130 in the control group[15].
A formal power calculation was conducted using an online genetic power calculator. The parameters were set as follows: (1) Assuming a multiplicative genetic model for the risk allele (f); (2) An odds ratio (OR) of 2.0 (representing a conservative estimate lower than the OR = 2.36 observed in our final results); (3) A risk allele frequency of 0.45 based on pilot data, a two-sided alpha level of 0.05; and (4) A statistical power (1-β) of 80%. Assuming a case-to-control ratio of approximately 1:1, the calculation indicated that a minimum of 106 participants per group was required to detect this association.
Our final enrollment of 115 DR patients and 130 T2DM controls met and exceeded this calculated requirement, ensuring adequate power for our primary genetic analysis. The healthy control group (n = 58) was included to establish population-specific baseline genotype and allele frequencies, providing a broader context for our findings.
Demographic and anthropometric data were systematically collected for all participants. Data encompassed age, sex, body mass index, duration of diabetes, and family history of diabetes. Smoking status was also recorded. Additionally, systolic and diastolic blood pressure (DBP) were measured for each participant using an electronic automated brachial sphygmomanometer after a 30-minute resting period.
Comprehensive laboratory testing was performed, including a complete blood count (CBC) [with differentials for neutrophil percentage, absolute neutrophil count, absolute lymphocyte count, and derived neutrophil-to-lymphocyte ratio (NLR)], fasting plasma glucose (FPG), lipid profile [total cholesterol, triglycerides (TG), high-density lipoprotein cholesterol (HDL-C), and low-density lipoprotein cholesterol (LDL-C)], serum uric acid and serum 25(OH)D levels.
All assays were performed in accordance with standardized automated protocols: (1) Flow cytometry for CBC; (2) Hexokinase method for FPG; (3) Immunoassay for 25(OH)D; (4) Enzymatic methods for uric acid (uricase), cholesterol (cholesterol oxidase), and TG (glycerophosphate oxidase–peroxidase method); and (5) Direct surfactant clearance method for HDL-C and LDL-C.
Following the CBC analysis, three systemic inflammatory indices were derived to evaluate the subclinical inflammatory status of the participants: (1) The NLR was calculated by dividing the absolute neutrophil count by the absolute lymphocyte count; (2) The platelet-to-lymphocyte ratio (PLR) was calculated as the absolute platelet count divided by the absolute lymphocyte count; and (3) The systemic immune-inflammation index (SII) was calculated as: SII = (absolute neutrophil count × absolute platelet count)/absolute lymphocyte count.
Genomic DNA was extracted from EDTA-anticoagulated whole blood samples obtained from all participants using the non-enzymatic salting-out method. The region encompassing the FokI polymorphism (rs2228570) was amplified by polymerase chain reaction (PCR) using the following primers: (1) Forward: 5’-GCA ACC TCA GGA AAG CGA TT-3’; and (2) Reverse: 5’-TCT GAG CCA GCT ATG TAG GG-3’. PCR was performed in a 50 μL reaction mixture containing 1× TSE102 Gold Mix, 10 pmol of each primer, and 1 μL of DNA template. The amplification protocol consisted of an initial denaturation at 94 °C for 2 minutes; followed by 5 cycles of denaturation at 94 °C for 30 seconds, annealing at 50 °C for 40 seconds, and extension at 72 °C for 1 minute; and subsequently, 35 cycles of denaturation at 94 °C for 30 seconds, annealing at 55 °C for 40 seconds, and extension at 72 °C for 1 minute; with a final extension at 72 °C for 10 minutes.
The resulting PCR products were purified and subsequently subjected to bidirectional Sanger sequencing. Sequencing reactions were performed using the BigDye Terminator v3.1 Cycle Sequencing Kit, and products were analyzed on an ABI 3500 Genetic Analyzer. The generated electrophoretograms were manually reviewed, and genotype calling for the FokI locus was performed by comparing the sequencing results with the reference sequence (NCBI).
Statistical analyses were conducted using SPSS version 25.0 (IBM Corp., Armonk, NY, United States). Data visualizations were performed using GraphPad Prism version 9.0 (GraphPad Software, San Diego, CA, United States). Continuous variables were compared between groups using the independent samples t-test, while categorical variables, including genotype and allele frequencies, were analyzed by Pearson’s χ² test. Logistic regression analysis was employed to assess the associations among VD deficiency (VDD), VDR FokI polymorphisms, and DR risk, with results expressed as ORs and 95%CI. Differences in clinical characteristics among FokI genotypes were compared using one-way analysis of variance. A two-tailed P < 0.05 was considered statistically significant.
The baseline characteristics and clinical profiles of the study participants are summarized in Table 1. No statistically significant differences were observed among the control, T2DM, and DR groups regarding sex, age, and body mass index (P > 0.05). As expected, significant differences were noted in several diabetes-related parameters. Patients in the DR group had a significantly longer duration of diabetes compared with the T2DM group (P < 0.001). Both systolic blood pressure and DBP were significantly higher in the T2DM and DR groups compared with the control group (P < 0.001 for both). Regarding lipid profiles, serum levels of TG and HDL-C were significantly different among the three groups (P = 0.001 and P < 0.001, respectively), whereas total cholesterol and LDL-C showed no significant differences. A significant stepwise decline in serum 25(OH)D levels was observed from the Control group to the T2DM group and further to the DR group (P < 0.001). Furthermore, systemic inflammatory markers, including NLR, PLR, and SII, all demonstrated a progressive increasing trend from controls to T2DM patients, with the highest levels observed in the DR group (NLR: P = 0.002; PLR: P = 0.016; SII: P = 0.029).
| Observational index | Healthy control group | Type 2 diabetes mellitus group | Diabetic retinopathy group | P value |
| Male | 33 (57.90) | 81 (62.31) | 76 (66.09) | 0.479 |
| Age (years) | 50.43 ± 12.53 | 51.57 ± 11.24 | 53.52 ± 10.87 | 0.191 |
| Body mass index (kg/m2) | 22.29 ± 4.18 | 22.98 ± 3.25 | 23.12 ± 3.38 | 0.319 |
| Diabetes duration (years) | - | 8.00 (2.00, 14.25) | 12.00 (6.00, 18.00) | < 0.001a |
| Family history of diabetes | - | 25 (19.23) | 24 (20.87) | 0.749 |
| History of diabetic retinopathy | - | 5 (3.85) | 21 (18.26) | < 0.001a |
| Systolic blood pressure (mmHg) | 113.30 ± 18.89 | 120.79 ± 19.93 | 128.11 ± 23.21 | < 0.001a |
| Diastolic blood pressure (mmHg) | 70.88 ± 4.52 | 77.81 ± 9.14 | 78.89 ± 10.57 | < 0.001a |
| Serum uric acid (μmol/L) | 335.92 ± 97.22 | 351.10 ± 89.81 | 353.00 ± 94.05 | 0.490 |
| Total cholesterol (mmol/L) | 4.97 ± 0.87 | 4.58 ± 0.93 | 4.56 ± 1.33 | 0.045a |
| Triglycerides (mmol/L) | 1.36 ± 0.64 | 2.42 ± 1.98 | 2.46 ± 2.48 | 0.001a |
| Low-density lipoprotein cholesterol (mmol/L) | 2.93 ± 0.78 | 2.79 ± 0.82 | 2.68 ± 1.18 | 0.277 |
| High-density lipoprotein cholesterol (mmol/L) | 1.53 ± 0.43 | 1.04 ± 0.29 | 1.02 ± 0.33 | < 0.001a |
| Serum 25-hydroxy vitamin D, nmol/L | 65.56 ± 12.78 | 57.89 ± 18.84 | 51.53 ± 21.81 | < 0.001a |
| Fasting plasma glucose (mmol/L) | - | 7.85 ± 3.14 | 7.90 ± 2.96 | 0.898 |
| Neutrophil-to-lymphocyte ratio | 1.52 ± 0.54 | 1.71 ± 0.73 | 1.93 ± 0.85 | 0.002a |
| Platelet-to-lymphocyte ratio | 107.35 ± 43.04 | 112.84 ± 42.98 | 127.26 ± 45.22 | 0.016a |
| Systemic immune-inflammation index | 331.91 ± 151.42 | 376.17 ±194.71 | 422.72 ± 263.50 | 0.029a |
As presented in Table 2, patients with DR exhibited significantly lower 25(OH)D levels compared with both the T2DM and healthy control groups (P < 0.001). Correspondingly, the prevalence of VDD was markedly higher in the DR group (54.78%) than in the T2DM (31.54%) and control (20.69%) groups (Figure 1).
| Vitamin D status | Diabetic retinopathy | Type 2 diabetes mellitus | Healthy control | Odds ratio (95%CI) | P value |
| VDD | 63 (54.78) | 41 (31.54) | 12 (20.69) | 3.09 (1.90-5.01) | < 0.001a |
| Non VDD | 52 (45.22) | 89 (68.46) | 46 (79.31) | Reference | - |
The genotype distribution of the VDR FokI polymorphism is summarized in Table 3. The ff genotype predominated in DR patients (51.30%), followed by Ff (27.83%) and FF (20.87%). In contrast, the Ff genotype was the most frequent in T2DM patients, with ff and FF observed in 36.92% and 23.85%, respectively. Among healthy controls, the Ff genotype accounted for 43.10%, followed by FF (37.93%) and ff (18.97%) (Figure 2).
| Genotype | Diabetic retinopathy (n = 115) | Type 2 diabetes mellitus (n = 130) | Control (n = 58) | Odds ratio (95%CI) | P value |
| ff | 59 (51.30) | 48 (36.92) | 11 (18.97) | 2.36 (1.27-4.39) | 0.007a |
| Ff | 32 (27.83) | 51 (39.23) | 25 (43.10) | 1.09 (0.57-2.08) | 0.792 |
| FF | 24 (20.87) | 31 (23.85) | 22 (37.93) | Reference | |
| Allele frequency | |||||
| f | 150 (65.22) | 147 (56.54) | 47 (40.52) | 1.67 (1.18-2.36) | 0.004a |
| F | 80 (34.78) | 113 (43.46) | 69 (59.48) | Reference |
Clinical and biochemical characteristics of the study participants stratified by VDR FokI genotype (FF, Ff, and ff) are summarized in Table 4. The distribution of baseline variables, including sex, age, blood pressure, lipid profiles, serum uric acid, FPG, and systemic inflammatory indices (NLR, PLR, SII), was compared across the three genotype groups. Notably, no statistically significant differences were found in most clinical parameters among the FF, Ff, and ff genotype groups (P > 0.05; Table 4).
| Variables | FF (n = 77) | Ff (n = 108) | ff (n = 118) | P value |
| Male | 48 (62.34) | 70 (64.81) | 72 (61.02) | 0.838 |
| Age (years) | 52.62 ± 11.98 | 54.56 ± 12.34 | 52.19 ± 11.91 | 0.318 |
| Body mass index (kg/m2) | 22.32 ± 3.25 | 22.68 ± 3.58 | 23.16 ± 3.20 | 0.218 |
| Systolic blood pressure (mmHg) | 116.85 ± 6.22 | 115.95 ± 5.37 | 117.05 ± 5.01 | 0.290 |
| Diastolic blood pressure (mmHg) | 76.12 ± 4.39 | 76.28 ± 4.50 | 76.84 ± 4.70 | 0.492 |
| Serum uric acid (μmol/L) | 340.98 ± 81.86 | 346.95 ± 91.15 | 356.91 ± 100.89 | 0.473 |
| Total cholesterol (mmol/L) | 4.68 ± 1.02 | 4.70 ± 1.04 | 4.58 ± 1.19 | 0.702 |
| Triglycerides (mmol/L) | 2.07 ± 2.13 | 2.17 ± 1.81 | 2.41 ± 2.24 | 0.471 |
| Low-density lipoprotein cholesterol (mmol/L) | 2.80 ± 0.82 | 2.82 ± 0.93 | 2.72 ± 1.10 | 0.742 |
| High-density lipoprotein cholesterol (mmol/L) | 1.19 ± 0.42 | 1.13 ± 0.39 | 1.07 ± 0.36 | 0.100a |
| Serum 25-hydroxy vitamin D (nmol/L) | 59.08 ± 24.46 | 57.89 ± 18.87 | 54.61 ± 16.56 | 0.244 |
| Fasting plasma glucose (mmol/L) | 7.16 ± 3.15 | 7.06 ± 3.01 | 7.57 ± 2.64 | 0.382 |
| Neutrophil-to-lymphocyte ratio | 1.60 ± 0.53 | 1.72 ± 0.81 | 1.84 ± 0.81 | 0.073 |
| Platelet-to-lymphocyte ratio | 110.61 ± 40.27 | 113.48 ± 42.82 | 113.26 ± 44.36 | 0.885 |
| Systemic immune-inflammation index | 351.10 ± 156.80 | 392.70 ± 234.59 | 397.65 ± 234.66 | 0.291 |
This case-control study demonstrates that VDD and the VDR FokI ff genotype are independently associated with an increased risk of DR in a Southwest Chinese population with T2DM. Our findings further elucidate the complex interplay among genetic predisposition, nutritional status, and microvascular complications in diabetes.
The significantly higher prevalence of VDD in DR patients observed in our study (OR = 3.09, P < 0.001) is consistent with accumulating evidence from diverse ethnic populations[16-19]. The robustness of this association underscores the potentially pivotal role of VD status in the development of DR. Beyond the established correlation, our study revealed a significant gradient of decreasing serum 25(OH)D levels from controls to T2DM patients and finally to the DR group (P < 0.001). The role of VD in the pathogenesis of DR extends beyond its classical functions in mineral metabolism[20]. Immunohistochemical and molecular studies have confirmed that nuclear VDR is functionally expressed in multiple retinal cell types, including vascular endothelial cells, pericytes, and retinal pigment epithelial cells[6]. This ocular expression pattern provides an anatomical foundation for direct VD signaling within the retina, suggesting that local VDR activation may play a role in maintaining retinal homeostasis and mitigating diabetic microvascular damage.
A distinct inverse relationship was evident in our cohort: As VD status progressively declined from controls to T2DM patients, and finally to the DR group, the levels of systemic inflammatory indices (NLR, PLR, and SII) increased in parallel. This association provides a clinical correlate to the postulated anti-inflammatory mechanisms of VD, suggesting that the pro-inflammatory state is intrinsically linked to – and potentially driven by – VDD.
Several biological mechanisms may underlie this association. Firstly, VD exerts potent anti-inflammatory effects by suppressing pro-inflammatory cytokines such as IL-1β, TNF-α, and IL-6, which are elevated in DR and contribute to blood-retinal barrier disruption[21,22]. Secondly, its anti-angiogenic effects via inhibition of VEGF expression directly counter a key driver of diabetic macular edema and proliferative DR[23]. Thirdly, VD bolsters antioxidant defense systems, thereby mitigating oxidative stress in retinal tissues subjected to hyperglycemic damage[24]. Additionally, emerging evidence suggests VD may protect retinal pericytes from apoptosis and modulate mitochondrial function in retinal neurons[25]. Collectively, these pleiotropic actions position VD as a crucial modulator in the retinal microenvironment; its deficiency disrupts multiple protective pathways and thereby accelerates the progression of DR.
The association between the FokI ff genotype and elevated DR risk observed in our cohort (OR = 2.36, P = 0.007) aligns with broader epidemiological research[26,27], wherein this genetic variant has been consistently implicated as a risk factor for multiple diabetes-related complications across diverse populations. The functional significance of this polymorphism stems from its effect on VDR structure – the shorter 424-amino acid protein (encoded by the F allele) exhibits enhanced transcriptional activity compared with the full-length variant (encoded by the f allele). This suggests that the ff genotype, which produces the less active full-length isoform, may compromise receptor efficacy. This structural alteration may influence the receptor’s interaction with co-activators, DNA-binding affinity, or nuclear localization, ultimately modifying VD signaling efficiency.
Although our study demonstrates a significant association between the FokI polymorphism and DR risk, the precise underlying molecular mechanisms remain to be fully elucidated. Future research should explore whether this association is mediated through the dysregulation of inflammatory pathways or metabolic signaling cascades, which are known to be involved in DR pathogenesis and are potentially modulated by VD signaling in other tissues[28-30].
Population-specific associations observed may also be influenced by linkage disequilibrium between the FokI polymorphism and other functional variants in the VDR gene. Beyond direct transcriptional effects, the functional consequences of the FokI polymorphism may extend to modulating inflammatory and angiogenic pathways relevant to DR. Consequently, future mechanistic studies are warranted to explore whether these effects are mediated by alterations in non-genomic VDR actions or crosstalk with other signaling cascades.
The concurrent assessment of VD status and VDR genotype in our study elucidates their potentially synergistic roles in DR risk. Notably, both factors remained independently significant in multivariate analysis, suggesting distinct contributions to disease pathogenesis. This observation supports a model in which environmental factors (VD status) and genetic determinants (VDR polymorphisms) modulate individual susceptibility to microvascular complications.
From a clinical standpoint, the combination of biochemical and genetic markers could improve risk stratification for DR. T2DM patients presenting with VDD and the FokI ff genotype exhibited particularly high DR risk in our cohort, potentially identifying a subgroup that would benefit from more frequent ophthalmological screening and targeted risk factor modification.
Several strengths of the present study warrant mention, including the use of bidirectional Sanger sequencing for accurate genotype determination, standardized chemiluminescence immunoassays for VD measurement, and careful matching of study groups. However, certain limitations merit discussion. First, the cross-sectional design precludes causal inference, and the sample size, though sufficient for detecting strong genetic effects, may be underpowered for identifying modest associations or gene-environment interactions.
Second, unmeasured confounders – such as physical activity, detailed sun exposure, dietary habits, and other genetic variants – may have partially influenced the observed associations. We did not account for individual-level sunlight exposure, dietary VD intake, or sampling season, factors known to affect serum 25(OH)D levels[31]. Although participants were recruited throughout the year, which may help mitigate seasonal variation, residual confounding cannot be ruled out. Future research should incorporate comprehensive assessments of sun exposure and lifestyle factors. Third, the absence of data on DR severity staging (e.g., non-proliferative vs proliferative DR) limited our ability to examine whether the identified factors are associated with specific stages of disease progression. Finally, the single-center design and focus exclusively on Han Chinese participants restricts the generalizability of our findings to other ethnic populations.
Our findings underscore several promising avenues for future research. Prospective longitudinal studies tracking VD status and genetic factors from diabetes onset could clarify their temporal relationship with DR development. Intervention trials examining whether VD supplementation benefits specific genetic subgroups would have significant clinical relevance for precision medicine. Mechanistic studies exploring VDR function in specific retinal cell types and the impact of the FokI polymorphism on retinal gene expression profiles are needed to elucidate the underlying biological processes.
If confirmed in larger, prospective cohorts, the combination of VD status and VDR genotype might eventually contribute to a multifactorial risk model for DR. Meanwhile, expanding genetic analyses to include additional VDR polymorphisms (e.g., BsmI, ApaI, TaqI) and genes involved in VD metabolism would offer a more comprehensive understanding of the genetic determinants of DR risk. Additionally, investigating whether VDR polymorphisms modify responses to anti-VEGF therapy or other DR treatments represents an intriguing pharmacogenetic research avenue.
This study demonstrates significant associations among VDD, the VDR FokI polymorphism, and DR risk in our study population. Our findings strengthen the evidence supporting the relevance of VD pathway alterations in DR pathogenesis. Future research should focus on elucidating the underlying mechanisms of these associations and assessing targeted interventions for high-risk individuals identified by both VD status and genetic profile.
The authors would like to thank the researchers and studies whose work contributed to the foundation of this article. Additionally, we acknowledge the support provided by the First Affiliated Hospital of Kunming Medical University in facilitating this research.
| 1. | Jin C, Lai Y, Li Y, Teng D, Yang W, Teng W, Shan Z; China National Diabetes and Metabolic Disorders Study Group and the Thyroid Disorders, Iodine Status and Diabetes Epidemiological Survey Group. Changes in the prevalence of diabetes and control of risk factors for diabetes among Chinese adults from 2007 to 2017: An analysis of repeated national cross-sectional surveys. J Diabetes. 2024;16:e13492. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 18] [Reference Citation Analysis (0)] |
| 2. | Shyam M, Sidharth S, Veronica A, Jagannathan L, Srirangan P, Radhakrishnan V, Sabina EP. Diabetic retinopathy: a comprehensive review of pathophysiology and emerging treatments. Mol Biol Rep. 2025;52:380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 3. | Li H, Liu X, Zhong H, Fang J, Li X, Shi R, Yu Q. Research progress on the pathogenesis of diabetic retinopathy. BMC Ophthalmol. 2023;23:372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 54] [Reference Citation Analysis (0)] |
| 4. | Zhang D, Zhang Y, Kang J, Li X. Nonlinear relationship between diabetes mellitus duration and diabetic retinopathy. Sci Rep. 2024;14:30223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 5. | Argano C, Mirarchi L, Amodeo S, Orlando V, Torres A, Corrao S. The Role of Vitamin D and Its Molecular Bases in Insulin Resistance, Diabetes, Metabolic Syndrome, and Cardiovascular Disease: State of the Art. Int J Mol Sci. 2023;24:15485. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 101] [Reference Citation Analysis (1)] |
| 6. | Song YS, Jamali N, Sorenson CM, Sheibani N. Vitamin D Receptor Expression Limits the Angiogenic and Inflammatory Properties of Retinal Endothelial Cells. Cells. 2023;12:335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 18] [Reference Citation Analysis (0)] |
| 7. | Yang J. Genetically Predicted Serum 25-Hydroxyvitamin D Concentrations in Related to Type 2 Diabetes Mellitus: A Mendelian Randomization Study. Endocrinol Diabetes Metab. 2025;8:e70050. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 8. | Usategui-Martín R, De Luis-Román DA, Fernández-Gómez JM, Ruiz-Mambrilla M, Pérez-Castrillón JL. Vitamin D Receptor (VDR) Gene Polymorphisms Modify the Response to Vitamin D Supplementation: A Systematic Review and Meta-Analysis. Nutrients. 2022;14:360. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 155] [Cited by in RCA: 123] [Article Influence: 30.8] [Reference Citation Analysis (1)] |
| 9. | Awasthi R, Manger PT, Khare RK. Fok I and Bsm I gene polymorphism of vitamin D receptor and essential hypertension: a mechanistic link. Clin Hypertens. 2023;29:5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 10. | Jurutka PW, Remus LS, Whitfield GK, Thompson PD, Hsieh JC, Zitzer H, Tavakkoli P, Galligan MA, Dang HT, Haussler CA, Haussler MR. The polymorphic N terminus in human vitamin D receptor isoforms influences transcriptional activity by modulating interaction with transcription factor IIB. Mol Endocrinol. 2000;14:401-420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 212] [Cited by in RCA: 242] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 11. | Jiao J, Li Y, Xu S, Wu J, Yue S, Liu L. Association of FokI, TaqI, BsmI and ApaI polymorphisms with diabetic retinopathy: a pooled analysis of case-control studies. Afr Health Sci. 2018;18:891-899. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 12. | Neves JPR, Queiroz DJM, Araújo EPDS, Lacerda LM, Nascimento RAFD, Carvalho AT, Costa MJC, França AKTDC, Persuhn DC, Silva AS, Diniz ADS, Gonçalves MDCR. Variants rs1544410 and rs2228570 of the vitamin D receptor gene and glycemic levels in adolescents from Northeast Brazil. Nutr Hosp. 2020;37:21-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 13. | Liu Z, Liu L, Chen X, He W, Yu X. Associations study of vitamin D receptor gene polymorphisms with diabetic microvascular complications: a meta-analysis. Gene. 2014;546:6-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 36] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 14. | ElSayed NA, Aleppo G, Aroda VR, Bannuru RR, Brown FM, Bruemmer D, Collins BS, Hilliard ME, Isaacs D, Johnson EL, Kahan S, Khunti K, Leon J, Lyons SK, Perry ML, Prahalad P, Pratley RE, Seley JJ, Stanton RC, Gabbay RA; on behalf of the American Diabetes Association. 2. Classification and Diagnosis of Diabetes: Standards of Care in Diabetes-2023. Diabetes Care. 2023;46:S19-S40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2057] [Cited by in RCA: 1712] [Article Influence: 570.7] [Reference Citation Analysis (5)] |
| 15. | Li L, Chen X, Yi X. Association between vitamin D receptor polymorphisms and diabetic retinopathy in Uygur Chinese with type 2 diabetes. Ophthalmic Genet. 2025;46:261-266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 16. | Chackochan A, Reghunath SR, Guddattu V, Thunga G, Nagri SK, Shenoy RP, Shenoy VP, Acharya LD. Vitamin D status and its association with diabetic complications in type 2 diabetes mellitus: A case-control study. Nutr Health. 2025;2601060251359708. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 17. | Hung KC, Chang LC, Chang YJ, Ho CN, Wu JY, Liu WC, Chen IW. Vitamin D deficiency and diabetic retinopathy risk in patients with newly diagnosed type 2 diabetes mellitus: a retrospective analysis. Front Nutr. 2025;12:1614287. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 18. | Melake A, Alamnie G, Mekonnen M. Impact of Vitamin D Deficiency and VDRTaqI Polymorphism on Diabetic Retinopathy Risk Among T2DM Ethiopian Population. Food Sci Nutr. 2025;13:e70197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 19. | Dervis N, Carniciu S, Spinean A, Jurja S. Synergistic Association of Glycemic Variability and Severe Vitamin D Deficiency with Proliferative Diabetic Retinopathy. Nutrients. 2025;17:3210. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 20. | Wimalawansa SJ. Physiology of Vitamin D-Focusing on Disease Prevention. Nutrients. 2024;16:1666. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 46] [Reference Citation Analysis (1)] |
| 21. | Padovani-Claudio DA, Morales MS, Smith TE, Ontko CD, Namburu NS, Palmer SA, Jhala MG, Ramos CJ, Capozzi ME, McCollum GW, Penn JS. Induction, amplification, and propagation of diabetic retinopathy-associated inflammatory cytokines between human retinal microvascular endothelial and Müller cells and in the mouse retina. Cell Signal. 2024;124:111454. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 22. | Fenercioglu AK. The Anti-Inflammatory Roles of Vitamin D for Improving Human Health. Curr Issues Mol Biol. 2024;46:13514-13525. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 71] [Cited by in RCA: 61] [Article Influence: 30.5] [Reference Citation Analysis (0)] |
| 23. | Dervis N, Jurja S, Cioti C, Stoica AM. Protective Role of Vitamin D Supplementation in Proliferative Diabetic Retinopathy in a Type 1 Diabetes Premenopausal Woman: A Case Report from the region of Dobrogea, Romania. Rom J Ophthalmol. 2025;69:460-463. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 24. | Yang X, Qi X, Zuo K, Huang Y, Bian X, Wang J, Yu H, Feng Q, Lei X, Chen T. Vitamin D alleviation of oxidative stress in human retinal pigment epithelial cells. Int Ophthalmol. 2024;44:314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 25. | Lu L, Lu Q, Chen W, Li J, Li C, Zheng Z. Vitamin D(3) Protects against Diabetic Retinopathy by Inhibiting High-Glucose-Induced Activation of the ROS/TXNIP/NLRP3 Inflammasome Pathway. J Diabetes Res. 2018;2018:8193523. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 122] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 26. | Vania A, Samatra DPGP, Adnyana IMO, Saraswati MR, Darwinata AE, Widyadharma IPE. Vitamin D receptor FokI polymorphism as a risk factor for painful diabetic neuropathy in type 2 diabetes mellitus patients. J Neurogenet. 2025;39:7-15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 27. | Zhao Y, Liu Z, Feng S, Yang R, Ran Z, Zhu R, Ma L, Wang Z, Chen L, Han R. The association between vitamin D receptor gene polymorphism FokI and type 2 diabetic kidney disease and its molecular mechanism: a case control study. BMC Med Genomics. 2024;17:288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 28. | Chen J, Wang Q, Li R, Li Z, Jiang Q, Yan F, Ye J. The role of Keap1-Nrf2 signaling pathway during the progress and therapy of diabetic retinopathy. Life Sci. 2024;338:122386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 29. | Lu S, Cai J. Targeting therapy of PI3K/AKT signaling pathway via non-coding RNAs in diabetic retinopathy. Naunyn Schmiedebergs Arch Pharmacol. 2025;398:11493-11504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 30. | Liu L, Gao Y, Yao S. Transthyretin-Regulated Diabetic Retinopathy Through the VEGFA/PI3K/AKT Pathway. Invest Ophthalmol Vis Sci. 2024;65:45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 31. | Delrue C, Speeckaert MM. Vitamin D and Vitamin D-Binding Protein in Health and Disease. Int J Mol Sci. 2023;24:4642. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 100] [Cited by in RCA: 84] [Article Influence: 28.0] [Reference Citation Analysis (1)] |